Abstract
Background:
Large-cell neuroendocrine lung carcinoma (LCNEC) is a rare subtype of lung cancer that lacks standard treatment at present.
Purpose:
This study aimed to investigate the treatment status, failure pattern, survival outcome, and gene expression profile of LCNEC in China.
Design:
This is a real-world retrospective study combined with transcriptome sequencing of LCNEC tumors.
Methods:
Patients with newly diagnosed LCNEC at Beijing Chest Hospital from 2015 to 2022 were retrospectively reviewed. Treatment, failure pattern, and survival were analyzed. Transcriptome sequencing of LCNEC and non-small-cell lung cancer was conducted for differentiated expressed genes exploration and enrichment analysis.
Results:
In all, 151 eligible patients met the criteria: stage I (24.5%), II (9.9%), IIIA (13.9%), and IIIB–IV (51.7%). Median progression-free survival (PFS) and overall survival (OS) were 7.9 and 17.8 months for an entire cohort of patients. For stage I/II and IIIA patients receiving radical operation or chemoradiation, 47 out of 77 cases developed treatment failure with 2-year cumulative systemic/distant failure (SF), locoregional failure (LRF), and overall failure rates of 65.2%, 52.7%, and 30.8%, respectively. Failure incidence increased with stage development. Stage III disease presented with a significantly higher cumulative SF rate (2-year, 57.3% vs 29.7%; p = 0.010) but a similar LRF rate (2-year, 41.5% vs 37.6%, p = 0.369) than stage I/II, achieving favorable SF-free survival and comparable LRF-free survival. Adding adjuvant chemotherapy to surgery reduced distant dissemination which translated into survival benefit (2-year SF, 53.7% vs 41.3%, p = 0.055; 2-year OS, 37.1% vs 79.9%, p < 0.001). For advanced LCNEC, immunochemotherapy and chemotherapy alone achieved PFS of 10.3 and 4.7 months, respectively (p = 0.045). Differential gene expression analysis revealed that antigen presentation/processing, chemokine signaling, CXCR4, and IFN-γ pathways were upregulated in LCNEC, suggesting the vulnerability of LCNEC to immunotherapy. Besides, MMP9, AGT, COL1A2, COL1A1, and CXCL9 may play vital roles in the LCNEC pathogenesis.
Conclusion:
LCNEC is a highly aggressive disease and incorporation of immunotherapy might be an effective treatment option.
Introduction
Large-cell neuroendocrine lung carcinoma (LCNEC) is a highly aggressive pulmonary neuroendocrine tumor that accounts for 2%–3% of non-small-cell lung cancer (NSCLC).1,2 Like small-cell lung cancer (SCLC), LCNEC has a predominant prevalence in elderly heavy smokers with a fairly poor prognosis.3,4 Over the past decade when molecularly targeted therapies and immune checkpoint inhibitors (ICIs) have dramatically improved the prognosis of NSCLC,5,6 little progress in management, however, has been achieved in patients with LCNEC. The lack of an encompassing pathological definition and high-quality clinical trials made LCNEC an area of unmet need.
No standard of care exists due to an absence of phase III and/or randomized clinical trials in LCNEC.4,7–11 Generally, for localized stage I–IIIA disease, surgical resection is considered the mainstay of treatment.3,12–14 Raman et al. reviewed 6092 patients with localized LCNEC from the National Cancer Database (2004–2015) and the 5-year overall survival (OS) for cases undergoing an operation for stage I, II, and IIIA LCNEC were 50%, 45%, and 36%, respectively. 12 Surgical resection was associated with significant survival benefit in comparison with radiation in stage I disease (HR = 0.70, 95%CI, 0.57–0.86) and chemoradiation in stage II–IIIA disease (stage II, HR = 0.47, 95%CI, 0.35–0.63; stage IIIA, HR = 0.69, 95%CI, 0.60–0.81). 12 However, the role of adjuvant therapy is unclear in early LCNEC. Data from primarily retrospective analyses showed adjuvant chemotherapy was associated with improved OS in early-stage LCNEC.12,14–16 At present, the failure pattern of localized LCNEC has not been fully investigated, which might be useful in better management of local treatment and systemic therapy in this group of patients. Altieri et al. reported that even after radical resection, half of early-stage LCNEC patients recurred mostly within the first 2 years of follow-up. 17
For advanced LCNEC, platin-based systemic chemotherapy is considered the mainstay of therapy. Whether to classify and treat LCNEC using NSCLC- or SCLC-like regimens remains controversial at present.4,8,18 Two recent multicenter prospective studies evaluated the efficacy of SCLC chemotherapy (cisplatin and etoposide or irinotecan) in advanced LCNEC, and median OS rates were 7.7 and 15.1 months, respectively.7,10 Besides, the clinical activity of ICIs in advanced LCNEC has not been fully defined.11,19–21 Since most available data were collected from case reports and small retrospective series, or population-based studies of Western countries, the treatment, survival, and failure pattern of LCNEC from a large cohort of Chinese LCNEC patients was lacking.
