Abstract
Salivary gland tumors are rare and very heterogeneous types of head and neck cancer. Among them, salivary duct carcinoma (SDC) is one of the most aggressive, with poor prognosis and few recommendations for treatment. Numerous targetable mutations have been described in SDC, especially for Human Epidermal growth factor receptor 2 (HER2), up to 56% according to the literature. This target is interesting due to the development of several forms of anti-HER2 therapy in other cancers. Finally, the antibody-drug conjugate trastuzumab-deruxtecan (T-Dxd) significantly improves progression-free survival and objective response rate, with tolerable side effects. Few data have been published concerning anti-HER2 therapies for SDC, mainly concerning T-Dxd, most of the studies described treatment with docetaxel or trastuzumab alone. As patients with SDC are often young and without comorbidities, they receive a few different treatments. That is why case reports about patients with SDC treated by T-Dxd can facilitate molecular testing of these tumors and improve our knowledge about this rare disease. This report describes a 44-year-old man with HER2-positive locally advanced SDC who experienced disease progression just after surgery. Molecular analysis showed positive androgen receptors, MLH1 mutation, and HER2 positivity. He had received several different treatments, including endocrine therapy, chemotherapy, immunotherapy, and anti-HER2 therapy. The longest duration of treatment was observed with T-Dxd: 13 months, allowing local treatment of localized progression (cervical lymph nodes, bone metastasis) at the same time. This report and other published data highlight the need for better implementation of recommendations for molecular testing regarding a rare and aggressive salivary tumor. In addition, these data should increase the development of clinical research in this small patient population.
Molecular characteristics of salivary duct carcinoma
Salivary duct carcinoma (SDC) is a rare tumor of the parotid gland accounting for approximately 5%–10% of all salivary gland malignancies. 1 It is known as the most aggressive histology among many different salivary tumor subtypes. SDCs are frequently compared to breast ductal carcinoma, and they share specific immunohistological features. Among them, an androgen receptor (AR) is expressed in more than 90% of patients, allowing endocrine therapy as first-line treatment. 2 The recent literature suggests that 7%–56% of SDCs have shown a positive immunohistochemical (IHC) staining for the human epidermal growth factor receptor 2 (HER2) encoded by the ERBB2 gene.3–5 HER2 is a member of the epidermal growth factor receptor family and is overexpressed in a broad range of malignancies. Since the breakthrough of trastuzumab (tz), a monoclonal antibody directed to HER2, in the early 90s, anti-HER2 therapies have been approved, particularly HER2-positive breast and gastric cancer.6,7 Despite the scarce occurrence of SDC, several trials have focused on HER-positive SDC with interesting results in terms of response rate and progression-free survival (PFS).8–10 More recently, third-generation anti-HER2 agent trastuzumab-deruxtecan (T-Dxd) has shown impressive results in breast and gastric cancer.11–14
There are very few published studies about SDC patients treated with T-Dxd. In Destiny-PanTumor02, a total of 24 patients with head and neck cancer were included, 19 of whom had salivary gland cancer. 15 An objective response rate of 41.7% was observed, with 12.5% experiencing drug-related interstitial lung disease or pneumonitis.
Another phase I study including eight SDCs with HER2 overexpression, showed tumor shrinkage but with very few details, and some case reports have described stable tumor or response for patients already treated with anti-HER2 therapy (trastuzumab monotherapy, trastuzumab emtansine, or tucatinib).16–19 However, more real-life data are required to build a clinical trial in the hope of obtaining a major response, as has been observed with this molecule in other types of cancer.
Case presentation
The reporting of this study conforms to the CARE guidelines (Supplemental Material). 20
The patient was a 42-year-old Caucasian man, without comorbidities, who had been consulted for unexplained sudden right facial paralysis, improved on steroids. A few days later, computed tomography (CT) of the face found intraparotid calcified nonspecific lesions. One month after the first episode of facial paralysis due to clinical re-aggravation, the patient underwent a right parotidectomy with ipsilateral cervical node dissection.
