Abstract
Background:
Skin disorders are major adverse events associated with necitumumab plus gemcitabine and cisplatin (Neci + GC) administration. However, the prognostic effect of skin disorders in patients with lung squamous cell carcinoma (LSCC) administered Neci + GC is unclear.
Objectives:
We examined this prognostic effect in patients with LSCC, and the usefulness of minocycline administration.
Design:
This was a sub-analysis of the retrospective multicenter NINJA study.
Methods:
We performed a landmark survival analysis according to the presence of skin disorders at Day 30 of treatment and examined the usefulness of minocycline for treating skin disorders.
Results:
Among the 93 patients, 62 (66.7%) had a skin disorder at Day 30. Nineteen, 30, and 13 patients experienced Grade 1, 2, and 3 skin disorders, respectively. The overall survival (OS) and progression-free survival (PFS) of patients with skin disorders at Day 30 were longer than those of patients without skin disorders (median OS: 434 vs 278 days, p = 0.0201; median PFS: 148 vs 82 days, p = 0.0835). Multivariable analysis showed that a skin disorder at Day 30 was an independent prognostic factor for both OS (p = 0.0044) and PFS (p = 0.0514). Of the 62 patients with skin disorders at Day 30, 38 (61.3%) were taking minocycline, and their time to treatment failure (TTF) was better than that in patients not taking minocycline (median TTF: 148 vs 101 days, p = 0.0495).
Conclusion:
A skin disorder within 30 days was a favorable prognostic factor for patients with LSCC administered Neci + GC. Additionally, minocycline administration may be beneficial in patients who develop skin disorders within 30 days.
Introduction
Necitumumab plus gemcitabine and cisplatin (Neci + GC) therapy has become an optional first-line treatment for patients with advanced lung squamous cell carcinoma (LSCC) since the international phase III trial (the SQUIRE study) 1 and the Japanese phase Ib/II study (the JFCM study) 2 elucidated its clinical benefit in patients with untreated advanced LSCC.
In the retrospective multicenter NINJA study, we demonstrated the effectiveness of Neci + GC therapy after combined immune checkpoint inhibitor (ICI) treatment, which is the current standard therapy for patients with LSCC. 3 Another retrospective study demonstrated the clinical benefits of Neci + GC therapy after ICI combination treatment. 4 Furthermore, a phase II NESSIE study (Japan Registry of Clinical Trials jRCTs051200138) is underway, evaluating the efficacy of Neci + GC therapy after combined ICI therapy. Thus, Neci + GC therapy may improve the prognosis of patients with LSCC who have previously been treated with ICI combination therapy.
Skin disorders are frequently observed as adverse events specific to anti-epidermal growth factor receptor (EGFR) antibodies, including necitumumab.1–4 The presence of skin disorders has been reported as a favorable prognostic factor during the administration of panitumumab and cetuximab, which are the same anti-EGFR antibodies as necitumumab and are used to treat colorectal cancer.5–9 However, the prognostic effect of skin disorders in patients with LSCC treated with Neci + GC therapy remains unclear.
In this study, we examined the frequency and prognostic effect of Neci + GC-induced skin disorders in patients with LSCC by performing a sub-analysis of the NINJA study. We also examined the usefulness of minocycline administration for Neci + GC-induced skin disorders.
Materials and methods
Study design
This was a sub-analysis of the multicenter, retrospective, observational NINJA study. This manuscript was prepared according to the STROBE Checklist (Table S1). The protocol was reviewed and approved by the institutional review board of Teikyo University (No. 22-002, Date: August 2, 2022; UMIN000048656), and the procedures were performed in compliance with the relevant laws and institutional guidelines. We used an opt-out method to explain the study to the patients and obtain their consent, and the study content was disclosed using a method prescribed by each participating institution.
The inclusion criteria were (i) pathologically diagnosed LSCC; (ii) an age of 20 years or older; (iii) postoperative recurrence, recurrence after radical concurrent chemoradiotherapy (CRT), or stage IV cancer; (iv) initiation of Neci + GC therapy between November 1, 2019, and March 3, 2022; (v) ICIs as the previous therapy (ICI combination chemotherapy, ICI–ICI combination therapy, or ICI monotherapy); and (vi) administration of Neci + GC therapy before fourth-line therapy.
