Abstract
Background:
Standard of care (SoC) for patients with advanced triple-negative breast cancer (TNBC) whose tumors express PD-L1 (combined positive score ⩾ 10) is chemotherapy plus anti-PD-(L)1 inhibitors; however, prognosis and survival for most patients is poor. Datopotamab deruxtecan (Dato-DXd), a novel antibody-drug conjugate comprising a humanized anti-TROP2 IgG1 monoclonal antibody conjugated to a potent topoisomerase I inhibitor payload via a plasma-stable, cleavable, tetrapeptide-based linker, has shown preliminary activity as mono or combination therapy in advanced/metastatic TNBC.
Objectives:
TROPION-Breast05 is an ongoing randomized, open-label, multicenter phase III study. The primary objective is to demonstrate the superiority of Dato-DXd in combination with durvalumab (an anti-PD-L1 antibody) versus SoC treatment in patients with PD-L1-high locally recurrent inoperable or metastatic TNBC.
Methods and design:
Patients (⩾18 years) will be randomized 1:1 to receive Dato-DXd (6 mg/kg intravenously (IV) every 3 weeks (Q3W)) plus durvalumab (1120 mg IV Q3W) or investigator’s choice of chemotherapy (ICC; paclitaxel, nab-paclitaxel, or gemcitabine plus carboplatin) plus pembrolizumab (200 mg IV Q3W). In selected countries, patients will also be randomized (1:1:1) to a third arm of Dato-DXd monotherapy. The primary study endpoint is progression-free survival (PFS) per blinded independent central review (Dato-DXd plus durvalumab arm vs ICC plus pembrolizumab arm). Overall survival is a key secondary endpoint; other secondary endpoints include PFS (investigator-assessed), objective response rate, duration of response, clinical benefit rate at Week 24 (all assessed in the Dato-DXd plus durvalumab arm vs ICC plus pembrolizumab arm), patient-reported outcomes, and safety.
Ethics:
The study is approved by independent ethics committees or institutional review boards at each study site. All patients will provide written informed consent.
Discussion:
TROPION-Breast05 will assess the potential role of Dato-DXd with or without durvalumab in patients with PD-L1-high advanced or metastatic TNBC. The findings of this trial could lead to a new treatment option for these patients.
Trial registration:
ClinicalTrials.gov identifier: NCT06103864 (Date of registration: 27 October 2023).
Plain language summary
Standard treatment for people with TNBC that cannot be removed with surgery or has spread to other parts of the body and whose cancer cells express a protein called PD-L1 is chemotherapy plus anti-PD-(L)1 inhibitors. However, additional treatment options are needed to improve treatment outcomes and minimize side effects.
Dato-DXd, an antibody-drug conjugate (ADC), consists of an antibody (datopotamab) linked to an anti-cancer drug (deruxtecan). The antibody attaches to a protein found on the surface of breast cancer cells called TROP2. The antibody is then taken inside the cancer cells, the anti-cancer drug is released, and the cells are killed. By releasing the anti-cancer drug inside the cell, the treatment is directed to the cancer site with the goal of minimizing side effects.
In a study called TROPION-PanTumor01, 40% of people with TNBC who received Dato-DXd (and who had not previously received ADCs) had a reduction in their tumor size of at least 30%. These results were promising and it is thought that combining Dato-DXd with a PD-L1 inhibitor may make cancer cells more susceptible to being targeted by the body’s immune system. In another study called BEGONIA, Dato-DXd was evaluated in combination with durvalumab, which blocks the PD-1/PD-L1 interaction. In people with TNBC, treatment with Dato-DXd and durvalumab resulted in a decrease in tumor size of at least 30% in 79% of people. The safety profile of Dato-DXd and durvalumab was tolerable and manageable; the most common D-DXd side effects were nausea and mouth sores, known as stomatitis.
Based on data from TROPION-PanTumor01 and BEGONIA, the phase III TROPION-Breast05 will compare Dato-DXd alone and in combination with durvalumab vs standard-of-care treatment for people with TNBC whose cancer cells express PD-L1, and whose cancer cannot be removed by surgery, or their cancer has spread to other parts of the body.