Therefore, we conducted this study aiming to assess the real-world status of treatment options, failure pattern, and survival of LCNEC among Chinese LCNEC patients at our institute; besides, we also performed gene-expressing sequencing of LCNEC tumors in comparison to NSCLC among Chinese LCNEC patients at our institute.
Materials and methods
Study design and patient eligibility
This is a real-world retrospective study conducted at Beijing Chest Hospital. Consecutive patients with pathologically confirmed LCNEC from 2015 to 2022 were initially considered. Pathological diagnosis of each sample was re-confirmed by two pathologists independently. Inclusion criteria were as follows: pathologically diagnosed LCNEC with typical morphology and immunophenotype according to WHO classification; mixed LCNEC and SCLC of lung origin as well as mixed LCNEC and NSCLC with a predominating LCNEC component; stage I–IV disease; no prior surgery, radiation, or systemic chemotherapy; and at least one measurable lesion for advanced disease. Exclusion criteria were as follows: patients with ambiguous diagnoses and cases with incomplete clinicopathologic or follow-up information.
Medical records of eligible patients were reviewed including time, age of diagnosis, gender, smoking histology, site of primary tumor, AJCC 8th edition staging, node, metastasis, pathological staining, and molecular alternations. Treatment material, response, and survival of each patient were also collected. Follow-up visits were conducted every 3 months in the first 2 years post-operation, every 3–6 months thereafter for patients receiving surgery, and every 2–3 months for patients receiving other treatments.
Targeted DNA sequencing
Tissues with tumor proportions ⩾10% were selected for extraction of gDNA using QIAamp DNA FFPE Tissue Kit (Qiagen, Valencia, CA, USA). DNA quantification was performed using a Qubit 2.0 fluorimeter (Thermo Fisher Scientific, USA). A panel of 60 commonly reported genes in lung cancer was used for the profiling of DNA (Supplemental Table 1). Targeted sequencing was performed using Nextseq500 (Illumina, San Diego, CA, USA) with paired-end reads. LCNEC cases in this study were divided into two groups based on the molecular results according to Natasha Rekhtman’s study 22 : the SCLC-like group (TP53 + RB1 co-mutations/deletions), and the NSCLC-like group (STK11, KRAS, and KEAP1 mutations).
Transcriptome sequencing
A total of six tumor tissues were collected from three LCNEC patients, and 16 samples from 10 NSCLC patients following informed consent and ethical approval. Total RNA was extracted using the RNeasy Mini Kit (Qiagen), ensuring the integrity of the RNA with an RNA Integrity Number ⩾ 7.0 as assessed by the Agilent 2100 Bioanalyzer. RNA-seq libraries were prepared using the TruSeq RNA Sample Preparation Kit v2 (Illumina), which involved mRNA enrichment via poly-T oligo-attached magnetic beads, followed by fragmentation and cDNA synthesis. The cDNA was then subjected to end-repair, adenylation of 3′ ends, and ligation of indexed adapters. The library quality was validated on an Agilent 2100 Bioanalyzer and quantified using the KAPA Library Quantification Kit. Equimolar amounts of indexed libraries were pooled and sequenced on an Illumina NovaSeq 6000 platform to generate 150 bp paired-end reads. Raw sequencing data were processed using FastQC for quality control, and adapter sequences were removed with Trimmomatic. Clean reads were aligned to the human reference genome (GRCh38) using HISAT2, and feature counts were obtained with HTSeq-count.
Programmed death-ligand 1 expression
The FFPE tumor sample of each patient was stained using programmed death-ligand 1 (PD-L1) antibody (clone 22C3, Dako 22C3 PharmDx Assay; Agilent, Santa Clara, CA, USA). PD-L1 expression was determined using the tumor proportion score (TPS). PD-L1 positivity was defined as the demonstration of at least partial membrane staining with moderate or strong intensity in a minimum of 1% of tumor cells.
