Abstract
Background:
Despite progress in treatment, many metastatic renal cell carcinoma (mRCC) patients still experience progression after first-line tyrosine kinase inhibitor (TKI), necessitating effective second-line options. While guidelines endorse combination therapies, accessibility limitations often restrict therapy to TKI monotherapy.
Objectives:
Existing decision-making relies on limited evidence, lacking direct comparisons between the leading second-line options (cabozantinib and nivolumab) which surpass everolimus in advanced mRCC. To address this gap, this study compares the efficacy of TKI versus nivolumab in second line while investigating factors influencing outcomes.
Design:
This was a retrospective cohort study.
Methods:
Turkish Oncology Group Kidney Cancer Consortium includes more than 1000 mRCC patients from 13 centers in Türkiye. It has the largest national data. We extracted 214 patients treated with a TKI in the first line and nivolumab or TKI in the second line.
Results:
The median overall survival (OS) and time to treatment failure (TTF) were similar in the TKI-TKI and TKI-immune checkpoint inhibitor (ICI; 41.1 and 44.8 months, p = 0.446 for OS; 27.4 and 29.8 months, p = 0.857 for TTF). The presence of previous nephrectomy for TTF made a significant difference in univariable and multivariable analysis. Bone metastases negatively affected TTF in both univariable and multivariable analyses. In the neutrophil-to-lymphocyte ratio (NLR)-high group, OS and TTF were longer in patients treated with TKI-ICI than in the TKI-TKI. In multivariable analysis, NLR was an independent prognostic factor for OS and TTF to select ICI in the second-line.
Conclusion:
Our analysis revealed no significant difference in OS between patients receiving ICIs or TKIs as second-line therapy. In the subgroup of patients with elevated NLR, ICI therapy was found to cause no improvement in OS. This finding suggests the potential utility of NLR as a biomarker to guide targeted selection of ICI therapy among patients progressing after first-line TKIs. Furthermore, our study identified other noteworthy prognostic factors influencing outcomes, including the presence of bone or liver metastases, Eastern Cooperative Oncology Group performance status, and International Metastatic Renal Cell Carcinoma Database Consortium risk score.
Introduction
Renal cell carcinoma (RCC) is the primary cause of 95% of all kidney cancers.1,2 About 30% of patients with RCC require systemic therapy for metastatic disease. 3 The current standard of care for metastatic RCC (mRCC) includes targeted therapies that focus largely on vascular endothelial growth factor (VEGF)/VEGF receptors (VEGFR)-directed angiogenesis, with or without immune checkpoint inhibitors (ICIs). 4 Tyrosine kinase inhibitors (TKIs), such as sunitinib, sorafenib, pazopanib, axitinib, cabozantinib, or tivozanib, inhibit the VEGF/VEGFR pathway. Pembrolizumab, nivolumab, and avelumab are ICIs used to treat mRCC and several tumors. 4
Recently, guidelines have recommended combination therapies, especially for patients in the intermediate-poor risk group, according to the International Metastatic Renal Cell Carcinoma Database Consortium (IMDC). 4 However, combination therapies are still not accessible for many, and these patients are treated with TKI monotherapy in the first-line. Although these drugs have been shown to enhance often progression-free survival (PFS), a sizable portion of patients treated with first-line TKI still experience drug resistance and disease progression.5,6 This circumstance highlights the requirement to choose the treatment agents to be administered during the post-TKI stage of treatment. For instance, the phase III METEOR trial demonstrated that cabozantinib outperformed everolimus in terms of PFS, objective response rate (ORR), and overall survival (OS) in patients with mRCC who had disease progression after receiving a TKI.7–9 Nivolumab, on the other hand, demonstrated superior OS and ORR as compared to everolimus following progression in first-line TKI. 10 As a standard second-line therapy following first-line TKI, nivolumab can, therefore, be said to be competitive with cabozantinib. 11
Despite progress in targeted therapies, many mRCC patients still experience disease progression after first-line TKI treatment, necessitating effective second-line options. While guidelines endorse combination therapies for certain risk groups, accessibility limitations often restrict therapy to TKI monotherapy. Although these initial TKIs improve PFS, drug resistance remains a challenge. Existing decision-making relies on limited and anecdotal evidence, lacking direct comparisons between the leading second-line options (cabozantinib and nivolumab) which surpass everolimus in advanced mRCC. To address this gap and improve patient outcomes, this study compares the efficacy of TKI versus nivolumab in second-line therapy while investigating prognostic and predictive factors influencing treatment effectiveness in this specific post-TKI population. Prognostic biomarkers are anticipated to offer insights into cancer outcomes, including OS, regardless of the treatment applied. By contrast, predictive biomarkers are meant to indicate how a therapeutic intervention will influence the patient’s response. 12