Keywords
Introduction
Triple-negative breast cancer (TNBC) is defined by tumor cells lacking the expression of estrogen receptors (ER) and progesterone receptors (PR), and not have amplification or overexpression of the human epidermal growth factor receptor 2 (HER2). 1 TNBC is the most aggressive breast cancer subtype and is associated with a shorter time to relapse, increased risk of developing metastasis, and greater molecular heterogeneity compared with other subtypes. 2 Although TNBC accounts for approximately 15% of breast cancer diagnoses, it is responsible for over a third (~35%) of all breast cancer-related deaths.2–4 Furthermore, while around 5% of patients with TNBC present with metastatic disease, approximately 25% of patients with localized disease will subsequently develop distant metastasis.1,5,6
Historically, advanced or metastatic TNBC had limited treatment options, and the standard of care was chemotherapy. Unfortunately, the prognosis for patients with advanced or metastatic TNBC who received single-agent chemotherapy was poor, with a median overall survival (OS) of approximately 12 months. 7 However, immune checkpoint inhibitors in combination with chemotherapy have recently emerged as a new treatment option in this setting. 8 In the phase III KEYNOTE-355 study (NCT02819518), median progression-free survival (PFS; 9.7 vs 5.6 months, respectively; hazard ratio (HR) 0.65 (95% confidence intervals (CI): 0.49–0.86); p = 0.0012) and OS (23.0 vs 16.1 months, respectively; HR 0.73 (95% CI: 0.55–0.95); p = 0.0185) in patients with a combined positive score (CPS) of ⩾10 were significantly longer for patients who received chemotherapy plus pembrolizumab (an anti-programmed cell death 1 (PD-1) antibody) compared with patients who received chemotherapy plus placebo.8,9 The addition of immunotherapy to chemotherapy did not result in unexpected toxicities but did result in immune-mediated adverse events (AEs) that are well-recognized to occur with programmed cell death (ligand) 1 (PD-(L)1) inhibitors. 10 Based on the results of this study, chemotherapy plus pembrolizumab was subsequently approved by the Food and Drug Administration (FDA) in the United States for patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ⩾ 10) as determined by an FDA-approved test. 11 In the European Union, chemotherapy plus pembrolizumab is approved for patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ⩾ 10) and who have not received prior chemotherapy for metastatic disease. 12 In the phase III IMpassion130 study (NCT02425891), the combination of nab-paclitaxel with atezolizumab (an anti-PD-L1 antibody) prolonged PFS compared with nab-paclitaxel plus placebo in the intention-to-treat (ITT) population (7.2 vs 5.5 months, respectively; HR 0.80 (95% CI: 0.69–0.92); p = 0.002) and the PD-L1-positive subgroup (7.5 vs 5.0 months, respectively; HR 0.62 (95% CI: 0.49–0.78); p < 0.001). 13 However, no significant difference was observed in the median OS. 14 In addition, the combination of paclitaxel plus atezolizumab did not demonstrate improvements in PFS or OS compared with paclitaxel monotherapy in patients with advanced TNBC in the IMPassion131 trial (NCT3125902). 15 Following the changes to the treatment landscape and the results of the IMPassion130 and IMPassion131 trials, the approval of atezolizumab was voluntarily withdrawn in the United States for this indication. 16 Nab-paclitaxel plus atezolizumab was approved by the European Union for the treatment of patients with unresectable locally advanced or metastatic TNBC whose tumors have PD-L1 expression ⩾1% (PD-L1 stained tumor-infiltrating immune cells covering ≥1% of the tumor area) and who have not received prior chemotherapy for metastatic disease.13,17
Given the limitations of systemic chemotherapy in the treatment of advanced TNBC, the ability of ADCs to selectively deliver cytotoxic agents directly to breast cancer cells, thereby reducing systemic exposure and toxicity, 18 is of particular interest. Over the last 5 years, topoisomerase-based ADCs have demonstrated improved efficacy compared with chemotherapy in all breast cancer subtypes. 19 ADCs consist of an antibody directed against a specific tumor antigen, the cytotoxic drug (payload), and a linker connecting the payload and antibody. 20 Upon binding to the cell-surface antigen, the ADC is internalized by the tumor cell, the payload is released into the cytoplasm, and the cell dies via apoptosis. 20 One such transmembrane protein is trophoblast cell-surface antigen 2 (TROP2), which is overexpressed in TNBC,21–23 and represents a potential target for directing ADCs to TNBC tumors. Validation of TROP2 as an ADC target in TNBC was demonstrated in the phase III ASCENT study (NCT02574455), which reported longer median OS (12.1 vs 6.7 months, respectively; HR 0.48 (95% CI: 0.38–0.59); p < 0.001) and PFS (5.6 months vs 1.7 months, respectively; HR 0.41 (95% CI: 0.32–0.52); p < 0.001) with sacituzumab govitecan (an anti-TROP2 ADC) compared with chemotherapy. 