Abstract
Background:
To date, no specific studies have reported the use of dynamic serum tumor markers (STMs) as prognostic factors in patients with advanced non-small-cell lung cancer (NSCLC) who receive first-line immunotherapy. Therefore, it is unclear whether STMs can be used as a prognostic factor for first-line immunotherapy in advanced NSCLC.
Objectives:
To elucidate the role of STMs in monitoring immunotherapy response in advanced NSCLC. Patients were treated with first-line programmed cell death-1/programmed cell death ligand-1 inhibitors at four Chinese centers.
Design:
This was a multicenter retrospective study.
Methods:
Blood samples were collected at baseline and after 6–8 weeks of treatment. Computed tomography scans were used to evaluate treatment efficacy according to Response Evaluation Criteria in Solid Tumors (RECIST) 1.1. Post-treatment drops in STMs [Serum carcinoembryonic antigen (CEA), neuron-specific enolase (NSE), cytokeratin fragment 19 (CYFRA21-1), carbohydrate antigen 19-9 (CA19-9), and carbohydrate antigen 125 (CA125)] were decreased ⩾20% (Group C) over baseline was used as cutoff level for defining a marker response. If STMs were increased by ⩾20% after treatment, the therapeutic effect was limited (Group A). Patients with STM changes between a 20% increase or decrease were enrolled in Group B. In univariate and multivariate stepwise Cox regression analyses, STMs and RECIST responses were analyzed for their impact on progression-free survival (PFS) and overall survival (OS).
Results:
The analysis included 716 patients. By multivariate analysis, CEA, NSE, CYFRA21-1, CA19-9, and CA125 (Group A versus Group B and Group A versus Group C) were associated with significant differences in PFS. Similar results were observed in the OS analysis. Similar results were observed in the adenocarcinoma subgroup analyses. In squamous cell carcinoma subgroup analyses, there was no statistical difference in PFS (p = 0.147) or OS (p = 0.068) between Group A and Group B for CA125.
Conclusion:
The increase and decrease in serum levels of STMs might be reliable prognostic factors for immunotherapy efficacy in NSCLC patients.
Introduction
Lung cancer remains the deadliest malignancy worldwide.1,2 Non-small-cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer cases. 3 NSCLC also comprises several pathological subtypes, such as lung squamous cell carcinoma (LUSC) and lung adenocarcinoma (LUAD). 4 Most patients with NSCLC are diagnosed at an advanced stage and have no chance of surgical treatment. 3
Chemotherapy or targeted therapy improves the prognosis of some subtypes of lung cancer, but up to 90% of patients inevitably relapse, with 5-year survival rates below 20%.2–4 Numerous clinical studies have demonstrated that the use of immune checkpoint blockade targeting programmed cell death-1/programmed cell death ligand-1 (PD-1/PD-L1) has revolutionized the treatment of advanced lung cancer.5–10
PD-L1 expression is a simple biomarker for predicting the efficacy of immunotherapy in NSCLC.8–11 Recently, several studies used tumor immune-infiltrating cells or immune-related genes to predict immunotherapy efficacy; however, its principle is complicated and its cost is high, making it unsuitable for large-scale clinical applications.12,13
Serum carcinoembryonic antigen (CEA), neuron-specific enolase (NSE), cytokeratin fragment 19 (CYFRA21-1), carbohydrate antigen 19-9 (CA19-9), and carbohydrate antigen 125 (CA125) are common lung cancer markers. Their value as predictive and prognostic factors has been confirmed in many studies.14,15 CEAs are glycosyl phosphatidyl inositol cell-surface-anchored glycoproteins that are most widely found in gastric, pancreatic, lung, breast, and medullary thyroid carcinomas.15,16 High CEA levels can also be detected in some non-neoplastic conditions and smokers. 16 CYFRA 21-1 is a cytokeratin-19 fragment with high expression in pulmonary tissues.14,15 It is a sensitive and specific marker of NSCLC, especially LUSC, which correlates with tumor size, lymph node status, and disease stage.14,15 NSE is an enzyme found in mature neurons and cells of neuronal origin, which is also produced by small-cell lung cancer (SCLC) with a specificity of approximately 85% and is useful for the prognosis of survival, monitoring of treatment, and prediction of relapse.17,18 CA19-9 is usually attached to O-glycans on the surface of cells and is also a tumor marker used primarily in the management of pancreatic cancer and lung cancer. 