Abstract
Colorectal cancer is a leading cause of cancer-related mortality worldwide. Approximately 3–5% of colorectal tumors harbor human epidermal growth factor receptor (HER2) amplification which is associated with a higher incidence of intracranial metastasis and overall worse outcomes. In the setting of refractory metastatic RAS wild-type tumors, evidence supports the use of various HER2-blocking agents, including monoclonal antibodies, tyrosine kinase inhibitors, and novel antibody–drug conjugates. With a number of relatively active agents clinically available and even more in development, it is crucial for clinicians to familiarize themselves with the mechanisms of action, efficacy data, and safety profiles of these treatments. In this review, we aim to summarize key findings from past and ongoing trials with anti-HER2 agents in metastatic colorectal cancer.
Background
Colorectal cancer (CRC) is the third most common malignancy diagnosed in men and the second in women worldwide. Despite advances, CRC remains one of the leading global causes of mortality accounting for 9% of all cancer-related death. 1 Exploration of the genomic landscape of this disease allowed therapeutic expansion to include targeted therapies and immunotherapy. Approximately 3–5% of CRC harbor amplification of human epidermal growth factor receptor 2 (HER2) or overexpression of HER2 protein. Accumulating evidence supports the therapeutic benefit of HER2 blockade therapies in patients with metastatic CRC overexpressing HER2, resulting in a recent accelerated approval by the Food and Drug Administration for tucatinib in combination with trastuzumab for this patient population. Despite the inclusion of HER2-targeted therapy in the guidelines, a real-world study on treatment patterns showed its underutilization in patients with metastatic CRC. 2 In this comprehensive review, our objective is to summarize pivotal studies on the efficacy and safety of anti-HER2 agents in the context of CRC.
Molecular and clinical features of HER2-amplified CRC
HER2, also known as ERBB2, is a member of the receptor tyrosine kinase family, along with epidermal growth factor receptor (EGFR), HER3, and HER4. These receptors possess an extracellular ligand-binding domain, a transmembrane domain, and an intracellular tyrosine kinase domain. Unlike other family members, HER2 does not have identified ligands, making it the preferred receptor for heterodimerization with other HER family members. This heterodimerization leads to the autophosphorylation of tyrosine kinase residues, triggering pivotal signaling involved in cell survival and proliferation, including RAS-RAF-ERK and PI3K-PTEN-AKT pathways.3,4 In various types of tumors, such as breast, gastroesophageal, and CRCs, HER2 gene amplification typically is associated with an overexpression of HER2 protein, resulting in its constitutional activation and consequent carcinogenesis.5–8
In CRC, genomic studies on tumor laterality reveal that gene alterations are distributed along a continuum throughout the colorectal tract. 9 In particular, HER2 amplification is more frequently observed in left-sided tumors located distal to the splenic flexure.10,11 In addition, HER2 amplifications in CRC are primarily associated with KRAS/BRAF wild-type tumors (incidence ranging from 2.1% to 5.2%) versus KRAS/BRAF-mutant tumors (incidence ranging from 0.2% to 2.1%). 12 Clinically, HER2-amplified CRCs show distinctive features and outcomes. These tumors are associated with a higher incidence of metastatic disease in the central nervous system. In addition, patients with HER2-amplified CRC tumors appear to exhibit worse outcomes. In the early stages of disease following resection, HER2 amplification is associated with a shorter disease-free survival.13–15 In the metastatic setting, RAS/RAF wild-type HER2-overexpressed CRC is associated with lower overall survival (OS) and poor response to anti-EGFR therapy.7,16
Diagnostic criteria for HER2-overexpression in CRC
The terminology ‘HER2 positivity’ or ‘HER2-expressing tumor’ has been commonly utilized in both past and present studies. However, these terms lack specificity and may lead to misinterpretation. It is important to differentiate between HER2 mutations (a therapeutic target in non-small-cell lung cancer) 17 and HER2 amplifications (which are targeted in breast, gastric, and CRCs).5,18–20 In CRC, the benefits of anti-HER2 therapy are better documented in tumors with HER2 genomic amplification and/or HER2 protein overexpression. As such, clinicians should familiarize themselves with its definition criteria across the different diagnostic platforms.
