Abstract
Background:
Despite advances in the treatment of early triple-negative breast cancer (TNBC), patients with residual invasive disease after neoadjuvant therapy have a high risk of disease recurrence and worse survival outcomes than those who have pathological complete response (pCR). Improving outcomes in early TNBC remains an unmet need requiring new adjuvant treatment approaches. Datopotamab deruxtecan (Dato-DXd) is an antibody–drug conjugate comprising a humanized anti-trophoblast cell-surface antigen 2 immunoglobulin G1 (IgG1) monoclonal antibody attached via a plasma-stable, cleavable linker to a potent topoisomerase I inhibitor payload, with activity observed in advanced TNBC.
Objectives:
TROPION-Breast03 is an ongoing phase III study evaluating the efficacy and safety of Dato-DXd alone or combined with durvalumab versus standard-of-care therapy as adjuvant treatment in patients with stage I–III TNBC with residual invasive disease at surgical resection following neoadjuvant treatment.
Methods and design:
Eligible patients, aged ⩾18 years, will be randomized in a 2:1:2 ratio to receive Dato-DXd [6 mg/kg intravenously (IV) every 3 weeks (Q3W); eight cycles] and durvalumab (1120 mg IV Q3W; nine cycles), Dato-DXd monotherapy (6 mg/kg IV Q3W), or investigator’s choice of therapy (ICT; capecitabine, pembrolizumab, or capecitabine and pembrolizumab). The primary endpoint is invasive disease-free survival (iDFS) for Dato-DXd and durvalumab versus ICT. Key secondary endpoints include safety, distant disease-free survival, and overall survival for Dato-DXd and durvalumab versus ICT and iDFS for Dato-DXd monotherapy versus ICT.
Ethics:
TROPION-Breast03 will be approved by the independent ethics committees or institutional review boards at each study site. All study participants will provide written informed consent.
Discussion:
TROPION-Breast03 will help define the potential role of Dato-DXd in the treatment of patients with early-stage TNBC who do not have pCR after neoadjuvant therapy.
Trial registration:
ClinicalTrials.gov identifier: NCT05629585 (registration date: 29 November 2022).
Plain language summary
Triple-negative breast cancer (TNBC), in which cells do not have estrogen or progesterone receptors or high levels of human epidermal growth factor receptor 2, is the most aggressive breast cancer subtype. TNBC is difficult to treat and associated with high risk of recurrence despite standard systemic therapy (treatment targeting the entire body), which can include chemotherapy alone or in combination with immunotherapy (treatment targeting the immune system). To reduce the risk of recurrence, standard systemic treatment is often followed by surgical removal of the patient’s tumors and additional systemic treatment. Dato-DXd is an antibody-drug conjugate, which is an anticancer drug (DXd) connected to an antibody (datopotamab) by a stable linker. Datopotamab binds to TROP2, a protein found on breast cancer cells, and is taken into the tumor cell where the linker breaks, releasing DXd to kill the cell. By delivering DXd directly to cancer cells, Dato-DXd reduces exposure in the rest of the body, reducing the risk of side effects. Since Dato-DXd can recruit immune cells to cancer sites, it may work better combined with durvalumab, a drug that blocks the activity of a protein called PD-L1, making cancer cells more susceptible to being killed by immune cells. The TROPION-Breast03 study will compare Dato-DXd, alone or combined with durvalumab, with standard-of-care therapy in patients with TNBC that has not spread to parts of the body away from the original tumor site(s), but with cancer cells remaining at the time of surgery after initial systemic therapy. It will assess how well each treatment works and describe any side effects. We plan to recruit 1,075 eligible adults who will be randomly assigned in a 2:1:2 ratio to: • Dato-DXd + durvalumab • Dato-DXd alone • Standard-of-care therapy • Patients will receive treatment until they complete the planned course of therapy (8 or 9 cycles), their cancer returns, side effects become unacceptable, or they choose to stop.