Statistical analysis
Locoregional failure (LRF) was defined as a disease progression or recurrence in the tumor lesion and/or drainage area of the hilum and mediastinal lymph nodes, independent of the involvement of distant lymph nodes and sites. Systemic failure (SF) was defined as distant dissemination, irrespective of the LRF. Progression-free survival (PFS) was defined as the time interval from the date of treatment to the date of progression, relapse, last follow-up, or death from any cause. OS was defined as the time interval from the date of treatment to the date of death from any cause or last follow-up. Locoregional failure-free survival (LRFS) was defined as the interval from the date of treatment to the date of LRF occurrence, last follow-up, or death from any cause, regardless of SF. Systemic failure-free survival (SFS) was defined as the period from the date of treatment to the date of SF occurrence, last follow-up, or death from any cause, with no regard to LRF. Estimates of survivals were calculated using the Kaplan–Meier method and survival differences were assessed using the log-rank test. Cox regression was used for multivariate analysis of indicators with p values <0.05 in the univariate regression for PFS. A two-tail p < 0.05 was determined to be with statistical difference. IBM SPSS (version 26.0; IBM, USA) was used for statistical analysis and GraphPad Prism (version 9.0; Graphpad Software, USA) was used to graph the statistics.
Differential gene expression analysis was performed using the DESeq2 R package, and the resulting p-values were adjusted for multiple testing using the Benjamini–Hochberg procedure. To identify pathways and function annotations, Gene Ontology, Kyoto Encyclopedia of Genes and Genomes (KEGG), and Gene Set Enrichment Analysis (GSEA) were conducted by the “cluster Profiler” package of R software (version 4.2.0). An adjusted p-value <0.05 was considered as the threshold. The Enrichment Score (ES) was calculated for each predefined gene set, and the Normalized Enrichment Score (NES) was derived to account for the size of the gene set. The significance of the NES was assessed using a permutation test, with 1000 permutations to generate a null distribution of ES. The p-values were adjusted for multiple hypothesis testing using the Benjamini–Hochberg procedure to control the false discovery rate. The leading-edge analysis was performed to identify the core genes contributing most to the enrichment of each gene set. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies statement. 22
Results
Patient baseline characteristics
From 2015 to 2022, 151 patients with histologically confirmed LCNEC diagnosed at Beijing Chest Hospital were retrospectively reviewed. Patient characteristics are summarized in Table 1. The median age was 64 (range, 32–84) years. The male-to-female ratio was 14.1:1. Stage I, II, IIIA, and IIIB–IV diseases accounted for 24.5%, 9.9%, 13.9%, and 51.7%, respectively. The majority of patients had a smoking history (80.1%) and presented peripheral lesions (76.2%). 44 cases (29.1%) were diagnosed with combined LCNEC: with SCLC (n = 21), with squamous carcinoma (n = 18), and adenocarcinoma (n = 5; Figure 1(a)). Median Ki-67 was 80% (Figure 1(b)). PD-L1 was expressed in 59.4% of patients with a median TPS of 1% (range, 0%–95%; Figure 1(c)): 46.4% cases had moderate PD-L1 expression (TPS ⩾ 1%, <50%) and 13.0% cases had strong PD-L1 expression (TPS ⩾ 50%).
Patient baseline characteristics.

CRT, concurrent chemoradiation; CT, chemotherapy; LCNEC, large-cell neuroendocrine lung carcinoma; S, surgery.

Pathological diagnosis, Ki-67 index, and PD-L1 expression in LCENC. (a) Pathological subtypes of LCNEC patients; (b) The Ki-67 index of LCNEC tumors; (c) PD-L1 TPS of tumors by immunohistochemistry staining.
Treatment, survival, and prognostic analysis of LCNEC
Among 68 patients with stage I–IIIA LCENC, 60 cases had curative-intent operation including 30 cases received surgery alone and 30 cases had surgery followed by adjuvant chemotherapy, 4 elderly patients had chemotherapy alone and the remaining 4 cases refused any anti-cancer treatment. For patients with stage IIIB–IV (n = 83), 8 cases had concurrent chemoradiation, 9 cases had surgical resection with perioperative chemotherapy, 56 cases received systemic chemotherapy, 2 cases received first-line EGFR-TKI, and 8 cases refused anti-cancer therapy.
Among patients with localized LCNEC who had curative surgery (n = 69), 38 cases were given a total cycle of 146 adjuvant chemotherapies: SCLC regimen (etoposide–platinum, n = 19), and NSCLC regimens including pemetrexed- (n = 9), gemcitabine-(n = 2), and paclitaxel/docetaxel-platinum (n = 8) doublet chemotherapy. In addition, one patient with stage III combined LCNEC and adenocarcinoma harboring EGFR-19DEL progressed 1 month after administration of icotinib and received surgery subsequently. For patients with inoperable localized or advanced LCNEC who received systemic chemotherapy (n = 60), 26 patients received an SCLC regimen (etoposide–platinum), 32 cases received NSCLC regimens chemotherapy, and 2 patients harboring EGFR-L858R mutation received icotinib as first-line treatment.
