Abstract
The introduction of the cyclin-dependent kinases 4 and 6 (CDK4/6) inhibitors abemaciclib and ribociclib to the adjuvant setting marks a significant advancement in the treatment of hormone-receptor-positive, human epidermal growth factor receptor 2-negative early breast cancer (HR+, HER2− EBC). Despite significant strides in early detection and treatment, many patients continue to face the risk of disease recurrence, highlighting the need for more effective adjuvant therapies. These CDK4/6 inhibitors, combined with adjuvant endocrine therapy, have shown promising efficacy in reducing recurrence rates while maintaining a manageable safety profile, as evidenced by the monarchE and NATALEE trials. This paper explores the integration of adjuvant CDK4/6 inhibitors into clinical practice, focusing on disease-free survival and safety outcomes. Key considerations in selecting between abemaciclib and ribociclib are discussed, including patient risk profiles, efficacy and safety profiles, treatment duration, and individual patient preferences. In addition, we discuss managing adverse events to prevent premature discontinuation, with strategies that include dose holds, dose reductions, proactive symptom management, and patient education. The paper also highlights strategies to enhance patient medication adherence and the involvement of multidisciplinary care teams to support treatment delivery. As research continues to evolve, additional follow-ups of the monarchE and NATALEE trials and future trials will further refine patient selection and treatment sequencing, ultimately improving outcomes and enhancing the quality of life for patients with HR+, HER2− EBC.
Plain language summary
Early breast cancer that is hormone-receptor-positive and human epidermal growth factor receptor 2 -negative (HR+, HER2− EBC) can be challenging to treat due to the risk of cancer returning after completing treatments such as surgery, chemotherapy, and anti-estrogen therapy. New treatments, called CDK4/6 inhibitors, such as abemaciclib and ribociclib, offer hope in reducing this risk. These treatments work by interfering with cancer cell growth and have shown promising results in recent studies. This expert review explains how these treatments are being used to help reduce the risk of cancer coming back. Choosing between abemaciclib and ribociclib depends on individual patient needs and preferences, treatment duration, and potential side effects. Managing these side effects is important to ensure patients continue their treatment without interruption. Healthcare teams, including doctors, nurses, and pharmacists, play a crucial role in supporting patients through education and symptom management. As research progresses, these treatments are expected to improve how early breast cancer is managed, leading to better outcomes and quality of life for patients.
Introduction
Breast cancer is the most frequently diagnosed cancer and the leading cause of cancer death among women, representing nearly one in four cancer cases and one in six cancer deaths among women globally. 1 According to global estimates, there were 2.3 million new breast cancer cases in 2022, accounting for almost 12% of all newly diagnosed cancers, with 666,000 resulting deaths. 1 Breast cancer is a heterogeneous disease and treatment concepts have evolved over the last two decades to account for underlying differences in disease biology. Several classifications have been developed to group tumors accordingly. Luminal-like tumors, which are hormone receptor-positive (HR+) and human epidermal growth factor receptor-2-negative (HER2−), account for the majority (~70%) of all breast cancers.2,3 Most women with HR+, HER2− tumors will present with early-stage breast cancer (EBC) and receive multimodal treatment that includes locoregional and systemic therapies. 4
The mainstay of systemic therapy for luminal EBC is 5–10 years of adjuvant endocrine therapy (ET) with or without neoadjuvant (before surgery), or adjuvant (after surgery) chemotherapy. The use of chemotherapy generally reduces the risk of recurrence by about 30% in selected patients.5–7 Adjuvant ET significantly reduces the risk of locoregional recurrence, distant metastatic recurrence, and contralateral breast cancer while improving overall survival (OS).8,9 Tamoxifen is generally indicated for premenopausal women with a low risk of recurrence or those intolerant to an aromatase inhibitor (AI). For premenopausal women with higher-risk disease (see Section “Who is at risk of recurrence?” for discussion about higher-risk definition), ovarian function suppression (OFS) paired with an AI or tamoxifen is preferred to further reduce recurrences and improve survival.10,11 In postmenopausal women, AI therapy provides a lower risk of recurrence compared to tamoxifen alone, particularly in patients with higher-risk disease. 9 Specifically, 5 years of treatment with an AI lowers the 10-year breast cancer mortality rate by ~15% compared to 5 years of tamoxifen, and by nearly 40% compared to no endocrine treatment. 8
What are the unmet needs in the adjuvant setting?
While systemic therapies are highly effective, many women have a persistent risk of distant recurrence and death from the disease despite adjuvant therapy. 12 Unlike many solid tumors, luminal breast cancer is characterized by a persistent risk of recurrence that extends over decades, with nearly 50% of recurrences occurring more than 5 years after diagnosis.13,14
The risk of distant recurrences has been shown to persist for an extended period following adjuvant ET. Data from the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) database showed that even after 5 years of adjuvant ET, patients experience distant recurrences across 20 years of follow-up. 12 The risk of distant recurrence was strongly correlated with the original anatomic stage, with 20-year distant recurrence risks ranging from 10% to 57%. For instance, stage II patients with T2N0 disease (tumor diameter ⩽2.0 to 5.0 cm and no lymph node involvement) had a 20-year recurrence risk of 29%. This risk rose to 57% with the involvement of four to nine lymph nodes.
In a real-world cohort including 3133 patients, those with stage II EBC had a 10-year risk of invasive disease recurrence of 41%. Notably, in the node-negative subgroup, the 10-year risk of recurrence also remained high at 36%. 15 The risk of recurrence has also been confirmed in a more recent cohort of patients treated with adjuvant ET. Data from the control arms of trials investigating the addition of cyclin-dependent kinases 4 and 6 (CDK4/6) inhibitors to ET among a diverse population of patients with stage II and III EBC indicated 3-year invasive disease recurrence rates ranging from 11% to 22% with adjuvant ET alone.16–19
The role of adjuvant ET extended for up to 10 years has been investigated in several trials and data suggest that continuation of ET has a modest reduction in the risk of recurrence in patients at higher risk.20–23 Despite this benefit, no additional survival benefit was noted so far, and extended therapy was associated with a higher incidence of bone pain, bone fractures, and new-onset osteoporosis. 24 Nevertheless, the risk of recurrence persists even with extended ET. Therefore, it is essential to identify patients at risk of recurrence to make informed treatment recommendations that balance potential benefits against toxicity and the impact on quality of life.
Who is at risk of recurrence?
Tumor size and nodal status are well-established prognostic factors in breast cancer. 12 However, the risk of recurrence is not evenly distributed across anatomic stages, and several other prognostic factors are known. These include a high-risk genomic assay result, a high histologic grade, lymphovascular invasion, elevation of the proliferation marker Ki-67, low or lack of expression of estrogen receptor and progesterone receptor, overexpression of HER2, premenopausal status, or younger age, as well as the presence of residual disease following neoadjuvant therapy.12,14,25,26 Prior therapies or refusal thereof will also help define a patient’s risk of recurrence. Several tools exist that integrate clinicopathological and molecular information to estimate the risk of recurrence and quantify the potential benefit of different adjuvant therapies. However, these tools can yield discordant risk predictions at the individual patient level. For instance, in a direct comparison of several genomic assays, fewer than 40% of tumors were classified similarly by all tests. 27 While the tools may produce inconsistent results, they can help physicians and patients contextualize therapeutic options. For this paper “higher-risk disease” is defined as in the clinical trials with adjuvant CDK4/6 inhibitors.16–19
What is the role of CDK4/6 inhibitors in EBC?