In this study, 75 years or older was defined as elderly according to the Japanese guidelines for lung cancer treatment, and 75 years was used as the cutoff. 10
One patient had Grade 4 infusion-related reactions immediately after the initiation of necitumumab administration, followed by the scheduled administration of gemcitabine and cisplatin in the first cycle on Day 1; this patient was excluded from the survival analysis.
Objectives
The primary objective of this study was to demonstrate the prognostic effect of Neci + GC-induced skin disorders in patients with LSCC. The secondary objective was to examine the usefulness of minocycline administration for continuing Neci + GC therapy.
Landmark survival analysis
Patients were divided into two groups according to the presence of Neci + GC-induced skin disorders at Day 30. We examined the association of the presence of skin disorders up to Day 30 with clinical characteristics and overall survival (OS) and progression-free survival (PFS). We also performed a survival analysis according to the grade of the skin disorder at Day 30 (⩾Grade 2, Grade 1, and Grade 0). Rashes, paronychia, and dry skin were defined as skin disorders.
A landmark survival analysis was performed to examine how Neci + GC-induced skin disorders affect the subsequent prognosis. We used a landmark survival analysis to account for lead-time bias and underreporting of skin disorders due to early treatment discontinuation.5,11 The landmark was set as 30 days from the date of the first administration of Neci + GC therapy based on previous reports.5,11 Patients who had a survival event within 30 days of Neci + GC therapy administration were excluded from the survival analysis.
Furthermore, to examine the usefulness of minocycline administration for skin disorders associated with Neci + GC therapy, a landmark survival analysis of OS, PFS, and time to treatment failure (TTF) with and without minocycline administration was performed in patients with skin disorders at Day 30.
Statistical analysis
This study is a sub-analysis that was not planned when the NINJA study was planned, and is an analysis aimed at exploratory analysis rather than validation proof. Pearson’s Chi-square test was used to analyze differences in clinical characteristics according to the presence of Neci + GC-induced skin disorders at Day 30. OS was defined as the period from the first administration of Neci + GC therapy to death; PFS was defined as the period from the first administration of Neci + GC therapy to disease progression