Abstract
Introduction:
Uveal melanoma (UM) is a subtype of melanoma arising from the ocular region. Despite various local therapies available, a significant portion of patients develop distant metastases, primarily to the liver. While cutaneous melanoma is very sensitive to immunotherapy, UM is known to be less responsive and patients were excluded from pivotal clinical trials. To date, there is no standard first line therapy for metastatic UM and clinical trial participation is encouraged. While UM is considered a radio-resistant tumor, there is a role for radiotherapy (RT) as palliative treatment and possibly for immune sensitization. This a retrospective analysis aimed at addressing the role of combination checkpoint inhibitors (ICI) with RT as a synergistic treatment in metastatic UM patient. We hypothesized that concurrent RT would improve the clinical response to immunotherapy.
Methods:
Retrospective chart review of patients with metastatic UM treated with ICI at Ella Lemelbaum Institute between 2015 and 2021. Patients’ electronic medical records were analyzed for baseline characteristics, response rate and survival data. Patients were grouped according to receipt of concomitant RT. Study was approved by local IRB and statistical analyses done using Stata V.17
Results:
Thirty-nine patients were treated with immunotherapy. Fifty percent were treated with anti-programmed cell death (PD)-1 and 50% with anti-PD1– anti CTLA4 combination therapy. Nine patients were treated concomitantly with immunotherapy and external beam RT or with stereotactic body RT (group A) and 29 patients were treated with immunotherapy alone (group B). Overall response rate was significantly higher in group A (44% versus 10%, p = 0.004). Median progression-free survival was longer for patients in group A (22 months versus 3 m, Hazard Ratio (HR) = 0.37, p = 0.036). Median overall survival was also longer for group A (26 months versus 7.5 m, HR = 0.34, p = 0.03). Toxicity was comparable between the groups.
Conclusions:
RT may improve response to immunotherapy with ICI in metastatic UM patients and may confer an advantage in survival. Further prospective, larger studies are warranted.
Introduction
For many years, malignant melanoma has been considered a fatal disease with very limited treatment options available for metastatic disease. Recent advances in the field of immunotherapy has revolutionized the outcomes for patients with metastatic melanoma with a significant increase in overall survival (OS) seen in the last decade. 1
While the majority of malignant melanoma cases arise from the skin (cutaneous), there are other subtypes of melanoma that arise from mucosal membranes and from the ocular region. 2
These subtypes are known to be less responsive to commonly used immunotherapy in comparison to cutaneous melanoma (CM) and therefore there is still an unmet need for improved treatment regimens for these histological subtypes.3–5
Of particular interest are Melanomas arising from the ocular region, with uveal melanoma (UM) being the most common subtype of ocular Melanoma. 6 Despite the fact that UM is the most common subtype of ocular Melanoma, it is still a rare disease with an incidence of 1.3–8.6 cases per million per year. 2 The main risk factors for UM include older age, fair skin, light eye color and inability to tan. 7
While UM can sometimes arise in the iris or ciliary body, the vast majority (90%) of cases arise from the choroid region. 2 It has been previously established that this anatomic location is associated with a higher risk to metastasize leading to a poorer prognosis.8,9
Choice of local therapy for early stage disease is dependent on tumor location, extent, size and visual acuity. The most common therapy in use today is plaque radiotherapy (RT) and only a small number of patients undergo Enucleation – a method preserved for more advanced cases. In cases of massive extraocular extension, orbital exenteration is performed. 10
Despite the various local therapies available, a significant portion of patients may develop distant metastases with a rate as high as 62% over 35 years. 9 The most common metastatic site is the liver accounting for about 90% of the cases, with other less common sites being the lungs, bone, skin, subcutaneous tissues and lymph nodes (8).
Treatment options for metastatic disease are very limited and immunotherapy has been studied in small series with low response rates.5,11–14
To date, there is no standard first line of treatment for metastatic UM and clinical trial participation is encouraged. Acceptable treatment options include local direct liver therapy, immune checkpoint inhibitors (ICIs), MEK inhibitors, cytotoxic chemotherapy and recently – Tebentafusp, a new class of T-cell–redirecting bispecific fusion proteins, that showed improved OS for patients with HLA-A*02:01 compared to the control group (composed of single-agent pembrolizumab, ipilimumab or dacarbazine). 15
The role of ICI is well established in metastatic CM, as a single agent or in combination therapy.3,16 However, there is only scarce data about its use in UM5,11–13 as these patients were excluded from the pivotal clinical trials. The biology of UM is different from CM, expressing a low tumor mutational burden and a limited number of neoantigens. 17 Therefore UM is considered immunologically as a ‘cold tumor’ with a low response rate to ICI. Clinical experience shows that although response rate is low, some patients do have a prolonged benefit from ICI therapy.
