Abstract
Background:
Current research presents conflicting evidence on whether pre-existing interstitial lung disease (ILD) serves as a risk factor for radiation pneumonitis (RP) and checkpoint inhibitor pneumonitis (CIP) in patients with lung cancer.
Objectives:
This study aims to systematically evaluate the impact of pre-existing ILD on the risk of developing RP and CIP in lung cancer patients.
Design:
A systematic review and meta-analysis was conducted using a random-effects model.
Data sources and methods:
PubMed, Embase, and Web of Science were searched to identify relevant studies. A random-effects model was applied to estimate the risk and incidence of RP and CIP in lung cancer patients with pre-existing ILD compared to those without ILD. Sensitivity analyses were performed to assess the robustness of the pooled findings, and potential publication bias was evaluated using Begg’s and Egger’s tests.
Results:
A total of 12 studies involving 2576 patients were included in the RP risk assessment, while 29 studies with 8037 patients were analyzed for CIP risk. The pooled results indicated that pre-existing ILD significantly increased the risk of developing any-grade RP (odds ratio (OR): 3.63, 95% confidence interval (CI): 2.26–5.83) and severe RP (OR: 6.10, 95% CI: 2.68–13.86) in lung cancer patients. Subgroup analyses identified stereotactic body radiation therapy as the modality associated with the lowest risk of any-grade RP in these patients. Similarly, pre-existing ILD was associated with a significantly higher risk of any-grade CIP (OR: 3.86, 95% CI: 2.65–5.61) and severe CIP (OR: 3.24, 95% CI: 2.07–5.07), with anti-programmed cell death 1 therapies showing the highest CIP risk.
Conclusion:
Pre-existing ILD markedly increases the risk of both RP and CIP in lung cancer patients. These findings underscore the critical importance of thorough ILD evaluation and the development of personalized treatment strategies to mitigate these risks prior to initiating cancer therapy.
Keywords
Introduction
Lung cancer remains one of the leading causes of cancer-related morbidity and mortality worldwide, contributing significantly to annual cancer diagnoses and deaths.1,2 This burden is particularly pronounced among individuals with a history of smoking, exposure to environmental pollutants, or genetic predispositions, which contribute to the complex etiology of the disease. 3 Despite these challenges, significant advancements in lung cancer treatment have been achieved in recent years. For early-stage disease, surgical resection remains the cornerstone of curative therapy. 4 However, as lung cancer is often diagnosed at advanced stages, a comprehensive and multifaceted therapeutic approach is required. 4 Treatment options, such as chemotherapy, targeted therapy, radiotherapy, and immunotherapy, each play critical roles by employing distinct mechanisms to combat tumor growth and metastasis, particularly in advanced-stage disease.
Radiotherapy, a cornerstone of cancer treatment, is highly effective at controlling tumor progression and alleviating symptoms. However, its application is often limited by the risk of radiation pneumonitis (RP), a potentially severe complication that restricts therapeutic dosing. Similarly, immune checkpoint inhibitors (ICIs) have revolutionized lung cancer management by enabling immune-mediated tumor destruction, significantly reshaping the treatment landscape. 5 However, ICIs are associated with immune-related adverse events, most notably checkpoint inhibitor pneumonitis (CIP), which can pose significant challenges in clinical practice. 5 Given these risks, careful patient selection and monitoring are crucial to ensure both safety and efficacy. Identifying risk factors for RP and CIP is, therefore, a critical step toward minimizing adverse events and advancing precision oncology.
Interstitial lung disease (ILD) encompasses a diverse group of pulmonary disorders characterized by varying degrees of inflammation and fibrosis in the lung interstitium. 6 Radiographic studies indicate that ILD is present in approximately 14% of treatment-naïve patients with advanced non-small-cell lung cancer (NSCLC), highlighting a significant overlap between these conditions. 7 The coexistence of lung cancer and ILD presents unique challenges in clinical management, requiring careful balancing of overlapping symptoms and complex treatment protocols to optimize outcomes. 8 The prevalence of lung cancer is higher among patients with pre-existing ILD compared to the general population, while lung cancer patients also exhibit an increased incidence of ILD, underscoring the bidirectional relationship between these diseases.9–11 This overlap not only complicates diagnosis and treatment but also exacerbates the risks of treatment-related complications, such as RP and CIP.
