Abstract
Background:
The impact of adjuvant or neoadjuvant chemotherapy in the treatment of craniofacial bone sarcomas has not been clarified. This study aimed to assess whether survival outcomes differed between patients who underwent adjuvant or neoadjuvant chemotherapy.
Methods:
A retrospective search for adult patients diagnosed with malignant neoplasms of the craniofacial bones (International Classification of Diseases 10 codes C41.0–C41.1), within the past 20 years from the access date 28 April 2022, was conducted using the TriNetX network (TriNetX, Cambridge, MA, USA). Cohort I included patients who underwent adjuvant chemotherapy and cohort II included patients with neoadjuvant chemotherapy. A refined search for individuals that received common chemotherapeutic agents, such as methotrexate, doxorubicin, cisplatin, and/or ifosfamide, was conducted and patients were assigned to cohort A (adjuvant chemotherapy) and cohort B (neoadjuvant chemotherapy). Following matching for age and sex, Kaplan–Meier analysis was performed, and risk ratio, odds ratio (OR), and hazard ratio were calculated.
Results:
Patients were assigned to two cohorts, with 181 patients each after matching. In cohorts I and II, 55 and 41 patients died, respectively. No significant differences were found between the two cohorts regarding the 5-year survival probability (I: 59.87% versus II: 68.45%; p = 0.076; log-rank test), or the risk of dying (I: 0.304 versus II: 0.227; risk difference: 0.077; p = 0.096). The risk analysis before matching for age and sex showed a significant survival benefit in cohort II (OR: 1.586; p = 0.0295; risk difference: 0.093). After a refined query to identify patients treated with methotrexate, doxorubicin, cisplatin, and/or ifosfamide, the two cohorts included 47 patients, respectively. In cohort A (adjuvant chemotherapy), 19 patients died, whereas 12 patients died in cohort B (neoadjuvant chemotherapy) within 5 years after diagnosis. Further analysis indicated a greater survival in cohort B, but the survival probability between the cohorts did not differ significantly (A: 43.55% versus B: 54.49%; p = 0.171).
Conclusion:
The use of neoadjuvant chemotherapy may improve survival rates in patients with surgically treated craniofacial bone sarcomas. Due to the retrospective nature of this study, randomized controlled studies are required to derive treatment recommendations.
Keywords
Introduction
The management of rare malignant bone tumors is challenging and requires the involvement of an interdisciplinary team of healthcare specialists encompassing radiologists, oncologists, pathologists, and surgeons. Osteosarcomas are considered the most common primary malignant bone tumors with incidence rates of 0.2/100,000 per year. Moreover, chondrosarcomas are among the most frequently occurring bone sarcomas in adults with incidence rates of approximately 0.1/100,000 per year, whereas Ewing sarcomas are the most common malignant bone tumors in children and less frequently encountered during adulthood.1–3
Osteosarcomas most frequently occur in the long bones of extremities, while the skull, face, or jaws also present common localizations (8%). 4 The incidence of osteosarcomas peaks in the second decade of life, often affecting adolescents and young adults. Craniofacial osteosarcomas predominantly occur in adult patients.4,5 Since approximately 15–20% of patients present with clinically detectable metastases at the time of initial diagnosis, micrometastases may be presumed in a majority of the remaining patients.6,7 The most frequent metastatic sites are the lungs, followed by distant bones. 8 Based on morphological findings, osteosarcomas can be classified as high grade or low grade. High-grade craniofacial osteosarcomas predominantly present a chondroblastic subtype and seem to have a lower risk for distant metastases. 3 Low-grade malignancies, such as low-grade osteosarcoma and parosteal osteosarcoma, may show high-grade components. 9 The majority of craniofacial osteosarcomas present with localized disease, and only a minor number of patients (11%) demonstrate metastases at the time of initial diagnosis. 10
The importance of surgical resection in the treatment of craniofacial bone sarcomas has been pointed out in the National Comprehensive Cancer Network and European Society for Medical Oncology guidelines, but the efficiency of chemotherapies remains controversial. Preoperative neoadjuvant therapy is recommended in high-grade osteosarcomas, whereas primary treatment of low-grade tumors consists of surgical resection. 8 The responsiveness to chemotherapy is considered a prognostic factor. 11 Yet, there is no evidence that neoadjuvant chemotherapy improves survival, provided that postoperative chemotherapy is performed. 12 Common multi-agent chemotherapies include high-dose methotrexate, doxorubicin, cisplatin, and/or ifosfamide.10,13 Limited 5-year survival rates of approximately 68–71%4,12 highlight the need for improved and standardized treatment protocols.