or death; and TTF was defined as the period from the first administration of Neci + GC therapy to the discontinuation of Neci + GC therapy. The Kaplan–Meier method was used to plot survival curves. Cox proportional hazards regression analysis was used to examine the risk factors. The factors analyzed in the multivariable analysis other than skin disorders at Day 30 were age, sex, performance status, and stage, which were obvious prognostic factors in chemotherapy for lung cancer. JMP Pro (version 16.0; SAS Institute, Cary, NC, USA) was used for all statistical analyses.
Results
A participant flow diagram is illustrated in Figure 1.
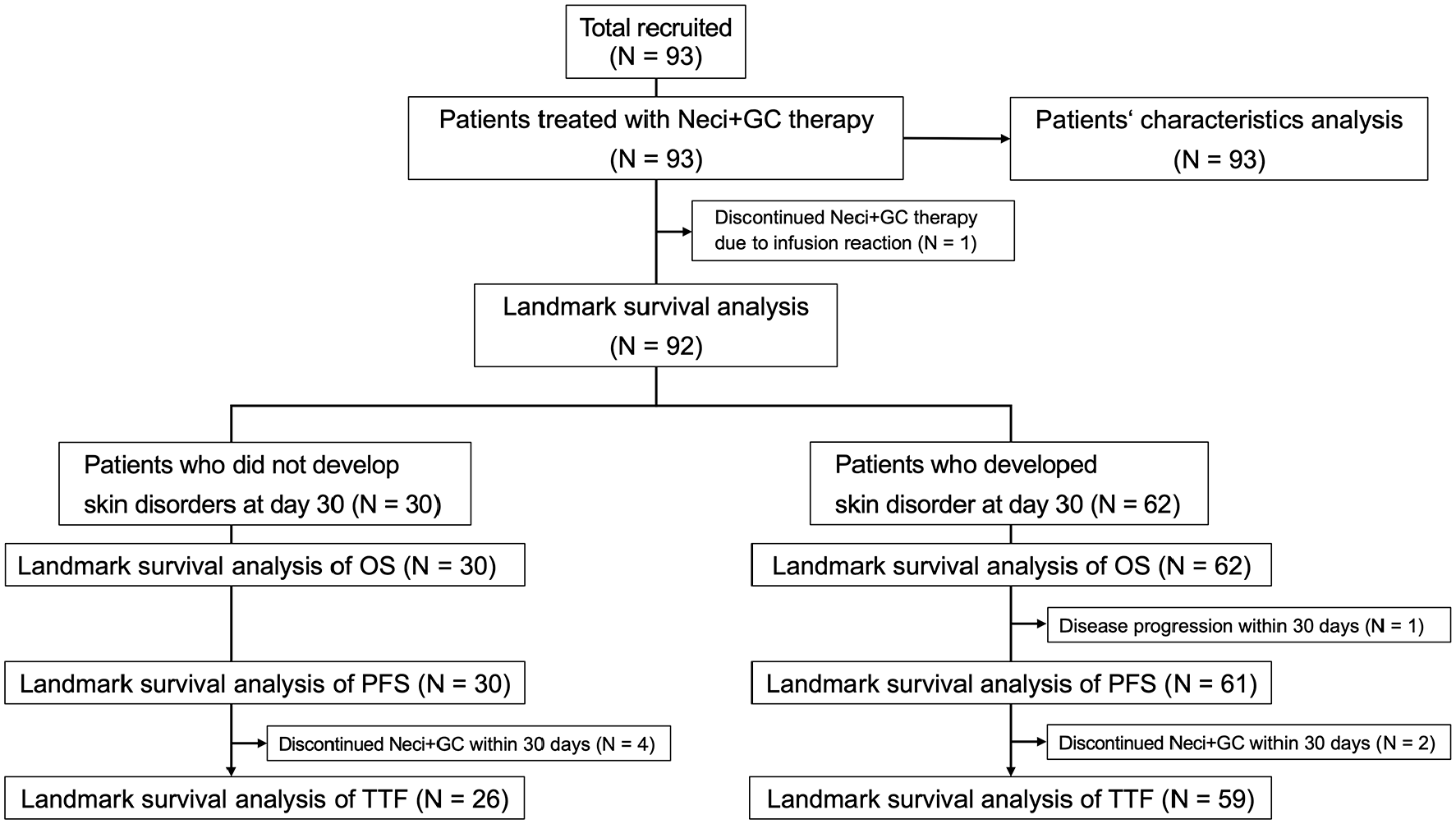
A flow diagram of the patients’ characteristics and landmark survival analyses.
Patient characteristics
The characteristics of the 93 patients are summarized in Table S2. Eight patients (8.6%) were older than 74 years, and 78 (83.9%) were men. Five (5.4%), 48 (51.6%), and 40 (43.0%) patients were diagnosed with postoperative recurrence, recurrence after CRT, and stage IV disease, respectively. Programmed death-ligand 1 expression was <1%, 1%–49%, and ⩾50% in 21 (22.6%), 37 (39.8%), and 22 (23.7%) patients, respectively. Skin disorders as immune-related adverse events of previous ICI therapy were observed in 23 patients (24.7%). The median follow-up period was 405 days (range: 39–893 days). During the follow-up period, the outcome events of OS, PFS, and TTF were observed in 49, 81, and 86 patients, respectively.
Neci + GC-induced skin disorder
The median elapsed period to the appearance of Neci + GC-induced skin disorders was 14 days (range: 1–105 days), and skin disorders up to Day 30 were observed in 62 patients (66.7%). Sixteen patients (17.2%) developed skin disorders after 30 days, and 15 (16.1%) did not develop skin disorders (see Table S3 which demonstrates the management of Neci + GC-induced skin disorders).
The patients’ clinical characteristics according to the presence of skin disorders at Day 30 are shown in Table 1. A total of 19, 30, and 13 patients experienced Grade 1, 2, and 3 skin disorders, respectively, at Day 30. There were no significant differences between the two groups.
Patient clinical characteristics according to the presence of skin disorders at Day 30.

BSC, best supportive care; CRT, chemoradiotherapy; ICI, immune checkpoint inhibitor; irAE, immune-related adverse event; Neci + GC, Necitumumab plus gemcitabine and cisplatin; PD-L1, programmed death-ligand 1; PS, performance status; TPS, tumor proportion score.