Keywords
Introduction
Breast cancer has the highest incidence of any cancer worldwide, accounting for significant global morbidity and mortality; it was diagnosed in almost 2.3 million people and accounted for approximately 685,000 deaths in 2020. 1 Triple-negative breast cancer (TNBC), which is characterized by the absence of the estrogen receptor (ER) and progesterone receptor (PR) and lack of amplification/overexpression of human epidermal growth factor receptor 2 (HER2), accounts for approximately 15% of breast cancer diagnoses and is the most aggressive subtype. 2 While most patients with TNBC present without overt metastatic disease, approximately 30% of patients with early-stage disease will relapse with distant metastasis, with a median time to distant metastasis of approximately 2 years from diagnosis.2–6 Once patients develop advanced or metastatic TNBC, median overall survival with recommended therapies ranges from 8.1 to 24.2 months and the rate of survival at 5 years is approximately 13%.7,8
TNBC is challenging to treat due to a relative lack of actionable molecular targets such as ER, PR, and HER2.9–11 Standard recommended treatment for early-stage TNBC includes neoadjuvant chemotherapy with an anthracycline- and taxane-containing regimen followed by surgical resection and pathologic response guided adjuvant therapy.12–14 Patients with TNBC who have pathologic complete response (pCR), defined as lack of invasive and in situ residual cancer cells in breast tissue and lymph nodes, have improved event-free survival [EFS; hazard ratio (HR), 0.18] and overall survival (OS; HR, 0.20) compared with those with residual disease; 15 however, pCR has been reported in only 22–51% of patients with TNBC treated with neoadjuvant chemotherapy alone.16–18 Patients with TNBC with residual disease have 5- to 10-year recurrence rates ranging from 40% to 60%.19–21 In this setting, guidelines recommend adjuvant therapy, as it has been demonstrated to reduce the risk of recurrence and distant metastasis.12,22 The ECOG-ACRIN EA 1131 study showed that in patients with stage II or III TNBC and residual disease after neoadjuvant chemotherapy, adjuvant capecitabine was associated with improved outcomes compared with observation, with 3-year OS of 69.4%, recurrence-free survival of 53.4%, and invasive disease-free survival (iDFS) of 53.5%. 23 Furthermore, the CREATE-X study showed that in patients with stage I–IIIB TNBC and residual disease after neoadjuvant chemotherapy, adjuvant capecitabine improved 5-year disease-free survival (69.8% versus 56.1%) and overall survival (78.8% versus 70.3%) compared with no further systemic treatment. 24
Several clinical trials have shown that the addition of immunotherapy to chemotherapy regimens for early-stage TNBC is associated with improvement in both pCR and prognosis. 25 Many immunotherapies block the interaction of programmed cell death-1 (PD-1) with its ligand (PD-L1), reducing the ability of cancer cells to avoid anti-cancer immune responses. 26 Pembrolizumab, an immunotherapy targeting PD-1, is one such agent approved in early-stage TNBC. 27 Based on the results of the KEYNOTE-522 trial, the approved regimen comprises neoadjuvant therapy with both pembrolizumab and chemotherapy prior to surgery followed by adjuvant pembrolizumab monotherapy.27,28 This regimen is now the guideline-recommended treatment for high-risk early-stage TNBC. 29 Compared with neoadjuvant chemotherapy and surgery without adjuvant treatment, the addition of neoadjuvant/adjuvant pembrolizumab improved pCR rate from 56.2% to 63.4% and improved EFS at 3 years from 76.8% to 84.5% among patients with stage II or III TNBC.28,30 Despite these improvements, distant metastatic recurrence rates ranged from 8.7% to 35.0% among patients with residual disease depending on the extent of the disease. 30 Thus, new therapeutic options are still needed to improve outcomes in patients with early-stage TNBC who have residual disease after neoadjuvant therapy even when pembrolizumab is included in the regimen.