Proposed treatment regimens of osteosarcomas comprise pre- and/or postoperative systemic therapies. 14 The aim of this study was to examine the impact of adjuvant and neoadjuvant chemotherapy in the management of osteosarcoma of the craniofacial skeleton. Hence, 5-year survival rates of patients that underwent adjuvant chemotherapy were compared to patients who underwent neoadjuvant chemotherapy. To evaluate treatment strategies of craniofacial bone sarcomas, a retrospective case–control study was performed. Data were retrieved from the TriNetX global health research network (TriNetX, Cambridge, Massachusetts, USA). The TriNetX platform promotes research on highly relevant medical areas15,16 by providing continuously updated real-world data on more than 250 million patients supplied by more than 120 healthcare organizations (HCOs) worldwide.
Patients and methods
Data acquisition, inclusion and exclusion criteria
The TriNetX network was accessed on 28 April 2022. This query was run on the COVID-19 Research Network, the platform of a group of 68 HCOs. The database was searched for the electronic medical records of patients at least 18 years of age at most recent occurrence, diagnosed with malignant neoplasms of the skull and face or mandible according to the International Classification of Diseases (ICD)-10 codes C41.0–C41.1, up to 20 years before the access date. Patients were eligible that underwent a combination of surgical treatment (Healthcare Common Procedure Coding System D7111–D7999) and chemotherapy. A total of 278 patients who underwent surgical resection before chemotherapy were grouped into cohort I, and 184 treated with chemotherapy before surgery were assigned to cohort II. Propensity score matching was applied to reduce confounding variables and equate groups based on similar covariate distributions, as demonstrated in the modified Consolidated Standards of Reporting Trials flow chart 17 (Figure 1). Characteristics such as current age and gender distribution of all individuals were retrieved, and one-to-one matching was performed. Following propensity score matching, cohorts I and II of 181 patients each were obtained (Table 1).

Modified Consolidated Standards of Reporting Trials flow chart. 17
Patient characteristics before and after propensity score matching of cohort I (adjuvant chemotherapy) and cohort II (neoadjuvant chemotherapy).

Percentage refers to the gender distribution within the respective cohorts. p Value refers to the comparison between both cohorts (log-rank test).
SD, standard deviation.
A refined search was conducted that limited the query criteria to patients that received chemotherapy using commonly applied chemotherapy drugs. Subjects diagnosed with ICD-10 codes C41.0–C41.1 over a 20-year period prior to the access date that received surgery and chemotherapy with methotrexate, doxorubicin, cisplatin, and/or ifosfamide were included and electronic patient records were retrieved from the TriNetX platform on 29 April 2022. Cohort A included 93 patients that underwent adjuvant chemotherapy, whereas cohort B included 47 patients that received neoadjuvant chemotherapy. Propensity score matching was performed, as previously described. After matching, both cohorts contained 39 patients.
Data analysis
After propensity score matching of the two cohorts, the primary outcome was defined as ‘death’ after 5 years of the initial diagnosis. The evaluation was limited to a period of 5 years after diagnosis, as this time frame was considered the most relevant for an evaluation of treatment outcome. Subsequently, statistical analysis was performed using Kaplan–Meier analysis. Risk ratios (RR), odds ratios (OR), and hazard ratios (HR) were calculated and a log-rank test performed to compare the risk of death between the two groups with a 95% confidence interval (CI). p Values below 0.05 were defined as statistically significant.