CDK4/6 inhibitors have been practice-changing for the treatment of advanced breast cancer (ABC). Three CDK4/6 inhibitors—palbociclib, ribociclib, and abemaciclib—are approved and are now considered the standard of care in combination with ET for the first-line (and second-line) treatment of HR+, HER2− ABC.28,29 The success of the CDK4/6 inhibitors in ABC prompted their evaluation for the adjuvant treatment of EBC. This review examines the evidence of adjuvant CDK4/6 inhibitors in the EBC setting and their clinical applicability as a treatment escalation option for patients at higher risk of recurrence. We will also share some insights into our practices and provide best practice recommendations where applicable.
Treatment efficacy and safety
Which trials investigated the use of adjuvant CDK4/6 inhibitor therapy?
To date, four major trials—PALLAS, 16 PENELOPE-B, 17 monarchE,18,30 and NATALEE19,31—have explored the role of CDK4/6 inhibitor therapy in the adjuvant EBC setting (Table 1).
Trials for CDK4/6 inhibitors in HR+, HER2− EBC.

ABCSG/AFT, Austrian Breast & Colorectal Cancer Study Group/Alliance Foundation Trials; ALN, axillary lymph node; BID, twice daily; CDK4/6, cyclin-dependent kinase 4/6; CPS-EG, Clinical-Pathologic Scoring System; CT, chemotherapy; DDFS, distant disease-free survival; DRFS, distant relapse-free survival; Dx, diagnosis; EBC, early breast cancer; ET, endocrine therapy; GBG, Global Benefits Group; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; iDFS, invasive disease-free survival; LN, lymph node; mos, months; N0, node-negative; NACT, neoadjuvant chemotherapy; NCT, National Clinical Trial; NSABP, National Surgical Adjuvant Breast and Bowel Project; NSAI, nonsteroidal aromatase inhibitor; OS, overall survival; pCR, pathological complete response; QD, once daily; RT, radiation therapy; wks, weeks.
PALLAS is an open-label, phase III trial in which patients were randomly assigned to receive standard ET with or without palbociclib for 2 years (125 mg once daily for 3 weeks, followed by 1 week off). The trial included pre- and postmenopausal women or men with stage II or stage III EBC. 16
PENELOPE-B is a placebo-controlled, phase III trial in which patients were randomly assigned to receive standard ET with or without palbociclib for 13 cycles (125 mg once daily for 3 weeks, followed by 1 week off). The trial included EBC patients with residual disease after neoadjuvant chemotherapy and considered at higher risk by the clinical-pathological stage-estrogen/grade (CPS-EG score), defined as CPS-EG score of ⩾3, or score 2 if nodal status at surgery is ypN+. 17
MonarchE is an open-label, phase III trial in which patients were randomly assigned to receive standard ET with or without abemaciclib for 2 years (150 mg twice daily, continuously). The trial included pre- and postmenopausal women or men with stage II or III EBC and node-positive disease (four or more positive axillary lymph nodes or one–three positive axillary lymph nodes and either tumor size ⩾5 cm, histologic grade 3, or Ki-67 ⩾20%). 30
NATALEE is an open-label, phase III trial in which patients were randomly assigned to receive a nonsteroidal aromatase inhibitor (NSAI) with or without ribociclib for 3 years (400 mg once daily for 3 weeks, followed by 1 week off). In the NATALEE trial, all patients (pre- and postmenopausal women or men) with stage II and III disease were eligible, except for those with node-negative stage IIA disease (T2N0) who were required to have other high-risk disease features (grade 3 disease or grade 2 disease plus Ki-67 ⩾20% or high genomic risk). 19
The PALLAS and PENELOPE-B trials failed to demonstrate a benefit of palbociclib plus ET over standard ET in terms of their primary endpoints—invasive disease-free survival (iDFS)—and therefore palbociclib is not considered for adjuvant treatment escalation. However, the monarchE and NATALEE trials achieved their primary endpoint, demonstrating a significant improvement in iDFS within their intent-to-treat (ITT) populations.
The ITT populations of the monarchE and NATALEE trials comprised patients with stage II and III EBC, with the NATALEE trial including a broader patient population. A more comprehensive breakdown of the inclusion criteria for these trials, as previously published, 32 is shown in Table 2.
Comparison of eligible populations in the monarchE and NATALEE trials by anatomic stage.
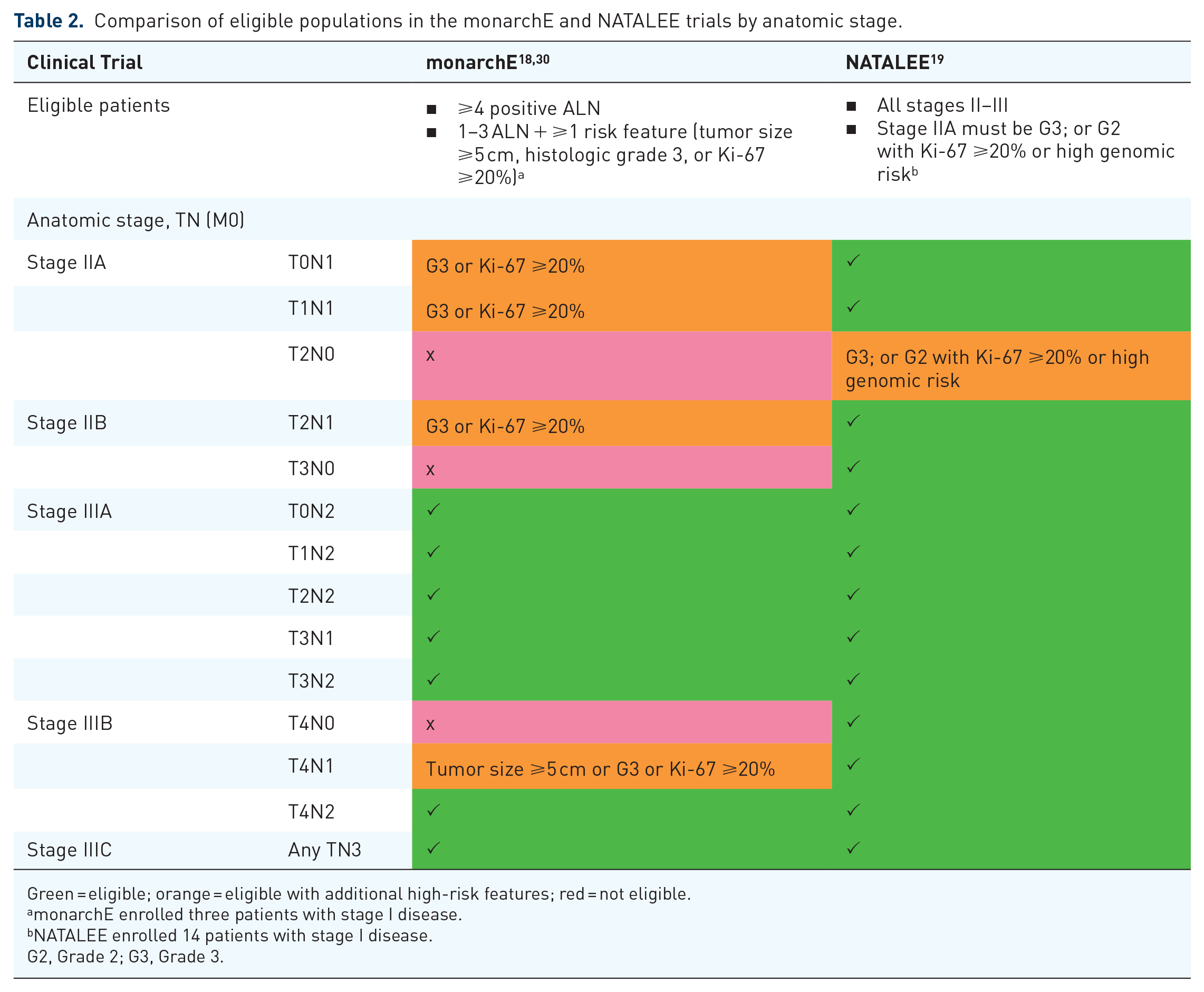
Green = eligible; orange = eligible with additional high-risk features; red = not eligible.
monarchE enrolled three patients with stage I disease.