Patients with pre-existing ILD experience more severe RP and worse clinical outcomes compared to those without ILD. 12 Similarly, they are at higher risk of developing severe CIP, further complicating the use of radiotherapy and immunotherapy in this population.13,14 These challenges necessitate a deeper understanding of the risks posed by pre-existing ILD in lung cancer patients undergoing treatment. However, current research on the association between pre-existing ILD and the risks of RP and CIP remains limited, relying primarily on small-scale retrospective studies with inconsistent findings. To address this critical gap, our meta-analysis systematically evaluates the occurrence patterns and risk differences of RP and CIP in lung cancer patients with pre-existing ILD. By synthesizing available evidence, this study seeks to guide clinical decision-making and ensure safer, more effective treatment strategies.
Methods
Study selection
This systematic review was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement (Table S1) 15 and was registered in the PROSPERO database (CRD42024609557). A comprehensive literature search was performed across PubMed, Embase, and Web of Science, encompassing each database’s inception through August 10, 2024. The search strategy incorporated keywords such as “lung cancer,” “interstitial lung disease,” “immune checkpoint inhibitor,” and “radiotherapy.” Both MeSH terms and free-text variations were employed to ensure the identification of all relevant studies. In addition, the reference lists of the included studies were reviewed to maximize comprehensiveness. The detailed search strategy is provided in Table S2.
Eligibility criteria
Studies were included based on the following criteria: (1) Study design: observational studies (cohort, case–control, cross-sectional) and clinical trials. (2) Participants: adult patients with a confirmed diagnosis of lung cancer and concurrent ILD. (3) Comparison: lung cancer patients without ILD. (4) Outcomes: reported incidence or risk (expressed as odds ratios (ORs) or hazard ratios (HRs)) of developing RP and/or CIP in patients with or without ILD. If ORs or HRs were not directly reported, they were indirectly calculated from the number of RP or CIP cases reported in the respective patient groups. Exclusion criteria included non-original research articles (e.g., reviews, meta-analyses, editorials, and conference abstracts) and articles not published in English.
Data extraction and quality assessment
Data extraction was independently conducted by two researchers using a standardized extraction form. The extracted data included study characteristics: author information, year of publication, country/region, study design, and sample size; patient demographics: age, gender distribution, and ILD subtype if available; treatment regimens: detailed information on radiation therapy and ICIs; outcome measures: the number and incidence of any-grade and severe RP/CIP in lung cancer patients with and without ILD. The quality of each observational study was assessed using the Newcastle–Ottawa Scale (NOS). 16 Studies were scored on the selection of study groups, comparability, and outcome assessment, with a score above 6 indicating high quality.
Statistical analysis
Statistical analyses were conducted using STATA 12.0 (StataCorp LP, College Station, TX, USA). The I2 statistic was calculated to assess heterogeneity among studies, with I2 values of 25%, 50%, and 75% corresponding to low, moderate, and high heterogeneity, respectively. 17 Due to the expected high heterogeneity in study populations, a random-effects model was applied to pool effect sizes and rates. The metan function was used to combine ORs/HRs and their 95% confidence intervals (CIs), evaluating the association between pre-existing ILD and the risk of any-grade and severe RP and CIP in lung cancer patients. The metaprop function was employed to pool rates, examining differences in the occurrence of any-grade and severe RP and CIP among lung cancer patients with and without ILD. Sensitivity analyses were performed with the metainf function to assess the robustness of the pooled results. Funnel plots were drawn using the metafunnel function, and publication bias was assessed using Begg’s and Egger’s tests with the metabias function. A p-value of <0.05 was considered statistically significant.
Results
General characteristics of the included studies
As of August 10, 2024, a total of 3998 studies were identified as potentially relevant to the occurrence of RP in lung cancer patients with pre-existing ILD. These included 983 studies from PubMed, 1919 from Embase, and 1096 from Web of Science. After removing 805 duplicates, 3193 studies remained for title and abstract screening. Following full-text review of 240 papers, 12 studies comprising 2576 patients were included in the final meta-analysis assessing RP risk.18–29 The literature selection process is depicted in Figure 1(a). All included studies were conducted in Asia, originating from China (six studies), Japan (four studies), and South Korea (two studies). All were retrospective studies published between 2012 and 2024, with sample sizes ranging from 49 to 690. Nine studies18–20,22,24,25,27–29 included both NSCLC and small-cell lung cancer (SCLC) patients, two studies21,23 focused exclusively on NSCLC patients, and one study 26 included only SCLC patients. The predominant radiotherapy types were stereotactic body radiation therapy (SBRT) and intensity-modulated radiation therapy (IMRT) or volumetric modulated arc therapy (VMAT), with four studies addressing each type. The quality assessment, based on the NOS, showed that seven studies scored above 6, qualifying them as high quality (Table S3). The basic characteristics and populations of the included studies are summarized in Table 1.