Results
Assessment, allocation, and matching
A total number of 462 patients with ICD-10 codes C41.0–C41.1 who received surgical treatment and chemotherapy from 68 HCOs met the query criteria and were assessed. Subsequently, 278 individuals from 20 HCOs that received chemotherapy after surgical tumor resection were allocated to cohort I. Cohort II included 184 patients from 17 HCOs, where chemotherapy occurred prior to surgical treatment. Cohort I (adjuvant chemotherapy) included 278 patients [118 females (42.4%), 160 males (57.6%)] with a mean age of 65.1 years ± 14.7 standard deviation (SD). Cohort II (neoadjuvant chemotherapy) included 184 patients [95 females (51.6%), 89 males (48.4%)] with a mean age of 63.1 years ± 16.7 SD. After propensity score matching, cohorts I and II included 181 patients, respectively. In cohort I, 95 females (52.5%) and 86 males (47.5%) were included with a mean age of 64.3 years ± 16.0 SD. Cohort II (neoadjuvant chemotherapy) included 92 females (50.8%) and 89 males (49.2%) with a mean age of 63.5 years ± 16.5 SD. The groups did not differ significantly in gender distribution or age (p = 0.752 and p = 0.647, respectively; log-rank test). Table 1 shows the patient characteristics of both cohorts before and after matching.
After a refined search for patients diagnosed with ICD-10 codes C41.0–C41.1, who received surgical treatment and chemotherapy using the four selected chemotherapeutic drugs, a total of 140 patients from 68 HCOs met the query criteria and were grouped into two cohorts. Cohort A (adjuvant chemotherapy) included 93 patients from 17 HCOs [32 females (34.4%) and 61 males (65.6%)] with a mean age of 65.0 years ± 13.0 SD. Cohort B (neoadjuvant chemotherapy) included 47 patients from 11 HCOs [26 females (55.3%), 21 males (44.7%)] with a mean age of 57.8 years ± 19.2 SD. Next, propensity score matching was conducted. Thereafter, cohorts A and B consisted of 39 patients, respectively (Table 2). In cohort A, 23 females (59.0%) and 16 males (41.0%) were included with a mean age of 65.3 years ± 14.7 SD. Cohort B included 18 females (46.2%) and 21 males (53.8%) with a mean age of 61.3 years ± 17.1 SD. The groups did not differ significantly in gender distribution or age (p = 0.257 and p = 0.281, respectively; log-rank test).
Patient characteristics before and after propensity score matching of cohort A (adjuvant chemotherapy) and cohort B (neoadjuvant chemotherapy).

Percentage refers to the gender distribution within the respective cohorts. p Value refers to the comparison between both cohorts (log-rank test).
SD, standard deviation.
Risk analysis and patient survival
The results before propensity score matching were evaluated to examine the risk of death within 5 years after the initial diagnosis. The risk analysis before matching demonstrated a significant difference in survival between cohort I (adjuvant chemotherapy) and cohort II (neoadjuvant chemotherapy) with a risk difference of 0.093%. The OR of 1.586 (95% CI: 1.045, 2.406) was statistically significant (p = 0.0295). The HR was 1.53 (95% CI: 1.07, 2.18). In addition, matching for age and sex was conducted, respectively. Matching for sex resulted in an OR of 1.5 (95% CI: 0.88, 2.56; p = 0.136), an HR of 1.52 (95% CI: 0.96, 2.42) and a risk difference of 0.076. After matching for age, the risk difference was 0.101, the OR was 1.67 (95% CI: 0.98, 2.85; p = 0.059) and the HR was 1.62 (95% CI: 1.03, 2.56).