NATALEE enrolled 14 patients with stage I disease.
G2, Grade 2; G3, Grade 3.
There are notable differences in their baseline characteristics (Table 3). The monarchE trial had a greater representation of stage III disease compared with the NATALEE trial (74% vs 60%). The monarchE trial also had a much higher proportion of patients with N2 or N3 disease compared with the NATALEE trial (60% vs 19%). More than 1/4 (28%) of the NATALEE population had node-negative disease. The monarchE trial also enrolled more patients with the higher-grade disease (39% vs 20% grade 3 disease). Estimates from real-world databases, including the Surveillance, Epidemiology, and End Results program, indicate that between 11% and 25% of patients with HR+, HER2− EBC meet the eligibility criteria for the monarchE trial, while 31%–43% meet the criteria for the NATALEE trial.33–35 This suggests that the NATALEE trial enrolled a population that is more representative of the EBC population in clinical practice. Given that more than 30% of EBC patients present with stage II disease and about 10% present with stage III disease in real life, there remains a significant unmet need for the broader population of patients with stage II and III disease.3,36
Key characteristics of the study population in the monarchE and NATALEE trials.
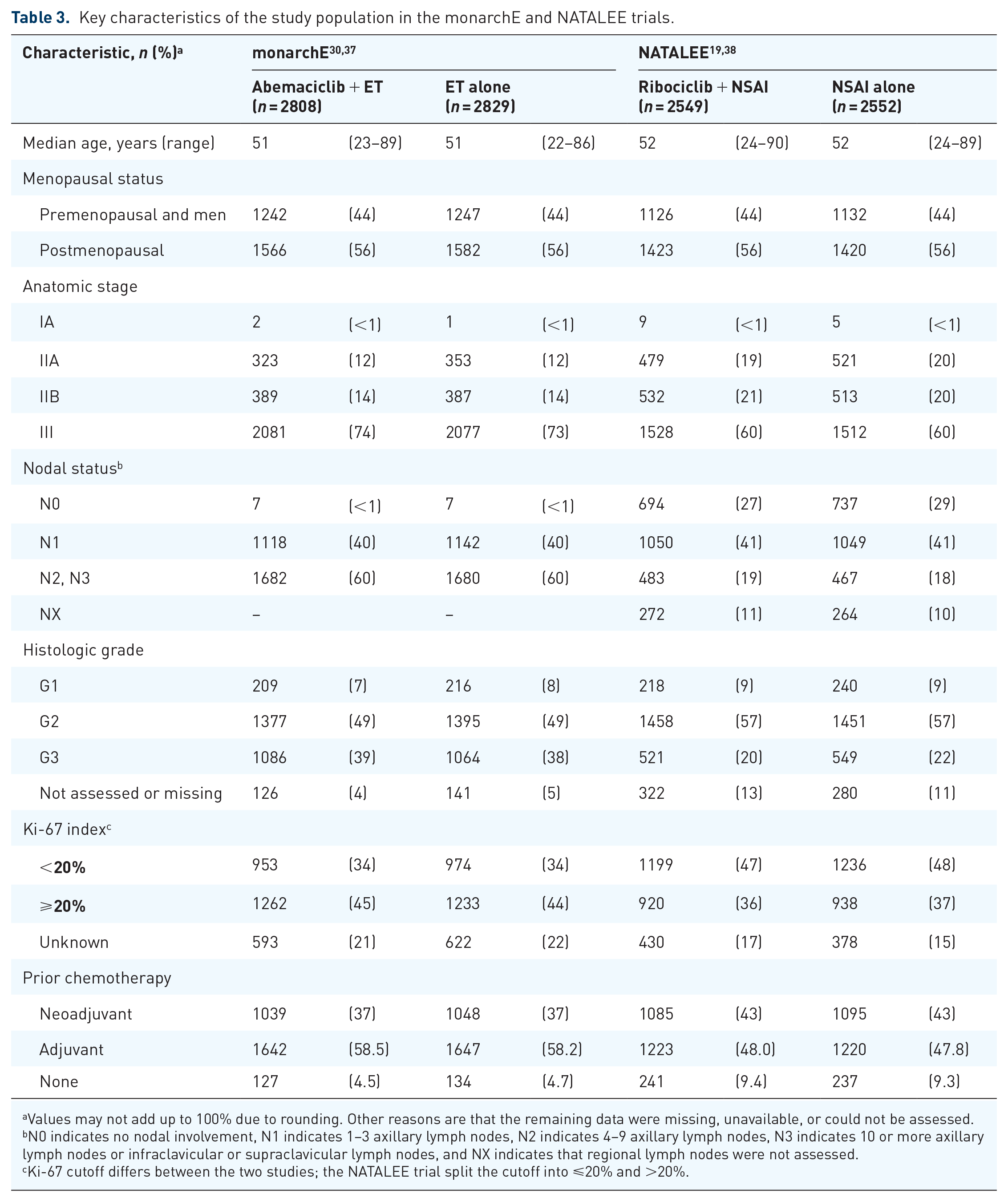
Values may not add up to 100% due to rounding. Other reasons are that the remaining data were missing, unavailable, or could not be assessed.
N0 indicates no nodal involvement, N1 indicates 1–3 axillary lymph nodes, N2 indicates 4–9 axillary lymph nodes, N3 indicates 10 or more axillary lymph nodes or infraclavicular or supraclavicular lymph nodes, and NX indicates that regional lymph nodes were not assessed.
Ki-67 cutoff differs between the two studies; the NATALEE trial split the cutoff into ⩽20% and >20%.
What is the benefit of combining adjuvant CDK4/6 inhibitors with ET?
While palbociclib plus ET failed to statistically improve iDFS compared with ET alone in both the PALLAS and PENELOPE-B trials,16,17 abemaciclib demonstrated a 32% reduction in the risk of developing an iDFS event in the monarchE trial, 18 and ribociclib showed a 28.5% reduction in the risk of developing an iDFS event in the NATALEE trial (Figure 1). 39

Summary of iDFS and DDFS from phase III adjuvant CDK4/6 inhibitor + ET trials.
In the PALLAS trial, at the final analysis (median follow-up of 31 months), 16 the 4-year iDFS was similar between the palbociclib and control groups (84.2% vs 84.5%) with a hazard ratio of 0.96 (95% confidence interval (CI), 0.81–1.14; p = 0.65). No significant differences were observed for other efficacy endpoints, and subgroup analyses did not show any differences by subgroup.
In the PENELOPE-B trial, at the final analysis (median follow-up of 42.8 months), 17 the 4-year iDFS was similar between the palbociclib and control groups (73.0% vs 72.4%) with a hazard ratio of 0.93 (95% CI, 0.74–1.17; p = 0.525). Palbociclib did not improve any other efficacy endpoint.