Materials and methods
Study design and participants
Patients included in the study were followed between September 2016 and December 2023. Turkish Oncology Group Kidney Cancer Consortium (TKCC) includes more than 1000 patients from 13 centers in Türkiye. It has the largest national data regarding the patients with mRCC. The number of centers and patients’ data are updated regularly. We extracted 214 patients treated with a TKI in the first-line and nivolumab or TKI in the second-line from the TKCC database. Patients with missing data for drugs or last control/exitus date were excluded from the study. The consort diagram explaining how patients were included is also indicated as Supplemental Figure 1. We used neutrophil and lymphocyte values in the first-line setting before starting treatment to calculate the neutrophil-to-lymphocyte ratio (NLR). This study’s reporting adheres to the guidelines set by Strengthening the Reporting of Observational studies in Epidemiology (STROBE) 13 (see Supplemental Table 1).
Related definitions and study endpoints
In this study, an evaluation was made in terms of TTF and OS differences between the groups. It was evaluated whether patients who received immunotherapy after first-line TKI would be superior in terms of TTF and OS compared to those who continued their treatment with TKI change.
Statistical analysis
In our analysis, the null hypothesis was that there is no significant difference in OS or time to treatment failure (TTF) between patients receiving 2L TKI and those receiving 2L ICI following 1L TKI for mRCC. The study was designed as a retrospective cohort analysis to compare the outcomes of the two treatment groups. Kaplan–Meier survival estimates and log-rank tests were utilized to compare OS and TTF. In addition, multivariate Cox proportional hazards regression models were employed to adjust for potential confounding factors, including IMDC risk scores, presence of metastases, and ECOG performance status. We used medians with interquartile range (IQR) and percentages to delineate the data for continuous and categorical variables, respectively. We compared medians and percentages using the Mann–Whitney U test and the Chi-square/Fisher’s exact test, respectively. The primary and secondary outcomes of the study were OS and TTF. We did survival analysis using the Kaplan–Meier estimates and performed a log-rank test to compare survival curves. For multivariable survival analysis, we used Cox’s proportional hazards regression models. We accepted a p-value of less than 0.05 for statistical significance. We used SPSS 27.0 for Mac (IBM Corp., Armonk, NY, USA) and R Studio (version 1.4.1106) for all statistical analyses.
Results
Patient characteristics
A total of 214 patients with clear cell mRCC were included in the analysis. A significant portion of the patients were male (78%). In total, 47.7% of the patients had Fuhrman grade 3–4 tumors. Sarcomatoid features were observed in 7.9% of all patients. In addition, 160 patients (74.8%) had a history of previous nephrectomy. In first-line systemic therapy, 60.3% of patients received sunitinib. The IMDC favorable patient rate was 11.7% in the entire group. The most common visceral metastasis site is the lung (53.3%). Central nervous system (CNS) metastasis was determined to be 29.9% in the whole group. CNS metastasis was also observed significantly more frequently in patients who received ICIs in second-line therapy than in those who continued with different TKIs (36.6% vs 19.3%, p = 0.006). In total, 89.3% of patients had an Eastern Cooperative Oncology Group (ECOG) 0 or 1 performance status. The NLR was observed to be significantly lower in patients who received immune checkpoint inhibitors (ICIs) in second-line therapy compared to those who continued with different TKIs (32.8% vs 55.4%, p < 0.001). In most patients in the TKI-TKI group, axitinib (n = 70, 84.3%) was given as a second-line treatment. In all, 37 and 35 patients received third-line therapy in the TKI-TKI and TKI-IO groups, respectively. Diarrhea (3.4%) and hypertension (3.4%) were the most common grade 3 or 4 adverse events in the TKI-TKI group. Two patients (1.5%) had grade 4 colitis, and one patient (<1%) had grade 3 pneumonitis in the TKI-IO group. The baseline characteristics and subsequent therapies are presented in Tables 1 and 2.