Regarding the management of the skin disorders up to Day 30 (Table S3), minocycline, topical steroids, and moisture were administered in 48 (51.6%), 66 (71.0%), and 72 patients (77.4%), respectively. In addition, minocycline, topical steroids, and moisture were administered as preemptive therapy in 15 (16.1%), 11 (11.8%), and 41 patients (44.1%), respectively.
In terms of therapy changes due to the presence of skin disorders, reduction and interruption of Neci + GC therapy was observed only in patients with ⩾Grade 2 skin disorders at Day 30 (Table S4). Neci + GC therapy was discontinued in one patient (2.3%) with a ⩾Grade 2 skin disorder, but in 15.8% (3/19) and 9.7% (3/31) of patients with Grade 1 skin disorders and without skin disorders, respectively. Treatment was discontinued in three patients who did not have a skin disorder at Day 30 because they developed a skin disorder thereafter.
Landmark survival analysis according to the presence of a skin disorder at Day 30
Kaplan–Meier survival curves based on the presence and grade of skin disorders are presented in Figure 2. The OS of patients with skin disorders at Day 30 was significantly longer than that of patients without skin disorders (median OS: 434 vs 278 days, p = 0.0201, Figure 2(a)). Furthermore, there was a trend that the PFS of patients with skin disorders at Day 30 was longer than that of patients without skin disorders (median PFS: 148 vs 82 days, p = 0.0835, Figure 2(b)). In the analysis based on the grade of skin disorders at Day 30, patients with ⩾Grade 2 skin disorders showed the longest OS and PFS compared with those with Grade 1 or 0 skin disorders (Figure 2(c) and (d)).

Survival curves of OS (a) and PFS (b) according to the presence of skin disorders at Day 30. Survival curves of OS (c) and PFS (d) according to the grade of skin disorders at Day 30.
The multivariable analysis showed that the presence of a skin disorder at Day 30 was an independent prognostic factor for both OS and PFS (OS: p = 0.0044, Table 2; PFS: p = 0.0514, Table 3). The hazard ratio (HR) of skin disorders was 0.62 for OS (95% confidence interval (CI): 0.45–0.86, Table 2) and 0.78 for PFS (95% CI: 0.62–1.00, Table 3) for every one grade increase in skin disorders.