Flowchart of study selection. (a) Literature screening process for studies investigating the relationship between pre-existing ILD and the risk of RP in lung cancer patients. (b) Literature screening process for studies examining the relationship between pre-existing ILD and the risk of CIP in lung cancer patients.
Basic characteristics of studies exploring the association between pre-existing ILD and the risk of RP in lung cancer patients.

3DCRT, 3D conformal radiotherapy; ADC, adenocarcinoma; CC, case–control study; DLCO, Diffusing capacity of the lungs for carbon monoxide; ECOG PS, Eastern Cooperative Oncology Group performance status; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; ILD, interstitial lung disease; IMRT, intensity-modulated radiation therapy; NA, not available; non-SCC, non-squamous cell carcinoma; NOS, Newcastle–Ottawa Scale; NSCLC, non-small-cell lung cancer; RO, retrospective study; RP, radiation pneumonitis; SBRT, stereotactic body radiation therapy; SCC, squamous cell carcinoma; SCLC, small-cell lung cancer; VMAT, volumetric modulated arc therapy.
Regarding CIP, after reviewing 117 full texts, 29 studies involving 8037 patients were included.13,14,30–56 The screening process is shown in Figure 1(b). Most studies originated from Asia, with Japan contributing 20 studies, China 6 studies, and South Korea 1 study. Two additional studies were conducted in the United States. These studies were published between 2018 and 2024, with study designs comprising 25 retrospective cohort studies, two case–control studies, one prospective cohort study, and one phase II clinical trial. Sample sizes ranged from 17 to 2570. Most studies exclusively enrolled NSCLC patients, while six studies31,33,38,40,44,53 included both NSCLC and SCLC patients. No study focused solely on SCLC patients. The most commonly utilized ICIs were anti-programmed cell death-1 (PD-1) monoclonal antibodies (mAb), featured in 11 studies, while only 2 studies evaluated anti-programmed cell death ligand-1 (PD-L1) mAb. Quality assessment revealed that eight studies scored above 6 points, classifying them as high quality (Table S4). Details of the included studies and their populations are summarized in Table 2.
Basic characteristics of studies exploring the association between pre-existing ILD and the risk of CIP in lung cancer patients.
ADC, adenocarcinoma; anti-PD-1, anti-programmed cell death-1; anti-PD-L1, anti-programmed cell death ligand-1; ATZ, atezolizumab; CC, case–control study; CIP, checkpoint inhibitor pneumonitis; CRT, chemoradiotherapy; DLCO, diffusing capacity of the lungs for carbon monoxide; DUR, durvalumab; ECOG PS, Eastern Cooperative Oncology Group performance status; ICI, immune checkpoint inhibitor; ILD, interstitial lung disease; IPI, ipilimumab; LCNEC, large-cell neuroendocrine carcinoma; NA, not available; NIVO, nivolumab; non-SCC, non-squamous cell carcinoma; NOS, Newcastle–Ottawa Scale; NOSP, not otherwise specified; NSCLC, non-small-cell lung cancer; PEMB, pembrolizumab; PO, prospective study; RO, retrospective study; SCC, squamous cell carcinoma; SCLC, small-cell lung cancer.
Correlation between pre-existing ILD and the risk of developing RP in lung cancer patients
To investigate the association between pre-existing ILD and the risk of developing RP in lung cancer patients, data from 11 studies were analyzed for any-grade RP risk. The heterogeneity test showed no significant heterogeneity (I2 = 40.3%, p = 0.080). Using a random-effects model, pre-existing ILD was shown to significantly increase the risk of RP (OR: 3.63, 95% CI: 2.26–5.83, Figure 2(a)). Sensitivity analysis confirmed the robustness of these findings (Figure S1(A)), and no significant publication bias was detected (Begg’s test: p = 0.119, Egger’s test: p = 0.099; Figure S1(B)).