19 CA125 is a glycoprotein that is increased in ovarian cancer, lung cancer, breast cancer, and other cancers and is currently used in the follow-up of patients with NSCLC and to evaluate response to therapy.14,15,20,21 Many studies have demonstrated that baseline and dynamic serum tumor marker (STM) levels can effectively predict the efficacy of chemotherapy and targeted therapy for lung cancer.22,23 However, only a few studies have reported the prognostic value of baseline and dynamic STMs for the efficacy of immunotherapy in NSCLC.24–27 According to a study published by Bello et al., 24 the reduction in serum levels of CYFRA21-1 or CEA might be a reliable biomarker for predicting nivolumab efficacy in patients with NSCLC. However, NSE was not significant for monitoring the efficacy of nivolumab. This study included only 70 patients who received nivolumab monotherapy and only 28 patients received first-line nivolumab monotherapy, which had a limited predictive effect on evaluating STMs. 24 A retrospective cohort study showed that decreasing leading STMs at first restaging predicts longer progression-free survival (PFS) and overall survival (OS) and identifies patients with favorable outcomes among initial radiological nonresponders in advanced NSCLC patients receiving immunotherapy. However, this study included only 32 patients who received first-line immunotherapy. 25 Recently, a retrospective study based on a Chinese population showed that dynamic changes in CEA, CA125, CYFRA21-1, and squamous cell carcinoma antigen (SCC-Ag) from baseline have prognostic value for patients with advanced NSCLC treated with immunotherapy. 26 A decrease in the serum levels of associated biomarkers was associated with favorable clinical outcomes. 26 However, this study included only 100 patients who received first-line immunotherapy. 26 In a validation study involving a large number of NSCLC patients receiving immune checkpoint therapy, Muller et al. 27 designed a model to accurately detect nonresponse in NSCLC, allowing for early and safe discontinuation of immunotherapy in a significant proportion of patients.
To date, no specific studies have reported the prognostic value of first-line immunotherapy efficacy using dynamic STMs in patients with advanced NSCLC. Therefore, whether STMs can be used as a prognostic factor in first-line immunotherapy for advanced NSCLC remains unclear. In the present study, we conducted a multicenter retrospective study to explore the prognostic value of dynamic changes in CEA, CYFRA21-1, NSE, CA19-9, and CA125 for first-line immunotherapy efficacy in advanced NSCLC.
Methods
Study design
This retrospective multicenter study included 716 patients with stage IIIB–IV NSCLC who received first-line immunotherapy between July 2017 and July 2021 at the Third Affiliated Hospital of Sun Yat-sen University, General Hospital of Southern Theater Command, the Third Affiliated Xiangya Hospital of Central South University, and Jiangxi Cancer Hospital (Figure 1).

Flowchart of this study.
Baseline covariates, including age, sex, histological type, clinical stage, smoking history, PD-L1 expression (22C3 PD-L1 antibody, Dako, Denmark), Eastern Cooperative Oncology Group Performance Status, metastatic sites (liver, lung, brain, bone, and adrenal), radiotherapy, and treatment (monotherapy or combination therapy) were collected.
Treatment regimen
Immunotherapy drugs included pembrolizumab with or without chemotherapy, nivolumab with or without chemotherapy, atezolizumab with chemotherapy, sintilimab with or without chemotherapy, camrelizumab with or without chemotherapy, and tirelizumab with chemotherapy. Chemotherapy regimens included platinum-based regimens with or without bevacizumab. The duration of immunotherapy was at least 6 weeks.
Treatment evaluation
The efficacy of immunotherapy was evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST, version 1.1), 28 including complete response (CR), partial response (PR), stable disease (SD), and progressive disease. Objective response rate (ORR) was defined as the percentage of CR + PR after immunotherapy. Efficacy was evaluated independently by two experienced doctors. Considering the possibility of pseudoprogression in immunotherapy, the determination of disease progression requires two consecutive radiological examinations.