Datopotamab deruxtecan (Dato-DXd) is a trophoblast cell-surface antigen 2 (TROP2)-directed antibody–drug conjugate (ADC) composed of a humanized anti-TROP2 immunoglobulin G1 monoclonal antibody attached to a highly potent topoisomerase I inhibitor payload (an exatecan derivative, DXd) via a plasma-stable, tumor-selective, cleavable linker. 31 ADCs are a class of anticancer agents that selectively deliver a cytotoxic payload to tumor cells, thereby minimizing systemic toxicity, and benefit from a robust local bystander effect with payloads like DXd that have high membrane permeability.32–35 TROP2 is a type one transmembrane glycoprotein expressed on several normal tissues 36 ; notably, it is of clinical interest as it is broadly expressed in several solid tumors, including TNBC, and has been associated with poor prognosis.37–39 Another TROP2-directed ADC, sacituzumab govitecan, has been approved as second-line and beyond therapy in advanced/metastatic TNBC based on the results of the phase III ASCENT trial,40,41 and is under evaluation as adjuvant therapy in the phase III SASCIA trial in patients with primary HER2-negative breast cancer and residual disease after standard neoadjuvant treatment.42,43
Preclinical evidence indicates that topoisomerase I inhibitors like DXd can enhance antitumor immunity by recruiting immune cells.44–46 Given that many solid tumors, including TNBC, are known to evade the immune system, 47 combination therapy with both Dato-DXd and an immune cell activating agent may be more effective than either therapeutic agent alone. The immune checkpoint inhibitor durvalumab is a human monoclonal antibody of the immunoglobulin G1 kappa subclass that blocks the interaction of PD-L1, but not programmed cell death ligand-2, with PD-1 on T cells and CD80 on immune cells. 48 Blockade of the interaction of PD-L1 with PD-1 and CD80 releases the inhibition of immune responses, including those that may result in tumor elimination. 48 Thus, the combination of Dato-DXd and durvalumab may lead to improved clinical responses through complementary pharmacodynamic effects: Dato-DXd increases the recruitment of immune cells to tumor sites, and durvalumab increases the activation of immune cells.44–46,48
Dato-DXd monotherapy has shown encouraging preliminary safety and efficacy in a variety of advanced solid tumors in TROPION-PanTumor01 (NCT03401385), an ongoing phase I first-in-human study. 49 In a subset of patients with advanced/metastatic TNBC who had not received prior topoisomerase I inhibitor treatment, Dato-DXd resulted in a confirmed overall response rate of 44% and a manageable safety profile. 50 Dato-DXd has also been investigated in combination with durvalumab in the phase Ib/II BEGONIA study (NCT03742102). 51 Preliminary data from BEGONIA suggest that the combination has clinical activity in TNBC, with an objective response rate of 79%, median duration of response of 15.5 months, and median progression-free survival of 13.8 months as first-line therapy for locally advanced or metastatic TNBC.51,52 In addition to results from TROPION-PanTumor01 and BEGONIA, the ongoing TROPION-Breast02 trial (NCT05374512) is evaluating Dato-DXd as first-line monotherapy in advanced/metastatic TNBC 53 ; however, Dato-DXd has yet to be evaluated as adjuvant treatment in TNBC. Evaluation of Dato-DXd with or without durvalumab in early-stage TNBC is warranted given the unmet need to prevent relapse and disease progression.
Here we describe the design of TROPION-Breast03, which is investigating whether Dato-DXd, alone or in combination with durvalumab, has clinical benefit as adjuvant treatment in patients with stage I–III TNBC who do not have pCR at the time of surgery following neoadjuvant therapy.