Baseline characteristics of all patients.

Percentages after excluding missing data.
CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; IO, immunotherapy; IQR, inter-quartile range; NLR, neutrophil-to-lymphocyte ratio; TKI, tyrosine kinase inhibitors.
Subsequent therapies.
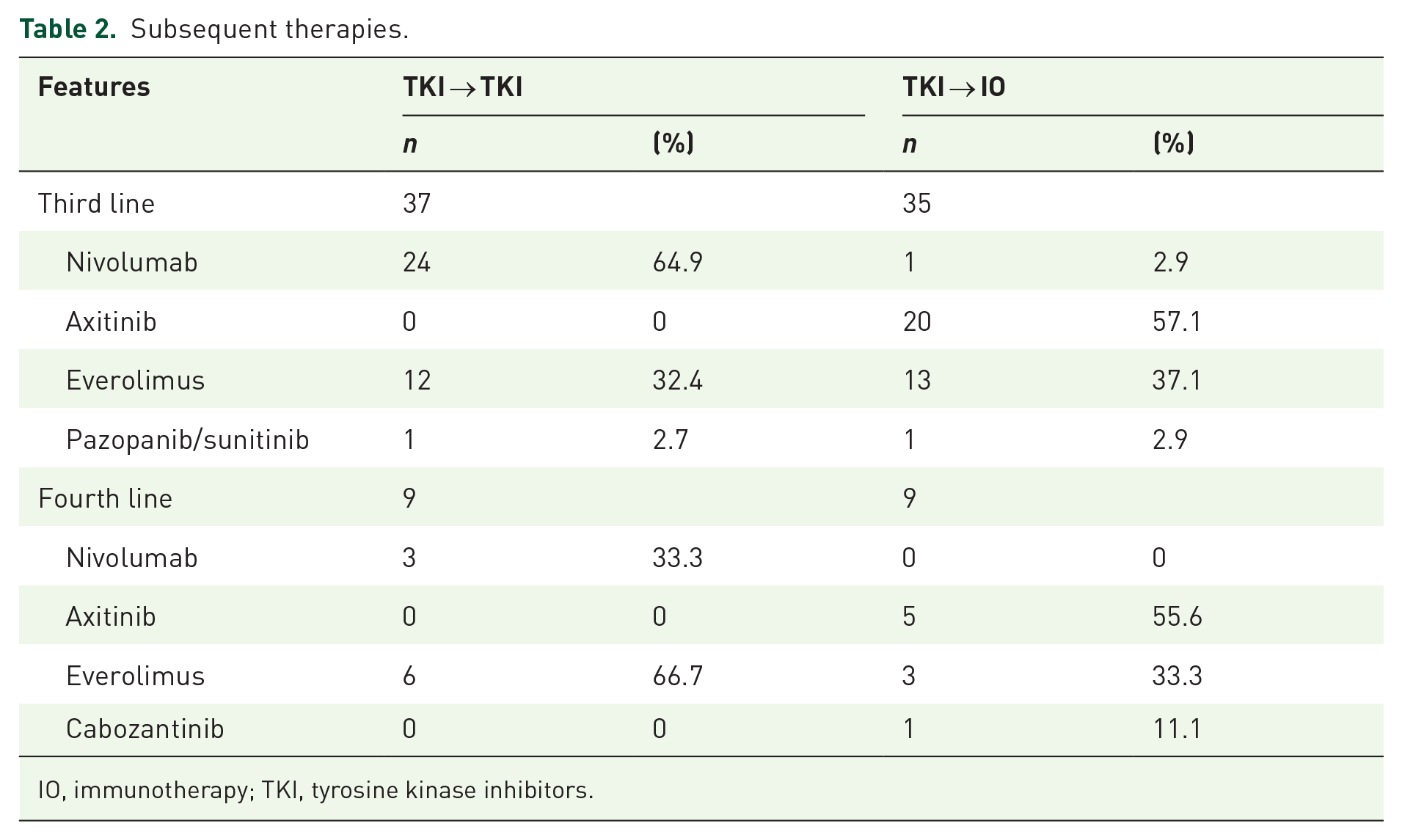
IO, immunotherapy; TKI, tyrosine kinase inhibitors.