At the histologic level, HER2 protein overexpression is assessed using immunohistochemistry (IHC) performed on the tumor tissue. Based on the staining intensity, HER2 expression will be categorized as positive (score 3+), negative (score 0 or 1+), or equivocal (2+). In cases where the IHC score is 2+, a reflex fluorescence in situ hybridization (FISH) is performed. A HER2/CEN17 ratio of 2 or higher in the FISH result is considered positive in this setting. 21 Although classically considered HER2 negative, tumors that are IHC 1+ or IHC 2+/FISH negative are being re-categorized as ‘HER2-low’. It is due to the clinical benefit associated with trastuzumab deruxtecan (T-DXd) in breast cancers, 5 which has yet to be demonstrated in CRC tumors.
At the genomic level, tissue- and, to some extent, blood-based next-generation sequencing (NGS) will allow for simultaneous detection of HER2 amplifications and relevant mutations, along with other actionable genetic variants. HER2 status assessment from the MOUNTAINEER trial suggested high inter-test reliability between centrally confirmed different testing methods. Specifically, there was a 92.6% concordance between IHC/FISH and tissue-based NGS with an 81% concordance between blood- and tissue-based NGS, and a 79.5% concordance between IHC/FISH and blood-based NGS testing. 22 There are multiple factors that can explain some of the result discrepancies, including the quality of the tumor sample, varying levels of circulating tumor DNA (ctDNA) shedding in the blood, and the specific characteristics of the NGS panel used for analysis.23,24 Moreover, HER2 expression within the same tumor may vary across different time points owing to intratumor heterogeneity and exposure to prior anti-EGFR and anti-HER2 therapies. 25 As such, careful consideration is advised to ensure reliable determination of HER2 status in patients.
Current evidence on anti-HER2 therapy in CRC
Multiple mechanistic strategies are available for inhibiting HER2 pathways in CRC. Monoclonal antibodies (mAb) directly block HER2-mediated signaling by binding to its extracellular domain (trastuzumab) or by inhibiting its dimerization (pertuzumab). 20 In addition, zanidatamab, an engineered immunoglobulin G1 bispecific mAb, targets two distinct extracellular domains similar to the combined effect of trastuzumab and pertuzumab, leading to dual HER2 blockade with receptor internalization and downregulation. Moreover, zanidatamab triggers an antibody-dependent T-cell-mediated cytotoxic effect on HER2-overexpressing cells. 26 Orally available tyrosine kinase inhibitors (TKI) such as tucatinib, neratinib, and lapatinib block the phosphorylation of the intracellular tyrosine kinase domain, thereby reducing pathway signaling. Trastuzumab emtansine (T-DM1) and T-DXd are antibody–drug conjugates (ADCs) that combine a cytotoxic payload (e.g. a topoisomerase inhibitor in T-DXd) covalently linked to the trastuzumab-like antibody. The trastuzumab-like moiety facilitates endocytosis through the HER2 receptor, leading to intracellular linker cleavage and the release of the drug payload. This results in direct cytotoxic effects on cancer cells and indirect effects on neighboring cells through a bystander effect.8,27,28
Preclinical studies with tumor grafts or xenografts observed limited activity of HER2-targeted monotherapy while suggesting a synergistic effect of dual anti-HER2 blockade.29,30 These findings led to several phase II studies exploring the clinical benefit of various anti-HER2 regimens in metastatic CRC. When selecting and sequencing treatment options for these patients, clinicians should consider individual patient characteristics, the genomic profile of tumors, and prior HER2 therapy exposure. Notably, RAS mutations are known predictors of resistance to anti-EGFR therapies. 31 Early trials have demonstrated a lack of activity with direct HER2-inhibiting mAb in RAS-mutant CRCs.20,22,32 Table 1 summarizes the key findings of clinical trials with various anti-HER2 agents in patients with refractory metastatic CRC.
Summary of key clinical trial findings with anti-HER2 agents in metastatic CRC treatment.