A large retrospective series of anti-programed death-1 (PD-1) and anti-programed death ligand-1 agents in UM showed a poor outcome with only 3% overall response rate (ORR), and a 10- month median OS. The authors concluded that this treatment rarely confers durable remissions in patients with metastatic UM and clinical trial enrollment should be prioritized in this population. 14
Combination immunotherapy with anti-PD1 and anti-cytotoxic T-lymphocyte associated antigen-4 (CTLA-4) was tested in a single arm phase II study and showed a modest improvement in OS over historical benchmarks of chemotherapy (median OS 12.7 versus 9.3 months) and a 3 month progression-free survival (PFS), with a manageable toxicity profile. 13 Response rate was evaluated in a prospective single arm phase II study that included 33 patients. The ORR was 18% with one patient achieving complete response (CR) and another five achieving partial responses (PRs). The median PFS was 5.5 months (95% confidence interval (CI) 3.4–9.5 months) and median OS was 19.1 months (95% CI 9.6 months to Not Reached (NR)). 12 These results are better compared with historical results of prior therapies including single-agent ICI. 14
RT has a well-established palliative role in the treatment of unresectable metastatic cancer, such as in the treatment of visceral, skeletal and brain metastasis. 18 Melanoma is considered a relatively radio-resistant tumor due to a broad shoulder in the cell survival curves. 19 However, there is still a role for palliative and adjuvant RT in melanoma. 18
There is now an increasing interest in the use of RT as an immune sensitizer. RT has the ability to affect the irradiated site, as well as distant sites, outside the radiation field. This is commonly known as the abscopal effect which is considered to be an immune mediated response. 20 It has been hypothesized that RT and ICI can work in a synergistic fashion with the main mechanism being the liberation of tumor neo-antigens which then enhance the immune system’s response to ICI treatment. 21 This hypothesis was tested in pre-clinical trials 22 and in small retrospective and prospective phase I/II clinical trials in metastatic melanoma.23,24 A comprehensive review by D’andrea and Reddy 25 included current data available in the field. Most of the clinical evidence about abscopal effects of radiation therapy have been reported in the form of case reports and small nonrandomized studies. 25 In these studies, abscopal effect was estimated in 21–38% of the patients receiving both immunotherapy and RT. This effect was noticed in both anti CTLA-4 and anti-PD-1 agents and in different types of radiation regimens, with hypofractionation being the most common protocol. Radiation was given to both intra and extra cranial sites and response was noticed in and outside the radiation site, both intra and extra cranial.26–28
Another radiation-related treatment option that is being examined in UM is the possibility of combination treatment of immunotherpay + trans-arterial radioembolization selective internal radiation (SIRT) to the liver. A retrospective analysis of 24 patients with UM and hepatic metastases described the results of treatment with yttrium-90 (Y90) SIRT. 29 Half of the patients were treated with immunotherapy within 3 months of SIRT and half received treatment other than immunotherapy. Response rates to therapy did not differ significantly between the groups; however, the addition of Immunotherapy to SIRT prolonged PFS and OS significantly compared to patients who had received therapy with SIRT alone or SIRT + non-immunotherapy treatment.
A prospective study published in early 2022. 30 further analyzed the utility of hepatic SIRT therapy with Y90 in 26 metastatic UM patients. Patients received concomitant therapy with SIRT Y90 + immunotherapy with ipilimumab–nivolumab combination therapy at varying doses. The primary objective of this study was to assess tolerability of this treatment regimen and indeed grade 3–4 hepatitis was noted in almost approximately 50% of the patients leading to dose reduction in the RT regimen to limit normal liver dose to 35 Gy. Responses to therapy were seen in 20% of patients with a very high disease control rate of 68%. Median PFS was 5.5 months and median OS 15 months.
UM patients are occasionally offered external beam RT for palliative reasons, usually for visceral or bone metastasis, or as a local liver metastatic treatment with or without a systemic therapy. Although currently there is a role for both external beam RT and ICI treatment in UM, there is no data about the effect of combining treatment in this poor prognosis disease.