Methods
Study design
TROPION-Breast03 (NCT05629585) is an ongoing phase III, global, multicenter, randomized, open-label, active-controlled, three-arm study (Figure 1) investigating the efficacy and safety of Dato-DXd with or without durvalumab compared with investigator’s choice of treatment (ICT; capecitabine and/or pembrolizumab) as adjuvant therapy in patients with stage I–III TNBC who have residual invasive disease at the time of surgery after neoadjuvant therapy. Eligible patients enrolled from approximately 300 sites in 16 countries across Asia, Europe, and the Americas will be randomized 2:1:2 among three study arms: combination therapy with Dato-DXd and durvalumab (Arm 1), Dato-DXd monotherapy (Arm 2), or ICT (Arm 3). Randomization will be stratified based on receipt of prior neoadjuvant pembrolizumab, receipt of prior neoadjuvant platinum chemotherapy, and extent of residual disease at the time of surgery (<1 cm in the absence of lymph node involvement versus ⩾1 cm or any lymph node involvement). A recruitment cap of 40% will be applied to patients who have not received neoadjuvant pembrolizumab, and a cap of 20% will be applied to patients who have residual disease <1 cm in the absence of lymph node involvement. The treatment assigned to patients will be determined by a randomization scheme administered via an Interactive Response Technology/Randomization and Trial Supply Management system. The randomization scheme will be software-generated and comprise a blocked randomization list for each stratum, with randomization codes assigned strictly sequentially.
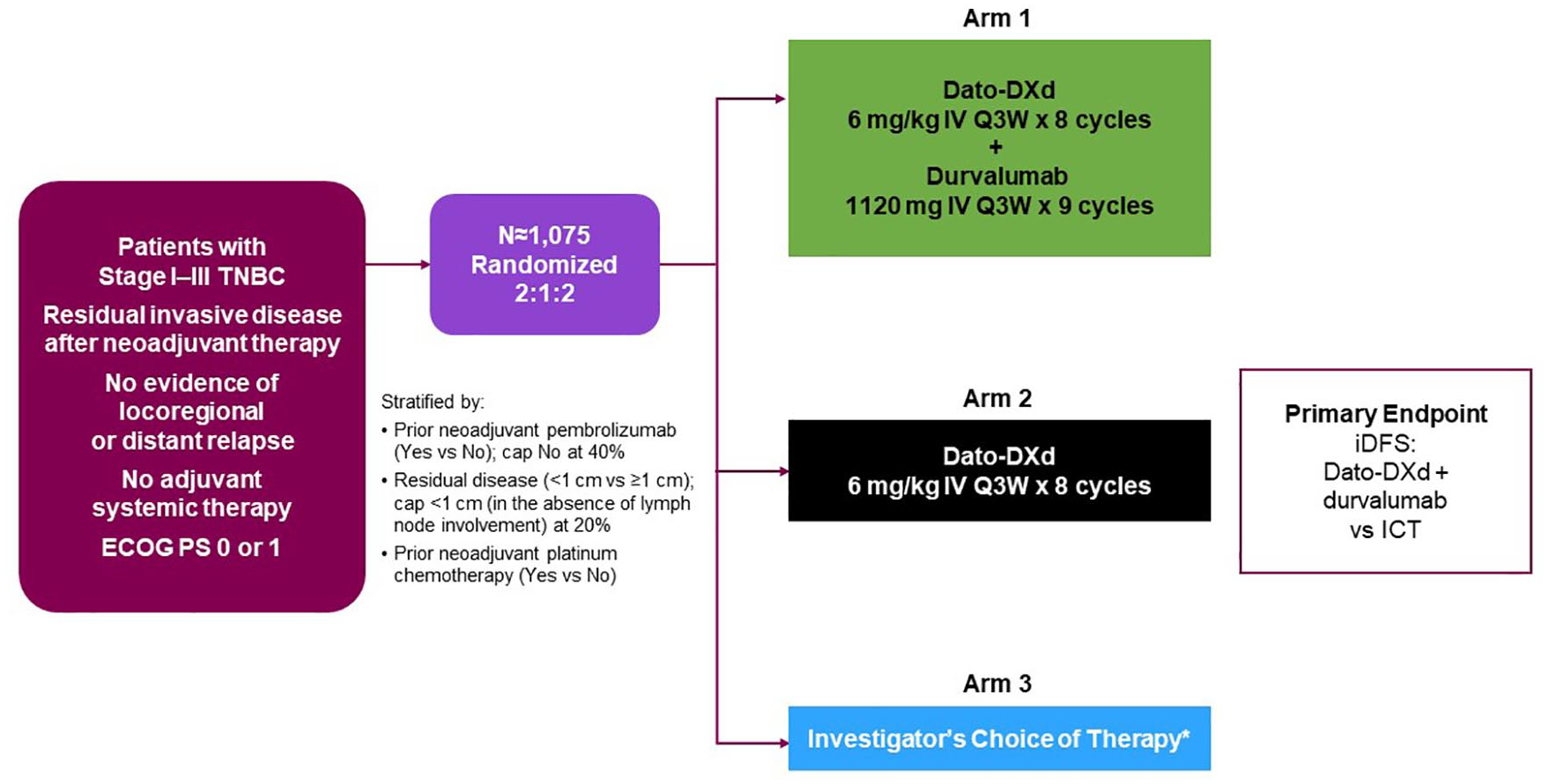
TROPION-Breast03 study design.