Next, statistical analysis was performed to compare the risk of death between the two cohorts after propensity score matching within a 5-year period after initial diagnosis, as demonstrated in Table 3. First, the risk of death and survival probability were compared between cohort I (adjuvant chemotherapy) and cohort II (neoadjuvant chemotherapy). In cohort I, 55 deaths were reported amongst 181 patients. In cohort II, 41 individuals died. This corresponds to a risk of death of 0.304 in cohort I compared to a risk of 0.227 in cohort II. The difference between risks of death was not statistically significant in a log-rank test (p = 0.096). The related RR and OR were 1.341 (95% CI: 0.947, 1.900) and 1.491 (95% CI: 0.931, 2.386), respectively. Kaplan–Meier analysis demonstrated a survival probability after 5 years of 59.87% in cohort I, compared to a 68.45% survival probability in cohort II (Figure 2). No statistical significance was found using a log-rank test (p = 0.076). The HR was 1.44 (95% CI: 0.961, 2.159).
Risk of death, RRs and ORs of cohort I (adjuvant chemotherapy) and cohort II (neoadjuvant chemotherapy) after propensity score matching.
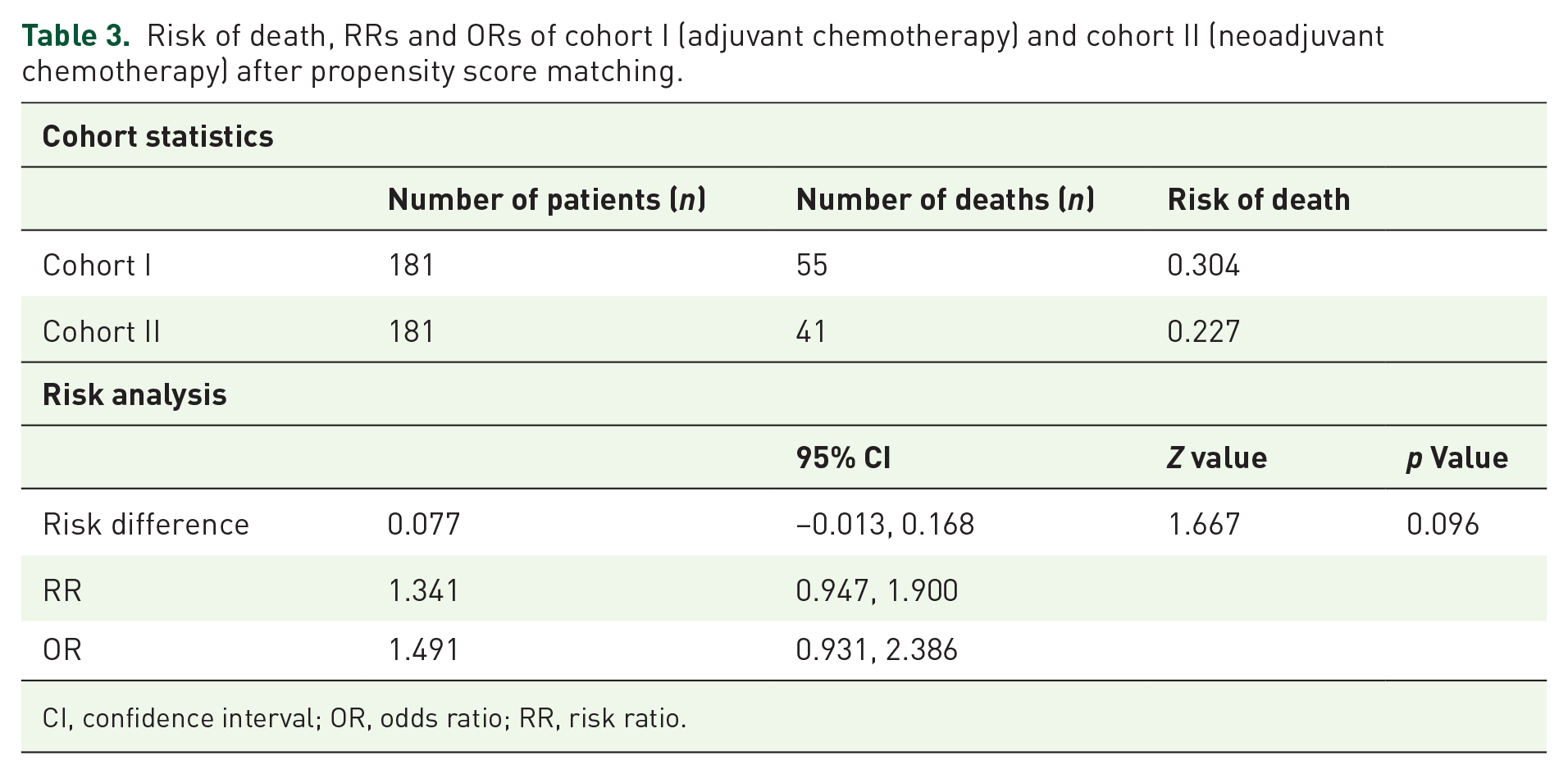
CI, confidence interval; OR, odds ratio; RR, risk ratio.