Forest plot of meta-analysis for RP risk in lung cancer patients with pre-existing ILD. (a) Pooled analysis showing the association between pre-existing ILD and the risk of any-grade RP. (b) Pooled analysis showing the association between pre-existing ILD and the risk of severe RP.
A subgroup analysis based on the radiotherapy technique was conducted to examine its impact on RP risk. The results demonstrated that patients receiving SBRT had a lower risk of any-grade RP (OR: 2.09, 95% CI: 1.09–4.01) compared to those undergoing IMRT or VMAT (OR: 3.36, 95% CI: 2.11–5.37; Figure S2). By contrast, patients treated with 3D conformal radiotherapy (3DCRT) or other radiotherapy techniques exhibited the highest risk of any-grade RP (OR: 13.35, 95% CI: 4.89–36.42; Figure S2). A subgroup analysis based on pathological type revealed that patients with SCLC had a lower incidence of any-grade RP (OR: 3.01, 95% CI: 0.96–9.44; Figure S3) compared to patients with NSCLC (OR: 3.46, 95% CI: 0.35–34.61; Figure S3).
Four studies specifically reported OR for severe RP risk in patients with pre-existing ILD, showing no heterogeneity (I2 = 0.0%, p = 0.785). The meta-analysis indicated that pre-existing ILD significantly increased the risk of severe RP (OR: 6.10, 95% CI: 2.68–13.86; Figure 2(b)). These findings were validated through sensitivity analysis (Figure S4(A)), and no publication bias was observed (Begg’s test: p = 0.089, Egger’s test: p < 0.001; Figure S4(B)). In summary, pre-existing ILD significantly increases the risk of both any-grade and severe RP in lung cancer patients.
Prevalence of RP in lung cancer patients with/without pre-existing ILD
We investigated the differences in RP incidence between lung cancer patients with and without pre-existing ILD. Eight studies reported the incidence of any-grade RP. Using a random-effects model, the incidence of any-grade RP was found to be 32% in patients without ILD (95% CI: 15%–49%; heterogeneity I2 = 98.42%, p < 0.001; Figure 3(a)), compared to a significantly higher 50% in patients with ILD (95% CI: 42%–57%; heterogeneity I2 = 0, p = 0.51; Figure 3(b)). For severe RP, the incidence was 