Survival outcomes
The median OS and TTF were similar in the TKI-TKI and TKI-ICI groups (41.1 and 44.8 months, p = 0.446 for OS; 27.4 and 29.8 months, p = 0.857 for TTF; Figure 1). On the other hand, the presence of previous nephrectomy for TTF made a significant difference in univariable analysis. This also remained significant in multivariable analysis. The presence of bone metastases in the univariable analysis showed a negative effect in terms of OS. In multivariable analysis, this did not maintain its significance. The presence of bone metastases in the univariable analysis showed a negative effect in terms of TTF. In multivariable analysis, this situation remained significant. The presence of liver metastases in the univariable analysis showed a negative effect in terms of OS. In the univariable analysis, according to IMDC risk scoring, being in the poor group showed a negative effect in terms of OS compared to being in the favorable or intermediate group. This situation remained significant in multivariable analysis. In the univariable analysis, according to IMDC risk scoring, being in the poor group showed a negative effect in terms of TTF compared to being in the favorable and intermediate groups.

(a) Overall survival. (b) Time to treatment failure.
In the NLR-high group, OS and TTF were longer in patients with mRCC treated with TKI-ICI than in the TKI-TKI group. In multivariable analysis, NLR was an independent predictive and prognostic factor for OS and TTF to select ICI in the second line (Figure 2). All univariable and multivariable survival analyses in subgroups are shown in Tables 3–5.
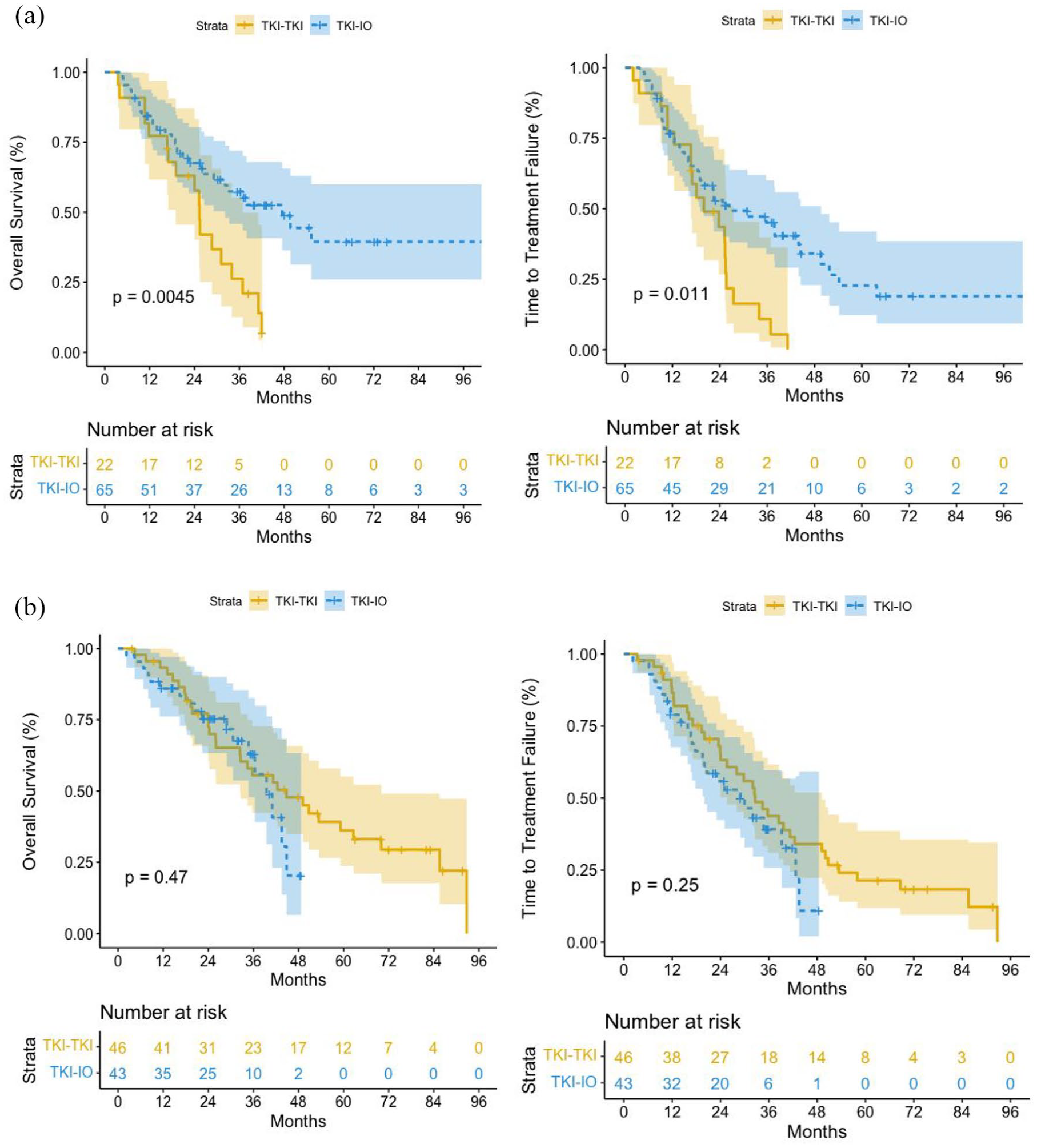
Overall survival and time to treatment failure in NLR high and low groups. (a) NLR-high group. (b) NLR-low group.
Univariate and multivariate analyses for overall survival in all patients.
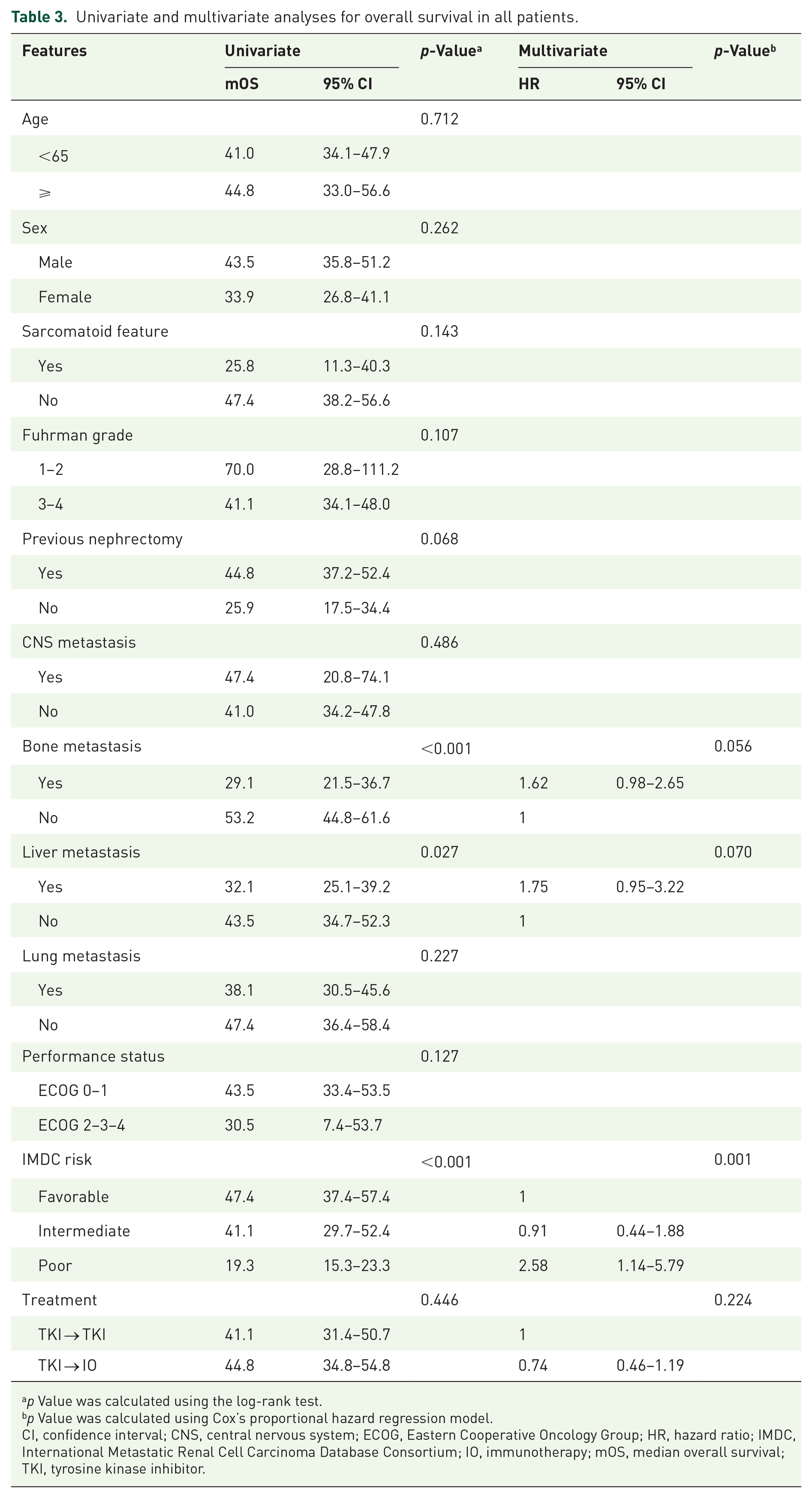
p Value was calculated using the log-rank test.
p Value was calculated using Cox’s proportional hazard regression model.
CI, confidence interval; CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; IO, immunotherapy; mOS, median overall survival; TKI, tyrosine kinase inhibitor.
Univariate and multivariate analyses for TTF in all patients.

p Value was calculated using the log-rank test.
p Value was calculated using Cox’s proportional hazard regression model.
CI, confidence interval; CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; IO, immunotherapy; mTTF, median time to treatment failure; TKI, tyrosine kinase inhibitor.
Multivariate analysis for overall survival and TTF in NLR-high group.

CI, confidence interval; CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; IO, immunotherapy; NLR, neutrophil-to-lymphocyte ratio; OS, overall survival; TKI, tyrosine kinase inhibitor; TTF, time to treatment failure.
Discussion
Our analysis revealed no significant difference in OS between patients receiving ICIs or TKIs as second-line therapy following first-line TKI treatment for mRCC. In the subgroup of patients with elevated NLR, ICI therapy did not demonstrate improved OS compared to TKI. This finding suggests the potential utility of NLR as a predictive biomarker to guide the targeted selection of ICI therapy amongst patients progressing after first-line TKIs. Furthermore, our study identified other noteworthy prognostic factors influencing patient outcomes, including the presence of previous nephrectomy, bone or liver metastases, ECOG performance status, and IMDC risk score. Notably, the presence of bone metastases significantly impacted TTF, highlighting its prognostic role in the disease course. These findings underscore the importance of considering both clinical characteristics and emerging biomarkers like NLR when making personalized treatment decisions for mRCC patients who have developed resistance to first-line TKI therapy. Comparing second-line therapies, TKI-TKI and TKI-ICI showed similar outcomes in OS and TTF. While both TKI and ICIs outperformed everolimus and showed no superiority over each other, variations in subgroup analyses suggest individualized evaluations might be useful.