STMs assay
STMs were collected before immunotherapy treatment and after 6–8 weeks. For the reported cohort, STM analyses were conducted using a cobas e 801immunoassay module (Roche Diagnostics, Rotkreuz, Switzerland) and the corresponding ElectroChemiLuminescence-ImmunoAssay kits acquired from Roche. According to the manufacturer’s instructions, the reference range was 0.00–5.00 ng/ml for CEA, 0.00–16.3 ng/ml for NSE, 0.00–3.30 ng/ml for CYFRA 21-1, 0.00–27.0 ng/ml for CA19-9, and 0.00–35.0 ng/ml for CA125. On the basis of the results from previous studies in advanced NSCLC patients treated with standard first-line chemotherapy and immunotherapy, a post-treatment drop in serum concentration ⩾20% (Group C) over baseline was used as the cutoff level for defining a marker response.14,24,26 If the STMs increased by more than 20% after treatment, the therapeutic effect was considered limited (Group A). Patients with an STM change between a 20% increase and a 20% decrease were enrolled in Group B. Therefore, we divided the treated population into three groups based on the ±20% cutoff.
Statistical analysis
All statistical analyses in this study were performed using R software v4.2.1 (https://www.r-project.org/, Auckland, New Zealand). Continuous variables are presented as mean ± standard deviation, and categorical variables are presented as numbers (%). The association between baseline STMs and PD-L1 expression levels and the association with dynamic changes in STMs and ORRs were calculated using a chi-square test. Univariate and multivariate analyses were performed to evaluate the prognostic impact on PFS and OS. PFS was calculated from the initiation of treatment to definite tumor progression, death, or the last follow-up. OS was calculated from the initiation of treatment to the date of death or last follow-up. All follow-up data were collected until 31 October 2022. PFS and OS curves were obtained using the Kaplan–Meier method and assessed using the log-rank test. The median, 95% confidence intervals (CIs), and p values from the log-rank tests are reported in the figures. The Cox proportional hazards regression model was used for univariate and multivariate analyses to assess the prognostic role of STMs adjusted for the possible confounding effect of all other factors included in the same model. All p values were two-sided, and values <0.05 were considered statistically significant.
Results
Patient characteristics
A total of 716 patients were enrolled in this study, including 390 with LUAD, 280 with LUSC, and 46 with other types of NSCLC (Table 1). The average age of the enrolled patients was 61.1 years, and the vast majority of patients were male. Nearly 10% more smokers or ex-smokers than nonsmokers were included in the study. According to the eighth edition Tumor, Lymph Node, Metastasi (TNM) staging of the International Lung Cancer Research Association, 29 86 patients with stage IIIB, 25 patients with stage IIIC, and 605 patients with stage IV were included. PD-L1 expression was positive (⩾1%) in most patients. A total of 451 patients were treated with pembrolizumab, 108 with nivolumab, 108 with sintilimab, and the rest with camrelizumab, atezolizumab, or tislelizumab. The mean value of baseline CEA was 57.29 ng/ml, NSE was 24.26 ng/ml, CYFRA21-1 was 15.69 ng/ml, CA19-9 was 45.99 ng/ml, and CA125 was 69.04 ng/ml (Table 1). Baseline CEA, NSE, CYFRA21-1, CA19-9, and CA125 levels were higher than normal in 409, 414, 618, 268, and 416 patients, respectively.
Characteristics of the patients at baseline.