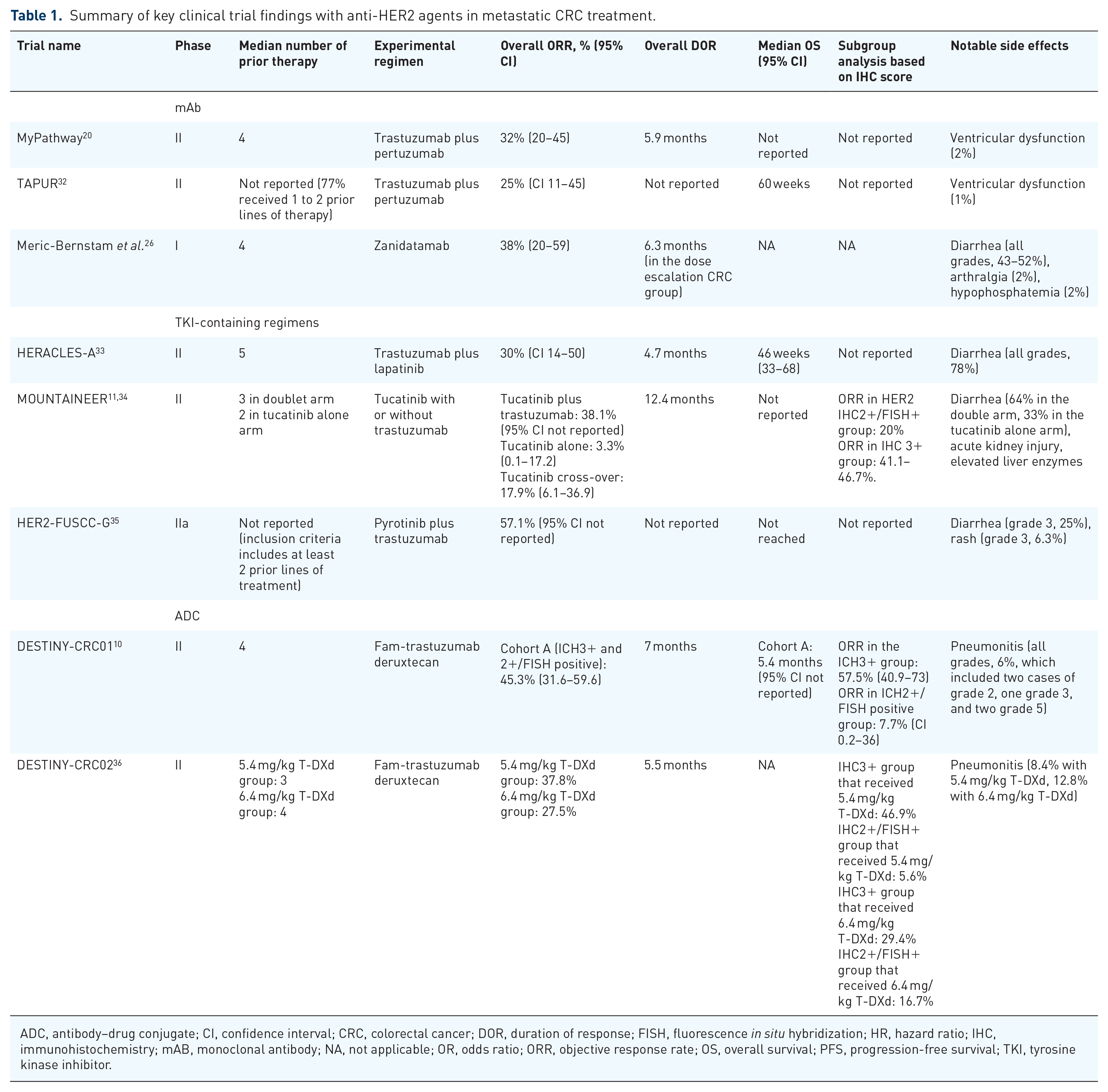
ADC, antibody–drug conjugate; CI, confidence interval; CRC, colorectal cancer; DOR, duration of response; FISH, fluorescence in situ hybridization; HR, hazard ratio; IHC, immunohistochemistry; mAB, monoclonal antibody; NA, not applicable; OR, odds ratio; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; TKI, tyrosine kinase inhibitor.
Monoclonal antibodies
Two phase II basket trials examined the clinical activity of trastuzumab plus pertuzumab in HER2-amplified metastatic CRC. In the MyPathway study with 57 patients whose disease progressed on standard-of-care therapies, trastuzumab plus pertuzumab doublet was associated with an overall objective response rate (ORR) of 32% (95% CI 20–45). 20 Similarly, the TAPUR study, which enrolled 28 patients, reported an ORR of 25% (95% CI 11–45). 32 In addition to promising efficacy signals in patients with refractory CRC, both studies revealed significant clinical implications. First, the clinical activity of anti-HER2 blockade was predominantly observed in patients with KRAS wild-type CRC. In the MyPathway trial, the KRAS wild-type subgroup exhibited a significantly higher response rate compared to KRAS-mutated tumors, with ORRs of 40% (95% CI 25–56) versus 8% (95% CI 0.2–36), respectively. Furthermore, the survival was numerically longer in the KRAS wild-type cohort, with a median OS of 14 months [95% CI 8–not estimated (NE)] versus 8.5 months (95% CI 3.9–NE) in the KRAS-mutated group. The lack of response in the TAPUR study among patients with mutated KRAS (ORR 0%) reinforces the rationale of reserving anti-HER2 antibodies exclusively for KRAS wild-type CRCs. Both trials also suggested that the combination of trastuzumab and pertuzumab is generally well tolerated in this patient population. Ventricular dysfunction, a known rare treatment-related adverse event (TRAE), occurred in only one patient (2%) in the MyPathway study and one patient (1%) in the TAPUR study.