Brain metastases are observed in approximately 10% of patients with mRCC. The increase in the use of more active systemic therapies and stereotactic radiation has shown potential to improve outcomes in cases with brain metastases; however, evidence remains limited, and these findings require validation in prospective studies. More active systemic therapies and increased use of stereotactic radiation have resulted in likely similar survival outcomes for patients without brain metastases in retrospective analyses. 14 ICIs have CNS penetration and efficacy, although the CNS has traditionally been an area considered difficult to reach by systemic therapies. On the other hand, VEGFR TKIs have very modest reported efficacy based on predictions of angiogenesis required for metastatic spread to the CNS. In a phase II study, no objective response could be demonstrated in patients receiving sunitinib, but CNS metastases remained stable in 31% of patients. 15 In this sense, the results obtained with ICIs seem more positive. In NIVOREN, a phase II study using second-line nivolumab in mRCC patients, an ORR of 23% was demonstrated in untreated, asymptomatic brain metastases. 16 The increase in the use of more active systemic therapies and stereotactic radiation has shown potential to improve outcomes in cases with brain metastases; however, evidence remains limited, and these findings require validation in prospective studies.
Recent research has shown that local and systemic inflammation, as well as decreased or increased myelopoiesis, play a role in survival responses to tumors. 17 Changes in peripheral blood biomarkers, such as the readily available NLR, may represent a systemic inflammatory response in patients. This situation can also be used to predict the response to the options to be used in treatment. Several studies have reported that NLR is a strong predictive biomarker associated with worse OS in various tumor types, including mRCC, in the pre-immunotherapy period.18–20 Currently, more and more studies are showing that NLR can be used in predicting treatment results with ICIs for different types of cancer. 21 Although several studies have investigated the predictive value of NLR in patients receiving immunotherapy for RCC, it is still difficult to confirm.22–25 Because some studies have shown that NLR is not a predictive factor and is not linked to the outcome of RCC patients treated with immunotherapy.25,26 Chen et al.’s 27 meta-analysis linked a high NLR before therapy to reduced OS. Our findings similarly showed shorter OS and TTF in the high NLR group. In patients with high NLR, ICIs demonstrated superior outcomes in OS and TTF compared to TKIs, suggesting that NLR may act as a predictive biomarker for selecting second-line IO treatment in mRCC.
The second most common site of metastasis in mRCC is the bones. 28 RCC bone metastases, being lytic, raise fracture risks and can increase morbidity, potentially causing spinal cord compression, hypercalcemia, and pain.29,30 Swanson et al. 31 examined nearly a thousand patients with RCC and found skeletal metastases mostly involving the spine, pelvis, and proximal femur. In addition to skeletal-related complications, the presence of bone metastases in mRCC has an adverse effect on the PFS and OS. 32 Santoni et al. 33 examined patients with mRCC and bone metastases and reported that age, ECOG, histology, the MSKCC prognostic score, the presence of extra-bony metastases, and the time from nephrectomy to the appearance of bone metastases are important prognostic factors. In our study’s subgroups, like prior research, bone metastasis was pivotal for TTF and OS, reinforcing its recognition as a poor prognostic indicator. Another important site of metastatic spread in mRCC is the liver, which is rarer than lung, lymph node, and bone metastases. Like the CNS and bones, liver metastases have also been associated with poor outcomes. 34 Metastases to the liver, brain, and pleura were associated with the shortest median OS (<18 months). 35 Our analysis revealed a link between liver metastasis and reduced OS.
Due to the known poor prognosis of mRCC, it is important to select appropriate prognostic factors, determine treatment options, and stratify patients in clinical trials to be able to predict survival. The most widely used model for prognosis in mRCC is the IMDC prognostic model. 36 The IMDC model was created based on prognostic data from patients treated with various targeted drugs. 37 Since its emergence, many studies have used the IMDC score to stratify patients at risk for using targeted agents. In our study, sub-analyses revealed a significant difference in both TTF and OS between IMDC risk groups. Currently, immune-based treatments constitute the gold standard for mRCC clear cell variants. Immune-based treatments consist of immunotherapy TKI combination or immunotherapy doublet treatments. 7 While immune-based combination therapies are becoming the standard first-line treatment for mRCC in many high-income countries, such as the United States and European nations, financial and logistical barriers limit their accessibility in low- and middle-income regions. In these settings, TKIs remain the predominant first-line therapy due to availability and reimbursement policies. Specifically, in Turkey, first-line IO-TKI or IO-IO combinations are not reimbursed, making TKI monotherapy the standard approach. This real-world scenario highlights the ongoing relevance of our study, as the majority of patients in these regions continue to require evidence-based guidance for selecting second-line therapies following TKI failure.
