Abstract
Background:
Immunotherapy blocking programmed death-1 (PD-1)/programmed death ligand-1 (PD-L1) has revolutionized the treatment of extensive-stage small-cell lung cancer (SCLC), but only with limited real-world efficacy data; evidence from immunotherapy for other pulmonary neuroendocrine carcinoma (PNEC) is scarce.
Objective:
The purpose of this study is to evaluate the efficacy of receiving PD-1/PD-L1 inhibitors in patients with advanced PNEC and explore factors related to survival prognosis, providing clues for treatment for patients with advanced PNEC.
Methods:
In all, 203 patients with advanced PNEC who received PD-1/PD-L1 inhibitors between January 2019 and December 2021 were retrospectively analyzed. Kaplan–Meier curves were constructed for progression-free survival (PFS) and overall survival (OS).
Results:
For the 203 patients, the objective response rate (ORR) was 48.3%, the disease control rate (DCR) was 83.3%, the median PFS (mPFS) was 6.0 months, and the median OS (mOS) was 13.1 months. Among them, the histology was 166 SCLC, 13 large-cell neuroendocrine carcinoma, and 24 other unspecified PNEC. Histologically, no significant difference was observed in PFS (p = 0.240) or OS (p = 0.845). In first-line (1L) treatment (N = 125), patients received chemoimmunotherapy and had an ORR of 64.8%, DCR of 92.0%, mPFS of 6.6 months, and mOS of 14.9 months. In second-line (2L) or later-line setting, the ORR, DCR, mPFS, and mOS were 21.8%, 69.2%, 4.4, and 9.4 months; immunotherapy plus small-molecule antiangiogenic agents showed significantly greater PFS than immunotherapy monotherapy or chemoimmunotherapy (6.4 vs 1.4 vs 3.7 months, p = 0.041). Patients without liver metastasis had superior PFS (7.0 vs 5.1 months, p < 0.001) and OS (19.2 vs 9.6 months, p < 0.001) than those with liver metastasis.
Conclusion:
In clinical practice, PD-1/PD-L1 inhibitors are effective in patients with advanced PNEC, regardless of the pathological histology. The efficacy of 1L immunochemotherapy is worthy of recognition, and the addition of small-molecule antiangiogenic agents to immunotherapy in 2L or later-line treatment provides a better survival trend.
Design:
Retrospective study.
Keywords
Introduction
In the fifth edition of the World Health Organization (WHO) Classification of Thoracic Tumors, pulmonary neuroendocrine neoplasms (PNEN) are classified into four categories: typical carcinoid (TC), atypical carcinoid (AC), small-cell lung cancer (SCLC), and large-cell neuroendocrine carcinoma (LCNEC). 1 With respect to the high heterogeneity in the biological characteristics of these four major subtypes, they can be further classified into low-intermediate grade pulmonary neuroendocrine tumor (PNET) with high differentiation, consisting of TC and AC, and low-differentiated high-grade pulmonary neuroendocrine carcinoma (PNEC), including SCLC and LCNEC. Notably, PNET, independent of tobacco exposure, is a less aggressive subtype of PNEN with a favorable prognosis. Conversely, PNEC is strongly associated with smoking and is a highly aggressive malignancy characterized by rapid tumor growth and early metastasis with an extremely poor prognosis.
In patients with extensive-stage SCLC (ES-SCLC), despite initial sensitivity to first-line (1L) chemotherapy, local recurrence or distant metastasis inevitably occurs, and there are limited alternative and subsequent therapeutic options. 2 With regard to advanced LCNEC, there is currently no approved therapy for these patients and, in fact, no prospective data are available due to its rarity. Based on small sample studies and case reports, chemotherapy remains the primary treatment modality, but the optimal regimen is still debated; usually, chemotherapy regimens similar to those used for SCLC are clinically recommended, whereas it exhibits a lower response rate compared to ES-SCLC.3,4
In recent years, immune checkpoint inhibitors (ICIs) targeting programmed death-1 (PD-1)/programmed death ligand-1 (PD-L1), either as a monotherapy or in combination with chemotherapy, have changed the treatment landscape for several thoracic malignancies, including non-small-cell lung cancer (NSCLC), SCLC, malignant mesothelioma, and thymic tumors. The PD-L1 inhibitors atezolizumab and durvalumab combined with chemotherapy, in the IMpower133 and CASPIAN trials, achieved an unprecedented median overall survival (mOS) of 12.0–13.0 months, leading to their approval as a standard 1L treatment for ES-SCLC.5,6 Subsequently, the CAPSTONE-1 and ASTRUM-005 trials reported mOS of 15.0–16.0 months.7,8 More recently, the EXTENTORCH and RATIONALE-312 trials reported an mOS of 14.0–16.0 months.9,10 Despite the favorable results of these clinical trials, real-world efficacy data are required to further complement these findings. However, the available real-world data are mostly obtained from relatively small cohorts.11,12 In addition, clinical evidence has indicated that LCNEC is also associated with a high tumor mutational burden, as is SCLC, and exhibits higher PD-L1 expression compared to SCLC.13,14 Dudnik et al. conducted a retrospective study of patients with advanced LCNEC and found that ICIs (n = 41) significantly improved mOS over chemotherapy (12.5 months vs 8.4 months). 15 A real-world study that evaluated the efficacy of nivolumab in 17 previously treated patients with advanced LCNEC achieved an objective response rate (ORR) of 29.4%, an mOS of 12.1 months, and a median progression-free survival (mPFS) of 3.9 months. 16 Collectively, these data suggest that ICIs are a potentially effective therapeutic option for treating advanced LCNEC.