12% in patients without ILD (95% CI: 8%–16%; heterogeneity I2 = 3.47%, p = 0.35; Figure 3(c)) and 41% in patients with ILD (95% CI: 24%–57%; heterogeneity I2 = 0, p = 0.69; Figure 3(d)). These findings highlight that pre-existing ILD significantly increases the incidence of both any-grade and severe RP in lung cancer patients.
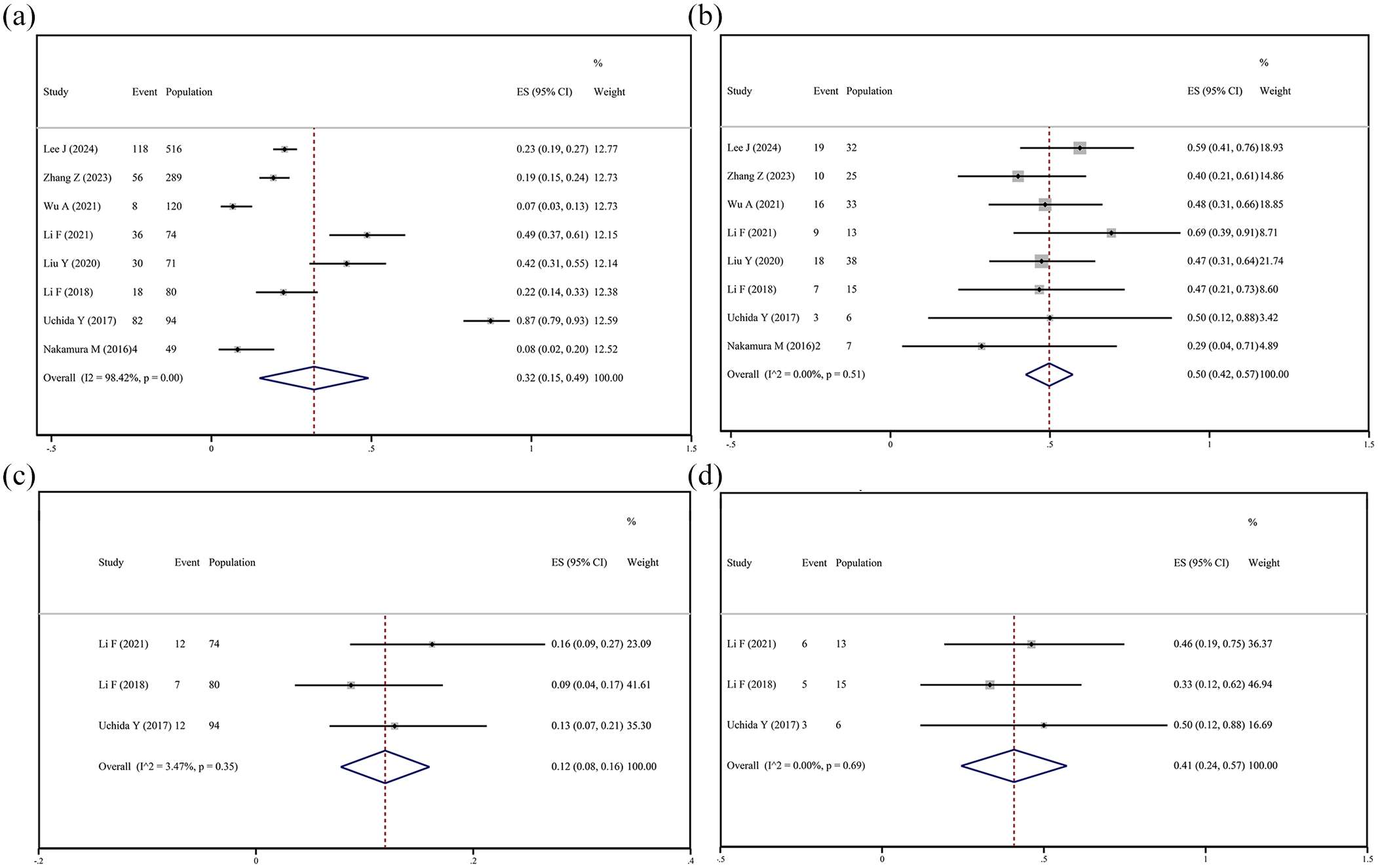
Forest plots of RP incidence in lung cancer patients with and without ILD. (a) Pooled prevalence of any-grade RP in lung cancer patients without ILD. (b) Pooled prevalence of any-grade RP in lung cancer patients with ILD. (c) Pooled prevalence of severe RP in lung cancer patients without ILD. (d) Pooled prevalence of severe RP in lung cancer patients with ILD.
Correlation between pre-existing ILD and the risk of developing CIP in lung cancer patients
We evaluated the risk of CIP in lung cancer patients with pre-existing ILD using data from 24 studies. The heterogeneity test revealed significant variability among the studies (I2 = 59.8%, p < 0.001). A meta-analysis using a random-effects model indicated that pre-existing ILD significantly increases the risk of developing any-grade CIP (OR: 3.86, 95% CI: 2.65–5.61; Figure 4(a)). Sensitivity analysis confirmed the robustness of these pooled results (Figure S5(A)), and no significant publication bias was observed (Begg’s test: p = 0.130, Egger’s test: p = 0.222; Figure S5(B)). A subgroup analysis based on the type of ICIs was conducted to evaluate their impact on any-grade CIP risk. As shown in Figure S6(A), patients treated with anti-PD-1 mAb (OR: 4.27, 95% CI: 3.04–5.99) had a higher risk of any-grade CIP compared to those receiving anti-PD-L1 mAb (OR: 3.29, 95% CI: 1.98–5.48). Subgroup analysis by pathological type indicated that patients with NSCLC had a lower incidence of any-grade CIP (OR: 3.63, 95% CI: 2.46–5.35; Figure S6(B)).

Forest plot of meta-analysis for CIP risk in lung cancer patients with pre-existing ILD. (a) Pooled analysis showing the association between pre-existing ILD and the risk of any-grade CIP. (b) Pooled analysis showing the association between pre-existing ILD and the risk of severe CIP.