Univariable and multivariable analyses of overall survival.

CI, confidence interval; CRT, chemoradiotherapy; HR, hazard ratio; PS, performance status.
Univariable and multivariable analyses of progression-free survival.
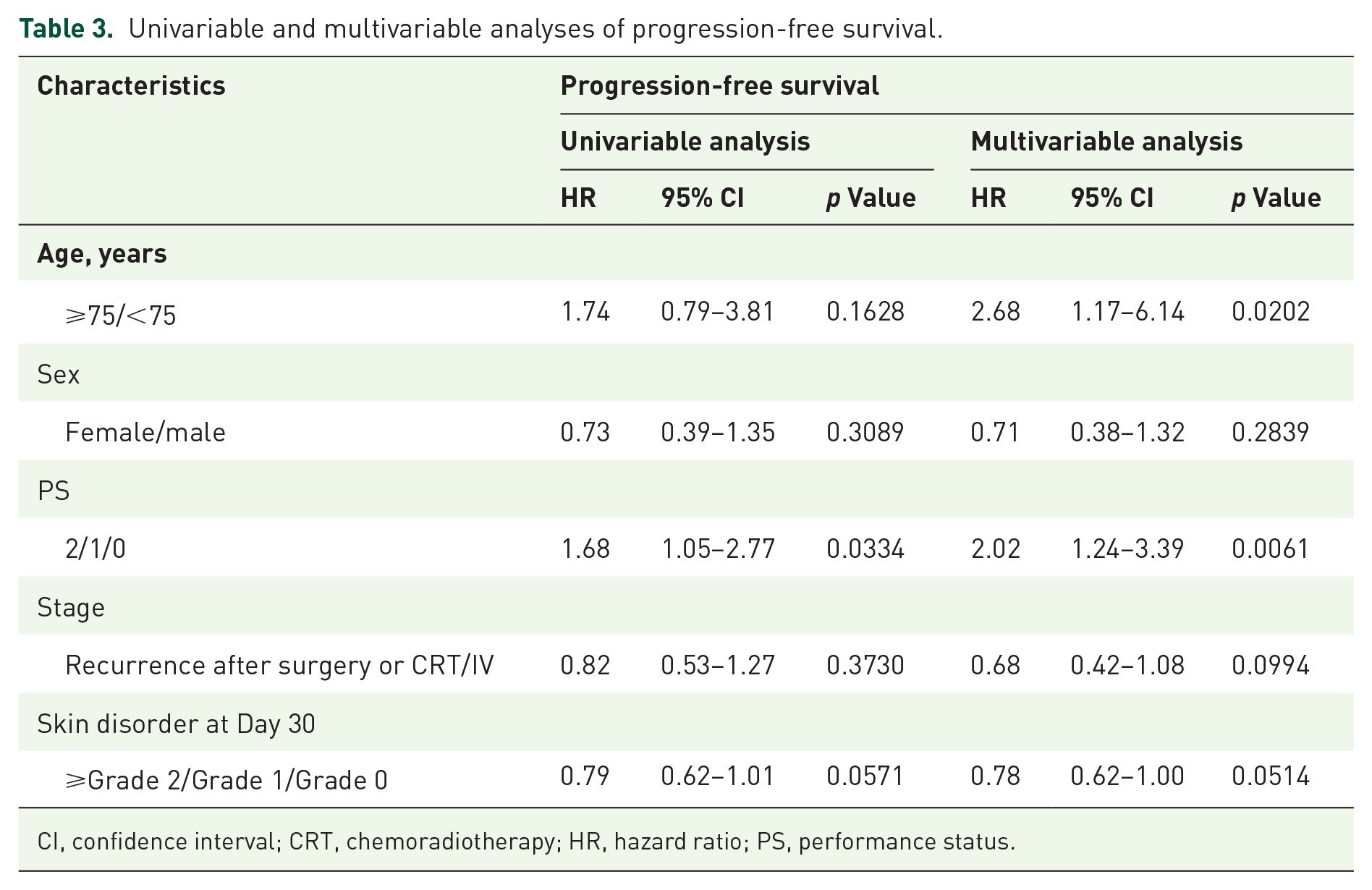
CI, confidence interval; CRT, chemoradiotherapy; HR, hazard ratio; PS, performance status.
An additional analysis according to the presence of skin irAE caused by previous immunotherapy was examined. In the patients with skin irAE caused by previous immunotherapy, HRs of skin disorder for OS and PFS were 0.29 (95% CI: 0.06–1.52, p = 0.1430) and 0.11 (95% CI: 0.02–0.57, p = 0.0082), respectively. In the patients without skin irAE caused by previous immunotherapy, HRs of skin disorder for OS and PFS were 0.46 (95% CI: 0.23–0.90, p = 0.0241) and 0.68 (95% CI: 0.40–1.14, p = 0.1387), respectively.
Usefulness of minocycline administration for Neci + GC-induced skin disorders
In the 62 patients with skin disorders at Day 30, 38 (61.3%) were treated with minocycline. Survival curves according to minocycline treatment are shown in Figure 3. The OS and PFS of patients treated with minocycline tended to be longer than those of patients not treated with minocycline (median OS: 479 vs 396 days, p = 0.1805, Figure 3(a); median PFS: 162 vs 134 days, p = 0.4868, Figure 3(b)). Moreover, in terms of TTF, minocycline treatment was associated with good prognosis (median TTF: 148 vs 101 days, p = 0.0495, Figure 3(c)).

Survival curves of overall survival (a), progression-free survival (b), and time to treatment failure (c) according to the administration of minocycline in patients with skin disorders at Day 30.
A further exploratory analysis according to the grade of skin disorders was performed. In the patients with Grade 1 skin disorders, HRs of minocycline administration for OS, PFS, and TTF were 0.45 (95% CI: 0.11–1.83, p = 0.2646), 0.45 (95% CI: 0.16–1.39, p = 0.1636), and 0.30 (95% CI: 0.09–0.98, p = 0.0457), respectively. In the patients with ⩾Grade 2 skin disorders, HRs of minocycline administration for OS, PFS, and TTF were 0.72 (95% CI: 0.30–1.72, p = 0.4634), 0.98 (95% CI: 0.48–2.00, p = 0.9555), and 0.71 (95% CI: 0.35–1.42, p = 0.3324), respectively.