Although clinical studies have demonstrated the efficacy of ICIs in SCLC, real-world data remain limited. However, owing to the rarity and limited number of clinical trials of LCNEC treated with ICIs, the therapeutic strategy remains controversial. Thus, this study was conducted to evaluate the real-world efficacy of PD-1/PD-L1 inhibitors in patients with advanced PNEC and to analyze potential factors that may affect the efficiency and prognosis.
Materials and methods
Patients
A total of 396 patients with PNEN who received PD-1/PD-L1 inhibitors at Zhejiang Cancer Hospital between January 2019 and December 2021 were screened. Of these, 203 eligible patients with advanced PNEC were enrolled. The inclusion criteria were histologically or cytologically confirmed PNEC, at least one measurable lesion, a stage IV imaging diagnosis, at least two cycles of PD-1/PD-L1 inhibitors therapy, and complete clinical and survival data. We further classified PNEC into SCLC, LCNEC, and unspecified PNEC, with the unspecified PNEC being the diagnosis for biopsy specimens that were difficult to identify due to severe tissue compression or extensive necrosis. In addition, the exclusion criteria were enrollment in clinical trials and receipt of non-standard regimens. Data collected included age, sex, Eastern Cooperative Oncology Group performance status (ECOG PS), smoking status, histology, lines of ICI therapy, ICI treatment regimens, number of organs with metastasis, baseline brain and liver metastases, duration of response, and outcome data. Figure 1 illustrates the patient selection process. Immunotherapy includes 1L and second-line or later line (⩾2L) of treatment, where the ⩾2L cohort specifically comprises patients who have previously received one or more lines of other treatments and then received immunotherapy after disease progression. In addition, ICI treatment regimens include monotherapy, combination chemotherapy, and combination small-molecule antiangiogenic therapy. Common anti-angiogenic agents include apatinib and anlotinib. Apatinib, a selective small-molecule tyrosine kinase inhibitor, effectively inhibits tumor angiogenesis by blocking vascular endothelial growth factor receptor (VEGFR-2) activity, while anlotinib not only achieves a similar anti-angiogenic effect by blocking VEGFR pathways but also contributes to a synergistic therapeutic impact by modulating the reprogramming of the tumor immune microenvironment. This study followed the principles of the Declaration of Helsinki, and individual consent for this retrospective analysis was waived. The research protocol received approval from the Ethics Committee of the Zhejiang Cancer Hospital (IRB No. 2023-432). The reporting of this study conforms to the statement of ESMO Guidance for Reporting Oncology real-world evidence (Supplemental File 1). 17