Noteworthy, Nakamura et al. 24 utilized ctDNA genotyping to inform patient enrollment and response evaluation to trastuzumab plus pertuzumab in KRAS wild-type HER2-amplified metastatic CRC. Among 30 participants enrolled in the phase II TRIUMPH study, 22 had a HER2 amplification in both tissue-based and ctDNA-based testings, 5 had a positive tissue testing only, and 3 with a positive ctDNA testing. Patients were then divided into a tissue-positive group and a ctDNA-positive group. The 22 patients with double positivity on tissue and ctDNA were accounted for in both groups. The primary endpoint was confirmed investigator-assessed ORRs associated with trastuzumab plus pertuzumab, which were, respectively, 30% (N = 8 out of 27) in the tissue-positive group and 28% (N = 7 out of 25) in the ctDNA-positive group. Of clinical significance, decreased ctDNA at week 3 of treatment correlated with a significantly longer progression-free survival (PFS) and OS. Moreover, alterations in the RTK/RAS/PI3K pathway genes were enriched among non-responders and upon disease progression.
The phase I basket trial of zanidatamab, a novel bispecific anti-HER2 mAB demonstrated promising results in patients with advanced HER2-expressing solid tumors. 26 The study enrolled 28 patients with KRAS wild-type metastatic CRC who received a median of four prior lines of therapy. Seven (25%) participants had received prior anti-HER2-targeted therapy including T-DXd. In the response-evaluable cohort with 26 patients with metastatic CRC, 10 had a confirmed objective response (ORR 38%, 95% CI 20–59), with a median duration of response of 5.6 months. In patients across all tumor types, diarrhea was the most common TRAE, with an incidence ranging from 52% (N = 24/46) in the dose-escalation cohort to 43% (N = 37/86) in the dose-expansion cohort. Grade 3 TRAEs included arthralgia (N = 1, 2%), hypertension (N = 1, 2%), and hypophosphatemia (N = 1, 2%).
IAH0968, a trastuzumab-like anti-HER2 mAb showed a promising safety profile in a first-in-human phase I trial. Among 18 heavily re-treated patients with HER2-positive solid tumors, only one dose-limiting toxicity (grade 4 arrhythmia) was recorded. 37 Notably, the study did not identify a maximum tolerated dose. A phase II portion of this basket trial is currently underway, evaluating the efficacy of IAH0968 as monotherapy (NCT04934514). In addition, an ongoing phase II/III study will examine IAH0968 combined with capecitabine plus oxaliplatin in HER2-positive metastatic CRCs (NCT05673512).
Oral tyrosine kinase inhibitors
Lapatinib is a novel dual inhibitor of HER2 and EGFR. 38 The HERACLES study was the first proof-of-concept, open-label phase II clinical trial evaluating the efficacy and safety of combined lapatinib and trastuzumab in patients with refractory metastatic CRC. 33 The study enrolled patients with KRAS exon 2 wild-type tumors, who had undergone a median of five prior lines of therapy. Among the 27 participants, 8 (30%, 95% CI 14–50) had an objective response, which included 7 cases of partial response and 1 complete response. Notably, 20 had a tumor with a HER2 IHC score of 3+, while seven had a score of 2+. Among all responders, only one had HER2 IHC of 2+, suggesting higher HER2 expression levels were more likely to respond to the treatment. Diarrhea was the most common adverse event in 21 (78%) patients. Grade 3 TRAEs were rare and included one (4%) case of rash and four (15%) of fatigue.