Kaplan–Meier survival curve of cohort I (adjuvant chemotherapy) and cohort II (neoadjuvant chemotherapy) after propensity score matching.
Next, the risk of death and survival probability were assessed in cohorts A and B, that included patients that were treated using a combination of surgical resection and chemotherapy using methotrexate, doxorubicin, cisplatin, and/or ifosfamide. First, we conducted a risk analysis before propensity score matching for cohorts A and B. The OR was 1.32 (95% CI: 0.6, 2.91; p = 0.49), an HR of 1.45 (CI: 0.76, 2.76), and a risk difference of 0.063. A further analysis to compare the risk of death between the two cohorts A and B after propensity score matching within a 5-year period after initial diagnosis was conducted, as demonstrated in Table 4. In cohort A, 19 deaths were reported among 39 patients. In cohort B, 12 individuals died. This corresponds to a risk of death of 0.487 in cohort A compared to a risk of 0.308 in cohort B. The difference between risks of death was not statistically significant (p = 0.105; log-rank test). The related RR and OR were 1.583 (95% CI: 0.895, 2.801) and 2.138 (95% CI: 0.847, 5.394), respectively.
Risk of death, RRs and ORs of cohort A (adjuvant chemotherapy) and cohort B (neoadjuvant chemotherapy) after propensity score matching.

CI, confidence interval; OR, odds ratio; RR, risk ratio.
The survival probability after 5 years derived from Kaplan–Meier analysis was 43.55% in cohort A and, thus, lower than the 54.49% survival probability in cohort B (Figure 3). The difference in survival probability between the two groups was not statistically significant (p = 0.932; log-rank test). The HR was 1.649 (95% CI: 0.799, 3.405). Overall, the results indicated a higher survival probability in patients, that received neoadjuvant chemotherapy (cohort B).

Kaplan–Meier survival curve of cohort A (adjuvant chemotherapy) and cohort B (neoadjuvant chemotherapy) after propensity score matching.
Discussion
The aim of this study was to elucidate the role of adjuvant and neoadjuvant chemotherapy in the management of craniofacial bone sarcomas by performing a retrospective data analysis via the TriNetX platform. Complete surgical tumor resection is considered a major factor to achieve optimum treatment outcome in bone sarcomas, while the effectiveness and time point of chemotherapy remain unclear. 14
This retrospective analysis found no statistically significant difference in the risk of death between patients that underwent adjuvant or neoadjuvant chemotherapy. However, the OR of 2.138 indicates an association between adjuvant chemotherapy and a greater risk of death within the first 5 years of initial diagnosis. The survival probability between the cohorts did not differ significantly. Yet, patients treated with neoadjuvant chemotherapy demonstrated higher survival rates of 54.49%, compared to a 43.55% survival probability in patients that received adjuvant chemotherapy. Furthermore, age and sex were relevant factors in determining outcome, as demonstrated by the risk analysis before propensity matching. The OR of 1.586 between cohorts I and II before matching indicates improved survival in the cohort that received neoadjuvant chemotherapy. The results were compared to previously reported studies. Current literature shows similar 5-year survival rates of approximately 77% in craniofacial osteosarcomas and approximately 55–70% in extracranial osteosarcomas.5,18,19 The observational EUROpean Bone Over 40 Sarcoma Study (EURO-B.O.S.S.) found a greater 5-year disease-free survival rate in patients that received neoadjuvant chemotherapy followed by postoperative chemotherapy, as compared to patients that received adjuvant chemotherapy only. However, treatment regimens were not randomized and selected based on clinical features and feasibility of surgical tumor removal. 10 Prospective randomized studies are required to clarify the benefits of neoadjuvant compared to adjuvant chemotherapy.