Till the last visit in June 2024, the median follow-up period was 24.0 months. The median PFS and OS were 7.9 (5.9–10.0) and 17.8 (13.5–22.2) months, respectively, for the entire cohort of patients. Female gender, advanced stage, lymph node involvement, and distant metastasis were associated with unfavorable OS (Table 2). Besides, surgery (HR = 0.316, 95%CI, 0.151–0.663, p = 0.002) and chemotherapy (HR = 0.447, 95%CI, 0.233–0.860, p = 0.016) significantly reduced death hazard compared with none treatment. Multivariate regression demonstrated gender, stage, and treatment as independent prognostic indicators for OS in LCNEC (Table 2).
Univariate and multivariate analyses of overall survival in patients with LCNEC.

LCNEC, large-cell neuroendocrine lung carcinoma; NSCLC, non-small-cell lung cancer; SCLC, small-cell lung cancer.
Failure incidence and patterns in localized LCNEC
Failure patterns were analyzed among patients with localized LCNEC who received curative surgery (n = 69) or chemoradiation (n = 8). Till the last visit, 61.0% of patients (47/77) had relapsed or progressive disease with SF being the main failure pattern (Figure 2(a)). In total, 25 cases had SF (32.5%), 15 cases had LRF (19.5%), and 7 cases with both LRF and SF (9.1%). Stage I and II LCNEC presented identical failure patterns and similar incidences (Figure 2(a)). Failure incidence increased with the tumor stage during the disease course. Patients with stage III disease had higher incidences of overall failure (OF; p = 0.044) and SF (p = 0.014) than those with stage I disease, but OF and SF were not significantly different with stage II disease; however, incidences of LRF did not differ with stage (Figure 2(b)).
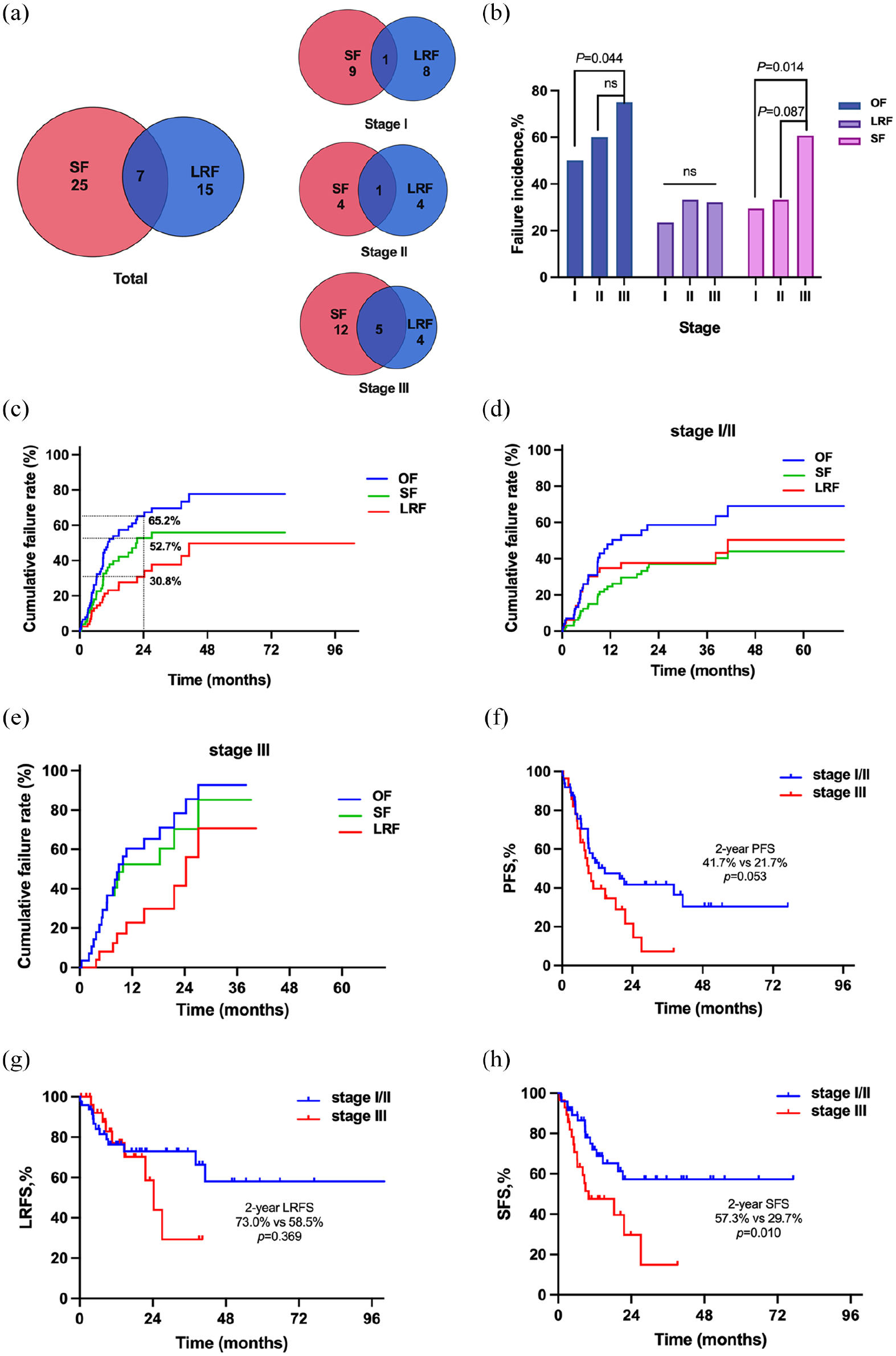
Failure patterns of localized LCNEC. (a) Venn plot of failure pattern of stage I–III LNCEC; (b) cumulative failure rates of localized LCNEC; (c) failure incidence and pattern according to stage; (d, e) cumulative failure rates of stage I/II (d) and III (e) LCENE; (f) comparison of 2-year cumulative failure rates between stage I/II and III LCNEC; (g, h) Comparison of LRF-free survival (g) and SF-free survival (h) between stage I/II and stage III LCNEC patients.