In the monarchE trial, the 5-year efficacy results from a prespecified OS interim analysis (median follow-up of 54 months) 18 demonstrated a significant iDFS benefit with a hazard ratio of 0.680 (95% CI, 0.599–0.772), with 5-year iDFS rates of 83.6% for abemaciclib plus ET compared with 76% for ET alone. This analysis showed a continuous benefit over time, with a 5-year absolute improvement in iDFS of 7.6% and in distant relapse-free survival (DRFS) of 6.7% compared with ET alone. The corresponding improvements were 6% and 5.3% at 4 years, and 4.8% and 4.1% at 3 years, respectively. OS data were immature at the 5-year analysis, with 208 and 234 total events in the abemaciclib plus ET and ET-alone groups, respectively. iDFS, DRFS, and OS were consistent across all subgroups.
In the NATALEE trial, the exploratory 4-year landmark analysis (median follow-up of 44.2 months), 39 representing the first analysis with all patients off ribociclib, demonstrated a significant iDFS benefit with a hazard ratio of 0.715 (95% CI, 0.609–0.840; p < 0.0001), with 4-year iDFS rates of 88.5% for ribociclib plus NSAI compared with 83.6% for NSAI alone. This analysis also shows a continuous benefit over time, with a 4-year absolute improvement in iDFS of 4.9% compared with NSAI alone. At 3 years, the absolute improvement in iDFS was 3.1%. 31 A significant absolute distant disease-free survival (DDFS) benefit of 2.8% over NSAI was also shown. A consistent benefit was observed across patient subgroups, including those with node-negative, stage II or III disease. OS data were also immature, with 105 and 121 total events in the ribociclib plus NSAI and NSAI-alone groups, respectively.
Why do we observe differences in efficacy between the CDK4/6 inhibitors in the adjuvant setting?
Although the reasons for the differences in outcomes between these trials are unclear, several reasons have been postulated, including differences in study populations, protocols, treatment schedules, treatment duration, and pharmacologic characteristics of the agents.16,17,40
The differences in study populations between the trials (as shown in Table 3) are apparent when comparing the iDFS rates at similar timepoints in the ET-alone groups, with 3-year iDFS rates of 89.4%, 84.4%, and 87.6% in the PALLAS, monarchE, and NATALEE trials, respectively. Despite these differences, neither lower- nor higher-risk patients benefited from adjuvant palbociclib. In terms of study protocols, strict stopping rules related to side effects in the PALLAS trial are believed to have contributed to a high discontinuation rate. In this trial, 42% of patients stopped palbociclib prematurely, with the majority (27.2%) discontinuing due to neutropenia and fatigue. 41 Comparatively, in the monarchE and NATALEE trials, 31% and 36%, respectively, did not complete the full treatment duration.39,42
There are ongoing questions regarding the impact of treatment duration, with some postulating that a longer duration may be needed to extend cell-cycle arrest and potentially induce senescence in more tumor cells. 32 While 1 year of palbociclib treatment in the PENELOPE-B trial did not result in clinical benefit, the PALLAS trial also did not achieve its primary endpoint with 2 years of treatment.16,17 By contrast, 2 years of treatment with abemaciclib has shown deepening benefits up to 5 years of follow-up, and 3 years of treatment with ribociclib has demonstrated continuous benefits up to 4 years of follow-up.18,39 Given the limited follow-up data available for ribociclib at this time, longer follow-up is warranted. Thus, questions remain about the true effect of treatment duration versus other factors.
The three CDK4/6 inhibitors also possess unique pharmacological characteristics, with subtle differences in kinase selectivity. Abemaciclib and ribociclib are more active against CDK4 than CDK6, while palbociclib inhibits CDK4 and CDK6 equally.43,44 Breast cancer cell lines have been associated with CDK4 gene dependency, whereas hematological cell lines, which are linked to neutropenia, have been associated with CDK6 gene dependency. 45 Abemaciclib is the most potent CDK4/6 inhibitor, as evidenced by its lowest IC50. It also shows affinity for CDK1 and CDK9, both of which are expressed in intestinal epithelial cells, suggesting that its gastrointestinal toxicity profile may be linked to this off-target inhibition.46–48 Ultimately, in the absence of head-to-head comparisons between the drugs, the exact reason for the differences in efficacy in the adjuvant setting remains scientific speculation.
Should we wait until OS data mature before incorporating adjuvant CDK4/6 inhibitors into practice?
Although OS is considered by many, including regulatory agencies, as the “gold standard” for incorporating a new therapy, there is currently sufficient evidence to adopt adjuvant CDK4/6 inhibitors into practice. Abemaciclib is approved for the adjuvant treatment of adults with HR+, HER2−, node-positive EBC who are at higher risk of recurrence. 49 It is also recommended by clinical practice guidelines as an option in patients with stage III or high-risk stage II EBC.4,50 Ribociclib has also received regulatory approval by the U.S. Food and Drug Administration (FDA) for the treatment of stage II and III EBC at higher risk of recurrence. 51 It is important to note that it can take over 10 years for an OS advantage to emerge in the adjuvant treatment of HR+ breast cancer, as shown in SOFT/TEXT 52 and the EBCTCG meta-analysis.8,11 Furthermore, disease-free survival (DFS) has been positively correlated with OS in HR+, HER2− EBC, which suggests that DFS may serve as a reliable surrogate endpoint for OS. 53 While OS has currently not reached statistical significance in the monarchE and NATALEE trials, the lower number of deaths in the treatment groups compared with the ET groups suggests that a survival signal may be emerging in both trials. At 54 months of follow-up, 208 of the 2808 (7.4%) in the abemaciclib plus ET group and 234 of the 2829 (8.3%) in the ET group had died (hazard ratio 0.903; 95% CI, 0.749–1.088). 18 At 44 months of follow-up for OS, 105 of the 2549 patients (4.1%) in the ribociclib plus NSAI group and 121 of the 2552 patients (4.7%) in the NSAI group had died (hazard ratio, 0.827; 95% CI, 0.636–1.074). 31 Ongoing monitoring of survival outcomes remains a priority as these agents are integrated into clinical practice.
What are the potential safety concerns with abemaciclib and ribociclib?
In the monarchE trial, the most common grade 3–4 adverse events (AEs) associated with abemaciclib were neutropenia (20%), leukopenia (11%), and diarrhea (8%; Figure 2).42,54 The incidence of grade 3–4 AEs was higher in the abemaciclib plus ET group (50%) compared with the ET-alone group (17%). The most frequent AEs of any grade with abemaciclib were diarrhea (84%), neutropenia (46%), and fatigue (41%). In the ET-alone group, the most frequent AEs of any grade were arthralgia (38%), hot flush (23%), and fatigue (18%). In terms of other important AEs, venous thromboembolic events (VTE) were more common with abemaciclib (2.5%) than ET alone (0.6%). The incidence of VTE was higher when abemaciclib was combined with tamoxifen (4.3%) compared to its combination with an AI (1.8%). Interstitial lung disease (ILD)/pneumonitis was also more common with abemaciclib (3.2%) than ET alone (1.3%).