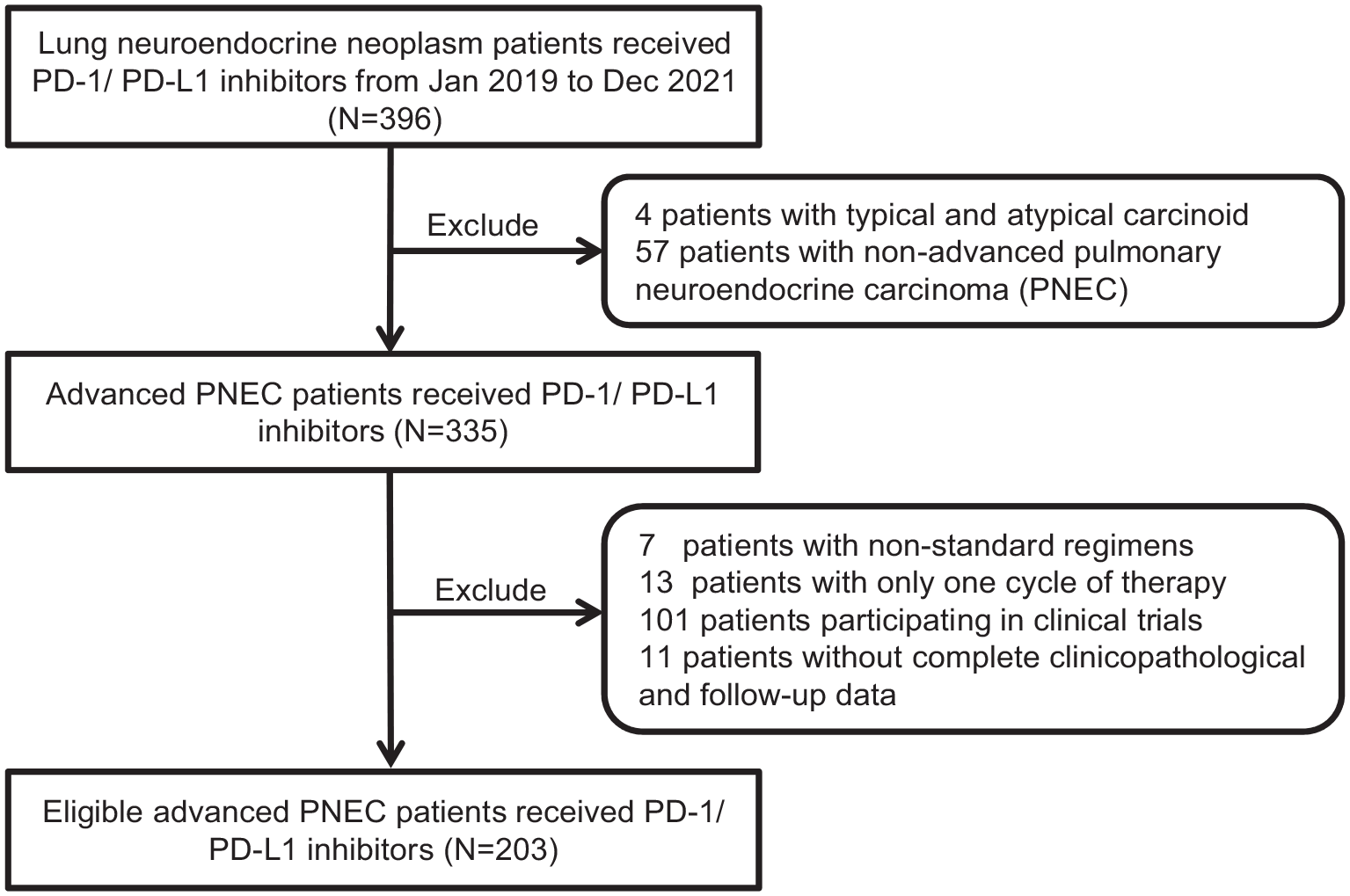
Study flowchart. A total of 396 patients with lung neuroendocrine neoplasm who received PD-1/PD-L1 inhibitors treatment were screened and 203 patients were finally enrolled.
Efficacy assessment and study endpoints
According to the Immune Response Evaluation Criteria In Solid Tumors (iRECIST), the efficacy of antitumor therapy is categorized into complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). 18 The primary endpoint was OS, defined as the period from the initiation of PD-1/PD-L1 inhibitors therapy until death from any cause or the latest follow-up date. Secondary endpoints included progression-free survival (PFS), ORR, and disease control rate (DCR). PFS was calculated as the time between the initiation of PD-1/PD-L1 inhibitors and disease progression or death due to different causes. The enrolled patients were followed up until October 18, 2023.
Statistical analyses
Categorical variables were presented as numbers and percentages, and continuous variables were presented as medians. Survival analysis was conducted using Kaplan–Meier and Log-rank methods. Cox regressions were used for prognostic analysis to obtain hazard ratios (HRs) and 95% confidence intervals (95% CIs). The multivariate Cox regression analysis obtained variables with p < 0.10 in the univariate analysis. Statistical significance was set at p < 0.05, and two-tailed p values were calculated in all reports. All analyses were performed using SPSS (version 26.0) and R (version 4.3.2).
Results
Patient characteristics
Table 1 shows the baseline characteristics of included patients. Of 203 patients, 64.5% (131/203) were age ⩽65 years, 91.1% (n = 185) were male, 78.3% (n = 159) were current or former smokers, and 76.4% (n = 155) had an ECOG PS ⩾1. Histologically, 166 (81.8%) patients had SCLC, 13 (6.4%) had LCNEC, and the remaining had unspecified PNEC. A total of 125 (61.6%) patients received PD-1/PD-L1 inhibitors plus chemotherapy as 1L treatment, while 78 patients (38.4%) received ⩾2L treatments, including 31 patients who received treatments beyond the 2L. In ⩾2L setting, 13 (16.7%), 37 (47.4%), and 28 (35.9%) patients received PD-1/PD-L1 inhibitors monotherapy, chemoimmunotherapy, and immunotherapy plus small-molecule antiangiogenic agents, respectively. The specific drugs and proportions of patients treated with PD-1 or PD-L1 inhibitors and small molecule anti-angiogenic drugs are detailed in Supplemental Tables S1 and S2. The metastatic organ count was ⩾3 in 105 (51.7%) patients, with 83 (40.9%) liver metastases and 72 (35.5%) brain metastases.
Baseline characteristics included advanced PNEC patients according to the lines of ICI treatment.