We sought to examine the possibility of a synergistic effect of combination ICI and RT in UM and hypothesized that the addition of RT to standard immunotherapy would improve ORR and prolong survival.
Methods
Patients and data
A retrospective chart review was performed. Inclusion criteria were metastatic UM patients, above the age of 18 years, treated at the Ella Lemelbaum Institute for Immuno-Oncology with ICI between the years 2015–2021. ICI could consist of anti-PD-1 therapy (nivolumab or pembrolizumab) or ipilimumab–nivolumab combination therapy, as first, second or a subsequent line. Patients that have received more than one line of immunotherapy (i.e., anti-PD-1, anti-CTLA4 or their combination) were only evaluated once for their best response. The population was split into cohorts (A and B) based upon whether they received external beam radiation (EBRT) or stereotactic body radiotherapy (SBRT) therapy within 6 months of commencing treatment. Patients who received RT with immunotherapy were defined as group A, patients who were treated with immunotherapy alone were defined as group B.
Medical records were analyzed for baseline patient characteristics, ORR defined as best response and for survival data.
Response was evaluated by the treating oncologist using clinical and radiological outcomes, based upon official measurements assessed by Israeli board certified radiologists or nuclear medicine experts using the iRECIST criteria. 31
Toxicity grading was done using the common terminology criteria for adverse events (CTCAE) v. 5. 32
Statistics
Records were analyzed for baseline parameters and response to therapy using a chi square test with a pre-defined alpha score of 0.05 for statistical significance. PFS and OS as defined by time from treatment initiation were assessed using Kaplan-Meier method and Cox-regression analyses. Toxicity grading was evaluated using the CTCAE v. 5 32 Toxicity comparison between the groups was done using chi square test. All statistical analyses were done with Stata v. 17 (Timberlake).
Results
Thirty-eight patients with metastatic UM were included in the study. Median age was 61 (range 32–90), 50% were female, 82% of the patients had an ECOG performance status of 0–1.
All baseline parameters (age, ECOG performance status, serum lactate dehydrogenase levels, disease stage and line of therapy) were comparable between the groups apart from a trend towards more use of ipilimumab–nivolumab combination in group A (p = 0.06). See Table 1 for baseline characteristics of both groups.
Baseline characteristics.

IO, immuno-oncology; RT, radiotherapy; ECOG PS, eastern cooperative oncological group performance status; LDH, lactate dehydrogenase; PD-1, programed death-1; anti-PD-1, pembrolizumab or nivolumab; UNL, Upper normal limit.
p = 0.06.
Single agent anti PD-1 therapy was given to 50% of the entire study population while ipilimumab–nivolumab combination was given to the other 50%. ORR for the entire cohort was 18% and median PFS was 3.5 months.
Nine patients of the entire 38 patients cohort received RT concomitantly with the immunotherapy treatment (group A), 29 patients did not receive RT (group B). All patients received the RT course within the first 6 months of therapy with a median time from treatment initiation to RT of 3 months (range 0–6 months).
Five patients were treated with liver SBRT – all received SBRT to the largest liver metastasis and one of the five also received SBRT to the second largest liver metastasis. The other four patients received different types of radiation – one patient received stereotactic radiosurgery for two symptomatic brain metastasis, one patient received EBRT to an intra-luminal metastasis in the stomach and the last two patients received radiation to bone metastasis, one SBRT and one EBRT. All patients were treated with palliative intent. Median RT dose given to the planning target volume was 24 Gy (8–60 Gy), which was given in a median number of three fractions (1–5). Please refer to Table 2 for RT regimen details.
RT regimen details.

PTV, planning tumor volume; GTV, gross tumor target volume; RT, radiotherapy; SBRT, stereotactic body radiotherapy; SRS, stereotactic radiosurgery; FSR, fractionated stereotactic radiotherapy.
ORR was significantly higher in group A at 44% compared to 10% in group B (p = 0.004). Responses were more substantial in group A patients with three out of the four patients (75%) who responded achieving a CR, while in group B, there were no CRs at all. For all four patients who responded in group A the response was seen in both the irradiated and the non-irradiated lesions. When looking at ORR in relation to RT only in the patients treated with the combination of ipilimumab–nivolumab in both groups, the results remain similar: an ORR of 57% (4/7) in the patients treated in combination with RT compared to 9% (1/11) in patients treated with ipilimumab–nivolumab combination without RT (p = 0.026 for response). Please refer to Table 3 for response data for both groups and Tables 4 and 5 for specific data about response with regard to the systemic treatment given.