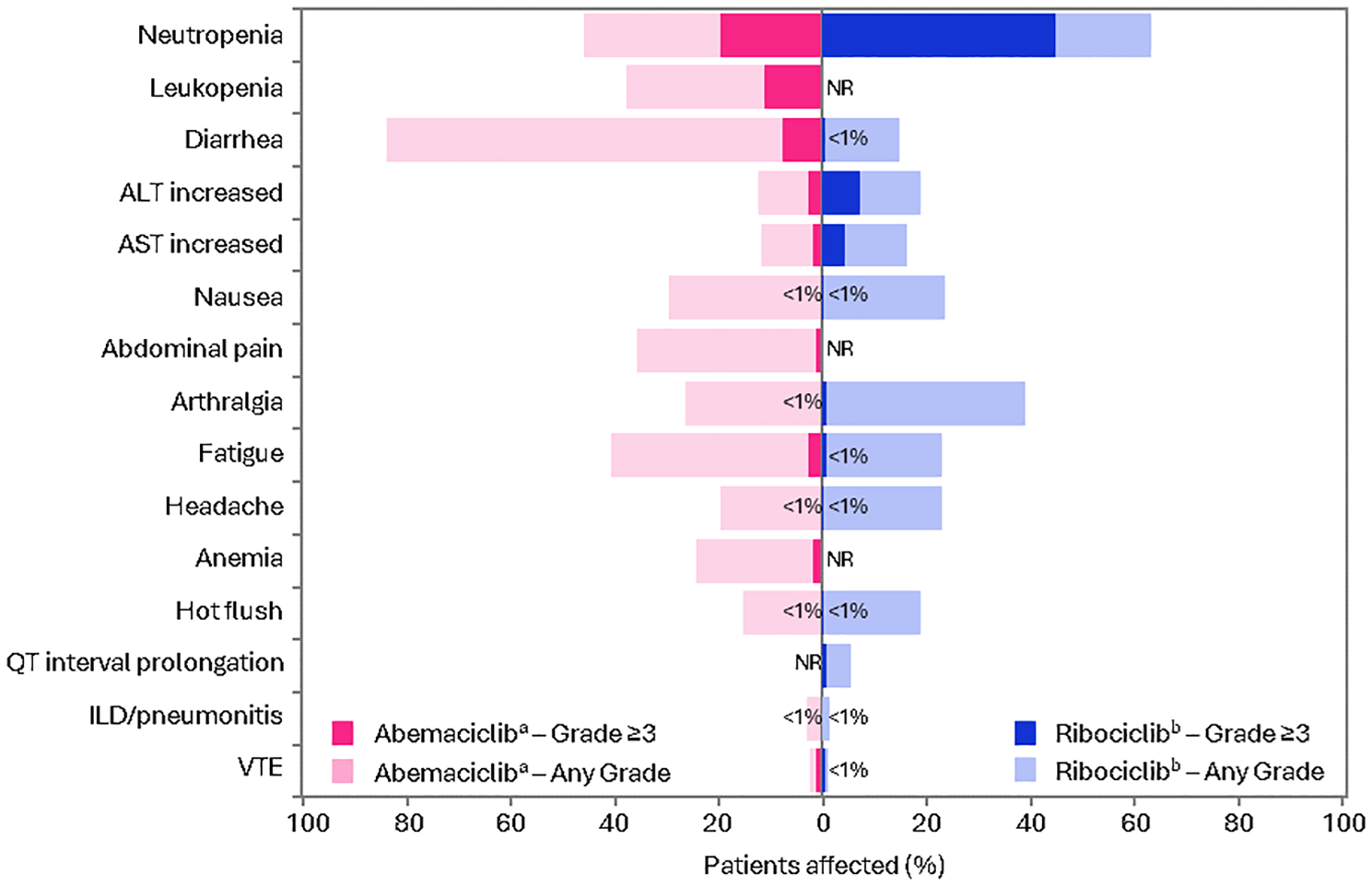
Incidence of most common AEs from the monarchE and NATALEE trials.
In the NATALEE trial, the most common grade 3–4 AEs associated with ribociclib were neutropenia (44%) and elevated liver enzymes (9%; Figure 2).19,31,39 The incidence of grade 3–4 AEs was higher in the ribociclib plus NSAI group (62%) compared with the NSAI-alone group (18%). The most common AEs of any grade with ribociclib were neutropenia (63%), arthralgia (39%), and elevated liver enzymes (alanine aminotransferase, 19%; aspartate aminotransferase, 16%). In the NSAI-alone group, the most frequent AEs of any grade were arthralgia (44%), hot flush (20%), and headache (17%). QTc interval prolongation was more common with ribociclib (5.4%) than NSAI alone (1.6%). The rate of VTE was slightly higher with ribociclib (1.1%) than NSAI alone (0.5%). ILD/pneumonitis was also more common with ribociclib (1.6%) than NSAI alone (0.9%).
Could AEs be managed with dose adjustments?
In the monarchE trial, dose interruptions and reductions (to 100 mg twice daily and 50 mg twice daily) were allowed (Table 4). A total of 62% of patients experienced dose interruptions due to AEs, and 43% had dose reductions. These dose adjustments were mainly related to diarrhea, neutropenia, or fatigue. Abemaciclib was discontinued in 19% of patients due to AEs. AEs occurred early during treatment and were managed with medications (e.g., anti-diarrheal medication) and dose adjustments; most patients stayed on treatment.
Dose adjustments and discontinuations in the monarchE and NATALEE trials.

2808 Patients were assigned to abemaciclib (continuous dosing) plus ET, and 2791 were evaluated for safety. At the overall survival interim analysis (data cutoff: July 1, 2022), all patients were no longer on study treatment.
2549 Patients were assigned to ribociclib (3 weeks on, 1 week off dosing) plus ET, and 2524 were evaluated for safety. At the 4-year landmark analysis (data cutoff: April 29, 2024), all patients were no longer on study treatment.
As reported for ribociclib in the second interim analysis (date cutoff: January 11, 2023).
Only a small proportion of patients with dose reductions discontinued abemaciclib due to AEs (8.9%, n = 249). By contrast, 266 of 515 (52%) discontinued without a prior dose reduction. Sixty-seven of 76 (88%) who discontinued treatment during the first month did so without a prior dose reduction.
AE, adverse event; BID, twice daily; ET, endocrine therapy; NR, not reported.
In the NATALEE trial, a dose reduction to 200 mg once daily was allowed (Table 4). A key feature of the NATALEE trial was a lower starting dose of ribociclib (400 mg once daily) as compared to the starting dose in the ABC setting (600 mg once daily) to improve safety and adherence. The lower starting dose resulted in fewer dose-related toxicities compared to a pooled analysis of the MONALEESA-2, -3, and -7 trials in the ABC setting; for instance, 62% of patients in the NATALEE trial experienced all-grade neutropenia, compared to 74% in the pooled MONALEESA analysis. 55 Twenty-two percent of patients experienced dose reductions, and ribociclib was discontinued in 20% of patients due to AEs. Close to half of these discontinuations were related to elevated liver enzymes. Similar to the monarchE trial, most discontinuations occurred early during treatment, with a median time to ribociclib discontinuation of 4 months. The rates of NSAI discontinuation due to AEs were similar between the ribociclib plus NSAI group (5.3%) and NSAI-alone group (4.9%) suggesting that adding ribociclib had limited impact on NSAI discontinuations.
Administering adjuvant CDK4/6 inhibitors
What is the timing and duration of adjuvant CDK4/6 inhibitor therapy?
Based on the eligibility criteria of the trials, patients should be initiated on adjuvant abemaciclib within 12 weeks of ET (initiation) after their last non-endocrine adjuvant treatment, 30 while those on adjuvant ribociclib should be initiated within 12 months of starting standard (neo)adjuvant ET (Figure 3). 19 Adjuvant abemaciclib is administered for 2 years, while adjuvant ribociclib is administered for 3 years. After completing adjuvant treatment with a CDK4/6 inhibitor, patients should continue their standard ET as indicated (5–10 years total). Concurrent adjuvant radiation treatment with abemaciclib or ribociclib was not allowed, and the start of adjuvant CDK4/6 inhibitor therapy was recommended 14 days after the completion of adjuvant radiation therapy.

Administration of adjuvant CDK4/6 inhibitors.