The selected ICD-10 codes C41.0–C41.1 in this study included various rare malignant neoplasms of bones of skull and face other than osteosarcoma, such as chondrosarcoma, Ewing sarcoma, and other rare forms of bone cancer – for example, malignant odontogenic tumors – that may require different treatment regimens. While osteosarcomas are considered the most common malignant bone tumors and account for approximately 10–20% of bone tumors, chondrosarcomas are the second most common malignant osseous malignancy in adults.1–3 In contrast to osteosarcomas, chondrosarcomas require primary surgical resection since chemotherapy is considered ineffective. Their limited responsiveness to chemotherapies has been assigned to their slow cell division and poor vascularization. 20 Evidence for the use of chemotherapies in chondrosarcomas is limited, but mesenchymal and dedifferentiated chondrosarcomas may respond to chemotherapies such as anthracycline and alkylating agents. In these cases, combined surgery and chemotherapy may be beneficial.21–23 Since the search query applied in this study only included tumors that were treated with a combination of surgical resection and chemotherapy, the entity of chondrosarcoma was not primarily addressed in this study. However, it cannot be excluded that patients diagnosed with chondrosarcoma and treated with the combination of surgery and chemotherapy formed part of the study cohort.
While Ewing sarcomas are frequently encountered malignant bone tumors in children and adolescents, they are less common during adulthood.1–3 The proposed treatment regimen for Ewing sarcomas compares to that of osteosarcoma using pre- and/or postoperative chemotherapy. Yet, this retrospective data included patients above 18 years so that the entity of Ewing sarcoma recedes into the background of this study. Other malignant odontogenic tumors, also comprised in ICD-10 code C41.1, such as ameloblastic carcinoma, ameloblastic odontosarcoma, or ameloblastic sarcoma, are treated by surgical resection with some beneficial reports of neoadjuvant or adjuvant chemotherapy dependent on tumor entity.24,25 Hence, the management of rare odontogenic malignancies may compare to the treatment of osteosarcomas.
Limitations of the study arise from the nature of retrospective data analysis based on ICD-10 codes. Differences in tumor entity, stage, and grading could not be retrieved. However, the results from histopathological analysis and staging are important factors when deciding on the treatment regimen including the need for adjuvant or neoadjuvant therapies. Furthermore, data on the surgical resection margin were not available, owing to the retrospective nature of this study. Yet, the complete surgical resection is an important factor with regard to treatment outcome and overall survival. As a further limitation of this study, data on the risk of relapse and the relapse-free survival between the two cohorts could not be retrieved. When assessing the results following adjuvant or neoadjuvant chemotherapy, the adherence to treatment as well as the treatment intensity and protocol need to be considered. In this study, the adherence to treatment protocol was assumed for both cohorts, independent of the time point of chemotherapy. Cohorts A and B included patients with craniofacial bone malignancies treated with combined surgery and chemotherapy using methotrexate, doxorubicin, cisplatin, and/or ifosfamide. Based on the incidence of craniofacial malignancies and the inclusion criteria applied in this retrospective study, cohorts A and B included tumor entities that are considered responsive to these chemotherapeutics. Consequently, craniofacial osteosarcomas may be considered the major tumor entity included in this retrospective study, whereas the number of patients diagnosed with tumor entities other than craniofacial osteosarcomas may be negligible.