24 Based on the results of this study, sacituzumab govitecan was approved in the United States and European Union and is recommended by international treatment guidelines for patients with unresectable or metastatic TNBC who have received two or more prior systemic therapies.25–27 Additionally, based on the results of the phase III DESTINY-Breast04 study, the anti-HER2 ADC trastuzumab deruxtecan is approved in the United States and European Union for patients with unresectable or metastatic HER2-low breast cancer who have received prior chemotherapy in the metastatic setting or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy.28–30
Datopotamab deruxtecan (Dato-DXd) is a TROP2-directed ADC in clinical development. The anti-TROP2 antibody is conjugated to a potent topoisomerase I inhibitor payload (an exatecan derivative) via a tetrapeptide-based, plasma-stable, tumor-selective cleavable linker. 31 Dato-DXd binds to the cell surface of TROP2-expressing cells, is internalized into tumor cells, and trafficked to the lysosome, leading to payload release. 31 DXd subsequently accumulates in tumor cells, inducing DNA damage and apoptosis. 31 The payload has a short systemic half-life that reduces systemic toxicity and also exhibits a bystander tumor effect via payload efflux to surrounding tumor cells.32,33 Preliminary safety and efficacy of Dato-DXd monotherapy were observed in patients with advanced, unresectable, or metastatic TNBC in the ongoing phase I first-in-human TROPION-PanTumor01 study (NCT03401385). 34 In patients with advanced, unresectable, or metastatic TNBC, Dato-DXd demonstrated a manageable safety profile, an objective response rate (ORR) of 32%, and a disease control rate of 80%. The median duration of response (DoR) was 16.8 months (95% CI: 5.6–not calculable (NC)). Median PFS by blinded independent central review (BICR) was 4.4 months (95% CI: 3.0–7.3). In patients with metastatic TNBC who had not received topoisomerase I inhibitor-based ADCs, the ORR was 40%. 34 The most common treatment-related AEs (TRAEs) were stomatitis (72.7%), nausea (65.9%), and vomiting (38.6%). Rates of any grade and grade ⩾3 gastrointestinal (diarrhea 15.9%, 0%) and hematological TRAEs were low (anemia 15.9%, 2.3%; neutropenia 20.5%, 2.3%). No grade 4 or 5 TEAEs were reported. For adverse events of special interest (AESIs), no cases of interstitial lung disease (ILD)/pneumonitis were reported. The most common ocular surface event was dry eye (n = 7), all of which were grade 1 or 2. In total, 32 patients had stomatitis, all of which were treatment-related. Grade ⩾3 stomatitis was observed in five patients. 34
Animal models have demonstrated that the combination of PD-(L)1 inhibitors and Dato-DXd had additional benefit compared with Dato-DXd monotherapy. 35 Moreover, data from preclinical studies have shown that immunotherapy activity may be increased when combined with ADCs due to immunomodulatory effects in the tumor microenvironment, such as increased PD-L1 expression, dendritic cell activation, and enhanced neoantigen formation. 36 ADCs can also trigger immunogenic cell death and T-cell infiltration, complementing the ability of immune checkpoint inhibitors to enhance the antitumor function of those T-cells. 37 In the phase Ib/II BEGONIA study (NCT03742102), Dato-DXd was investigated in combination with durvalumab (an anti-PD-L1 monoclonal antibody) as first-line treatment in patients with unresectable locally advanced or metastatic TNBC. The confirmed ORR was 79% (95% CI: 67–88), the median DoR was 15.5 months (95% CI: 9.9–NC), and the median PFS was 13.8 months (95% CI: 11–NC). 38 The safety profile of the combination was manageable, with no new safety signals reported. The most common AEs were nausea and stomatitis (each in 65% of patients). 38 Results from a subsequent study, ISPY2.2, have also demonstrated efficacy for the combination of neoadjuvant Dato-DXd and durvalumab in patients with HER2-negative breast cancer subtypes, including TNBC, with no new safety signals identified. 39 Given these data, it is thought that the combination of Dato-DXd and durvalumab may lead to improved clinical responses through complementary recruitment of immune cells to tumor sites and increased activation of immune cells by durvalumab (Figure 1).40–43 Building on these initial results, we describe the design of the ongoing phase III TROPION-Breast05 study (NCT06103864) which is investigating Dato-DXd with or without durvalumab versus the current standard of care (chemotherapy plus pembrolizumab), in the first-line treatment of patients with PD-L1-high locally recurrent inoperable or metastatic TNBC.