Pathological analysis revealed ductal adenocarcinoma, 10/29 tumoral nodes including one in extranodal extension (T2N3bMx according to AJCC January 2018). Assessment of tumor extension after surgery with CT scan and positron emission tomography (PET) imaging revealed right cervical nodes and at least two liver metastases, no brain metastasis. Complementary tumor analysis reported a tumor overexpressing AR (85% of positive cells) and HER2, with strong circumferential membrane staining 10% of the tumor cells by IHC, classified 1 + and was scored as amplified by dual-color dual-hapten in situ hybridization (ratio of 282/66 = 4.27). Furthermore, the tumor was proficient MissMatch Repair, a finding confirmed by molecular biology (microsatellite stability profile).
Due to the positive AR and the absence of symptoms, first-line (L1) treatment was continuous bicalutamide associated with monthly triptorelin. Assessment after 2 months of treatment with cervical magnetic resonance imaging (MRI) and PET-imaging showed local and metastatic progression: cervical nodes, subcutaneous lesion, liver metastasis, and the emergence of secondary bone locations on the sternum, sacrum, left costal arch, the right humerus, and body of the C7 vertebra. We then started systemic treatment with several combinations of therapeutics allowing a very limited partial response and/or stable disease and duration of treatment (DT) from 2 to at most 6 months (Figure 1). As second-line treatment, the patient received tz–carboplatin–paclitaxel with DT of 4 months. It is worth noting that material was sent for screening of a large panel of genes (Foundation One; Roche; Cambridge, MA, USA) analysis during L2 treatment. Results confirmed HER2 positivity and showed mutation in GRM3 (p.