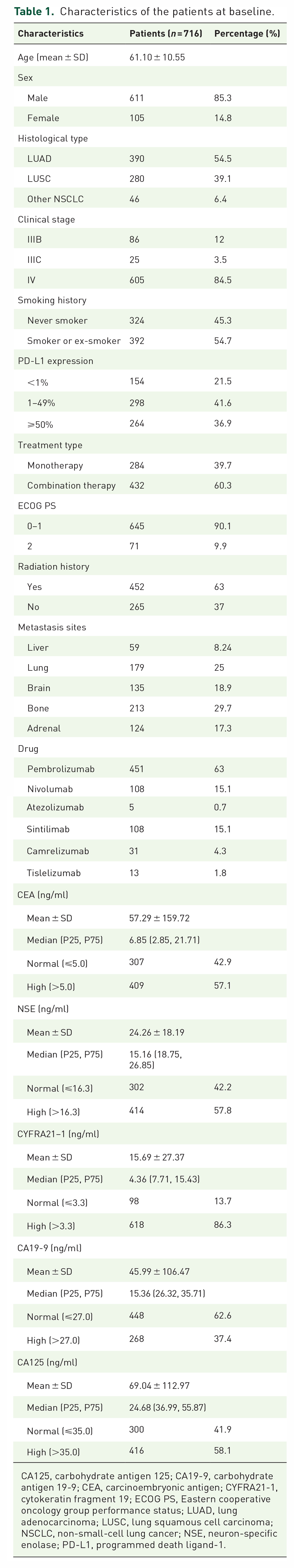
CA125, carbohydrate antigen 125; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin fragment 19; ECOG PS, Eastern cooperative oncology group performance status; LUAD, lung adenocarcinoma; LUSC, lung squamous cell carcinoma; NSCLC, non-small-cell lung cancer; NSE, neuron-specific enolase; PD-L1, programmed death ligand-1.
Association with dynamic changes in STMs and survival
Analysis of the whole population
Overall, the median PFS and OS of the 716 patients were 398 days (95% CI: 352–540 days) and 418 days (95% CI: 678–797 days), respectively. By univariate analysis, dynamic changes in CEA (Group A versus Group B and Group A versus Group C), NSE (Group A versus Group B and Group A versus Group C), CYFRA21-1 (Group A versus Group B and Group A versus Group C), CA19-9 (Group A versus Group B and Group A versus Group C), and CA125 (Group A versus Group B and Group A versus Group C) were associated with significantly different PFS and OS between subgroups (Tables 2, 3 and Figures 2, 3).
Prognostic factors for progression-free survival in patients with advanced NSCLC receiving first-line immunotherapy.*

More details in Supplemental Table S1.
CA125, carbohydrate antigen 125; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin fragment 19; ECOG PS, Eastern cooperative oncology group performance status; HR, hazard ratio; LUAD, lung adenocarcinoma; LUSC, lung squamous cell carcinoma; NSCLC, non-small-cell lung cancer; NSE, neuron-specific enolase; PD-L1, programmed death ligand-1; PFS, progression-free survival.
Prognostic factors for overall survival in patients with advanced NSCLC receiving first-line immunotherapy.*

More details in Supplemental Table S2.
CA125, carbohydrate antigen 125; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin fragment 19; ECOG PS, Eastern cooperative oncology group performance status; LUAD, lung adenocarcinoma; LUSC, lung squamous cell carcinoma; NSCLC, non-small-cell lung cancer; NSE, neuron-specific enolase; OS, overall survival; PD-L1, programmed death ligand-1.

Kaplan–Meier curves of progression-free survival in the whole population. Kaplan–Meier curves were based on dynamic changes in CEA (a), NSE (b), CYFRA21-1 (c), CA19-9 (d), and CA125 (e) levels.

Kaplan–Meier curves of overall survival in the whole population. Kaplan–Meier curves were based on dynamic changes in CEA (a), NSE (b), CYFRA21-1 (c), CA19-9 (d), and CA125 (e) levels.
By multivariate analysis, dynamic changes in CEA (Group A versus Group B and Group A versus Group C), NSE (Group A versus Group B and Group A versus Group C), CYFRA21-1 (Group A versus Group B and Group A versus Group C), CA19-9 (Group A versus Group B and Group A versus Group C), and CA125 (Group A versus Group B and Group A versus Group C) were associated with significantly different PFS and OS between subgroups (Tables 2 and 3).