In addition, in the 62 patients with skin disorders at Day 30, discontinuation, reduction, and interruption of Neci + GC therapy were observed in 0% (0/38), 18.4% (7/38), and 15.8% (6/38) of patients treated with minocycline, respectively. Conversely, in patients not treated with minocycline, discontinuation, reduction, and interruption of Neci + GC therapy were observed 16.7% (4/24), 0% (0/24), and 0% (0/24) of patients, respectively.
Discussion
This study examined the significance of skin disorder with necitumumab, the first anti-EGFR antibody with proven efficacy in lung cancer. In this study, we found that a skin disorder on Day 30 after the initiation of Neci + GC therapy was a favorable prognostic factor for subsequent OS and PFS. We also showed that minocycline administration may be helpful in continuing Neci + GC therapy in patients who develop skin disorders.
The primary analysis of the NINJA study also reported a favorable prognosis for patients treated with Neci + GC who develop skin disorders; however, it did not consider when the skin disorders appeared. 3 Therefore, it was unclear whether the number of patients with skin disorders increased because of successful Neci + GC therapy and long-term necitumumab administration, or whether the Neci + GC-induced skin disorder itself was a prognostic factor. In the current study, by performing landmark survival and multivariable analyses, we elucidated that a skin disorder at Day 30 may be an independent predictor of subsequent good prognosis in patients with LSCC treated with Neci + GC.
We demonstrated that the presence of a skin disorder within 30 days after Neci + GC therapy initiation was a favorable prognostic factor for both OS and PFS. In colorectal cancer, the presence and a higher grade of skin disorder with panitumumab or cetuximab treatment, which is also anti-EGFR antibodies similar to necitumumab, are associated with a good prognosis.5–9 A meta-analysis based on several clinical trials of panitumumab or cetuximab treatment for colorectal cancer revealed that the occurrence of skin toxicity is a favorable prognostic factor for both survival (HR: 0.51, p < 0.0001) and progression (HR: 0.58, p < 0.0001). 12 Our results are consistent with those of previous studies. Therefore, skin disorders during Neci + GC therapy should be noted but considered a favorable prognostic factor. The management of skin disorders is important in ensuring that Neci + GC therapy can be successfully continued despite their presence.
In lung cancer, EGFR tyrosine kinase inhibitors (EGFR-TKIs) have been used for EGFR-mutant lung cancer before anti-EGFR antibodies such as necitumumab. EGFR-TKIs are known to have a high incidence of adverse skin disorders like necitumumab. Several studies, including meta-analyses, have shown an association between skin disorders and favorable prognosis for erlotinib and gefitinib.13–17 There is a report suggesting skin disorder as a favorable prognostic factor for afatinib. 18 Although no studies have analyzed the prognostic significance of skin disorders for osimertinib, it is possible that skin disorder may be a favorable prognostic factor like anti-EGFR antibody. Therefore, we hypothesize that skin disorder may be a useful prognostic factor for both anti-EGFR antibodies and EGFR-TKIs.
The mechanism underlying skin disorders caused by anti-EGFR antibodies is that these antibodies affect the EGFR of keratinocytes in the skin. 19 The inhibition of EGFR signaling in keratinocytes causes skin disorders by inducing abnormal migration and maturation of keratinocytes and promoting the synthesis of inflammatory chemokines (such as CCL2 and CCL5) and cytokines (such as IL-6 and IL-7).19,20 In addition, it has been reported that the IL-8 level in the blood is decreased in patients with skin disorders due to the inhibition of EGFR signaling.21,22 IL-8 plays an important role in tumor angiogenesis and progression in several cancers, including in lung cancer.23–27 Based on these findings, a decrease in IL-8 is considered a reason that a skin disorder is a good prognostic factor. 21 Thus, necitumumab may have a mechanism similar to that of skin disorders, but further investigation is required.