Most failures occurred within the first 2 years among this group of patients: the 1-year cumulative OF, SF, and LRF rates were 52.3%, 37.9%, and 23.1%, and the corresponding 2-year rates were 65.2%, 52.7%, and 30.8%, respectively (Figure 2(c)). Compared with stage I/II diseases, those with stage III LCNEC had slightly higher 2-year cumulative OF rate (63.6% vs 45.6%, p = 0.060) and higher SF rate (70.3% vs 37.2%, p = 0.010), with stage I/II patients achieving favorable PFS (2-year, 41.7% vs 21.7%; p = 0.053; Figure 2(f)) and SFS (2-year, 57.3% vs 29.7%; p = 0.010; Figure 2(h)) compared to stage III patients. Noteworthy, 2-year cumulative LRF rate (41.5% vs 37.6%, p = 0.369) and LRFS (73.0% vs 58.5%, p = 0.369) did not differ with the stage (Figure 2(g)).
Adjuvant chemotherapy after surgery for localized LCNEC
Since SF was the main failure pattern especially in the first 2 years of treatment, we further investigated the value of adding chemotherapy to surgical resection. Adjuvant chemotherapy was associated with reduced distant dissemination (2-year cumulative SF rate, 53.7% vs 41.3%; p = 0.055, Figure 3(a)), whereas the locoregional control was comparable (Figure 3(b)). Systemic disease control was demonstrated to translate into survival benefits (2-year PFS, 42.9% vs 22.6%, p = 0.032; 2-year OS,79.9% vs 37.1%, p < 0.001; Figure 3(c) and (d)), and the benefit was independent of the stage (Figure 3(e) and (f)). However, survivals did not differ between adjuvant SCLC regimen and NSCLC regimen for patients after surgery (2-year OS, 70.3% vs 66.3%, p = 0.387; Figure 4(a)).

Adjuvant chemotherapy reduced SF and improved survival in LCENC after surgery. (a–d) Comparison of SF (a), LRF (b), PFS (c), and OS (d) between patients receiving surgery alone and those receiving surgery followed by adjuvant chemotherapy; (e, f) survival benefit of adjuvant chemotherapy among patients with stage I/II (e) and III LCNEC (f).

Survival analysis of LCNEC according to different chemotherapy regimens. (a, b) Comparison of OS between SCLC and NSCLC regimens as adjuvant chemotherapy for stage I–IIIA LCNEC (a) and as salvage regimens for stage IIIB–IV LCNEC (b); (c, d) comparison of PFS (c) and OS (d) between immunotherapy-containing treatment and non-immunotherapy-containing treatment in advanced stage IIIB–IV LCNEC.
Systemic chemotherapy plus ICIs for advanced LCNEC
The standard chemotherapy regimen for advanced LCNEC has not been defined yet. We compared survival outcomes between patients receiving SCLC regimens and NSCLC regimens. It revealed that SCLC regimens and NSCLC regimens achieved comparable overall response rates (ORR, 44.8% vs 51.7%, p = 0.465) and 2-year OS (27.0% vs 34.9%, p = 0.649; Figure 4(b)) in advanced LCNEC.