Eligibility criteria
Key inclusion and exclusion criteria are presented in Tables 1 and 2. Eligible patients include adults aged ⩾18 years with stage I–III TNBC with residual invasive disease in the breast and/or axillary lymph nodes at surgical resection following neoadjuvant therapy. Diagnosis of TNBC will be based on local laboratory results and defined according to the American Society of Clinical Oncology-College of American Pathologists guidelines:54,55 ER expression of <1% of tumor cells by immunohistochemistry (IHC), PR expression of <1% of tumor cells by IHC, and HER2 negative (IHC 0/1+, or 2+ intensity and in situ hybridization non-amplified). Eligible patients must have an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1; 56 adequate organ function; completed at least six cycles of neoadjuvant therapy containing an anthracycline and/or a taxane, with or without carboplatin, with or without pembrolizumab; and undergone surgical removal of all clinically evident disease in the breast and lymph nodes with no evidence of locoregional or distant relapse; radiological scans before treatment are not required and should be obtained as per local institutional practice. If radiotherapy is indicated, it must be completed at least 2 weeks prior to randomization. Eligible patients must not have received any adjuvant systemic therapy, have no known germline breast cancer gene 1/2 (BRCA1/2) mutations, and must be eligible for at least one of the ICT treatment options. A formalin-fixed paraffin-embedded tumor sample from residual invasive disease at the time of surgical resection must be available.
Key inclusion criteria.

ASCO/CAP, American Society of Clinical Oncology/College of American Pathologists; BRCA1/2, breast cancer gene 1/2; ECHO, echocardiogram; ECOG PS, Eastern Cooperative Oncology Group performance status; ER, estrogen receptor; FFPE, formalin-fixed paraffin-embedded; HER2, human epidermal growth factor receptor 2; IHC, immunohistochemistry; LVEF, left ventricular ejection fraction; MUGA, multigated acquisition; PR, progesterone receptor; TNBC, triple-negative breast cancer.
Key exclusion criteria.

AE, adverse event; CT, computed tomography; Dato-DXd, datopotamab deruxtecan; ILD, interstitial lung disease; PD-(L)1, programmed cell death (ligand)-1; QTcF, corrected QT interval by Fredericia’s formula; T-DXd, trastuzumab deruxtecan; TNBC, triple-negative breast cancer; TROP2, trophoblast cell-surface antigen 2.
Endpoints
The study endpoints are summarized in Tables 3 and 4.
Primary and secondary endpoints.

iDFS is defined as the time from randomization until the date of the first occurrence of one of the following events: ipsilateral invasive breast tumor (local) recurrence, regional invasive breast cancer recurrence (axilla, regional lymph nodes, chest wall, and skin of ipsilateral breast), or distant recurrence (metastatic breast cancer that has either been biopsy-confirmed or clinically diagnosed as recurrent invasive breast cancer); contralateral invasive breast cancer; second primary non-breast invasive cancer (other than squamous or basal cell skin cancer); or death from any cause.
DDFS is defined as the time from randomization to the date of the first distant recurrence, occurrence of second primary non-breast invasive cancer (other than squamous or basal cell skin cancer), or death from any cause.