Tucatinib is a highly selective and potent inhibitor of HER2. 39 The MOUNTAINEER study is an open-label phase II study that evaluated the safety and efficacy of trastuzumab plus tucatinib. 22 In this study, participants were randomized to receive either trastuzumab plus tucatinib or tucatinib alone. The study met its primary endpoint and was approved by the FDA on 29 January 2023, becoming the first approval in the HER2-expressing metastatic CRC. The final analysis with 86 patients in the doublet arm and 30 in the tucatinib cohort showed ORRs of 38.1% (95% CI not reported) and 3.3% (95% CI 0.1–17.2), respectively. Notably, 28 patients from the monotherapy group crossed over to receive trastuzumab plus tucatinib, as allowed by the study protocol, which resulted in an increased ORR of 17.9% (95% CI 6.1–36.9).
In addition, MOUNTAINEER investigators observed a lower ORR of 20% in patients with HER2 IHC2+/FISH+ tumors, while those with tumor IHC score of 3+ had an ORR ranging from 41.1 to 46.7%. Diarrhea was noted again as the most common adverse event with an incidence of 64% in the doublet arm and 33% in the tucatinib alone group. Tucatinib was associated with grade 3 acute kidney injury (N = 1), colitis (N = 1), elevated serum aspartate aminotransferase (N = 2), and alanine aminotransferase (N = 2). There were two (2%) cases of asymptomatic decreased left ventricular ejection fraction which led to tucatinib dose modification or discontinuation.
Pyrotinib is a pan-ErbB receptor TKI. The phase II HER2-FUSCC-G trial combined pyrotinib with trastuzumab for patients with metastatic CRC after at least two prior lines of therapy. 35 In 14 patients with RAS wild-type status, the ORR was 57.1%. The median PFS was 7.5 months and OS was not reached as of yet. There were no grade 4 TRAEs reported in the study. The most common grade 3 TRAEs included diarrhea, which occurred in four cases (25%), and rash, which occurred in one case (6.3%). An ongoing phase I/II study will examine the safety and tolerability of pyrotinib with capecitabine in HER2-positive metastatic CRC (NCT04227041).
Antibody–drug conjugates
The phase II HERACLES-B trial was the first to evaluate the efficacy of ADC for the treatment of HER2-amplified metastatic CRC. 40 In this single-arm study, 31 heavily pretreated patients received pertuzumab in combination with T-DM1. The study did not meet the primary endpoint of ORR, which was 9.7% (N = 3). Nevertheless, a high proportion of patients (67.7%, N = 21) achieved a stable disease with this regimen.
T-DXd has emerged as a new promising ADC option for HER2-amplified tumors and has demonstrated clinical activity in metastatic breast and gastroesophageal tumors.5,18 In previously treated HER2-expressing metastatic CRCs, two phase II trials have reported similar efficacy and safety data on T-DXd.
In DESTINY-CRC01, patients with metastatic CRC with at least two prior lines of treatment received T-DXd at 6.4 mg/kg. 10 HER2 expression was assessed by a central laboratory, and patients were divided into three cohorts based on their HER2 IHC scores. There were no objective responses among participants with HER2 IHC 2+/FISH negative (cohort B), or IHC 1+ (cohort C). In cohort A, 40 patients were included with HER2 IHC 3+ tumors and 13 with IHC 2+/FISH-positive tumors. The IHC 3+ tumors, compared to the IHC 2+/FISH-positive group, were associated with significantly higher ORR (57.5%, 95% CI 40.9–73.0 versus 7.7%, 95% CI 0.2–36.0, respectively) and numerically higher median PFS (not reached, 95% CI 5.4–NE versus 4.1 months, 95% CI 1.3–5.5, respectively).
In DESTINY-CRC02, T-DXd was administered at two different doses (5.4 and 6.4 mg/kg) to 121 patients with metastatic CRC. 36 Consistent with the findings in the DESTINY-CRC01 trial, the ORRs were higher in patients with HER2 IHC 3+ tumors compared to those with HER2 IHC 2+/FISH-positive tumors. In the 5.4 mg/kg dosing group, ORR was 46.9% (95% CI 34.3–59.8) for IHC 3+ patients and 5.6% (95% CI 0.1–27.3) for those with HER2 IHC 2+/FISH-positive tumors. Similarly, in the 6.4 mg/kg dosing group, the ORRs were 29.4% (95% CI 15.1–47.5) and 16.7% (95% CI NE due to only one event occurring in six participants) for HER2 IHC 3+ and HER2 IHC 2+/FISH-positive patients, respectively. Notably, among the 14 patients with mutated RAS tumors, all of whom had HER2 IHC 3+ tumors, four patients achieved an objective response, resulting in an ORR of 28.6% (95% CI 8.4–58.1).