In addition, 11 studies evaluated the relationship between pre-existing ILD and severe CIP risk. For these, no significant heterogeneity was identified (I2 = 4.5%, p = 0.400). A random-effects meta-analysis revealed that pre-existing ILD significantly increases the risk of severe CIP (OR: 3.24, 95% CI: 2.07–5.07; Figure 4(b)). Sensitivity analysis further confirmed the stability of these findings (Figure S7(A)), and publication bias tests showed no significant bias (Begg’s test: p = 0.640, Egger’s test: p = 0.782; Figure S7(B)). Subgroup analysis of ICI types for severe CIP indicated that patients receiving anti-PD-1 mAb faced a higher risk (OR: 3.91, 95% CI: 2.17–7.08) compared to those treated with anti-PD-1/anti-PD-L1 mAb therapies (OR: 2.78, 95% CI: 1.21–6.39; Figure S8(A)). Similarly, subgroup analysis by pathological type showed that patients with NSCLC had a lower incidence of severe CIP (OR: 3.10, 95% CI: 1.85–5.19; Figure S8(B)). In summary, pre-existing ILD significantly increases the risk of both any-grade and severe CIP in lung cancer patients.
Prevalence of CIP in lung cancer patients with/without pre-existing ILD
We compared the incidence of CIP in lung cancer patients with and without pre-existing ILD. The results showed that the incidence of any-grade CIP was 11% in patients without ILD (95% CI: 9%–13%; heterogeneity I2 = 86.39%, p < 0.001; Figure 5(a)), whereas it was significantly higher at 29% in those with ILD (95% CI: 23%–34%; heterogeneity I2 = 78.31%, p < 0.001; Figure 5(b)). For severe CIP, the incidence was 5% in patients without ILD (95% CI: 3%–6%; heterogeneity I2 = 61.38%, p = 0.01; Figure 5(c)), compared to 12% in patients with ILD (95% CI: 8%–15%; heterogeneity I2 = 0, p = 0.630; Figure 5(d)). These findings clearly demonstrate that pre-existing ILD significantly increases the risk of both any-grade and severe CIP in lung cancer patients.
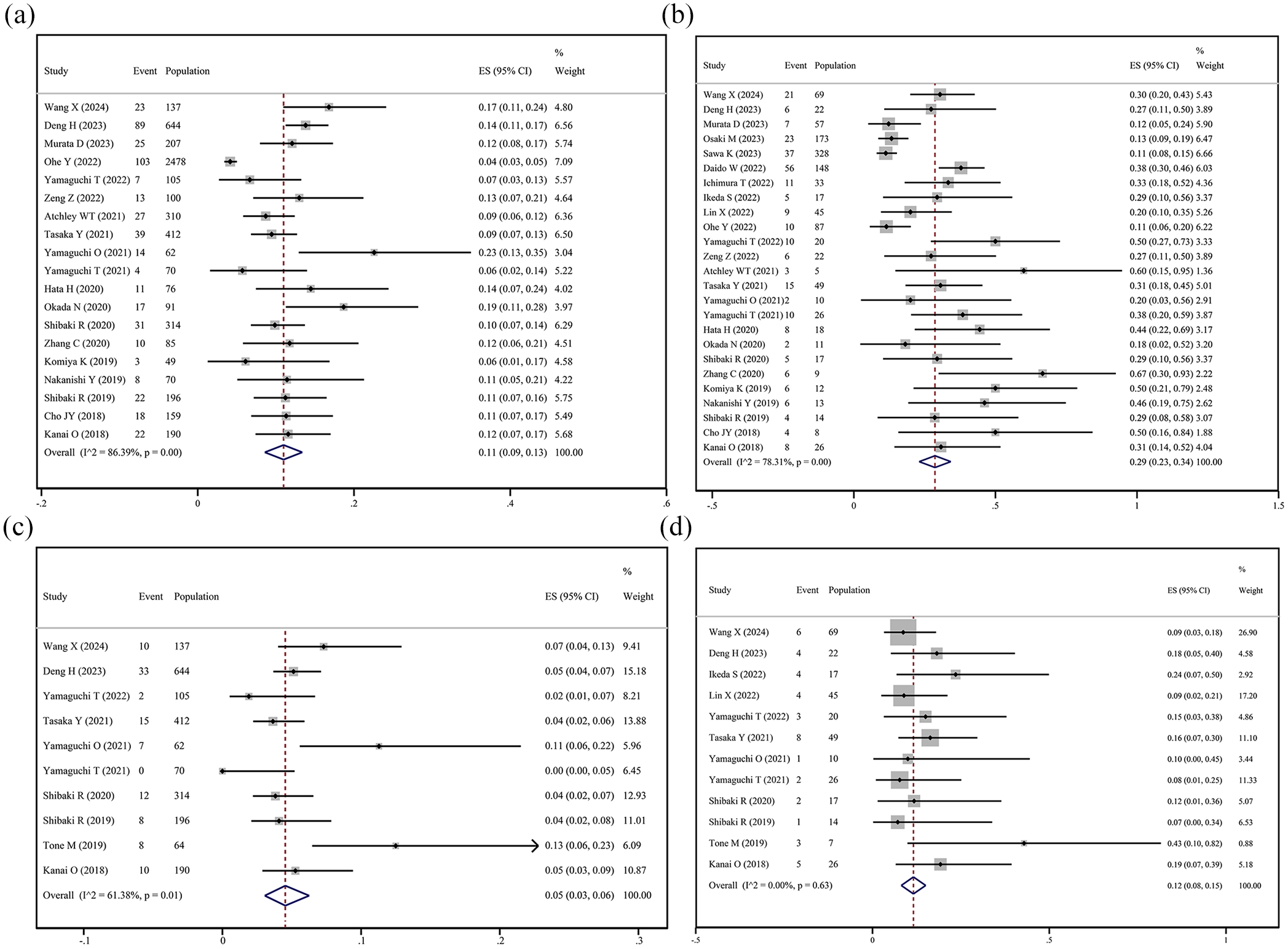