Noteworthy, eight patients had incorporated ICIs into systemic chemotherapy as first-line treatment in this study: five cases with anti-PD-1 antibody and four cases with anti-PD-L1 blockade infusion. Six patients reached partial remission (PR), one reached stable disease, and the ORR was 75% (6/8). Three out of six patients who got PR had negative PD-L1 expression which might indicate no association between PD-L1 expression and ICI response. Patients receiving ICIs plus chemotherapy had much more favorable PFS in comparison to those receiving chemotherapy alone (median PFS, 10.3 vs 4.7 months, p = 0.045; Figure 4(c)). Adding ICIs to chemotherapy shows a tendency of prolonged OS (2-year OS, 87.5% vs 20.1%, p = 0.056; Figure 4(d)). However, these results should be interpreted cautiously since the number of patients receiving ICIs was quite small in this retrospective study.
Molecular feature and targeted therapy for LCNEC
In this study, 74 patients had targeted sequencing using a next-generation sequencing technique based on tumor samples. A total of 71 abnormalities were detected in 52 patients (70.3%). TP53 (26/74, 35.1%) was the most frequently mutated gene in LCNEC. RB1 mutation was detected only in one patient. Five patients (two pure LCNEC and three combined LCNEC) harbored EGFR alternation including L858R mutation (n = 4) or EGFR-19 deletion (n = 1). ALK rearrangement was detected in one patient with pure LCNEC and one patient with combined LCNEC. Patients were subdivided into NSCLC-like LCNEC (n = 48) and SCLC-like LCNEC (n = 26) according to the molecular classification of LCNEC in the published study. 23 No difference in survival was observed between the two molecular groups (2-year OS, 54.6% vs 41.8%, p = 0.677; Supplemental Figure 2).
Among patients with driver gene positive LCNEC, two cases (stage IV) received first-generation EGFR-TKI as first-line treatment: one dead soon and another one was still alive (OS, 44.8 months); one patient (stage IIIA) received no targeted therapy after surgery and adjuvant chemotherapy was still alive with no tumor (disease-free survival [DFS], 13.3 months); one case (stage IIIA) had EGFR-TKI as perioperative therapy and was still on the visit (OS, 24.3 months) and one case (stage IIIA) had third-generation EGFR-TKI as second-line treatment but progressed rapidly and dead of the tumor.
Transcriptome sequencing and DEP analysis
We performed transcriptome sequencing on six tumor tissues from three patients with LCNEC and 16 tumor tissues from 10 patients with NSCLC. Results revealed significant differences in gene expression profiles between LCNEC and NSCLC (Figure 5(a)). In addition to neuroendocrine proteins or markers (CHGB, CHGA, NCF1, and IGF2), NR1H3, CGA, AGT, LTF, SLC8A, ZG16B, CXCL9, and other genes were differentially expressed genes (DEGs) in LCNEC than NSCLC (Figure 5(b)). KEGG pathway and GESA analysis revealed that upregulated DEGs were enriched in antigen presentation, cytokine activity, cytokine–cytokine receptor interaction, and IFN-γ signaling pathways (Figure 5(c) and (d)). Furthermore, histone and DNA methylation-associated genes were downregulated in LCNEC patients, indicating a limited role of methylation in LCNEC which may not be ideal candidates for methylation-based drug therapies (Figure 5(d)). Protein–protein interaction network analysis showed that MMP9, AGT, COL1A2, COL1A1, and CXCL9 might play vital roles in the pathogenesis of LCNEC (Figure 5(e)).