Indeed, the clinical trials of T-DXd in metastatic CRC have provided valuable insights into its clinical use. From the efficacy perspective, both trials observed similar response rates in patients who had received prior anti-HER2 exposure. In DESTINY-CRC01, ORRs were 43.8% for HER2-therapy-naïve patients and 45.9% in patients with prior anti-HER2-therapy. 10 In DESTINY-CRC02, respective ORRs were 41.2 and 40% for both groups. 36 This finding highlights the rationale of reserving T-DXd for use after tumor progression with other anti-HER2 therapies.
From the safety standpoint, T-DXd was associated with drug-related interstitial lung disease (ILD) in both trials, which is consistent with data from patients with breast and gastric cancers. In the DESTINY-CRC01 trial, five cases of ILD were attributed to T-DXd, and two of them resulted in deaths. In DESTINY-CRC02, the incidence of ILD was 12.8% in the 6.4 mg/kg group and 8.4% in the 5.4 mg/kg group, justifying the use of this lower dosage for clinical use. Clinicians must be vigilant about the potential development of ILD when using T-DXd, particularly in patients with preexisting pulmonary ailments. Monitoring and early management of any ILD-related symptoms are essential to minimize the risk of severe complications.
The activity and safety of additional ADCs are under investigation. Zanidatamab zovodotin or ZW49 is a novel ADC paired with cytotoxic payload with the auristatin toxin. In a first-in-human, dose-escalation, and dose-expansion study, ZW49 was administered to patients with HER2-positive advanced tumors with a median number of three prior lines of therapy. Preliminary analysis of the study data, which included 76 patients, indicated that the most common TRAEs associated with ZW49 included keratitis (in 42% of patients, with one case of dose-limited toxicity grade 2 keratitis), alopecia (25%), and diarrhea (21%). Importantly, pulmonary toxicities were not reported. Among 29 response-evaluable participants, the confirmed ORR was 28% across all tumor types. The CRC-specific outcome was not reported. 41
Disitamab vedotin, or RC48, is a novel investigational ADC linked with monotheyl auristatin E. In early phase studies, disitamab vedotin had shown similar therapeutic effects in patients with solid tumors that are HER2 positive and HER2 low. 42 Ongoing trials are exploring its safety and efficacy in metastatic HER2-expressing CRC when combined with fruquintinib (oral inhibitor of vascular endothelial growth factor receptors) (NCT05661357), or with immune checkpoint inhibitors including pembrolizumab (NCT05333809) or tislelizumab (NCT05493683).
Current and future perspectives
HER2 amplification represents a promising therapeutic target in 3–5% of CRC. Currently, in patients with chemorefractory HER2-positive, RAS wild-type metastatic CRC, evidence suggests the use of a doublet anti-HER2 regimen. Tucatinib plus trastuzumab regimen received the first FDA approval for this setting. In patients with prior exposure to doublet anti-HER2 regimens, data from DESTINY-CRC02 suggest the use of T-DXd at 5.4 mg/kg dosage maintains activity while improving the safety profile over a higher dose.
With many HER2-targeting agents already available and even more in development, there are still mounting clinical questions that warrant further investigational efforts. First, HER2 status as the sole predictor for treatment response requires further examination. Despite enrolling only patients with HER2-overexpressing tumors, a significant proportion of patients from the aforementioned trials did not derive benefit from anti-HER2 therapy. In addition, lower HER2 status (e.g. HER2 IHC 2+/ISH+) was associated with discordant clinical benefits when treated with T-DXd versus mAb combined with a TKI. Moreover, to advance therapy development and biomarker selection, it is a priority to explore mitigation strategies for known mechanisms of anti-HER2 resistance, such as point mutations in the HER2 protein kinase domain (particularly R784G and L755S point mutations) and concurrent alterations in the RTK/RAS/PI3K pathway.24,43 As patients with refractory CRC often suffer from comorbidities and polypharmacy, treatment selection and management of TRAEs of these varying agents will be essential to achieve durable clinical benefit. Lastly, as opposed to advanced breast and gastroesophageal tumors, the prospect of employing anti-HER2 therapy in the untreated CRC population remains to be explored. Additional data on proper treatment selection and sequencing are needed.