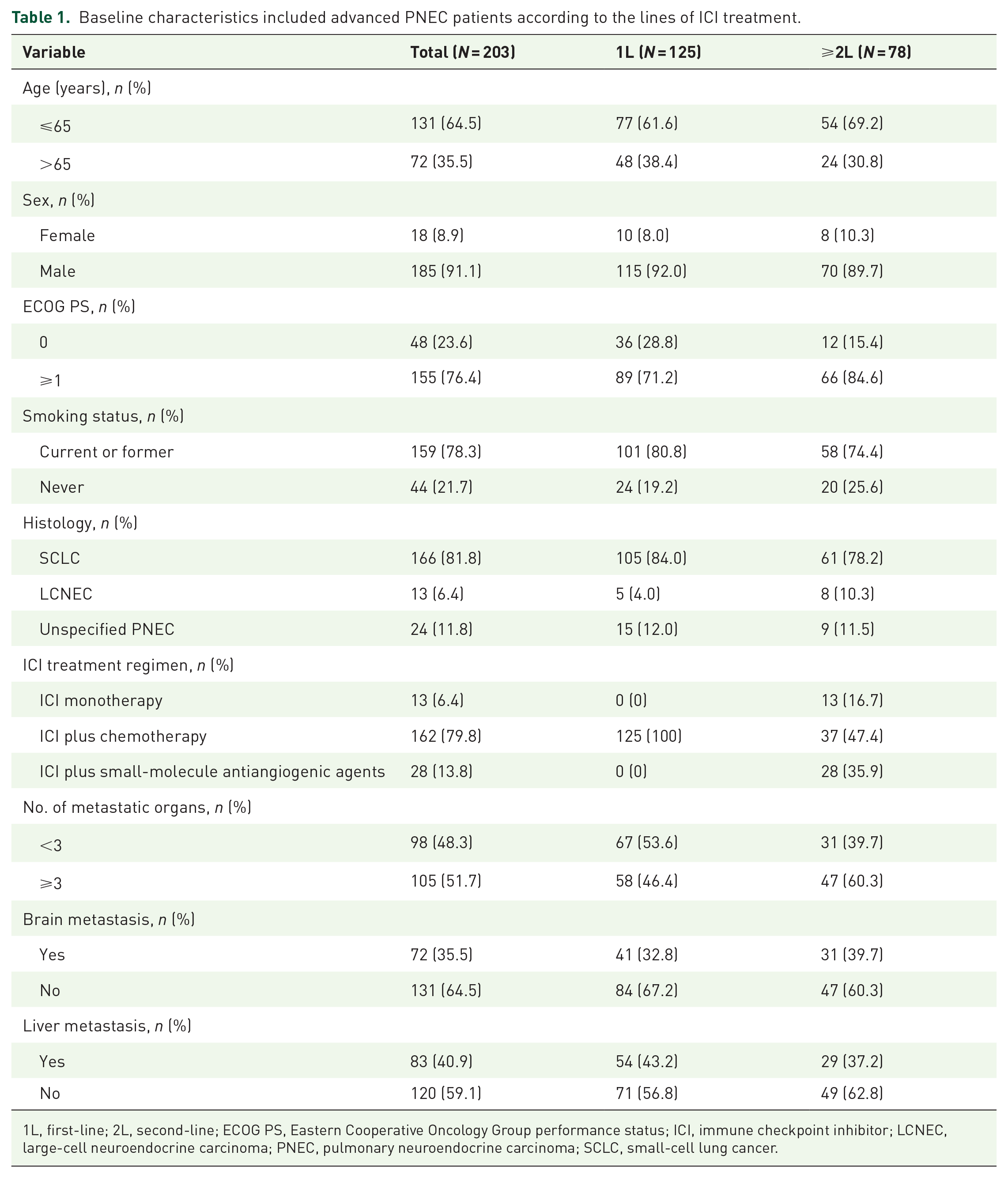
1L, first-line; 2L, second-line; ECOG PS, Eastern Cooperative Oncology Group performance status; ICI, immune checkpoint inhibitor; LCNEC, large-cell neuroendocrine carcinoma; PNEC, pulmonary neuroendocrine carcinoma; SCLC, small-cell lung cancer.
Efficacy and subgroup analysis of PD-1/PD-L1 inhibitors
Of the 203 patients, 1 (0.5%) achieved CR, 97 (47.8%) achieved PR, 71 (35.0%) achieved SD, and 34 (16.7%) achieved PD, with an ORR of 48.3% and a DCR of 83.3%. In addition, the ORR and DCR were 64.8% and 92.0% in the 1L setting, and 21.8% and 69.2% in the ⩾2L setting, respectively (Table 2).
Evaluation of efficacy in total, 1L, and ⩾2L patients.