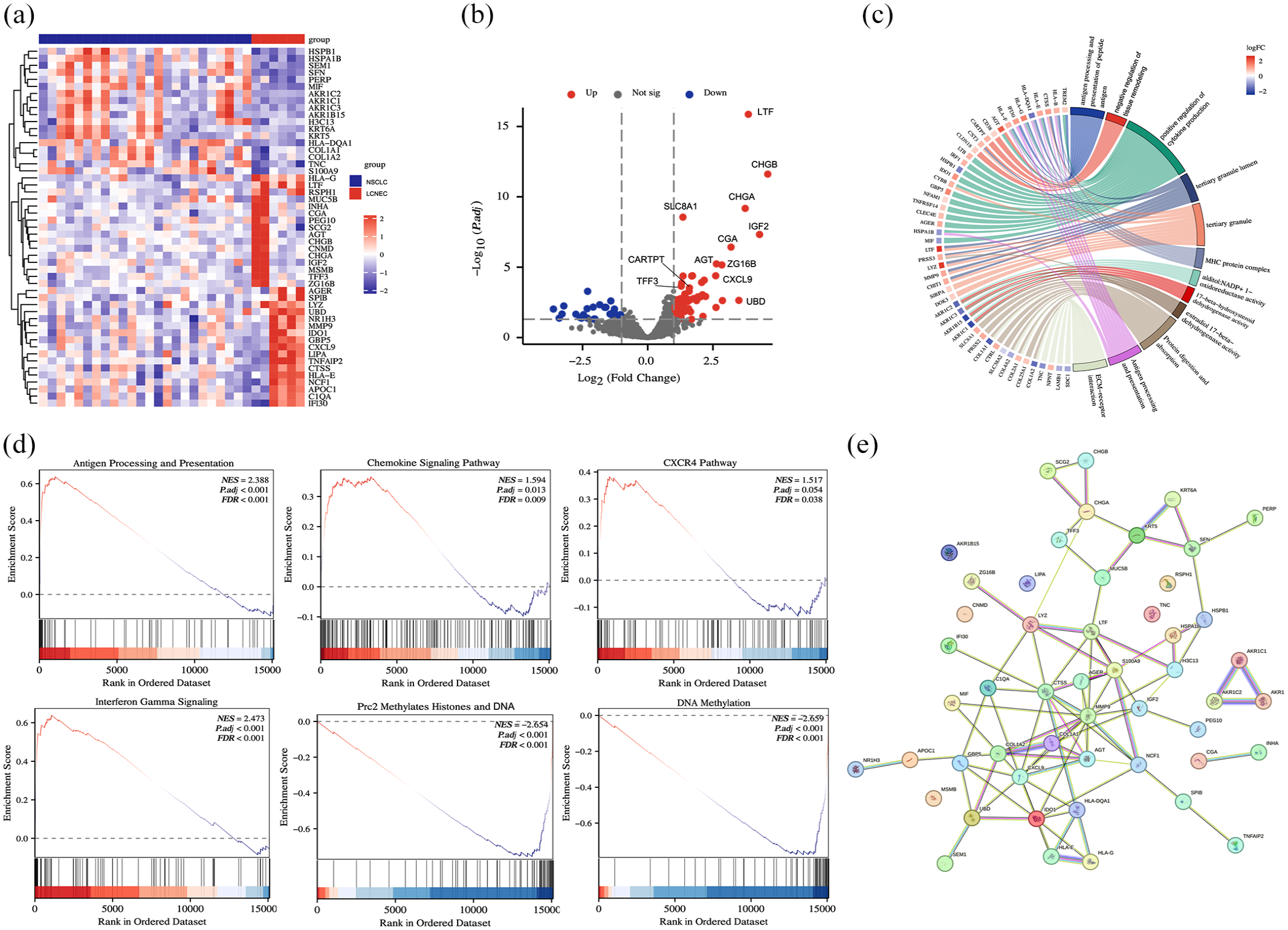
Gene expression analysis of LCNEC. (a) The heatmap chart of the top 50 DEGs in LCNEC versus NSCLC. (b) The volcano plots represented the DEGs between LCNEC and NSCLC. (c) The chord diagram of the DEPs-based functional enrichment pathway. (d) GSEA of antigen processing and presenting, chemokine signaling, CXCR4, IFN-γ, DNA, and histone methylation pathways. (e) PPI network analysis of upregulated DEGs in LCNEC.
Discussion
This is a comparably large sampled real-world study focused on the treatment, survival, and prognosis status of LCNEC among the Chinese population. Various treatment options including surgery, chemotherapy, targeted drugs, and immunotherapy were assessed and failure patterns of localized diseases were systemically investigated. Besides, transcriptome sequencing was conducted for differential expressed genes analysis. Our results indicate LCNEC is a highly aggressive systemic disease even in the early-stage phase and systemic chemotherapy is dispensable for the treatment; besides, it was demonstrated that antigen presentation/process, chemokine, CXCR4, and IFN-γ signaling pathways were upregulated in LCNEC, indicating the potential efficacy of ICI treatment.
Surgical resection is the backbone for the treatment of localized LCNEC.12,24 However, the significance of adjuvant chemotherapy and the optimal regimen have not been fully defined yet. In the present study, we analyzed the survival outcome as well as the failure pattern among localized LCNEC. Sixty-one percent of patients progressed or relapsed with SF being the main failure pattern. The 2-year cumulative SF rate was 52.7%. Besides, patients with stage III disease had a higher cumulative SF rate than those with stage I/II. Furthermore, our study demonstrated that adjuvant chemotherapy reduced distant dissemination (2-year cumulative SF rate, 53.7% vs 41.3%; p = 0.055) which translated into survival benefits (2-year PFS, 42.9% vs 22.6%, p = 0.032; 2-year OS,79.9% vs 37.1%, p < 0.001). In terms of regimens, no significant difference was observed between SCLC- or NSCLC-like regimens in our study, which was consistent with some reported studies.14,25,26 Conclusively, LCNEC is a highly aggressive malignancy even in the localized stage and distant dissemination is a main failure pattern of localized disease, indicating the important role of chemotherapy in this group of patients.