Forest plots of CIP incidence in lung cancer patients with and without ILD. (a) Pooled prevalence of any-grade CIP in lung cancer patients without ILD. (b) Pooled prevalence of any-grade CIP in lung cancer patients with ILD. (c) Pooled prevalence of severe CIP in lung cancer patients without ILD. (d) Pooled prevalence of severe CIP in lung cancer patients with ILD.
Discussion
Previous studies have reported mixed findings regarding whether pre-existing ILD is a risk factor for RP and CIP in lung cancer patients. Our meta-analysis addresses this uncertainty by demonstrating a significant association between pre-existing ILD and an increased risk of both RP and CIP. To our knowledge, this is the first large-scale meta-analysis to systematically explore this relationship in lung cancer patients. Our findings reveal that individuals with pre-existing ILD have a markedly higher susceptibility to RP, with a 3.63-fold increased risk for any-grade RP and an even greater 6.10-fold increase for severe RP.
A recent review by Bensenane et al. 57 summarized the risk factors for RP in lung cancer patients, including patient and tumor characteristics, pretreatment pulmonary function, and dosimetric factors. However, only one study in their review specifically addressed the association between pre-existing ILD and RP risk. In our subgroup analysis, we found that SBRT was associated with the lowest incidence of any-grade RP in ILD patients, followed by IMRT or VMAT, while the highest risk was observed in patients treated with 3DCRT. Consistent with our findings, Bensenane et al. 57 reported that SBRT and IMRT significantly reduced radiation-induced morbidity and lung function impairments in lung cancer patients, although their analysis primarily focused on patients without ILD. This suggests that SBRT and IMRT/VMAT are preferable options for patients with pre-existing ILD when technically feasible, although further studies are needed to confirm the safest treatment protocols.