(R352Q)-subclonal), TP53 (p.(G245S)), and MLH1 (p.(R325Q)). Because of this mutation in the MLH1 gene, we tried to include the patient in clinical trials with immunotherapy, but due to the COVID-19 pandemic, this was not possible. As third-line treatment (L3), Nivolumab 240 mg/2 weeks was prescribed. Brain imaging was performed during treatment to complete the assessment, despite the absence of neurological symptoms. It revealed a single vascularized lesion, suggestive of either a schwannoma or a metastasis. Unfortunately, immediately after the first assessment the tumor progressed for 3 months, with clinical deterioration, and we continued with standard chemotherapy in combination with anti-HER2 therapy.
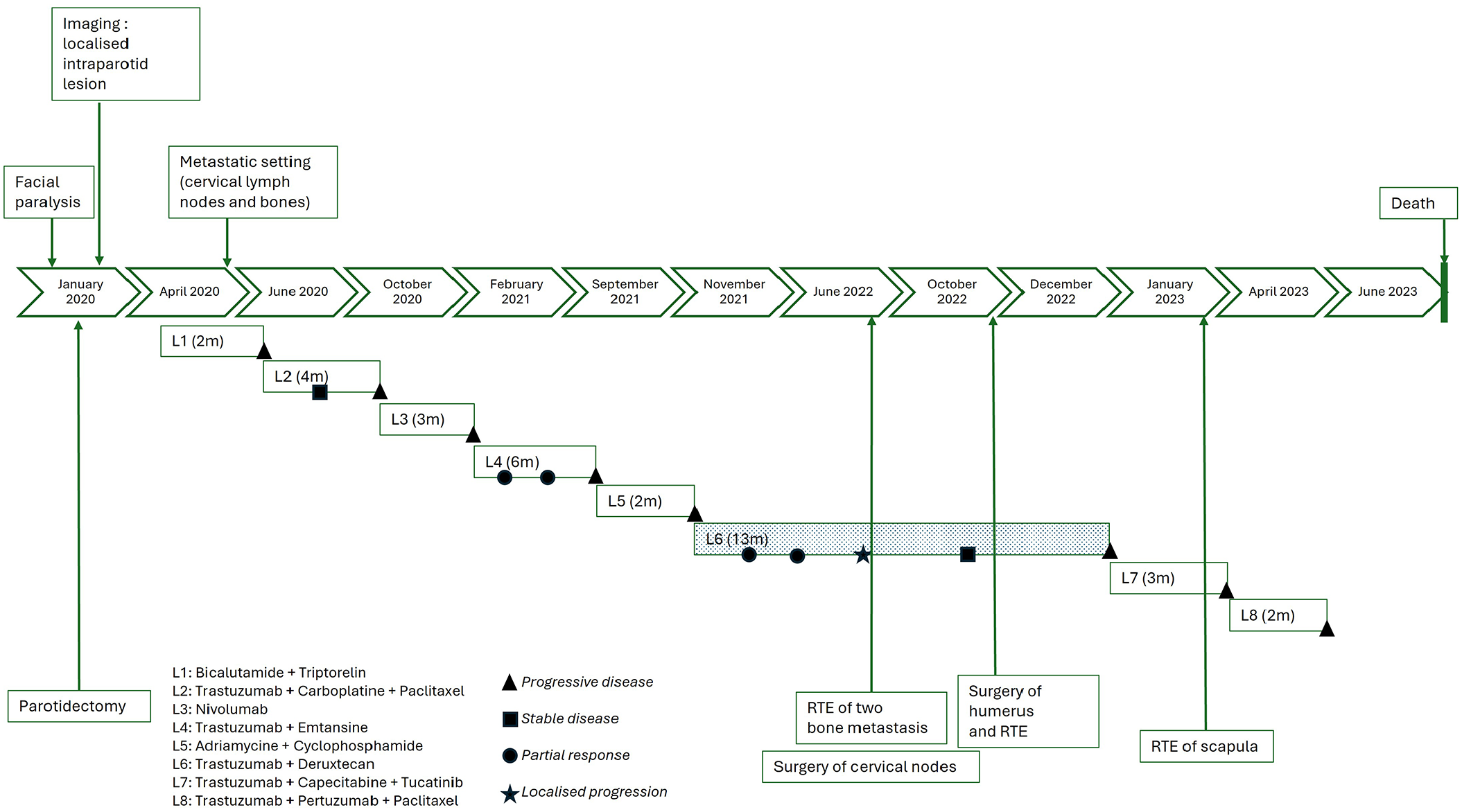
Treatment timeline.
Fourth-line treatment consisted of tz–emtansine, an antibody-drug conjugate (DT of 6 months), followed by adriamycin–cyclophosphamide. After 2 months of treatment with anthracycline and cyclophosphamide (L5), PET-imaging showed right supraclavicular adenopathy increased in size, and a pulmonary nodule and morpho-metabolic increase in bone lesions, mainly in the right humerus and left scapula.
At 22 months from diagnosis of metastatic disease and after all the recommended therapies had been applied, the disease progressed again. As the patient was Eastern Cooperative Oncology Group (ECOG) Performance Status (PS) 1 in good condition, we decided after approval by our institution and the national multidisciplinary tumor board meeting (MTBM) to prescribe T-Dxd as the sixth-line regimen (L6; Figure 2). The patient was informed that there were no validated guidelines about this use.