While this study contributes valuable insights into second-line treatment options for mRCC, several limitations merit consideration. The inherent limitations of the retrospective design include potential biases and data inconsistencies, despite being conducted in dedicated mRCC treatment centers. In addition, while comparisons were made between overall TKI and ICI groups, subgroup analysis within the TKI arm was not feasible. Furthermore, the cross-sectional nature of the study precludes the establishment of causal relationships between the observed associations and treatment response. Future prospective, controlled trials with larger sample sizes and subgroup analyses are warranted to confirm our findings and elucidate a causal link between NLR and treatment efficacy in this patient population. In conclusion, our study identified no significant difference in OS between second-line TKI and ICI therapy for patients with mRCC previously treated with first-line TKIs. This finding challenges the previously established advantage of both TKIs and ICIs over everolimus in this setting, highlighting the need for further studies to refine treatment selection strategies. While our analysis revealed the potential influence of established prognostic factors such as the presence of metastases and performance status, particularly within the NLR-high subgroup, these findings require validation in larger prospective trials.
Conclusion
The evolving landscape of first-line therapy for mRCC further necessitates research focusing on the impact of these changes on subsequent treatment lines. Notably, the prevalent trend of administering these agents in combination therapy during the first line underscores the critical need for high-quality evidence guiding the selection of second-line and beyond options. In addition, the lack of a widely reliable biomarker for individualizing treatment strategies necessitates further exploration of promising factors like NLR for their potential to optimize patient outcomes. Therefore, further research focusing on personalized medicine approaches and refining second-line treatment selection based on robust evidence are warranted to improve patient outcomes in the evolving treatment landscape of mRCC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251331540 – Supplemental material for Navigating second-line therapy in metastatic renal cell carcinoma: a comparative analysis of immune checkpoint inhibitors and tyrosine kinase inhibitors: a study of Turkish Oncology Group Kidney Cancer Consortium
Supplemental material, sj-docx-1-tam-10.1177_17588359251331540 for Navigating second-line therapy in metastatic renal cell carcinoma: a comparative analysis of immune checkpoint inhibitors and tyrosine kinase inhibitors: a study of Turkish Oncology Group Kidney Cancer Consortium by Musa Barış Aykan, Hatice Bölek, Emre Yekedüz, Elif Sertesen, Deniz Tural, Cengiz Karaçin, Mehmet Ali Nahit Şendur, Gökhan Uçar, Çağatay Arslan, Selver Işık, Saadet Sim, Özlem Nuray Sever, Bekir Hacıoğlu, Sema Sezgin Göksu, Mustafa Özgüroğlu, Nuri Karadurmuş and Yüksel Ürün in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251331540 – Supplemental material for Navigating second-line therapy in metastatic renal cell carcinoma: a comparative analysis of immune checkpoint inhibitors and tyrosine kinase inhibitors: a study of Turkish Oncology Group Kidney Cancer Consortium
Supplemental material, sj-docx-2-tam-10.1177_17588359251331540 for Navigating second-line therapy in metastatic renal cell carcinoma: a comparative analysis of immune checkpoint inhibitors and tyrosine kinase inhibitors: a study of Turkish Oncology Group Kidney Cancer Consortium by Musa Barış Aykan, Hatice Bölek, Emre Yekedüz, Elif Sertesen, Deniz Tural, Cengiz Karaçin, Mehmet Ali Nahit Şendur, Gökhan Uçar, Çağatay Arslan, Selver Işık, Saadet Sim, Özlem Nuray Sever, Bekir Hacıoğlu, Sema Sezgin Göksu, Mustafa Özgüroğlu, Nuri Karadurmuş and Yüksel Ürün in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