Regarding CIP, our analysis shows that lung cancer patients with pre-existing ILD are at a 3.86-fold higher risk for any-grade CIP and a 3.24-fold higher risk for severe CIP. These results are consistent with Zhang et al., 58 who reported a higher incidence of CIP in NSCLC patients with ILD compared to those without, based on a meta-analysis of 179 patients across 10 studies. Our subgroup analysis further indicates that anti-PD-1 therapies are associated with a higher risk of both any-grade and severe CIP compared to anti-PD-L1 therapies. Similarly, Khunger et al. 59 found that PD-1 inhibitors were correlated to a greater incidence of CIP than PD-L1 inhibitors in NSCLC patients. However, even anti-PD-L1 therapies, which appear to carry a relatively lower risk, may not be ideal for patients with pre-existing ILD. For instance, the AMBITIOUS phase II trial, which evaluated atezolizumab in NSCLC patients with ILD, was terminated early due to 23.5% of participants developing grade 3 or higher CIP. 60
The increased risk of RP and CIP in patients with pre-existing ILD is likely due to complex pathophysiological interactions. 61 ILD is characterized by chronic inflammation and fibrosis, which render lung tissue more vulnerable to damage from radiation and ICIs. 62 Fibrotic changes in ILD alter vascular architecture and immune responses, potentially amplifying the adverse effects of these treatments. 63 In addition, the reduced reparative capacity of fibrotic lungs may prolong recovery and exacerbate injury after treatment. Radiotherapy induces DNA damage in normal lung tissue, prompting alveolar capillary epithelial cells to release cytokines that activate immune cells and myofibroblasts. 64 This process recruits additional T cells and enhances local T-lymphocyte infiltration, exacerbating inflammation in fibrotic lung tissue. 64 Similarly, ICIs can intensify inflammatory responses in fibrotic lungs through T-cell recruitment and cytokine release, further aggravating CIP.65–67
From a clinical perspective, these findings underscore the importance of evaluating ILD status before initiating lung cancer treatment. Identifying ILD as a key risk factor allows healthcare providers to tailor therapeutic strategies accordingly. This may include advanced radiation techniques, such as proton therapy, to minimize exposure to healthy lung tissue, or selecting systemic therapies with a lower risk of pulmonary toxicity.68,69 A multidisciplinary approach involving pulmonologists and oncologists is essential to develop personalized treatment plans that optimize safety and efficacy. Proactive monitoring, including regular pulmonary function tests and imaging, is crucial for the early detection and management of pneumonitis. Preventive strategies, such as prophylactic corticosteroids, may also help reduce the incidence and severity of RP. 70 Furthermore, educating patients to recognize early symptoms of pneumonitis and encouraging open communication with healthcare teams can enable timely interventions, potentially improving outcomes and quality of life.
While our study is pioneering in exploring the association between pre-existing ILD and the risks of RP and CIP in lung cancer patients through a large-scale meta-analysis, several limitations should be acknowledged. Many of the included studies were retrospective in design, which may introduce selection and confounding biases. In addition, some effect estimates were derived from univariate analyses without adjustment for potential confounders, potentially resulting in biased pooled outcomes. Key clinical aspects, such as late-onset RP and ICI-induced acute exacerbations of ILD, were not adequately addressed in the included studies despite their potential impact on patient outcomes. Moreover, the lack of sufficient follow-up data for ILD patients treated with ICIs limits our understanding of disease progression and treatment responses. The absence of routine bronchoscopy or lung biopsies for many patients further restricts diagnostic precision and the ability to fully evaluate the underlying causes of respiratory complications. Another limitation lies in the subgroup analyses of pathological subtypes. Few studies exclusively focused on NSCLC or SCLC within certain subgroups, potentially leading to instability in the results. Furthermore, the absence of detailed information on ILD patterns and severity, radiotherapy protocols, specific ICI regimens, and treatment lines constrained our ability to perform a comprehensive assessment of RP and CIP risks across diverse patient subgroups. These limitations underscore the need for high-quality, well-designed prospective studies with detailed clinical data to enhance our understanding of these critical associations and improve patient outcomes in the future.
Conclusion
In conclusion, our study establishes pre-existing ILD as a significant risk factor for increased incidence and severity of RP and CIP in lung cancer patients. These findings underscore the importance of routine ILD screening and personalized treatment strategies to mitigate the risks associated with radiotherapy and immunotherapy in this high-risk population. Future research should focus on validating these results in larger, more diverse cohorts and developing targeted interventions to reduce adverse outcomes. Addressing these aspects will enhance therapeutic outcomes and improve the quality of life for patients with pre-existing ILD undergoing lung cancer treatment.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251338624 – Supplemental material for The impact of pre-existing interstitial lung disease on radiation and checkpoint inhibitor pneumonitis in lung cancer patients: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251338624 for The impact of pre-existing interstitial lung disease on radiation and checkpoint inhibitor pneumonitis in lung cancer patients: a systematic review and meta-analysis by Aimin Jiang, Haozheng Lu, Rui Zhao, Jupeng Yuan and Dawei Chen in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