How do you select the endocrine partner?
Based on the design of the trials, abemaciclib can be paired with either an AI or tamoxifen, while ribociclib must be paired with an NSAI. Interestingly, all premenopausal women in the NATALEE trial received OFS in conjunction with an NSAI, 19 whereas, in the monarchE trial, only half of the premenopausal women received OFS. 56 Approximately 30% of patients received tamoxifen in the monarchE trial. Given the elevated risk of VTE with the tamoxifen combination, patients with a prior VTE were excluded from enrolling in monarchE.
The NATALEE trial only allowed NSAIs because tamoxifen inherently prolongs the QTc interval, and this effect is additive when combined with ribociclib. The combination of ribociclib and tamoxifen is supported by data from the MONALEESA-7 trial for ABC but remains controversial due to the increased risk of QTc prolongation. 57 More data are needed in the adjuvant setting before this combination can be considered. It is worth noting that AIs have demonstrated superior efficacy over tamoxifen in patients at risk of recurrence in EBC.8,52
What are the best practices for monitoring and managing patients on adjuvant CDK4/6 inhibitors?
A standard monitoring approach based on prescribing information to help manage AEs associated with CDK4/6 inhibitors is recommended, using temporary dose interruptions and/or dose reductions as needed. The protocol-mandated assessments in the monarchE and NATALEE trials aligned with practice in the advanced setting. In this review, we provide general recommendations (Figure 3) and encourage readers to refer to the prescribing information and other resources on best practices for monitoring patients using CDK4/6 inhibitors.58,59
The most common AEs associated with abemaciclib and ribociclib include blood cell count abnormalities (i.e., neutropenia, leukopenia, anemia), abnormal liver function tests (LFTs), and diarrhea. QTc prolongation has also been reported with ribociclib. As such, monitoring for complete blood count, electrocardiogram (ECG) abnormalities, liver function, and diarrhea is important. Each CDK4/6 inhibitor product information includes specific guidance on monitoring. For example, abemaciclib and ribociclib both have recommended dose modifications for hematologic toxicities, liver toxicity, and ILD/pneumonitis. Ribociclib also includes dose modifications and management recommendations for QTc prolongation, while abemaciclib includes recommendations for diarrhea and VTEs.
In terms of the monitoring schedule, patients are typically followed every 2 weeks for the first 2 months, monthly from months 3 to 6, and then every 3 months until the end of treatment. Close monitoring in the first 6 months is critical as most AEs occur early during the treatment course. For example, in an analysis of hepatic safety for patients treated with ribociclib across multiple trials in ABC and EBC, including the NATALEE trial, the median time to grade ⩾3 elevated LFT ranged from 2.6 to 4.9 months. 60 Also, in a pooled analysis of ribociclib for the treatment of ABC, the median time to grade ⩾2 neutropenia was 17 days. 55 Likewise, the median time to all-grade diarrhea with abemaciclib was 8 days, with the incidence decreasing over time. Diarrhea was mainly managed with antidiarrheal medication (79%) and <25% of patients required dose modifications. 54 The early onset of these AEs highlights the role of dose interruptions and dose reductions.
Given the critical role of adherence to adjuvant treatment in maintaining continuous cell-cycle arrest, it is essential to consider how different toxicities might affect patient adherence. For instance, symptomatic toxicities such as diarrhea, nausea, fatigue, arthralgias, and asthenia can significantly lower patients’ quality of life, negatively affecting adherence to treatment.61,62 Premature discontinuation and poor adherence to adjuvant treatments have been associated with increased risk of recurrence and mortality.63,64 Therefore, it is vital to assess patient characteristics and their capacity to manage and tolerate these side effects to optimize adherence and enhance patient satisfaction.
Application to practice
How do you select between the available agents (abemaciclib and ribociclib)?
The decision-making process involves several factors, including the individual risk of recurrence, treatment efficacy, and other practical considerations.
Risk of recurrence
The monarchE and NATALEE trials highlight important differences in patient selection criteria that may influence treatment decisions. Notably, the NATALEE trial included a broader spectrum of risk profiles. We recommend adhering to the trial eligibility criteria when selecting patients, as outlined in Table 2.
Treatment efficacy
The benefit of treatment is a key determinant in discussing the risk–benefit of treatment with patients. Efficacy results are influenced by the maturity of the data. Both trials have now reported data with all patients off-study treatment. In the monarchE trial, the absolute improvement in iDFS at 4 years was 6.0% (86.0% vs 80%), which deepened to 7.6% (83.6% vs 76%) at 5 years. In the 4-year landmark analysis, ribociclib plus NSAI continued to demonstrate an iDFS benefit over NSAI alone, with an absolute improvement in iDFS of 4.9% (88.5% vs 83.6%).
While it is critical to note key differences between the pivotal trials when selecting a treatment option, cross-trial comparisons should be avoided due to differences in the study population, varying pre-specified endpoints, and statistical considerations. The monarchE trial involved “higher-risk” patients, while the NATALEE trial included a broader range of risk profiles, which may partly explain the different magnitude of benefits observed at similar follow-ups.
Practical considerations
Practical considerations that influence treatment decision-making include treatment initiation flexibility, duration, and toxicity profiles. Another consideration is the increased risk of QTc prolongation with ribociclib, requiring a careful medication review to avoid QTc-prolonging drugs. Abemaciclib may be considered where ECG availability is limited, as ribociclib requires ECGs in early treatment stages. Notably, the FDA updated its recommendations, now advising ECG assessments prior to treatment initiation, approximately Day 14 of the first cycle, and as clinically indicated, omitting the ECG at the start of the second cycle. 65 Comorbidities may also influence treatment choice; abemaciclib is less suitable for those with inflammatory bowel disease, while ribociclib requires caution when the risk of Torsade de Pointes is increased. Furthermore, endocrine partner choice varies; the NATALEE trial allowed only NSAIs while the monarchE trial allowed both tamoxifen and AIs. Thus, an indication for tamoxifen, such as an intolerance to NSAIs, would favor abemaciclib as the more suitable option. However, the risk of VTE must be considered, as abemaciclib is associated with a higher incidence of VTE when combined with tamoxifen compared to its combination with an AI. Meanwhile, the rate of VTE was slightly higher with ribociclib than NSAI alone. Lastly, the addition of OFS is required when using AI for premenopausal women to further reduce the risk of recurrences, which may also influence the choice of therapy.
What is important to patients when considering treatment options?
We know from our patients with HR+, HER2− EBC that they prefer CDK4/6 inhibitor treatment profiles that maximize efficacy while minimizing symptomatic AEs. 66 Treatment attributes related to monitoring or schedule, such as the number of blood tests, number of ECGs, and treatment schedule, do not appear to significantly influence treatment preference, although a shorter treatment duration is preferred. Another important consideration is the financial cost of CDK4/6 inhibitors, which can deter some patients from pursuing these therapies. These patient preferences are critical for shared decision-making when discussing the addition of a CDK4/6 inhibitor to adjuvant ET for eligible patients.
What strategies can be implemented to support adherence to adjuvant CDK4/6 inhibitors?
To effectively support adherence to adjuvant CDK4/6 inhibitors, it is crucial to understand the barriers and facilitators that influence patient compliance. A study of patients with ABC treated with CDK4/6 inhibitors identified several potential components of a multiprong intervention that could enhance adherence. These components include patient education, evidence-based strategies for symptom management, tips for open and assertive communication with healthcare providers, and information about available financial resources and support. 67
A significant concern is the premature discontinuation of both ET and CDK4/6 inhibitors due to toxicities. In the monarchE and NATALEE trials, approximately 6% and 3% of patients, respectively, discontinued both treatments because of AEs. One contributing factor may be the patient’s inability to differentiate between side effects caused by ET and those caused by the CDK4/6 inhibitor. To mitigate this issue, a potential strategy to consider is sequencing the therapies as was permitted in the adjuvant trials. This approach should allow patients to better identify the agent responsible for the toxicity, thereby reducing the likelihood of discontinuing both therapies prematurely.