Analysis of LUAD population
Overall, the median PFS and OS of the 390 patients were 446 days (95% CI: 372–528 days) and 760 days (95% CI: 704–931 days), respectively. By univariate analysis, dynamic changes in CEA (Group A versus Group B and Group A versus Group C), NSE (Group A versus Group B and Group A versus Group C), CYFRA21-1 (Group A versus Group B and Group A versus Group C), CA19-9 (Group A versus Group B and Group A versus Group C), and CA125 (Group A versus Group B and Group A versus Group C) in the LUAD population were associated with significantly different PFS and OS between subgroups (Tables 2, 3 and Supplemental Figures S1, S2).
By multivariate analysis, dynamic changes in CEA (Group A versus Group B and Group A versus Group C), NSE (Group A versus Group B and Group A versus Group C), CYFRA21-1 (Group A versus Group B and Group A versus Group C), CA19-9 (Group A versus Group B and Group A versus Group C), and CA125 (Group A versus Group B and Group A versus Group C) in the LUAD population were associated with significantly different PFS and OS between subgroups (Tables 2 and 3).
Analysis of LUSC population
Overall, the median PFS and OS of the 280 patients were 336 days (95% CI: 292–385 days) and 623 days (95% CI: 505–759 days), respectively. By univariate analysis, dynamic changes in CEA (Group A versus Group B and Group A versus Group C), NSE (Group A versus Group B and Group A versus Group C), CYFRA21-1 (Group A versus Group B and Group A versus Group C), CA19-9 (Group A versus Group B and Group A versus Group C), and CA125 (Group A versus Group B and Group A versus Group C) in the LUSC population were associated with significantly different PFS and OS between subgroups (Tables 2, 3 and Supplemental Figures S3, S4).
By multivariate analysis, dynamic changes in CEA (Group A versus Group B and Group A versus Group C), NSE (Group A versus Group B and Group A versus Group C), CYFRA21-1 (Group A versus Group B and Group A versus Group C), CA19-9 (Group A versus Group B and Group A versus Group C), and CA125 (Group A versus Group C) in the LUSC population was associated with significantly different PFS and OS between subgroups (Tables 2 and 3).
Association with baseline STMs and PD-L1 expression
PD-L1 expression was measured in all the patients. In the whole population, 562 (78.49%) patients had PD-L1 expression ⩾1%, and 264 (36.87%) patients had PD-L1 expression ⩾50% (Table 1). In the LUAD population, 315 (80.77%) patients had PD-L1 expression ⩾1% (Table 4). In the LUSC population, 214 (76.43%) patients had PD-L1 expression ⩾1% (Table 4). Surprisingly, only CA 19-9 was associated with PD-L1 expression in the LUAD population (p = 0.043). The remaining baseline STM concentrations did not correlate with PD-L1 expression.
The associations between STMs and PD-L1 expression levels in patients with advanced LUAD and LUSC.

CA125, carbohydrate antigen 125; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin fragment 19; LUAD, lung adenocarcinoma; LUSC, lung squamous cell carcinoma; NSE, neuron-specific enolase; PD-L1, programmed death ligand-1; STM, serum tumor marker.
Correlation between STM levels and tumor response
According to the dynamic change in serum CEA, NSE, CYFRA21-1, CA19-9, and CA125, there was a significant difference in ORR between the groups in both the LUAD population (Table 5). The same phenomenon can be seen in the LUSC population (Table 5).
The association between dynamic changes in STMs and ORRs.