1L, first-line; 2L, second-line; CR, complete response; DCR, disease control rate; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
With a median follow-up time of 37.5 months (95% CI: 32.7–42.2), mPFS was 6.0 months and mOS was 13.1 months in the overall population. For histological subgroups, no significant difference was observed in mOS (13.3 vs 13.3 vs 9.7 months, p = 0.845) and mPFS (6.2 vs 3.8 vs 5.7 months, p = 0.240) among patients with SCLC, LCNEC, and unspecified PNEC (Figure 2).
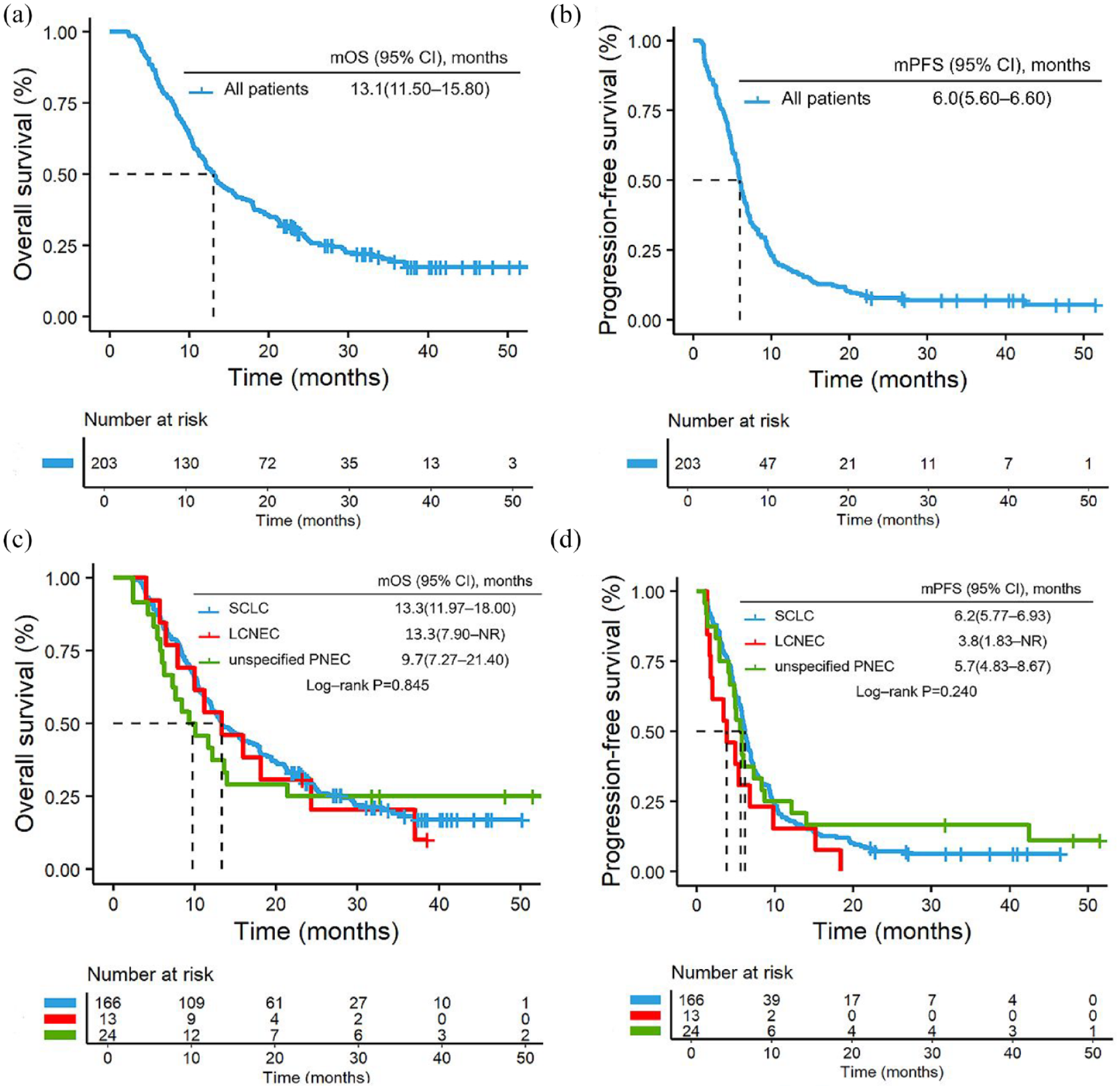
Kaplan–Meier analysis for OS (a) and PFS (b) in all patients receiving immunotherapy drugs; PFS and OS for different histological types. (c) The median OS of SCLC was 13.3 months, of LCNEC was 13.3 months and that of unspecified PNEC was 9.7 months. (d) The median PFS of SCLC was 6.2 months, of LCNEC was 3.8 months and that of unspecified PNEC was 5.7 months.