AE, adverse event; CTCAE, common terminology criteria for adverse events; Dato-DXd, datopotamab deruxtecan; DDFS, distant disease-free survival; ECOG PS, Eastern Cooperative Oncology Group performance status; EORTC, European Organisation for Research and Treatment of Cancer; GHS, global health status; HIV, human immunodeficiency virus; ICT, investigator’s choice of therapy; iDFS, invasive disease-free survival; OS, overall survival; PROMIS, Patient-Reported Outcomes Measurement Information System; QoL, quality of life; TTD, time to deterioration.
Exploratory endpoints.

IBCSF is defined as time from randomization until the date of the first occurrence of one of the following: ipsilateral invasive breast tumor (local) recurrence, regional invasive breast cancer recurrence (axilla, regional lymph nodes, chest wall, and skin of ipsilateral breast), or distant recurrence (metastatic breast cancer that has either been biopsy-confirmed or clinically diagnosed as recurrent invasive breast cancer); contralateral invasive breast cancer; or death from any cause.
ctDNA, circulating tumor DNA; Dato-DXd, datopotamab deruxtecan; IBCFS, invasive breast cancer free survival; ICT, investigator’s choice of therapy; PD-L1, programmed cell death ligand-1; TROP2, trophoblast cell-surface antigen 2.
Primary and key secondary endpoints
The primary endpoint is invasive disease-free survival (iDFS) for treatment Arm 1 (Dato-DXd + durvalumab) versus Arm 3 (ICT). iDFS is defined as time from randomization until the date of the first occurrence of one of the following events: ipsilateral invasive breast tumor (local) recurrence, regional invasive breast cancer recurrence (axilla, regional lymph nodes, chest wall, and skin of ipsilateral breast), or distant recurrence (metastatic breast cancer that has either been biopsy-confirmed or clinically diagnosed as recurrent invasive breast cancer); contralateral invasive breast cancer; second primary non-breast invasive cancer (other than squamous or basal cell skin cancer); or death from any cause.
Key secondary endpoints include distant disease-free survival (DDFS) and OS for Arm 1 versus Arm 3 and iDFS for Arm 2 (Dato-DXd monotherapy) versus Arm 3. DDFS is defined as the time from randomization to the date of the first distant recurrence, occurrence of second primary non-breast invasive cancer (other than squamous or basal cell skin cancer), or death from any cause. Note that DDFS excludes potentially non-lethal events including ipsilateral invasive breast tumor recurrence, regional invasive breast cancer recurrence, contralateral invasive breast cancer, and all in situ carcinomas. OS is defined as the time from randomization until the date of death due to any cause.
Additional secondary endpoints
Additional secondary endpoints include DDFS and OS for Arm 2 versus Arm 3; and iDFS and DDFS for Arm 1 versus Arm 2, as well as pharmacokinetics and immunogenicity of Dato-DXd. Patient-reported outcomes are also being investigated, including time to deterioration (TTD; defined as the time from the date of randomization to the date of deterioration) in physical function assessed by the Patient-Reported Outcomes Measurement Information System (PROMIS) Physical Function Short Form 8c;57,58 TTD in Global Health State (GHS)/Quality of Life (QoL) measured by the European Organisation for Research and Treatment of Cancer (EORTC) IL172 scale, which comprises 12 items for the GHS/QoL and role, emotional, cognitive, and social function scales from the EORTC QLQ-C30; 59 and fatigue measured via the PROMIS Fatigue Short Form 7a.60,61 Safety and tolerability of Dato-DXd with or without durvalumab relative to ICT will be assessed in terms of adverse events (AEs) graded by Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 (v5.0), 62 ECOG PS, clinical examination, and laboratory assessment.
Study procedures and assessments
Patients will receive treatment until they complete the planned course of therapy, their cancer returns, side effects become unacceptable, or they choose to stop. Disease recurrence will be determined per investigator assessment. Efficacy assessments, including medical history and physical examination, will be performed every 3 months during the first 2 years, every 6 months during years 3 through 5, and annually thereafter. Bilateral mammogram or magnetic resonance imaging will be performed annually timed from the last assessment prior to randomization, with additional imaging assessments for recurrent or metastatic disease at the discretion of the treating physician based on the local standard of care. If no breast tissue remains in patients who underwent bilateral mastectomy, a physical examination is sufficient, and imaging will not be required. Patients will be regularly assessed for at least 5 years of follow-up regardless of discontinuation of study treatment or initiation of subsequent anti-cancer therapy. Following an iDFS event, patients will be followed for survival until at least 5 years after the last participant is randomized.