A shared decision-making approach is essential for motivating patients to adhere to their treatment regimen. This involves educating patients about the potential benefits of treatment, helping them understand their actual risk of recurrence, and explaining how adjuvant treatment can significantly reduce this risk. It is also important to assure patients from the outset that any side effects will be addressed promptly through dose interruptions or reductions and regular follow-ups, ensuring they do not suffer for extended periods.
Should ribociclib be offered to all node-negative patients with high-risk features (i.e., T2N0, as per the NATALEE trial)?
The use of ribociclib in node-negative patients has been a point of contention.4,50 However, considering the demonstrated superiority in the ITT population, it is reasonable to offer ribociclib to the node-negative patients (i.e., T2N0) who meet the NATALEE eligibility criteria. In this T2N0 subgroup, which required patients to have either grade 3 disease or grade 2 disease plus Ki-67 ⩾20% or high genomic risk, the iDFS was improved with ribociclib plus NSAI compared to NSAI alone, with a 4-year absolute improvement in iDFS of 5.1% (92.1% vs 87.0%) and a hazard ratio of 0.666 (95% CI, 0.397–1.118). While statistical significance is unlikely due to the small sample size (n = 285), the data suggest a consistent benefit of ribociclib across the ITT population. Thus, it is important to engage in a shared decision-making process, considering not only the clinical benefits and risks but also the costs and the individual preferences and values of the patient.
What if my patient experiences ET-related AEs?
AEs associated with adjuvant ET include hot flashes, sexual dysfunction, weight gain, arthralgia, bone density loss, depression, cognitive dysfunction, and fatigue.68,69 In premenopausal patients, escalation with OFS will lead to a marked drop in estrogen levels, leading to more severe AEs. Since ribociclib is not indicated with tamoxifen, and there is an elevated risk of VTEs when combining abemaciclib with tamoxifen, along with the fact that AIs have shown superior efficacy over tamoxifen, using a combination of a CDK4/6 inhibitor, AI, and OFS may lead to more ET-related AEs in premenopausal patients. These AEs can significantly affect the quality of life and are key reasons for non-adherence to ET monotherapy, with a reported 20% rate of early discontinuations.10,70 This rate can be even higher after 5 years of treatment, ranging from 31% to 73%.71–73
Proactive management of these symptoms is essential to optimize treatment outcomes while balancing tolerability, adherence, and quality of life. We recommend referring to comprehensive reviews on various interventions—including hormonal and non-hormonal medications, non-pharmacological approaches, and complementary and alternative medicine—to help manage common AEs associated with adjuvant ET.74–76
Can I switch CDK4/6 inhibitors if my patient experiences AEs?
Switching CDK4/6 inhibitors has not been studied in the adjuvant setting and is not currently recommended, although it may occur in real-world practice. Management of CDK4/6 inhibitor-related AEs typically involves dose interruptions and dose reductions. Given the early onset of toxicities like diarrhea and neutropenia, close monitoring and early intervention are essential to improve tolerability. While a pragmatic clinical approach might consider switching CDK4/6 inhibitors, there is currently no evidence to support or refute this strategy. While this practice is common in ABC, we advocate for the collection of real-world evidence to evaluate the potential benefits of EBC.
Are adjuvant CDK4/6 inhibitors a replacement for (neo)adjuvant chemotherapy?
Adjuvant CDK4/6 inhibitors are not a replacement for (neo)adjuvant chemotherapy. The adjuvant CDK4/6 inhibitor trials were conducted as an add-on to standard-of-care chemotherapy, with most patients in both studies having received prior chemotherapy (88% in NATALEE and 95% in monarchE).19,30 In the node-negative subgroup of the NATALEE trial, 72% received prior chemotherapy. 77
What is the role of biological markers in selecting patients for adjuvant therapy?
As more patients receive adjuvant CDK4/6 inhibitor therapy, identifying predictors of efficacy and toxicity is crucial. While biological markers such as the Ki-67 score and genomic risk score serve as useful prognostic tools, their value as predictive biomarkers of response to CDK4/6 inhibitor therapy remains limited.
Initially, adjuvant abemaciclib was approved only for patients with a Ki-67 score of ⩾20%, but this requirement was eliminated after regulatory agencies recognized the benefits across the entire ITT population in the monarchE trial. In cohort 1 of the monarchE trial, which stratified patients by Ki-67 score, higher iDFS rates were observed in patients with Ki-67 ⩾20%, with a 5-year absolute improvement in iDFS of 9% (hazard ratio of 0.643, p < 0.001) in the Ki-67-high subgroup compared with 6.1% (hazard ratio of 0.662, p < 0.001) in the Ki-67-low subgroup. The consistent benefit across Ki-67 subgroups suggests that Ki-67 is not predictive of abemaciclib benefit. 18
A question arises whether there are patients for whom chemotherapy is not indicated but who might still be candidates for adjuvant CDK4/6 inhibitor therapy. Genomic risk is currently used in conjunction with clinicopathological markers to select candidates for chemotherapy. This approach is grounded in the understanding that gene expression profiles can predict chemotherapy benefits.78–81 Yet, there is limited evidence to support the use of genomic risk as a predictive marker of response to adjuvant CDK4/6 inhibitors. Data from the CORALLEEN trial suggest that some patients with high-risk HR+, HER2−, and luminal B EBC can achieve molecular downstaging of their disease with neoadjuvant ribociclib plus ET. 82 However, in a biomarker analysis of archived primary tumor tissue from the monarchE trial, transcriptome analyses demonstrated a similar magnitude of benefit across intrinsic molecular subtypes and in both high- and low-risk scores based on inferred 21-gene Oncotype-RNA signatures. 83 This suggests that genomic criteria that are useful for guiding chemotherapy decisions cannot solely determine the suitability of adjuvant CDK4/6 inhibitor therapy. The potential role of adjuvant CDK4/6 inhibitors as a substitute for chemotherapy will be further addressed by ongoing trials. For instance, the ADAPTcycle trial (NCT04055493) is evaluating the efficacy of ribociclib in combination with ET compared to standard-of-care chemotherapy (followed by adjuvant ET) in patients with HR+, HER2− EBC with intermediate genomic risk as defined by OncotypeDx®. 84
As more data become available, it is hoped that we will be able to better define the optimal cohorts that derive the most benefit from treatment, understand the effects on late recurrences, and determine the ideal duration of CDK4/6 inhibitor therapy.
What other adjuvant treatment options should be considered?
Other adjuvant therapy options for individuals with higher-risk HR+, HER2− EBC include capecitabine, olaparib, and bisphosphonates. The CREATE-X trial assessed capecitabine in both HR+ and triple-negative breast cancer (TNBC) patients, showing a significant DFS benefit in the ITT population, particularly in the TNBC subgroup. 85 However, the results were less promising for HR+ patients (DFS hazard ratio, 0.81; 95% CI, 0.55–1.17), limiting capecitabine’s role due to its unclear benefit in HR+ disease and the availability of other treatment options.