Owing to the retrospective nature of this study based on ICD-10 codes, the data did not elaborate on the differentiation of high-grade or low-grade osteosarcomas. Yet, the majority of osteosarcomas are considered high grade. 26 Low-grade osteosarcoma may not require pre- or postoperative chemotherapy. 8 Current protocols for high-grade osteosarcoma propose the administration of preoperative chemotherapy. The drug regimen of doxorubicin/cisplatin/HD-MTX (MAP) is most commonly used in children and young adult patients. 13 In older adults, the combined regimen of doxorubicin, cisplatin, and ifosfamide may be applied. MTX therapy may be performed in cases of poor response to non-MTX treatment.10,27 While neoadjuvant chemotherapy may facilitate preoperative tumor reduction, the pathohistological and immunohistological analysis of the resected specimen allows us to assess the response to the preoperatively administered regimen. These factors may implicate a benefit of neoadjuvant chemotherapy but there is no evidence to support a change in chemotherapy based on this alone.10,13 Future research should address the histological regression of craniofacial bone sarcomas following neoadjuvant chemotherapy based on pathohistological analysis of tumor specimen. Bone sarcomas present various histological types and genomic mutations. Hence, the application of systemic therapies in osteosarcomas remains challenging. 11
The inclusion criteria of neoadjuvant chemotherapy selected in this study did not differentiate between the sole administration of preoperative chemotherapy in contrast to the combined administration of pre- and postoperative chemotherapy. Yet, the standard treatment regimen of osteosarcomas labeled as neoadjuvant chemotherapy in previous studies includes the pre- and postoperative administration of chemotherapeutic agents. 10 Hence, the cohort of neoadjuvant chemotherapy included in this retrospective study may primarily consist of patients that received primary chemotherapy followed by postoperative chemotherapy.
To date, there is no evidence to apply a different treatment regimen for craniofacial osteosarcomas in comparison with bone malignancies of other locations. Yet, depending on the localization at the skull, face or jaws, surgical access, resection, and reconstruction may be especially demanding. Consequently, the administration of neoadjuvant chemotherapy for a defined period preoperatively may be beneficial for surgical resection.8,28 The treatment with adjuvant radiotherapy was not addressed in this study. Radiotherapy may be proposed if complete surgical resection cannot be performed due to tumor size, localization, metastasis, or other factors, or in case of positive margins after resection. 8 Proton-based therapies and stereotactic radiosurgery may permit improved multimodal treatment regimens.29,30
This retrospective study indicated greater survival rates following neoadjuvant chemotherapy in contrast to sole application of adjuvant chemotherapy. Craniofacial osteosarcomas that respond to neoadjuvant chemotherapy may benefit from the additional exposure to postoperative chemotherapy and demonstrate improved 5-year survival rates. The study of patients exposed to neoadjuvant and adjuvant chemotherapy with regard to staging, grading, and histological regression may permit the identification of individualized treatment regimens with the best survival rates and lowest risk of death. The real-world data were retrieved from healthcare providers all over the world, including countries in Europe, the Middle East, Africa, Asia, as well as North and South America. Discrepancies in the management of craniofacial bone sarcomas may be concealed in this multi-center retrospective study.
The rarity and complexity of craniofacial osseous sarcoma demands standardized treatment regimens. This retrospective study found no differences in the risk of death or survival rates between adjuvant and neoadjuvant chemotherapy in addition to surgical resection of craniofacial bone malignancies. However, the results indicated higher survival rates following neoadjuvant chemotherapy. When comparing treatment outcomes between adjuvant and neoadjuvant chemotherapies, differences in tumor entities, histological staging and grading, patient characteristics and treatment schedules must be considered. Hence, future studies should include the split analysis of different tumor entities such as chondrosarcoma and osteosarcoma. The treatment of bone sarcomas located at the craniofacial skeleton remains challenging. Patients’ quality of life depends on the preservation and restoration of function, including speech, swallowing, and chewing, as well as facial expression and aesthetics. Randomized controlled trials are required to derive recommendations for the management of bone sarcomas.