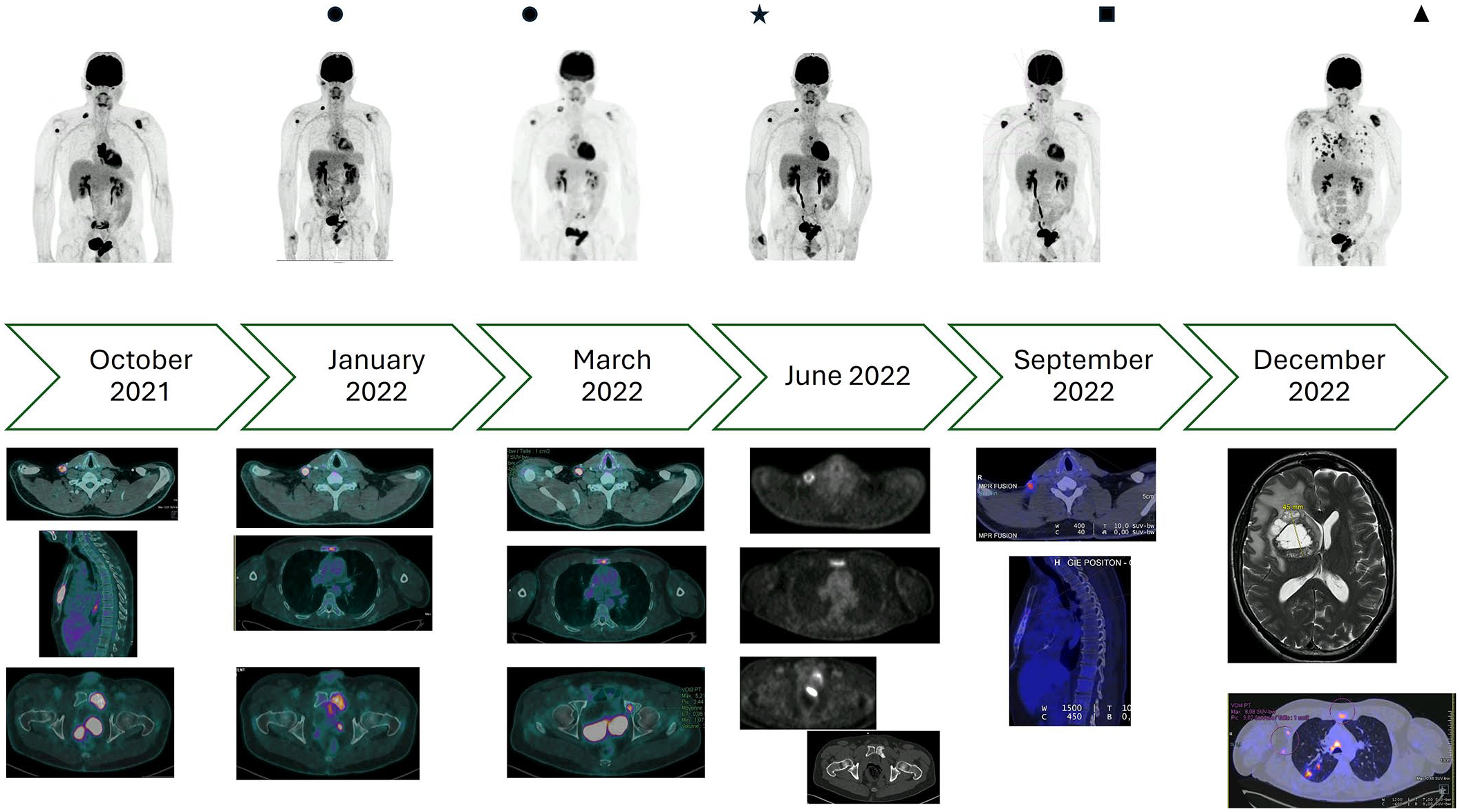
Focus on trastuzumab-deruxtecan period.
The first injection of T-Dxd was performed on November 4, 2021, with very good clinical and biological tolerance.
After four injections, PET-imaging highlighted partial response with partial regression of the primary right parotid lesion hypermetabolism and of known secondary bone lesions as well as pulmonary nodules. The right supraclavicular adenopathy remained stable. No new onset of hypermetabolic disease occurred.
After seven injections, the next evaluation found an almost complete regression of hypermetabolism of the right parotid space and overall stability of the remaining lymph node and bone lesions (Figures 1 and 2).
Unfortunately, in June 2022, after 11 injections of T-Dxd (duration of response of 5 months), PET-imaging confirmed progression of the right supraclavicular adenopathy, right jugulocarotid lymph nodes, and in the bone compartment on the sternum and sacrum. No other lesion increased, and no new onset occurred, the PFS was 8 months. We decided, in MTBM, due to the absence of threatening/symptomatic lesions and good tolerability to continue with T-Dxd and to carry out local treatments on isolated progressive lesions: the right supraclavicular adenopathy was surgically resected, and bone metastasis of the sternum and the sacrum were irradiated (30 Gy in 10 fractions each), while maintaining T-Dxd. Pathological analysis of resection consisted of seven lymph nodes, including five with extranodal extension, the HER2 IHC scored 1 + and fluorescence in situ hybridization amplified (Figure 3(a)).