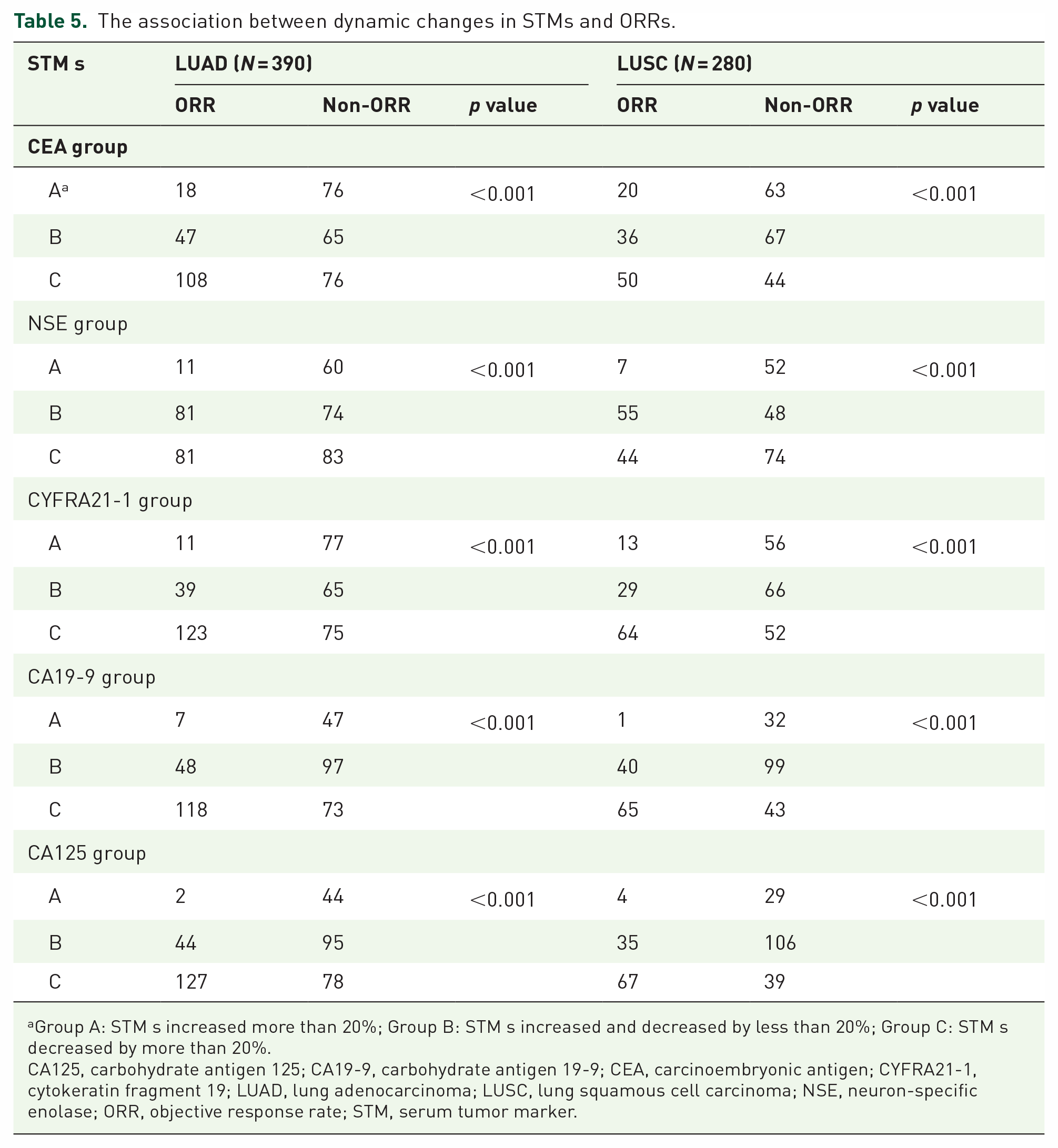
Group A: STM s increased more than 20%; Group B: STM s increased and decreased by less than 20%; Group C: STM s decreased by more than 20%.
CA125, carbohydrate antigen 125; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CYFRA21-1, cytokeratin fragment 19; LUAD, lung adenocarcinoma; LUSC, lung squamous cell carcinoma; NSE, neuron-specific enolase; ORR, objective response rate; STM, serum tumor marker.
Discussion
Immune checkpoint inhibitors, such as PD-1/PD-L1 inhibitors, can prolong survival in patients with advanced lung cancer and have demonstrated efficacy in several large-scale clinical studies.6,7,11 However, a substantial proportion of patients do not respond to PD-1/PD-L1 inhibitors or even experience serious adverse events that lead to treatment discontinuation. 30 By contrast, in a small percentage of patients who respond, immunotherapy appears to produce a long-term response with substantial survival benefits. 10 Therefore, the discovery of biomarkers with prognostic value will help in identifying patients who might benefit from such treatment. PD-L1 expression level, the most common predictor and prognostic factor of immunotherapy, is also limited. 31 Recently, many prognostic models based on tumor immune-infiltrating cells and immune-related genes have been developed, but they are complicated and expensive.12,13 Therefore, inexpensive, stable, and reliable biomarkers as prognostic factors of immunotherapy efficacy remain the focus of current research.