Response patterns in all patients (best response).

IO, immuno-oncology; RT, radiotherapy; ORR, overall response rate; PR, partial response; CR, complete response; SD, stable disease; PD, progressive disease.
Response patterns in IO combination patients.

Abbreviations: IO, immuno-oncology; RT, radiotherapy; Ipi, ipilimumab; Nivo, nivolumab; ORR, overall response rate; PR, partial response; CR, complete response; SD, stable disease; PD, progressive disease.
Response patterns in single agent anti-PD-1* patients.

RT, radiotherapy; ORR, overall response rate; PR, partial response; CR, complete response; SD, stable disease; PD-1, programed death-1.
Single agent anti-PD-1, pembrolizumab or nivolumab.
Median follow-up for the entire cohort was 8.5 months (1–64 months). Median follow-up time for group A was 26 months (4–45 months) and for group B was 8 months (1–64 months). PFS was significantly longer for group A with a median of 22 months compared to 3 months in group B (HR = 0.37 for progression or death, p = 0.036). Please refer to Figure 1 for Kaplan–Meier PFS survival curves. OS was calculated as time from the initiation of immunotherapy to death or data censorship. The OS was significantly longer for group A with a median of 26 months compared to 7.5 months for group B (HR = 0.34 p = 0.03). See Figure 2 for Kaplan–Meir OS curves.

Progression-free survival.
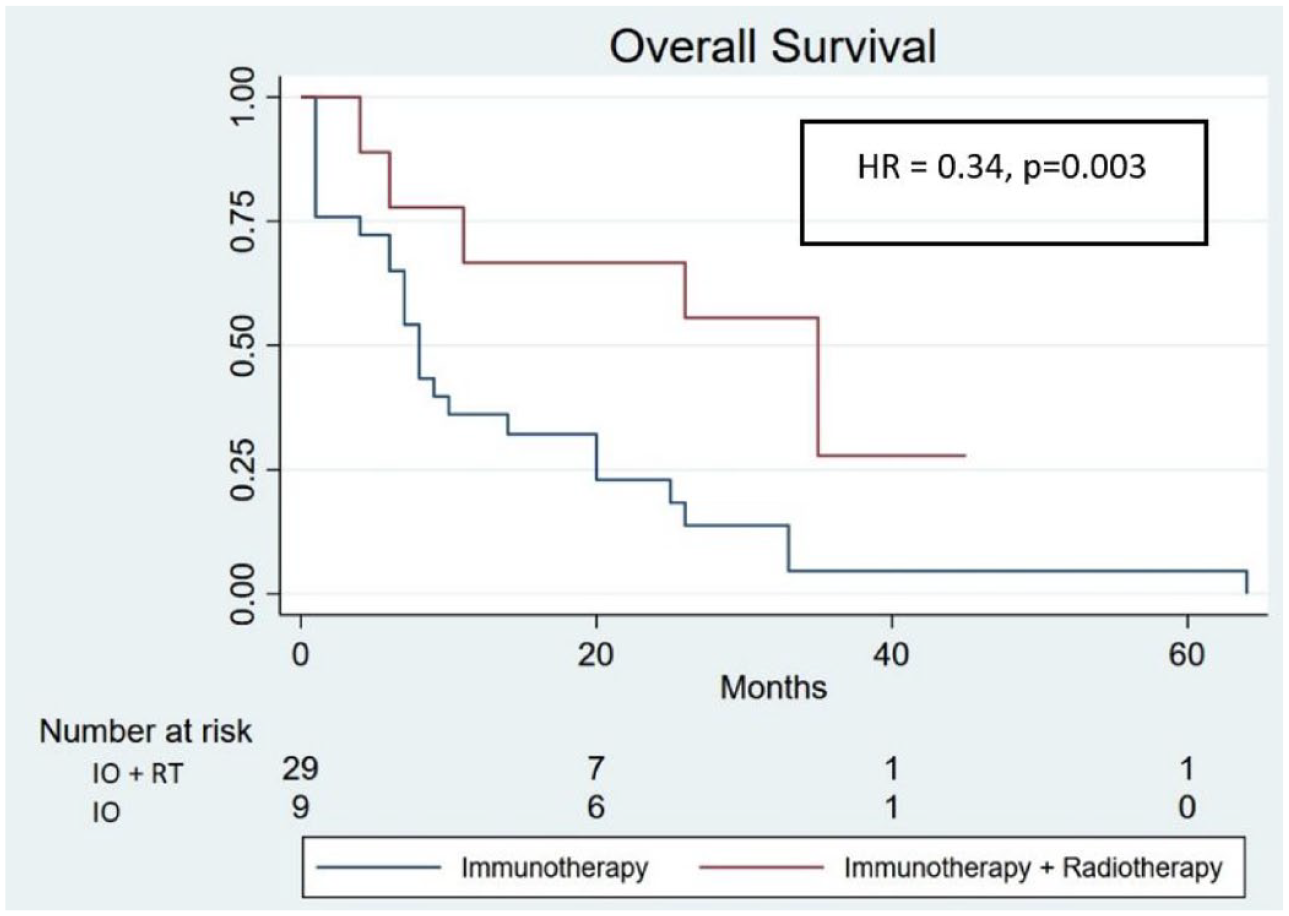
Overall survival.