Continuous assessment for safety and tolerability will begin at screening and continue up to 90 days after the last dose of the study drug. An independent data monitoring committee comprised of independent experts will review safety data and make recommendations to continue, amend, or stop the study based on safety findings. AEs will be graded by CTCAE v5.0 and toxicities treated according to Dato-DXd and durvalumab toxicity management guidelines provided to each study site. Collected information will include ECOG PS, vital signs, body weight, and physical examinations; laboratory assessments, ophthalmologic assessments, and cardiac testing will also be performed. If new or worsening pulmonary symptoms or radiological abnormality suggestive of interstitial lung disease (ILD) or pneumonitis is observed, Dato-DXd will be delayed, and a full investigation will be required. A daily oral care protocol for stomatitis prophylaxis will be given to all patients; steroid-containing mouthwash is highly recommended for participants randomized to Dato-DXd or Dato-DXd in combination with durvalumab, and prophylactic cryotherapy (e.g. ice chips or ice water help in the mouth) during Dato-DXd infusion should also be considered. For nausea prevention, prophylactic anti-emetic agents are recommended prior to Dato-DXd infusion and on subsequent days as needed.
Patient-reported outcomes will be recorded every week from Cycle 1, Day 1 for the first 12 weeks, then every 3 weeks thereafter until the end of treatment. Blood samples will be collected at various time points throughout the study and be used for pharmacokinetic, immunogenicity, and biomarker analysis.
Statistical methods
An intention-to-treat (ITT) population comprising all patients who are randomized to study intervention, regardless of treatment actually received, will be used for all efficacy analyses. The safety analysis set will include all patients who received any amount of randomized treatment.
Approximately 1400 participants will be screened/enrolled to achieve approximately 1075 participants randomized to study treatment. The study is powered to demonstrate the superiority of combination therapy with Dato-DXd and durvalumab (Arm 1) versus ICT (Arm 3) as measured by the primary endpoint of iDFS in the ITT population (Figure 1) at 2-sided 5% alpha level.
iDFS, DDFS, and OS will be assessed using a log-rank test adjusting for stratification factors at randomization. A stratified Cox proportional hazards model will be used to estimate the HR and associated 95% confidence intervals for each endpoint, and Kaplan–Meier plots will be presented by the treatment group. Safety data will be summarized descriptively.
Discussion
Patients with early TNBC who do not have pCR at the time of surgery following neoadjuvant therapy have a substantial risk of recurrence/progression and limited treatment options; TROP2-directed ADCs may represent an opportunity to expand treatment options in this setting. The phase III TROPION-Breast03 trial is evaluating the efficacy and safety of Dato-DXd with and without durvalumab as adjuvant treatment in patients with early-stage TNBC with the residual invasive disease at the time of surgery after neoadjuvant therapy. Enrollment began in November 2022.
TROPION-Breast03 complements the ongoing TROPION-Breast02 and BEGONIA studies which are evaluating Dato-DXd in patients with advanced/metastatic TNBC. While TROPION-Breast02 and BEGONIA will define the role of Dato-DXd, with or without durvalumab, in advanced/metastatic TNBC, the results of TROPION-Breast03 will help define the potential role of Dato-DXd, with or without durvalumab, in the treatment of patients with early-stage TNBC who do not have pCR after neoadjuvant therapy.
Footnotes
Acknowledgements
SWOG Cancer Research Network Clinical Trials Partnerships (SQOG CTP) is the lead academic group for the trial and was a key collaborator in its protocol development. Medical writing support for the development and submission of this manuscript, under the direction of the authors, was provided by Eric Exner of Ashfield MedComms (Milwaukee, WI, USA), an Inizio company, and was funded by AstraZeneca.