Mechanism of action Dato-DXd and durvalumab.
Methods
Study design
TROPION-Breast05 (NCT06103864) is a phase III, randomized, open-label, 3-arm, multicenter, international study assessing the efficacy and safety of Dato-DXd with and without durvalumab compared with investigator’s choice of chemotherapy (ICC; paclitaxel, nab-paclitaxel, or gemcitabine plus carboplatin) in combination with pembrolizumab in patients with PD-L1-high, locally recurrent inoperable, or metastatic TNBC (Figure 2). Patients will be enrolled from the Americas, Europe, Middle East, Africa, and Asia-Pacific regions and will be randomized 1:1 to Arm 1 (Dato-DXd plus durvalumab) or Arm 2 (ICC plus pembrolizumab), or, in selected countries (based on local feasibility and the current treatment landscape), 1:1:1 to Arm 1, Arm 2, and Arm 3 (Dato-DXd monotherapy). Arm 3 will close once approximately 75 patients have been randomized, after which all subsequent patients will be randomized into Arms 1 or 2.
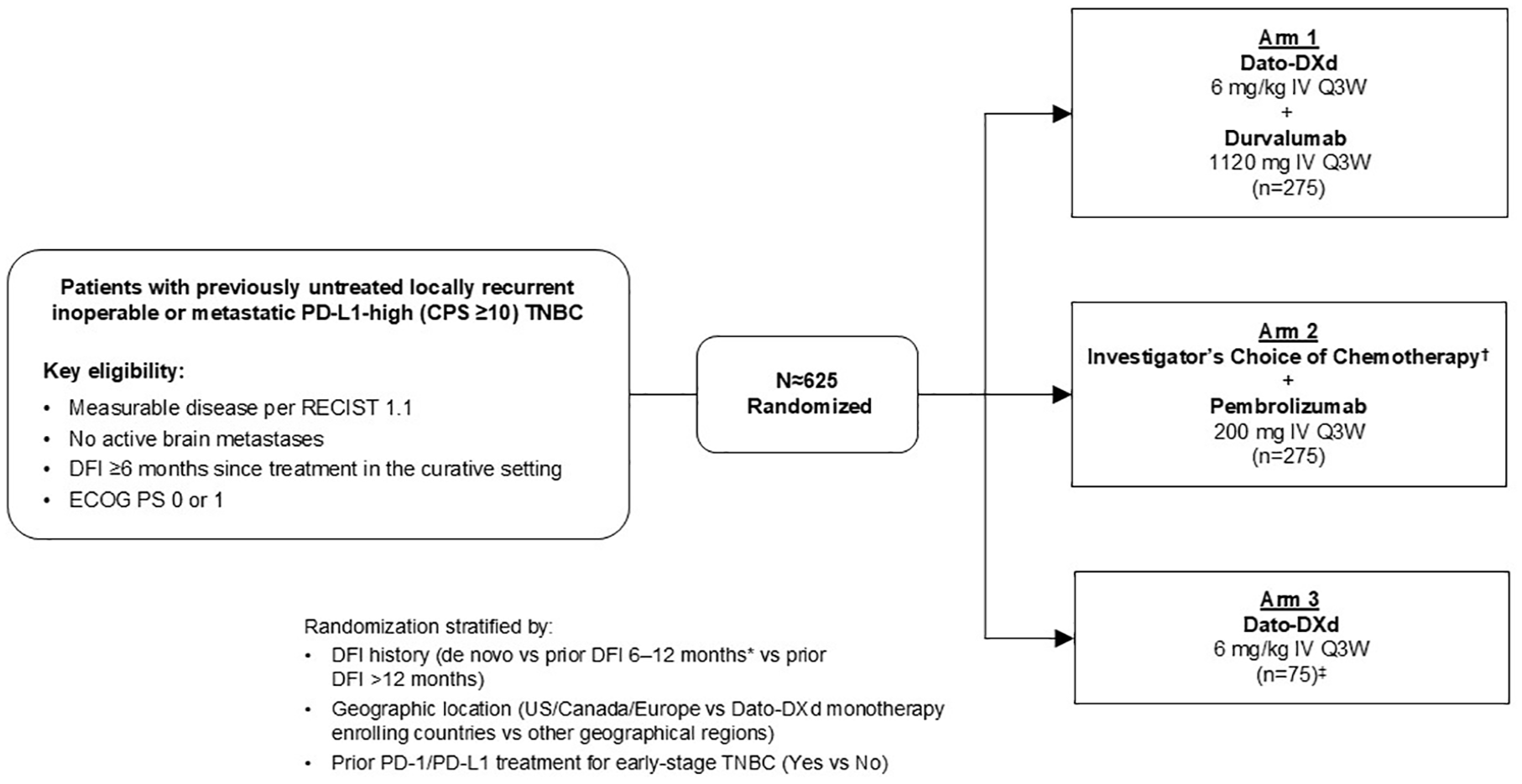
TROPION-Breast05 study design.