The discovery and large-scale clinical application of tumor markers in lung cancer have been ongoing for decades. Most studies have shown that tumor markers are highly sensitive for the diagnosis of malignant tumors.18,20,21,32 Some tumor markers can also predict the efficacy of chemotherapy and targeted therapy.14,15 However, the role of tumor markers in evaluating the efficacy of immunotherapy remains controversial.24–26,33 With the establishment of immunotherapy as the first-line treatment for advanced NSCLC, an increasing number of patients are receiving first-line immunotherapy. To date, no studies have focused specifically on the association between STMs and the efficacy of first-line immunotherapy. Previously published studies did not make a detailed distinction between the immunotherapy treatment lines, which may lead to bias in clinical applications.
In this study, we evaluated the dynamic changes in STMs routinely measured in clinical practice to explore their relationship with immunotherapy response in patients with advanced NSCLC. We confirmed that dynamic changes in CEA, NSE, CYFRA21-1, CA19-9, and CA125 levels correlated with the efficacy and prognosis of patients with advanced NSCLC treated with first-line PD-1/PD-L1 inhibitors. Similar results were observed in LUAD and LUSC subgroup analyses. Therefore, monitoring changes in STM levels may be a promising prognostic factor for patients with advanced NSCLC treated with immunotherapy.
Previous studies have demonstrated that dynamic reductions in serum CEA and CYFRA21-1 levels can predict the efficacy of immunotherapy,24,26 and our study also observed that a ⩾ 20% reduction in CEA or CYFRA21-1 levels was associated with better survival. This suggests a possible role as a marker for monitoring the tumor response during the initial phase of immunotherapy treatment. In LUAD and LUSC, a ⩾ 20% reduction in CEA and CYFRA21-1 was significantly associated with response to immunotherapy. Muller et al. and Moritz et al. have demonstrated that increasing concentration correlates with worse response and survival.27,34
Interestingly, NSE is a classic tumor marker in SCLC, and as reported in previous studies, dynamic changes in serum NSE levels do not predict immunotherapy efficacy in patients with advanced NSCLC. 24 In fact, when we divided our dataset using the grouping method of previous studies, 24 we were also unable to confirm the relationship between dynamic changes in serum NSE and clinical outcomes (PFS, p = 0.19; OS, p = 0.09, data not shown). However, we separated the dynamic changes in serum NSE by ±20% into three groups and group B (−20% to +20%) had the best PFS and OS compared to Groups C (reduction ⩾ 20%) and A (elevation ⩾ 20%). The underlying biological mechanisms are worth exploring in future studies. In addition, this is different from the results we found for serum CEA, CYFRA21-1, CA19-9, and CA125, which showed a positive correlation between the magnitude of decline and survival.
In addition, similar to the dynamic changes in serum CEA and CYFRA21-1, we demonstrate in the whole population, including subgroup analyses in the LUAD and LUSC populations, that a 20% reduction in CA19-9 or CA125 appeared to have longer PFS and OS, and a 20% increase in them appeared to have worse PFS and OS. When a 20% increase in serum CA19-9 level was observed, only seven patients with LUAD and one patient with LUSC achieved ORR. When a 20% increase in serum CA125 levels was observed, only two patients with LUAD and four patients with LUSC achieved ORR. In summary, the correlation between a 20% increase in CA19-9 or CA125 levels and a lower ORR and worse survival was highly significant. Therefore, in clinical practice, we can distinguish between populations with better or worse outcomes based on dynamic changes in CA19-9 or CA125 levels. This may help doctors make clinical decisions; for example, given the poor survival outcome and the extremely low probability of achieving manageable disease, patients may need to stop immunotherapy and change treatment modalities as early as possible in the absence of evidence of radiological response and a 20% increase in CA19-9 or CA125.