Toxicity was comparable between the two groups with 3/12 patients (33%) developing G3–4 toxicity in group A in comparison to 8/29 (28%) patients in group B (p = NS). All patients who developed G3–4 toxicities required the use of systemic corticosteroids. The use of combination ipilimumab + nivolumab was the cause of the G3–4 toxicities in all three patients in group A and for 5/8 (62%) patients in group B (with the other three patients receiving single agent Pembrolizumab). It is worth mentioning that all three patients who developed G3 4 toxicity in group A had achieved a response (two CR and one PR), while there were no responses seen in any of the eight patients in group B. Details regarding the different toxicities are available in Table 6.
Toxicities associated with immunotherapy in both groups.

IO, immunotherapy; RT, radiotherapy; G3–4, grades 3–4; GI, gastrointestinal.
Discussion
Immunotherapy has brought a new era in the treatment of metastatic melanoma with a significant prolongation of OS. Yet, there are unmet needs for subtypes that do not seem to benefit from these agents, mainly for mucosal and ocular melanomas. The worst prognosis to date is of UM patients for whom the novel drugs have had only a marginal effect on survival. The rarity of this disease along with its non-immunogenic nature has led to exclusion of these patients from the pivotal clinical trials, with only small phase II studies available demonstrating unfavorable results. A need for more efficient therapeutic options is dire, with the main goal of activating and targeting the immune response against UM metastasis. The notion of improving the effectivity of ICIs with the use of RT as an immunosensitizing tool is rapidly evolving, yet currently available data is scarce and include pre-clinical, case reports and small retrospective and prospective phase 1–2 studies in CM. While some data regarding SIRT + immunotherapy in UM is available, there are to our knowledge no significant data regarding external beam RT and immunotherapy efficacy in UM.
In this study, we sought to look for a first signal of such an interaction by reviewing UM patient files. Comparing those patients who received concomitant RT with ICI to those who received only standard ICI treatment. Although the number of patients in this study is small, the signal observed is intriguing with a statistically significant advantage to the combination of modalities (ICI + RT) in terms of response rate, response patterns, PFS and OS. It is worth mentioning that for all four patients who responded in group A (three were CR and one was PR), the responses were seen in the irradiated lesions as well as in lesions not included in the radiation field.
The major limitations of this study are of course its retrospective nature, the small sample size and the difference in the ICI protocol used by each group (with the ICI + RT group utilizing the ipilimumab + nivolumab combination more than the ICI group alone). Nevertheless, analysis of response rate in only those patients who received ipilimumab–nivolumab also showed a statistically significant advantage to addition of radiation to treatment. In addition, the fact that three patients of the nine in the combined modality group have achieved a CR is a strong hint at the possible benefits that such a combination might have on the outcomes of metastatic UM patients. Of note also is the difference in median follow-up time between the groups. It is worth emphasizing that the prolonged follow-up time in the group of patients receiving ICI + RT is attributed to the portion of patients with CR and significantly prolonged OS. The idea of adding localized palliative intent RT to ICI treatment seems to be safe and feasible. Due to the inherent limitations of this study, it may serve as hypothesis generating and warrants further prospective studies.