By contrast, the poly (ADP-ribose) polymerase inhibitor olaparib has shown significant promise for patients with germline BRCA1/2 mutations, as evaluated in the OlympiA trial. 86 Adjuvant olaparib reduced the risk of recurrence in a high-risk group, offering a targeted option beyond standard ET. One year of adjuvant olaparib improved DFS (hazard ratio 0.63; 95% CI, 0.5–0.78) and OS (hazard ratio 0.68; 95% CI, 0.47–0.97, p = 0.009) regardless of HR status. At 4 years, the absolute differences in iDFS and DDFS were 7.3% and 7.4%, respectively. Patients with HR+ disease were eligible if they met one of the following criteria: (1) they had no pathologic complete response (pCR) after neoadjuvant chemotherapy and a nuclear grade (CPS + EG) score of ⩾3 or (2) they had more than four positive lymph nodes following adjuvant chemotherapy.
The question of sequencing a CDK4/6 inhibitor after olaparib arises. This question was addressed at the 18th St. Gallen Consensus Conference on EBC. The consensus, which is not evidence-based, was to consider sequencing olaparib followed by abemaciclib. 87 Adjuvant WIDER (NCT05827081), an open-label phase IIIb trial, may address this question by allowing up to 36 months of ET prior to the initiation of ribociclib (ClinicalTrials.gov identifier: NCT05827081). This approach opens the door to sequencing olaparib followed by ribociclib. However, in the current absence of prospective evidence, sequencing decisions are guided by expert opinion and consensus.
Adjuvant bisphosphonate therapy may be considered to lower the risk of recurrence and mitigate the side effects of osteopenia or osteoporosis associated with AIs. 4 A meta-analysis conducted by the EBCTCG demonstrated a benefit irrespective of the HR status and bisphosphonate type or regimen. 88
The use of (neo)adjuvant immunotherapy is also of great interest. The KEYNOTE-756 (NCT03725059) 89 and CheckMate 7FL (NCT04109066) 90 trials are evaluating the role of neoadjuvant immunotherapy plus chemotherapy followed by adjuvant immunotherapy plus ET in patients with high-risk HR+, HER2− EBC. Event-free survival results are still immature in the KEYNOTE-756 trial, while the Checkmate 7FL trial showed significantly improved pCR rates. The potential advent of immunotherapy for the treatment of EBC raises the prospect of combination or sequencing strategies with adjuvant CDK4/6 inhibitors, though this also raises questions about the safety of combining such therapies.
What if my patient experiences disease recurrence after completing a full course of adjuvant CDK4/6i?
The optimal treatment strategy for patients who experience progression on adjuvant CDK4/6 inhibitor plus ET remains unclear. Several critical questions arise, including whether the recurrence is local or distant, whether it occurs early or late, the treatment-free interval, and whether there is a role for retreatment with the same or a different CDK4/6 inhibitor. Recent data from the ABC setting on retreatment with a different CDK4/6 inhibitor following disease progression from the phase III postMONARCH trial (switch to abemaciclib) 91 and the phase II MAINTAIN trial (switch to ribociclib) 92 provide some insights. In the postMONARCH trial, which included a small proportion (1%) of patients on a prior adjuvant CDK4/6 inhibitor, patients on abemaciclib plus fulvestrant had a significantly improved progression-free survival (PFS) compared with those on fulvestrant alone. The improvement seemed to predominantly favor the subgroup previously exposed to palbociclib rather than either ribociclib or abemaciclib. A similar benefit of switching to ribociclib was also noted in the MAINTAIN trial, where notably 84% of patients had previously received palbociclib.
Another consideration for patients experiencing early disease relapses (within 12 months of completing adjuvant therapy) is to prioritize blocking both the CDK4/6 and PIK3CA pathways in patients with PIK3CA-mutated tumors, as demonstrated in the phase III INAVO120 trial (NCT04191499). 93 The investigators reported a benefit in PFS from 7.3 to 15 months (hazard ratio of 0.43) and a trend in OS (hazard ratio of 0.64) for the inavolisib plus palbociclib plus fulvestrant treatment arm. In this trial, 1.9% of the study population had prior CDK4/6 inhibitor treatment in the adjuvant setting. Although this represents a small subset of patients typically seen in practice, it supports the principle of triple therapy blockade in the ABC patient population with poorer prognosis.
Innovative care models in the adjuvant setting
The integration of adjuvant CDK4/6 inhibitors with ET will require a multidisciplinary team to ensure effective and safe patient care. This approach is increasingly important given the expanded use of ribociclib, amplifying the need for patient follow-up. Pharmacists can assist in reviewing concomitant medications and potential drug–drug interactions, particularly those associated with QTc prolongation. They can also conduct medication reviews, monitor and manage side effects, adjust doses, educate patients, and handle reimbursement issues. Oncology nurses or nurse practitioners can coordinate laboratory and ECG monitoring, assess side effects, and help patients understand the dosing schedules of their oral medications.
Innovative care models are being implemented to reduce the burden on medical oncologists managing oral oncology treatments.94–101 Many of these models are associated with improved adherence, patient safety, and both patient and provider satisfaction. One example is the Medication Assessment by Pharmacist program, which incorporates pharmacists into a shared care model. This program aims to reduce outpatient visits for oncologists while adhering to monitoring and visit protocols for patients on CDK4/6 inhibitors. 94 The establishment of an appropriate care model will vary by center and jurisdiction.
Future directions
The decision-making process for determining prognosis and selecting optimal systemic therapy in HR+, HER2− EBC is complex. Extended follow-up of the monarchE and NATALEE trials should indicate whether a survival signal emerges over time. Future studies should focus on identifying biological markers and gene expression profiles that predict subgroups more or less likely to benefit from CDK4/6 inhibitor therapy. The ADAPTcycle trial (NCT04055493), 84 described earlier, and the ADAPTlate trial (NCT04565054) 102 are examples of such studies. The latter investigates the efficacy of adjuvant abemaciclib in patients with clinically or genomically high-risk HR+, HER2− EBC. Similar to Adjuvant WIDER (NCT05827081) involving the late introduction of ribociclib (ClinicalTrials.gov identifier: NCT05827081), ADAPTlate will also assess the effect of introducing abemaciclib at a later time. 102 The role of circulating tumor DNA (ctDNA) analysis as a marker for monitoring disease progression and treatment efficacy should also be further evaluated. An analysis of the monarchE trial found that ctDNA positivity was associated with a substantially increased risk of iDFS events. 103 Specifically, the study revealed that a total of 87% of the patients with ctDNA positivity at any timepoint (17% of the monarchE cohort) experienced an iDFS event versus 15% of those with persistent ctDNA negativity (83%), underscoring its potential role in improving risk stratification.
Conclusion
The integration of adjuvant CDK4/6 inhibitors, specifically abemaciclib and ribociclib, represents a significant advancement in the management of HR+, HER2− EBC, offering significant reductions in the rates of recurrence with a generally acceptable safety profile. Following the promising outcomes of the monarchE and NATALEE trials, a broader patient population is now considered for these therapies. The decision-making process regarding the selection of abemaciclib versus ribociclib should involve careful consideration of disease characteristics, recurrence risk, long-term efficacy, safety profiles, individual patient preferences regulatory approvals, accessibility, and cost implications. Furthermore, fostering a comprehensive approach that emphasizes patient education, effective symptom management, and clear communication can enhance adherence to therapy, ultimately improving treatment outcomes for eligible patients.
Footnotes
Acknowledgements
We would like to thank Novartis Pharmaceuticals Canada Inc. for supporting the development of this manuscript through an unrestricted educational grant to liV Agency Inc. We thank liV Agency Inc. for logistical and organizational support and medical writing support (Paul Heron and Scott Moffatt).