To the best of our knowledge, this is the largest cohort study available to assess the relationship between routinely measured STMs and the outcomes and prognosis of patients treated with immunotherapy. This study refines the prognostic power of STMs and strengthens their prognostic value by calculating dynamic changes in STMs using ±20% as a cutoff point and dividing the population into three groups. Notably, owing to the inclusion of a larger number of patients, this is the first study to use this classification method to determine the relationship between STMs and the prognostic value of immunotherapy.
Our study had some limitations. First, retrospective studies have natural limitations, but the inclusion of a large number of patients from multiple centers in our study, along with adjusting for possible confounding factors, makes the findings more reliable. Furthermore, the significant differences between subgroups based on the degree of dynamic changes in STMs strongly suggest that these biomarkers have a significant impact on the prognosis. Second, the STMs included in our study were elevated in many malignancies and were not highly specific. Therefore, our study excluded patients with a combination of other malignancies. Third, we analyzed only the five included STMs individually and did not analyze them in combination. However, considering that dynamic changes in individual tumor markers can effectively distinguish the treatment-benefit population, a complex permutation of STMs seems unnecessary.
Conclusion
In conclusion, we propose a new strategy to monitor the dynamics of STMs and highlight their importance as potential prognostic biomarkers in advanced NSCLC using first-line immunotherapy. This is the largest study to date to analyze the relationship between dynamic changes in STMs and efficacy, demonstrating that dynamic changes in CEA, NSE, CYFRA21-1, CA19-9, and CA125 can be used as reliable prognostic markers in patients with NSCLC treated with first-line immunotherapy. Increased or decreased levels of relevant serum biomarkers are correlated with worse or better clinical outcomes. Further prospective studies are needed to evaluate the role of these serum markers with different threshold values, as well as to further confirm these findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231206282 – Supplemental material for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359231206282 for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study by Xiongwen Yang, Yi Xiao, Yubin Zhou, Huiyin Deng, Zihao Yuan, Longyan Dong, Jun Lan, Hao Hu, Jian Huang and Shaohong Huang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231206282 – Supplemental material for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study
Supplemental material, sj-docx-2-tam-10.1177_17588359231206282 for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study by Xiongwen Yang, Yi Xiao, Yubin Zhou, Huiyin Deng, Zihao Yuan, Longyan Dong, Jun Lan, Hao Hu, Jian Huang and Shaohong Huang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-3-tam-10.1177_17588359231206282 – Supplemental material for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study
Supplemental material, sj-pdf-3-tam-10.1177_17588359231206282 for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study by Xiongwen Yang, Yi Xiao, Yubin Zhou, Huiyin Deng, Zihao Yuan, Longyan Dong, Jun Lan, Hao Hu, Jian Huang and Shaohong Huang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-4-tam-10.1177_17588359231206282 – Supplemental material for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study
Supplemental material, sj-pdf-4-tam-10.1177_17588359231206282 for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study by Xiongwen Yang, Yi Xiao, Yubin Zhou, Huiyin Deng, Zihao Yuan, Longyan Dong, Jun Lan, Hao Hu, Jian Huang and Shaohong Huang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-5-tam-10.1177_17588359231206282 – Supplemental material for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study
Supplemental material, sj-pdf-5-tam-10.1177_17588359231206282 for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study by Xiongwen Yang, Yi Xiao, Yubin Zhou, Huiyin Deng, Zihao Yuan, Longyan Dong, Jun Lan, Hao Hu, Jian Huang and Shaohong Huang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-6-tam-10.1177_17588359231206282 – Supplemental material for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study
Supplemental material, sj-pdf-6-tam-10.1177_17588359231206282 for Dynamic monitoring of serum tumor markers as prognostic factors in patients with advanced non-small-cell lung cancer treated with first-line immunotherapy: a multicenter retrospective study by Xiongwen Yang, Yi Xiao, Yubin Zhou, Huiyin Deng, Zihao Yuan, Longyan Dong, Jun Lan, Hao Hu, Jian Huang and Shaohong Huang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