In the 1L treatment group, the mPFS was 6.6 months, and the mOS was 14.9 months. Considering the histological types, SCLC had an mOS of 15.7 months, an mPFS of 6.9 months, a 1-year survival rate of 61.0%, and a 2-year survival rate of 23.8%. LCNEC had a mOS of 13.3 months, an mPFS of 6.8 months, a 1-year survival rate of 60.0%, and a 2-year survival rate of 20.0%. Among patients who received ICIs as the ⩾2L therapy, the mPFS and mOS were 4.4 and 9.4 months, respectively. Furthermore, patients treated with ICIs plus small-molecule antiangiogenic agents showed a significant improvement in mPFS (6.4 vs 1.4 vs 3.7 months, p = 0.041) compared to patients treated with immunotherapy alone or with chemotherapy, but there was no statistical difference in mOS (9.3 vs 9.5 vs 12.0 months, p = 0.930) (Figure 3). The OS of PD-1 and PD-L1 inhibitors showed no significant statistical difference (1L: 15.7 vs 24.7 months, p = 0.400; ⩾2L: 8.8 vs 14.2 months, p = 0.067), nor did the PFS (1L: 6.5 vs 7.0 months, p = 0.970; ⩾2L: 4.0 vs 3.8 months, p = 0.700), regardless of 1L or ⩾2L treatment. In addition, the type of small molecule anti-angiogenic agents did not impact the efficacy in ⩾2L therapies (mOS: 8.8 (Anlotinib) vs 9.9 (Apatinib) months, p = 0.548; mPFS: 6.8 (Anlotinib) vs 6.0 (Apatinib) months, p = 0.251) (Supplemental Figures S2 and S3).

Kaplan–Meier analysis for PFS and OS for different lines of ICI therapy, 1L histological types, and ⩾2L treatment regimens. (a) The median OS of 1L was 14.9 months and that of ⩾2L was 9.4 months. (b) The median PFS of 1L was 6.6 months and that of ⩾2L was 4.4 months. (c) The median OS of SCLC was 15.7 months, of LCNEC was 13.3 months and that of unspecified PNEC was 12.2 months in 1L. (d) The median PFS of SCLC was 6.9 months, of LCNEC was 6.8 months and that of unspecified PNEC was 5.6 months in 1L. (e) The median OS for those receiving monotherapy was 9.5 months, for those with added chemotherapy was 12.0 months, and for those receiving added antiangiogenic therapy was 9.3 months in ⩾2L. (f) The median PFS for patients receiving monotherapy was 1.4 months, for those with added chemotherapy was 3.7 months, and for those who received added antiangiogenic therapy was 6.4 months in ⩾2L.
Subgroup analyses revealed similar positive results in patients without liver metastases compared to those with baseline liver metastases (mOS: 19.2 vs 9.6 months, p < 0.001; mPFS: 7.0 vs 5.1 months, p < 0.001). However, when comparing patients with baseline brain metastases to those without, no differences were observed in mOS (12.2 vs 13.7 months, p = 0.766) and mPFS (5.8 vs 6.2 months, p = 0.484) (Figure 4). Patients with ⩾3 metastatic organs exhibited statistically inferior mOS and mPFS compared to those with fewer than three metastatic organs (mOS: 11.1 vs 18.1 months, p = 0.003; mPFS: 5.6 vs 6.8 months, p < 0.001). Moreover, further exploratory analysis revealed that even after excluding liver metastases, the number of metastatic organs still significantly impacted patient outcomes (Supplemental Table S3 and Supplemental Figure S1).

Kaplan–Meier analysis for PFS and OS in all patients with different numbers of organs with metastasis, liver metastasis, and brain metastasis. (a) The median OS of < 3 organs was 18.1 months and that of ⩾3 organs was 11.1 months. (b) The median PFS of < 3 organs was 6.8 months and that of ⩾3 organs was 5.6 months. (c) The median OS of liver metastasis was 9.6 months and 19.2 months for those with no liver metastasis. (d) The median PFS of liver metastasis was 5.1 months and that of no liver metastasis was 7.0 months. (e) The median OS of patients with brain metastasis was 13.7 months, compared to 12.2 months for those with no brain metastasis. (f) The median PFS of patients with brain metastasis was 6.2 months, and 5.8 months for those with no brain metastasis.
Prognostic analysis of advanced PNEC patients treated with PD-1/PD-L1 inhibitors
In the univariate Cox analyses, the lines of ICI therapy, ICI treatment regimen, number of organs with metastasis, liver metastases, and number of metastatic organs excluding liver metastasis showed a significant correlation with PFS, whereas sex, smoking status, lines of ICIs therapy, number of organs with metastasis, and liver metastases showed a significant correlation with OS. Multivariate Cox analyses found that lines of ICI therapy (HR: 2.01, 95% CI: 1.37–2.95, p = 0.000), the number of organs with metastasis (HR: 1.41, 95% CI: 1.01–1.97, p = 0.042), and liver metastases (HR: 0.62, 95% CI: 0.44–0.87, p = 0.005) were independent prognostic factors for PFS, while lines of ICI therapy (HR: 1.59, 95% CI: 0.14–2.22, p = 0.006) and liver metastases (HR: 0.49, 95% CI: 0.35–0.69, p = 0.000) were independent prognostic factors of OS (Table 3 and Supplemental Table S3).