Pathology evaluation. (a) Surgical resection of supraclavicular adenopathy, HER2 FISH showed an average HER2 gene copy number of 14 and a HER2/CEN17 ratio of 7 in 100 analyzed nuclei, consistent with HER2 positivity. (b) Immunohistochemistry on humerus biopsy revealed poorly differentiated carcinoma with morphologic features and IHC consistent with salivary duct carcinoma. HER2 IHC stain was scored 1+ with membranous staining.
In September 2022, PET-imaging showed a complete response on bone irradiated areas and avid inflammation in the right supraclavicular. The patient remained ECOG-PS 1, with good biological parameters, without cytopenia, with only grade 1 cytolysis caused by treatment, so we continued treatment with T-Dxd. Incapacitating pain in his right shoulder appeared during October, and imaging confirmed a fracture of the humerus on pathological bone (bone metastasis already visible on PET-imaging of September but without symptoms). The patient underwent intramedullary nailing in the humeral lesion followed by irradiation on the operating bed (30 Gy in 10 fractions). Analysis of this bone metastasis confirmed the cell type that was already known and revealed HER2 IHC score 1+ (Figure 3(b)).
Treatment with T-Dxd continued, without adverse events, notably no lung disease. Unfortunately, in January 2023, after 13 months of treatment, we found in imaging a major progression in known metastatic and new sites, mainly brain and lungs. Brain MRI confirmed at least two cerebral lesions, the largest measuring 41 mm and creating a mass effect on the ventricles, with deviation of the median line that made irradiation impossible (Figure 2).
The patient had psychomotor slowing, with little confusion with ECOG-PS 2, we tried two other lines of treatment at his request (consisting of tz–capecitabine–tucatinib (DT of 3 months), and then tz–pertuzumab–paclitaxel (DT of 2 months)), without success and the patient died in June 2023, 6 months after discontinuation of T-Dxd, with an overall survival of 40 months (Figure 1).
Discussion
This case report reveals a DT of 13 months with T-Dxd for this 44-year-old man with SDC HER2 positivity. Other treatments (endocrine therapy, chemotherapy, and immunotherapy) with or without anti-HER2 therapy (tz monotherapy, tz–emtansine, and lapatinib), allowed only from 2 to 6 months of DT. Our case report shows that local treatments (surgery or irradiation) are possible under T-Dxd, with preservation of a good quality of life.
It is interesting to compare cases in the literature (Table 1). Our patient was younger than the patients described in the published literature and had received anti-HER2 therapy before T-Dxd: tz monotherapy and tz with emtansine. It was similar in the other case reports, where patients were administered several anti-HER2 regimens; one patient received tucatinib.16–18 Contrary to the Boey et al. 17 case report where brain metastasis shrunk with T-Dxd, brain metastasis seemed in our patient to increase and become symptomatic during T-Dxd treatment.
Patient and treatment characteristics in the literature for salivary duct carcinoma treated with trastuzumab-deruxtecan.

DT, duration of treatment; T-Dxd, trastuzumab-deruxtecan; tz, trastuzumab; tz mono, tz monotherapy.
This case report shows a long DT with 13 months, it would be very interesting to obtain an update from other case reports to compare evolution at the end of T-Dxd and after: Which treatment for which tumoral evolution?