The optimal regimen for advanced LCNEC still remains unestablished.7,8,27 In the present study, SCLC regimens and NSCLC regimens achieved similar responses (p = 0.465) and survival (p = 0.465), which was in line with some previous studies (median OS, 7.0–12.6 months).4,15,27–29 Incorporation of ICIs might be a potential treatment option for advanced LCNEC. In a study of 34 LCNEC patients administered ICIs, the ORR was 29.4% and the median OS was 14.8 months. 30 Elizabeth Dudnik compared the outcome of LCNEC patients who received ICIs (n = 41) and patients who did not receive ICIs (n = 84), and the median OS was 12.5 and 8.4 months, respectively (p = 0.046). 11 In the present study, ICIs plus chemotherapy achieved significantly superior PFS than chemotherapy alone (10.3 vs 4.7 months, p = 0.045). In terms of biomarkers for predicting the efficacy of ICIs, PD-L1 expression might not be a reliable predictor. 11 However, it was reported that LCNEC with a high tumor mutation burden might benefit from ICIs. 19 These results suggest a positive impact of ICIs on OS in LCNEC.
Gene expression analysis in the present study also suggested that LCNEC might be vulnerable to immunotherapy. Compared to NSCLC for which ICIs have been a standard treatment option, LCNEC presented upregulated DEGs enriched in antigen-presenting and processing, chemokine, CXCR4, and IFN-γ signaling pathways. These results suggested the vulnerability of LCNEC to immunotherapy, in line with the favorable survivals of PD-1/PD-L1 blockade in LCNEC in the present study. However, the exact efficacy of immunotherapy in LCNEC needs validation from large-sample prospective clinical trials. Several reports suggested that epigenetic changes may play a key role in the pathogenesis of neuroendocrine neoplasms.31–33 Soldevilla et al. have assessed the epigenetic regulators microRNAs (miRNAs) miRNA in multiple endocrine neoplasias (MENs) including LCNEC, and they identified a signature of eight miRNAs that stratified patients in three prognostic groups (5-year OS, 80%, 66%, and 36%, respectively). 34 Consistently, our results revealed that epigenetic modification-related genes were upregulated in LCNEC. Besides, apart from traditional neuroendocrine proteins, we also found some potential markers for LCNEC, such as ARTX, also reported in a published study. 35
Limitation
This study has several limitations. Retrospective design and comparably small sample size of this study make some bias inevitable. Parts of our results should also be interpreted cautiously since the sample size was relevantly small. Currently, genotyping showed potential value in subtyping and guiding individualized treatment in LCNEC, 36 and no survival difference was observed between patients with SCLC-LCNEC and NSCLC-LCNEC in this study. The number of LCNEC cases available for the transcriptomic analysis was quite small; thus, the results should be considered exploratory. A large sample prospective study is on recruiting in our institute at present.
Conclusion
Our study suggested that LCNEC was a highly aggressive disease with a fairly poor prognosis. More intensive therapy including adjuvant chemotherapy and ICIs might be potential in an attempt to disease treatment. Large sample prospective studies are warranted in the future.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251324900 – Supplemental material for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China
Supplemental material, sj-docx-1-tam-10.1177_17588359251324900 for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China by Fei Qi, Minghang Zhang, Yi Han, Juan Du, Hongjie Yang, Hongmei Zhang, Yong Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-4-tam-10.1177_17588359251324900 – Supplemental material for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China
Supplemental material, sj-pdf-4-tam-10.1177_17588359251324900 for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China by Fei Qi, Minghang Zhang, Yi Han, Juan Du, Hongjie Yang, Hongmei Zhang, Yong Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tiff-2-tam-10.1177_17588359251324900 – Supplemental material for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China
Supplemental material, sj-tiff-2-tam-10.1177_17588359251324900 for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China by Fei Qi, Minghang Zhang, Yi Han, Juan Du, Hongjie Yang, Hongmei Zhang, Yong Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tiff-3-tam-10.1177_17588359251324900 – Supplemental material for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China
Supplemental material, sj-tiff-3-tam-10.1177_17588359251324900 for Treatment status, survival and gene expression analysis of large-cell neuroendocrine lung carcinoma: a real-world study in China by Fei Qi, Minghang Zhang, Yi Han, Juan Du, Hongjie Yang, Hongmei Zhang, Yong Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank all physicians and nurses who participated in this study as well as all enrolled patients at the Beijing Chest Hospital. We thank Professor Shaofa Xu for his contributions to language editing and some revisions of the article.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