Univariate and multivariate analyses of progression-free survival and overall survival in relation to the baseline characteristics of overall survival and progression-free survival.

Control group.
CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; ICI, immune checkpoint inhibitor; LCNEC, large-cell neuroendocrine carcinoma; PNEC, pulmonary neuroendocrine carcinoma; SCLC, small-cell lung cancer.
Discussion
Our study demonstrated that PD-1/PD-L1 inhibitors prolonged the PFS and OS of patients with advanced PNEC. The combination of PD-1/PD-L1 inhibitors and chemotherapy has shown satisfactory outcomes in treatment-naïve patients. In addition, small-molecule antiangiogenic agents combined with PD-1/PD-L1 inhibitors produced superior PFS compared with ICI monotherapy or immunochemotherapy in the ⩾2L settings. To our knowledge, this is the largest real-world cohort study of patients with advanced PNEC treated with PD-1/PD-L1 inhibitors in Asia.
In recent years, a growing body of research has investigated the efficacy and safety of PD-1/PD-L1 inhibitors in treatment-naïve and relapsed advanced PNEC, demonstrating satisfactory efficacy and well-tolerated toxicity. A phase II basket trial noted that of 32 patients with non-pancreatic neuroendocrine neoplasms, 18 had neuroendocrine carcinoma, with an ORR of 44% after dual immunotherapy. 19 Similarly, in our study, patients with advanced PNEC who received PD-1/PD-L1 inhibitors had an ORR of 48.3%, DCR of 83.3%, mPFS of 6.0 months, and mOS of 13.1 months. In addition, we found no significant differences in PFS or OS among patients with different histological types of PNEC. Our findings demonstrate that LCNEC, similar to SCLC, could also benefit from PD-1/PD-L1 inhibitors, which provides new evidence for the treatment of advanced LCNEC. This may be due to the similarity between SCLC and LCNEC. For instance, Derks et al. reported that the clinical characteristics of advanced LCNEC resemble SCLC as does the OS. 20 Miyoshi et al. sequenced 78 LCNEC samples and found that LCNEC had genomic characteristics similar to SCLC. 21 More prospective studies are needed to further explore the similarities and differences between SCLC and LCNEC.
For treatment-naïve patients, data from several phase III trials showed that the addition of PD-1/PD-L1 inhibitors to chemotherapy for patients with ES-SCLC had better response and survival as compared to chemotherapy alone, with ORR in the range of 60.2%–80.2%, mPFS of 4.8–5.8 months, and mOS of 12.3–15.8 months.5,8,22,23 In our study, the 1L ES-SCLC cohort also demonstrated similarly positive results, with an ORR of 64.8%, mPFS of 6.9 months, and mOS of 15.7 months. Compared to the CASPIAN and Impower133 studies, our patients are all of Asian descent, and after progressing on 1L immunotherapy, they received additional treatments, including immune rechallenge; moreover, our 1L population is relatively younger (median age: 61 vs 62 (CASPIAN) vs 64 (Impower133) years). Notably, Asian ethnicity and immune rechallenge appear to be associated with improved survival benefits.7,24 Besides the studies mentioned above, in the recent ESMO Asia Congress 2023, a phase IIIB study of 166 untreated Chinese patients with ES-SCLC showed an ORR of 76.4%, an mPFS of 6.3 months, and an mOS of 14.8 months after durvalumab plus platinum–etoposide. 25 These efficacy data are in excellent agreement with those from our 1L ES-SCLC cohort, providing further evidence of the efficacy of PD-1/PD-L1 inhibitors in a real-world population of patients with ES-SCLC. However, data on the efficacy of 1L ICIs in advanced LCNEC are scarce. A retrospective study based on the National Cancer Database showed that 37 patients with LCNEC treated with immunotherapy plus chemotherapy had significantly higher 12-month (34.0% vs 24.1%) and 18-month survival rates (29.1% vs 15.0%) than those with chemotherapy alone. 26 In another multicenter retrospective study including 10 treatment-naïve patients with LCNEC, pembrolizumab plus chemotherapy exhibited an ORR of 70%, DCR of 90%, mPFS of 5.5 months, and mOS of 13.0 months. 27 In our cohort, we observed that patients with advanced LCNEC who received 1L ICIs had an ORR of 60%, DCR of 80%, mPFS of 6.8 months, and mOS of 13.3 months. These data indicated that 1L chemotherapy combined with PD-1/PD-L1 inhibitors is an effective treatment option for patients with advanced PNEC. However, whether ICIs can replicate the efficacy of 1L SCLC treatments in LCNEC patients merits further evaluation.