The incidence of skin disorders in the NINJA study was 83.9% (78/93). 3 This is similar to the incidence of skin disorders in the SQUIRE (78.8% (424/538)) 1 and JFCM (97.8% (88/90)) studies, 2 indicating that skin disorders may be a frequent adverse event during Neci + GC therapy in patients previously treated with ICIs, as well as in first-line Neci + GC therapy. Additionally, 66.7% (62/93) of the patients in this study developed skin disorders by Day 30 of Neci + GC therapy; hence, skin disorders occurred in the early phase of therapy. These results indicate the need for early management of skin disorders in patients administered Neci + GC.
Among the patients with skin disorders induced by Neci + GC administration, those who received minocycline had a significantly better TTF and a trend toward better OS and PFS. One reason for the favorable survival of minocycline-treated patients might be that they had reduced or interrupted Neci + GC therapy rather than discontinued compared to patients not treated with minocycline. During panitumumab and cetuximab administration, the importance of skin treatment, including minocycline, has been reported; in particular, the efficacy of preemptive skin treatment has attracted attention.28–31 The STEPP study, a phase II randomized clinical trial, showed that preemptive skin treatment reduced the frequency of ⩾Grade 2 skin disorders (preemptive skin treatment group: 29% (14/48) vs control group: 62% (29/47)) and improved the quality of life of patients with colorectal cancer treated with panitumumab. 28 In the J-STEPP study, a Japanese phase II randomized clinical trial, preemptive skin treatment decreased the severity of skin disorders; the frequency of ⩾Grade 2 skin disorders in the preemptive skin treatment group was 21.3% (10/47) and that in the control group was 62.5% (30/48). 29 In this study, it was impossible to elucidate the usefulness of minocycline as a preemptive therapy because only 16.1% (15/93) of the patients received minocycline as a preemptive skin treatment. However, in the patients with Grade 1 skin disorders, there was a trend of a lower HRs of minocycline administration compared to in those with ⩾Grade 2 skin disorders, suggesting a possible benefit of minocycline administration at a milder stage of skin disorders. Furthermore, we demonstrated that preemptive or therapeutic minocycline may be useful for treating skin disorders induced by Neci + GC treatment. Therefore, the concomitant use of minocycline during Neci + GC therapy is important.
This study has several limitations. Although it was a multicenter study, the NINJA study was retrospective and included a limited number of patients. We expect that the prospective multicenter NESSIE study will elucidate the significance of skin disorders. In addition, the assessment and management of skin disorders differed among the institutions, and it is difficult to show a definitive association between minocycline and prognosis in this study alone. Therefore, a high-quality clinical trial that assesses the care of skin disorders is necessary, as in the STEPP and J-STEPP studies on colorectal cancer.28,29
Conclusion
In conclusion, skin disorders at Day 30 were a favorable prognostic factor in patients with LSCC treated with Neci + GC. Additionally, preemptive or therapeutic minocycline administration could successfully extend the duration of treatment in patients with LSCC who developed Neci + GC-induced skin disorders within 30 days. Therefore, the appropriate management of skin disorders caused by Neci + GC therapy may provide more benefits to patients undergoing this therapy.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241312503 – Supplemental material for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study