Randomization will be stratified based on geographic locations (USA, Canada, Europe vs Dato-DXd monotherapy enrolling countries vs other geographic regions), prior PD-1/PD-L1 treatment for early-stage TNBC (yes vs no), and disease-free interval (DFI, defined as the time between completion of treatment with curative intent (either date of primary breast tumor surgery or date of last dose of systemic anticancer therapy, whichever occurred last) and the first documented local or distant disease recurrence (either by biopsy or imaging)) history (de novo vs prior DFI 6–12 months vs prior DFI > 12 months). A 20% cap will be applied to patients who have a DFI of 6–12 months.
The study is being conducted in accordance with the Declaration of Helsinki, the International Council for Harmonization/Good Clinical Practice, and applicable regulatory requirements. All patients will provide written, informed consent. The reporting of this study conforms to the SPIRIT 2013 statement (Supplemental File). 45
Eligibility criteria
Inclusion and exclusion criteria are presented in Tables 1 and 2, respectively. In summary, eligible patients include adults (aged ⩾ 18 years) with PD-L1-high locally recurrent inoperable or metastatic TNBC and who are eligible to receive pembrolizumab plus one of the ICC regimens. TNBC is defined as a disease that is negative for both ER and PR (<1% of tumor cells positive on immunochemistry (IHC)), and negative for HER2 with 0 or 1+ intensity on IHC or 2+ intensity on IHC and negative in situ hybridization (ISH) per American Society of Clinical Oncology/College of American Pathologists HER2 guidelines.46,47 Patients must have PD-L1-high disease, with a CPS ⩾10 (based on the number of PD-L1 staining cells (tumor cells, lymphocytes, and macrophages) divided by the total number of viable tumor cells multiplied by 100) per the 22C3 PD-L1 assay as assessed by central laboratory testing. Patients must also have Eastern Cooperative Oncology Group performance status (ECOG PS) 0 or 1, measurable disease as per Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1), and adequate bone marrow reserve and organ function. Prior exposure to any treatment containing a chemotherapeutic agent targeting topoisomerase I and TROP2-directed therapy is not permitted. Patients with recurrent disease will be eligible if they have completed treatment for Stage I–III breast cancer and ⩾6 months have elapsed between completion of treatment with curative intent and the first documented recurrence. Patients with active brain metastases are ineligible; however, patients with treated, clinically inactive brain metastases who are no longer symptomatic and require no treatment may be included in the study. A formalin-fixed paraffin-embedded metastatic or locally recurrent inoperable tumor sample must be available. All patients must have a signed informed consent form (ICF) prior to study enrollment and before performing any protocol-specific procedures, including screening/baseline evaluations.
TROPION-Breast05 inclusion criteria.

ASCO/CAP, American Society of Clinical Oncology/College of American Pathologists; CPS, combined positive score; ECOG PS, Eastern Cooperative Oncology Group performance status; ER, estrogen receptor; FFPE, formalin-fixed paraffin-embedded; FISH, fluorescence in situ hybridization; HER2, human epidermal growth factor receptor 2; ICF, informed consent form; IHC, immunohistochemistry; PD-L1, programmed death ligand 1; PR, progesterone receptor; RECIST 1.1, Response Evaluation Criteria in Solid Tumors version 1.1; TNBC, triple-negative breast cancer.