For patients with relapsed PNEC, either SCLC or LCNEC, there is a dearth of treatment strategies available, and ICIs have made limited progress. ICI monotherapy has demonstrated unsatisfactory clinical benefits in patients with SCLC, and the approved indications for third-line therapies have been withdrawn.28,29 However, promising treatment outcomes have been observed in several cohorts that evaluated ICIs in patients with advanced LCNEC after progression to platinum-based 1L chemotherapy.15,30 Our study also analyzed the ⩾2L usage of ICIs and indicated promising clinical activity, achieving an ORR of 21.8%, a DCR of 69.2%, an mPFS of 4.4 months, and an mOS of 9.4 months. In addition, studies have reported that small-molecule antiangiogenic drugs (apatinib or anlotinib) plus ICIs also benefit patients with relapsed SCLC.31–33 Similarly, in our analysis, patients with relapsed PNEC who received ICIs plus small-molecule antiangiogenic therapy had significantly longer PFS than those who received ICI monotherapy or immunochemotherapy. These findings could indicate that antiangiogenic agents can reprogram the tumor immune microenvironment, converting the immunosuppressive status to an immunosupportive one.34–36 Therefore, PD-1/PD-L1 inhibitors may be a promising therapeutic regimen for pretreated PNEC, with better outcomes observed when combined with small-molecule antiangiogenic drugs. Recently, a phase III trial reported that benmelstobart (a PD-L1 inhibitor) plus anlotinib and chemotherapy reached the longest reported OS in untreated ES-SCLC, with a 7.4-month extension of survival over chemotherapy (19.3 vs 11.9 months), and an mPFS of 6.9 months, 37 which supported the combination of ICIs and small-molecule antiangiogenic agents as a promising therapeutic option in PNEC. In addition, these data provide encouraging evidence for this combination regimen to move from later lines to 1L treatment, which merits further detailed study.
Patients with liver metastasis had significantly poorer outcomes than those without liver metastasis in our study, and liver metastasis was an independent factor associated with worse OS. This was also revealed in several retrospective studies,15,26 possibly due to the immunosuppressive liver microenvironment and suppression of systemic immunity by liver metastasis.38,39 In addition, numerous studies have demonstrated that the number of organs with metastases is a poor prognostic factor.40,41 Consistent with most research findings, our study also found that patients with metastases in ⩾3 organs had inferior survival benefits compared to those with <3 organ metastases. Moreover, after excluding the liver metastases, the number of metastatic lesions still significantly impacts patient prognosis. This may be due to a poorer ECOG PS (ECOG PS ⩾2 subgroup: 4.2% vs 3.8%) and higher tumor burden in patients with multiple organ metastases compared to those with metastatic organ number <3, leading to inferior treatment outcomes and shorter PFS.
Our study has several limitations. One potential limitation is its single-institution retrospective design and the heterogeneity of patient characteristics. This design might be prone to selection bias; however, we attempted to minimize this bias using multivariate analyses. In addition, the cohort size of patients with LCNEC was small; therefore, large-scale studies should be conducted in the future. Recent studies have shown that different molecular subtypes of SCLC and LCNEC may have impacts on treatment and prognosis. Specifically, the SCLC-Y subtype or the SCLC-I subtype, as well as SCLC-like LCNEC, appear to have a better response and benefit from chemotherapy combined with ICIs.15,42,43 However, this molecular subtype-based treatment approach is not yet fully mature and is influenced by many factors. Therefore, we do not routinely conduct sequencing and analysis in clinical practice, and we also look forward to further exploration in the future. Finally, adverse event data were not reported because of the lack of complete records, which hindered the safety analysis of immunotherapy in patients with advanced PNEC.
Conclusion
In conclusion, our results demonstrate that PD-1/PD-L1 inhibitors are effective in patients with advanced PNEC in a real-world setting, regardless of histological type. The efficacy of 1L immunochemotherapy is worthy of recognition, and the addition of small-molecule antiangiogenic agents to PD-1/PD-L1 inhibitors in ⩾2L treatment produces a better survival trend. Furthermore, larger and prospective studies are required to confirm our findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241288130 – Supplemental material for Real-world efficacy of PD-1/PD-L1 inhibitors in patients with advanced pulmonary neuroendocrine carcinoma: a single-center analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241288130 for Real-world efficacy of PD-1/PD-L1 inhibitors in patients with advanced pulmonary neuroendocrine carcinoma: a single-center analysis by Wanchen Zhai, Ying Yu, Haicheng Wu, Qian Zhang, Yunfei Chen, Yehao Yang and Yun Fan in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359241288130 – Supplemental material for Real-world efficacy of PD-1/PD-L1 inhibitors in patients with advanced pulmonary neuroendocrine carcinoma: a single-center analysis
Supplemental material, sj-pdf-2-tam-10.1177_17588359241288130 for Real-world efficacy of PD-1/PD-L1 inhibitors in patients with advanced pulmonary neuroendocrine carcinoma: a single-center analysis by Wanchen Zhai, Ying Yu, Haicheng Wu, Qian Zhang, Yunfei Chen, Yehao Yang and Yun Fan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