In the published literature, we observed a few long responses with different anti-HER2 therapies, in Shukla et al. 18 10 months with tz–emtansine, and in Gazola et al. 16 12 months with tz–tucatinib–capecitabine. In the literature, there are very few data concerning T-Dxd in this kind of rare tumor.21,22 A pooled analysis from Takahashi et al. 19 consisted of 17 patients with salivary gland cancer with 9 cases of salivary duct cancer and 2 cases of adenocarcinoma treated with T-Dxd. The median duration of response was 17.6 months with an objective response rate of 58.8%. The MYTHOS trial (clinical trial identification: jRCT2011210017) study resulted in a poster and an oral presentation at ESMO 2024, involving 19 patients with PFS of 15.9 months (95% confidence interval (CI), 5.8–NE). 23 In the study by Haigh et al., 21 the authors summarized the prescription of anti-HER2 treatment in salivary gland cancers and showed that tz–emtansine was the main treatment used. In this retrospective cohort, only one patient received T-Dxd, with a disappointing result of 0.7 months, this patient had also a poor response with tz–emtansine: 1.5 months, highlighting a poor sensitivity to anti-HER therapies. In the study by Meric-Bernstam et al., 15 patients with HER2-expressing head and neck cancer were treated with T-Dxd. The objective response rate was 57.1% in the HER2 IHC 3+ group, compared to 16.7% and 25.0% in the HER2 IHC 2+ and 0 groups, respectively. PFS and duration of response were also superior in the IHC 3+ group versus IHC 2+ (23.4 vs 7.1 months and 22.1 vs 2.8 months, respectively).
The analysis of HER2 status at different stages of the patient’s disease history showed that positivity was maintained despite the selective pressure exerted by the multiple anti-HER2 treatments prescribed. This parameter is interesting because, in breast cancer, a change in HER2 expression is often observed following anti-HER2 treatment. For example, after neoadjuvant anti-HER2 therapy, a loss of positivity was observed in 21.4% of cases in a series of over 21,000 patients who initially had HER2-overexpressing breast cancer. 24
Some early-phase studies are ongoing with T-Dxd, mainly early stage and for advanced solid tumors. Few studies are dedicated to salivary gland cancer (Table 2). Some of these studies will test T-Dxd monotherapy, while others will try a combination with anti-ataxia telangiectasia and Rad3-related protein, anti-HER3, or bispecific antibody.
Ongoing clinical trials testing deruxtecan in advanced/metastatic settings.

ADC, antibody-drug conjugate; ATR, ataxia telangiectasia and Rad3-related protein; HER2, human epidermal growth factor receptor 2; T-Dxd, trastuzumab-deruxtecan.
Even if there is still a lack of data, it seems essential to perform HER2 analysis in cases of SDC as recommended in ESMO clinical practice guidelines with ESMO Scale for Clinical Actionability of Molecular Targets score II-B and in National Comprehensive Cancer Network guidelines version 2.2025.3,4 These simple, inexpensive tests are now routinely performed on a massive scale for breast and gastric cancers, thanks to effective, validated targeted therapies.6,7
In the cases reported in the literature, many patients achieve a response time of around 1 year with one anti-HER2 therapy or another, with acceptable toxicity, which is no mean feat for these multitreated patients.16–18,23 This strategy is even more important as mean age at diagnosis is around 65 years old, with no significant smoking or alcohol history, and therefore in good general condition.16–18,23 Thanks to the Food and Drug Administration’s accelerated approval of T-Dxd for unresectable or metastatic HER2-positive solid tumors, we will be able to treat our SDC patients and enhance both their quality of life and survival.
Conclusion
To conclude, further studies are needed to better define the histo-molecular profile, which is most likely to support HER2 positivity and obtain a good response to anti-HER2 therapy. In the whole panoply of anti-HER2 agents available, T-Dxd seems to have a place, given the good responses observed after other anti-HER2 treatments. Perhaps we need to keep the pressure on this HER2 receptor throughout the history of the disease, as is the practice in HER2-amplified breast and gastric cancers.11,14
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251334071 – Supplemental material for Trastuzumab-deruxtecan shows interesting disease control in a multitreated patient with ERBB2 amplified parotid carcinoma, a case report
Supplemental material, sj-pdf-1-tam-10.1177_17588359251334071 for Trastuzumab-deruxtecan shows interesting disease control in a multitreated patient with ERBB2 amplified parotid carcinoma, a case report by Camille Evrard, Jean-Michel Goujon, Claire Villalva and Matthieu Bainaud in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