TROPION-Breast05 exclusion criteria.

ADC, antibody-drug conjugate; Dato-DXd, datopotamab deruxtecan; HIV, human immunodeficiency virus; ILD, interstitial lung disease; IV, intravenous; PD-(L)1, programmed cell death (ligand)-1; TROP2, trophoblast cell-surface antigen 2.
Endpoints
Study endpoints are summarized in Table 3. The primary endpoint is PFS (Dato-DXd plus durvalumab vs ICC plus pembrolizumab), defined as the time from the date of randomization until the date of progressive disease (PD; per RECIST 1.1 as assessed by BICR) or death due to any cause. The key secondary endpoint is OS, defined as the time from randomization to death due to any cause. Other secondary endpoints include ORR, DoR, and clinical benefit rate at Week 24 per RECIST 1.1 by BICR and investigator assessment; PFS by investigator assessment; second PFS (defined as the time from randomization to the earliest progression event after first subsequent therapy, or death); time to first subsequent therapy; time to second subsequent therapy; and time to deterioration in breast and arm symptoms, pain, physical functioning, and global health scores. Pharmacokinetics and immunogenicity of Dato-DXd will also be investigated. Safety and tolerability parameters include AEs graded by Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 (v5.0), ECOG PS, vital signs, laboratory parameters, and ophthalmologic assessments.
TROPION-Breast05 endpoints.

PFS is defined as the time from the date of randomization until the date of objective progressive disease per RECIST 1.1 as assessed by BICR, or death.
For Dato-DXd plus durvalumab versus ICC plus pembrolizumab.
ORR is defined as the proportion of patients who have CR or PR, as determined by BICR/investigator assessment, per RECIST 1.1.
Clinical benefit rate at 24 weeks is defined as the percentage of patients who have a CR or PR or have SD, per RECIST 1.1, as assessed by BICR or per investigator assessment and derived from raw tumor data for at least 23 weeks after randomization.
AE, adverse event; BICR, blinded independent central review; CR, complete response; CTCAE, Common Terminology Criteria for Adverse Events; Dato-DXd, datopotamab deruxtecan; DoR, duration of response; ECOG PS, Eastern Cooperative Oncology Group performance status; EORTC, European Organization for Research and Treatment of Cancer; GHS, Global Health Status; ICC, investigator’s choice of chemotherapy; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PFS2, second progression-free survival; PR, partial response; PRO, patient-reported outcome; PROMIS, Patient-Reported Outcomes Measurement Information System; QoL, quality of life; RECIST 1.1, Response Evaluation Criteria in Solid Tumors version 1.1; SD, stable disease; TFST, time to first subsequent therapy; TSST, time to second subsequent therapy; TTD, time to deterioration.
Study procedures and assessments
Patients will enter screening on the day of signing the ICF and randomization will occur within 28 days of giving consent. Patients will receive study treatment until investigator-defined PD according to RECIST 1.1, or until unacceptable tolerability, withdrawal of consent, or another discontinuation criterion is met. Physical examination, ECOG PS, and vital signs will be assessed at the start of each treatment cycle prior to the infusion of study intervention, and for up to 90 days after the last dose. Tumor assessments per RECIST 1.1 will be undertaken using computed tomography (CT) or magnetic resonance imaging scans. Survival assessments will be conducted every 3 months following objective PD or treatment discontinuation until death, withdrawal of consent, or end of study. AEs will be continuously monitored from screening up to 90 days after the last dose of the study drug. Pulmonary function tests and high-resolution CT of the chest will be undertaken if pneumonitis or ILD is suspected, and ophthalmologic assessments will be conducted at screening, as clinically indicated, and at the end of treatment. A daily oral care protocol for stomatitis prophylaxis will be provided to all patients; a steroid-containing mouthwash (dexamethasone, or an alternative per local guidelines) is highly recommended for patients randomized to Dato-DXd or Dato-DXd in combination with durvalumab, and prophylactic cryotherapy (e.g., ice chips or ice water held in the mouth) during Dato-DXd infusion should also be considered.
Statistical methods
All efficacy analyses will be performed on the ITT population, defined as all patients randomized to study intervention. The safety analysis set will comprise all patients, including those in Arm 3, who received any randomized treatment, regardless of group assignment.