Supplemental material, sj-docx-1-tam-10.1177_17588359241312503 for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study by Fumihiko Kinoshita, Shigeru Tanzawa, Toshihiro Misumi, Hiroshige Yoshioka, Eisaku Miyauchi, Kiichiro Ninomiya, Yasunori Murata, Masafumi Takeshita, Masafumi Yamaguchi, Shunichi Sugawara, Yosuke Kawashima, Kazuki Hashimoto, Masahide Mori, Akihiko Miyanaga, Anna Hayashi, Hisashi Tanaka, Ryoichi Honda, Masafumi Nojiri, Yuki Sato, Ken Yamamoto, Ken Masuda, Toshiyuki Kozuki, Takahisa Kawamura, Takuji Suzuki, Teppei Yamaguchi, Kazuhiro Asada, Satoshi Tetsumoto, Hiroshi Tanaka, Satoshi Watanabe, Yukihiro Umeda, Kakuhiro Yamaguchi, Shoichi Kuyama, Kosuke Tsuruno, Yuki Misumi, Hiroshi Kuraishi, Ken Yoshihara, Akira Nakao, Akihito Kubo, Toshihiko Yokoyama, Kana Watanabe and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241312503 – Supplemental material for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study
Supplemental material, sj-docx-2-tam-10.1177_17588359241312503 for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study by Fumihiko Kinoshita, Shigeru Tanzawa, Toshihiro Misumi, Hiroshige Yoshioka, Eisaku Miyauchi, Kiichiro Ninomiya, Yasunori Murata, Masafumi Takeshita, Masafumi Yamaguchi, Shunichi Sugawara, Yosuke Kawashima, Kazuki Hashimoto, Masahide Mori, Akihiko Miyanaga, Anna Hayashi, Hisashi Tanaka, Ryoichi Honda, Masafumi Nojiri, Yuki Sato, Ken Yamamoto, Ken Masuda, Toshiyuki Kozuki, Takahisa Kawamura, Takuji Suzuki, Teppei Yamaguchi, Kazuhiro Asada, Satoshi Tetsumoto, Hiroshi Tanaka, Satoshi Watanabe, Yukihiro Umeda, Kakuhiro Yamaguchi, Shoichi Kuyama, Kosuke Tsuruno, Yuki Misumi, Hiroshi Kuraishi, Ken Yoshihara, Akira Nakao, Akihito Kubo, Toshihiko Yokoyama, Kana Watanabe and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359241312503 – Supplemental material for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study
Supplemental material, sj-docx-3-tam-10.1177_17588359241312503 for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study by Fumihiko Kinoshita, Shigeru Tanzawa, Toshihiro Misumi, Hiroshige Yoshioka, Eisaku Miyauchi, Kiichiro Ninomiya, Yasunori Murata, Masafumi Takeshita, Masafumi Yamaguchi, Shunichi Sugawara, Yosuke Kawashima, Kazuki Hashimoto, Masahide Mori, Akihiko Miyanaga, Anna Hayashi, Hisashi Tanaka, Ryoichi Honda, Masafumi Nojiri, Yuki Sato, Ken Yamamoto, Ken Masuda, Toshiyuki Kozuki, Takahisa Kawamura, Takuji Suzuki, Teppei Yamaguchi, Kazuhiro Asada, Satoshi Tetsumoto, Hiroshi Tanaka, Satoshi Watanabe, Yukihiro Umeda, Kakuhiro Yamaguchi, Shoichi Kuyama, Kosuke Tsuruno, Yuki Misumi, Hiroshi Kuraishi, Ken Yoshihara, Akira Nakao, Akihito Kubo, Toshihiko Yokoyama, Kana Watanabe and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359241312503 – Supplemental material for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study
Supplemental material, sj-docx-4-tam-10.1177_17588359241312503 for Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study by Fumihiko Kinoshita, Shigeru Tanzawa, Toshihiro Misumi, Hiroshige Yoshioka, Eisaku Miyauchi, Kiichiro Ninomiya, Yasunori Murata, Masafumi Takeshita, Masafumi Yamaguchi, Shunichi Sugawara, Yosuke Kawashima, Kazuki Hashimoto, Masahide Mori, Akihiko Miyanaga, Anna Hayashi, Hisashi Tanaka, Ryoichi Honda, Masafumi Nojiri, Yuki Sato, Ken Yamamoto, Ken Masuda, Toshiyuki Kozuki, Takahisa Kawamura, Takuji Suzuki, Teppei Yamaguchi, Kazuhiro Asada, Satoshi Tetsumoto, Hiroshi Tanaka, Satoshi Watanabe, Yukihiro Umeda, Kakuhiro Yamaguchi, Shoichi Kuyama, Kosuke Tsuruno, Yuki Misumi, Hiroshi Kuraishi, Ken Yoshihara, Akira Nakao, Akihito Kubo, Toshihiko Yokoyama, Kana Watanabe and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
This study was conducted by the NINJA study group under a funding contract with Nippon Kayaku Co., Ltd, Japan. The sponsor did not have any role in the study design; in the collection, analysis, and interpretation of the data; in the writing of the report; and in the decision to submit the article for publication. The authors thank the patients, their families, and all the investigators who participated in this study. We would like to thank Editage (![]() ) for English language editing and journal submission support. The authors have authorized the submission of this manuscript through Editage.
) for English language editing and journal submission support. The authors have authorized the submission of this manuscript through Editage.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