Approximately 1950 patients will be screened to enroll and randomize approximately 625 patients with PD-L1-high tumors to study treatment. The study is powered to demonstrate the superiority of Dato-DXd plus durvalumab versus ICC plus pembrolizumab as measured by the primary endpoint of PFS (by BICR) in the ITT population, using two-sided alpha level of 5.0%. If the PFS analysis meets statistical significance, OS will also be formally tested. To preserve the overall type 1 error (familywise error rate) at 5%, a multiple testing procedure will be implemented. No formal statistical comparisons between Arms 1 or 2 and Arm 3 (Dato-DXd monotherapy) are planned. The contribution of components will be evaluated descriptively for safety and efficacy endpoints in the Dato-DXd plus durvalumab arm relative to Dato-DXd monotherapy.
PFS and OS will be assessed using a log-rank test stratified by geographic location, DFI history, and prior PD-1/PD-L1 treatment for early stage TNBC. The HR and associated 95% CI will be estimated using a stratified Cox proportional hazards model, and Kaplan–Meier plots will be presented by the treatment group. Safety data will be summarized using descriptive statistics.
Discussion
Despite recent advances in treatment, such as the addition of immune checkpoint inhibitors to chemotherapy, survival remains poor for patients with advanced or metastatic TNBC, and novel treatment options are needed to improve clinical outcomes. TROP-2-directed ADCs have emerged as a new treatment option in this setting, and the combination of these agents with anti-PD-(L)1 checkpoint inhibitors may drive increased therapeutic benefit by improving long-term disease control while maintaining a manageable safety profile.
The phase III TROPION-Breast05 study, evaluating the efficacy and safety of Dato-DXd with or without durvalumab versus chemotherapy plus pembrolizumab in patients with PD-L1-high locally recurrent inoperable or metastatic TNBC, contributes to the wider field of studies investigating the combination of TROP2-directed ADCs and anti-PD(L)1 antibodies in TNBC. Several phase II trials investigating sacituzumab govitecan in combination with anti-PD-L1 antibodies pembrolizumab, atezolizumab, or avelumab in patients with advanced or metastatic TNBC are underway [ClinicalTrials.gov identifiers: NCT04468061; NCT03424005; NCT03971409], as well as ASCENT-04, an ongoing phase III trial investigating sacituzumab govitecan combined with pembrolizumab in patients with advanced or metastatic TNBC [ClinicalTrials.gov identifier: NCT05382286].
As described above, TROPION-Breast05 expands on the findings from the phase I/IIb BEGONIA study and complements the clinical program investigating Dato-DXd with or without durvalumab in patients with earlier stages of TNBC. For example, TROPION-Breast03 (NCT05629585) is an ongoing phase III study investigating Dato-DXd with or without durvalumab as adjuvant treatment in patients with early stage TNBC. 48 Furthermore, TROPION-Breast04 (NCT06112379) is an ongoing phase III study assessing Dato-DXd with or without durvalumab in the neoadjuvant setting in patients with treatment-naïve early stage TNBC. 49 Enrollment to TROPION-Breast05 started in November 2023 and the study is ongoing.
Supplemental Material
sj-doc-1-tam-10.1177_17588359251327992 – Supplemental material for TROPION-Breast05: a randomized phase III study of Dato-DXd with or without durvalumab versus chemotherapy plus pembrolizumab in patients with PD-L1-high locally recurrent inoperable or metastatic triple-negative breast cancer
Supplemental material, sj-doc-1-tam-10.1177_17588359251327992 for TROPION-Breast05: a randomized phase III study of Dato-DXd with or without durvalumab versus chemotherapy plus pembrolizumab in patients with PD-L1-high locally recurrent inoperable or metastatic triple-negative breast cancer by Peter Schmid, Mafalda Oliveira, Joyce O’Shaughnessy, Massimo Cristofanilli, Stephanie L. Graff, Seock-Ah Im, Sherene Loi, Shigehira Saji, Shusen Wang, David W. Cescon, Tina Hovey, Agata Nawrot, Karson Tse, Petra Vukovic and Giuseppe Curigliano in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Medical writing support for the development and submission of this manuscript, under the direction of the authors, was provided by Catherine Crookes, BSc. of Ashfield MedComms (Macclesfield, UK), an Inizio company, and was funded by AstraZeneca.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
