Abstract
Background:
The clinical impact of hypomagnesemia induced by necitumumab plus gemcitabine and cisplatin (GCN) as a second-line or later therapy is unclear.
Objective:
We aimed to evaluate the clinical characteristics and survival impact of hypomagnesemia induced by this therapy.
Design:
This was a sub-analysis of the retrospective multicenter NINJA study.
Methods:
Among the 93 patients enrolled in the NINJA study, this subanalysis included 75 patients with baseline serum magnesium concentrations.
Results:
The incidence of grade ⩾2 hypomagnesemia was 18.0% in the patients with normal baseline serum magnesium concentrations and 42.8% in those with low concentrations (p = 0.073). The discontinuation rates of GCN treatment owing to hypomagnesemia in each group were 0% and 7.1%, respectively (p = 0.187). The number of necitumumab doses and severity of hypomagnesemia were positively correlated (r = 0.389, p < 0.001). Patients who developed hypomagnesemia in fewer than 21 days after the first dose of GCN (n = 12) had significantly poorer progression-free survival (PFS) than those without the condition (n = 63; median: 4.1 vs 4.4 months, p = 0.048). A similar trend was observed for OS (median: 9.7 vs 15.7 months, p = 0.062). These results were maintained after multivariate analyses (PFS: hazard ratio (HR) 2.46, p = 0.014; OS: HR 2.78, p = 0.021).
Conclusion:
GCN as a second-line or later therapy may be tolerable regardless of the patient’s baseline serum magnesium concentration. On the other hand, early serum magnesium reduction with this therapy is associated with a poor prognosis. However, caution should be needed because our results lacked sufficient information for confounding variables other than those analyzed here that may influence the correlation between hypomagnesemia and survival.
Introduction
Necitumumab, a monoclonal antibody targeting the epidermal growth factor receptor (EGFR), has been approved in Japan for the treatment of patients with advanced lung squamous cell carcinoma (LSqCC). Based on the results of the international phase III SQUIRE Trial and the Japanese phase I/II JFCM Trial conducted in the first-line setting, necitumumab combined with gemcitabine and cisplatin (GCN) triple therapy—compared with gemcitabine and cisplatin dual therapy—significantly improved overall survival (OS) and progression-free survival (PFS) in patients with advanced LSqCC.1,2 In addition, immune checkpoint inhibitor (ICI)-based therapies have been established as the standard treatment for previously untreated advanced LSqCC. We therefore conducted a retrospective multicenter NINJA study to evaluate the efficacy and tolerability of GCN as a second-line or later treatment. We previously reported that GCN for patients pretreated with ICIs might be well tolerated and offer promising efficacy, regardless of treatment line. 3 Another retrospective study conducted at a single Japanese institution also supported the efficacy of GCN therapy in patients previously treated with ICIs. 4 Therefore, in clinical settings, GCN is considered one of the optional second-line treatment regimens for patients with advanced or recurrent LSqCC.
Although clinically significant survival benefits have been observed in patients who received GCN therapy, skin disorders and hypomagnesemia—which are characteristic adverse events related to anti-EGFR antibodies—were frequently observed, in turn influencing treatment continuation. The most common adverse events associated with necitumumab treatment in the NINJA study were skin disorders, with an incidence of 75.3% for all grades and 15.1% for grade ⩾3, followed by hypomagnesemia, with an incidence of 59.1% for all grades and 7.5% for grade ⩾3.
The mechanism of hypomagnesemia caused by anti-EGFR antibodies is thought to involve the downregulation of transient receptor potential melastatin 6 (TRPM6). TRPM6, present in the distal renal tubule, is an ion channel through which magnesium (Mg) passes into the cells from the renal tubular lumen. TRPM6 expression is upregulated by epidermal growth factor. Thus, antibodies targeting the EGFR reduce TRPM6 expression and prevent Mg reabsorption, resulting in treatment-related hypomagnesemia.5,6
Regarding the association between serum Mg levels and human disease, habitually low Mg intake is reported to increase the risk of illnesses such as hypertension, cardiovascular disease, type 2 diabetes, and osteoporosis, while increasing dietary Mg intake is reported to reduce the risk of stroke, heart failure, diabetes, and all-cause mortality.7,8 Furthermore, Sakaguchi et al. 9 reported that hypomagnesemia in patients with hemodialysis is a significant predictor of cardiovascular and non-cardiovascular mortality.
Regarding the association between anti-EGFR antibody-related hypomagnesemia and patient prognosis, an early reduction in serum Mg concentration caused by cetuximab monotherapy was associated with a poor prognosis in patients with colon cancer in the subanalyses of two randomized control trials.10,11 However, the mechanism for this association is unclear. Furthermore, one of the trials also demonstrated that the patients with hypomagnesemia at any point during cetuximab treatment had longer median OS versus those without hypomagnesemia, resulting in the opposite outcome. 11 In terms of necitumumab treatment in patients with lung cancer, the association between hypomagnesemia and survival has not yet been evaluated.
Therefore, herein, we assessed the clinical characteristics of hypomagnesemia induced by GCN therapy and its impact on survival in patients with advanced or recurrent LSqCC by performing a subanalysis of the NINJA study.
Materials and methods
Study design and patients
The NINJA study was a multicenter, non-interventional, retrospective cohort study, of which the eligibility criteria have been previously reported. 3 Among the 93 patients enrolled, 75 with a baseline Mg concentration measurement were included in this subanalysis.
Evaluations
We evaluated the incidence of hypomagnesemia according to the baseline serum Mg concentration, the correlation between the number of administrated necitumumab doses during the induction phase of GCN therapy and the severity of hypomagnesemia, and the association between hypomagnesemia induced by GCN treatment and survival, with the endpoints being PFS and OS.
Given the retrospective study design, the timings of radiographic evaluation, serum Mg measurement after the first GCN dose, and prophylactic Mg administration were not specified in the protocol; these were determined by the attending physician according to the local product labeling and proper usage guidelines for necitumumab.
PFS was defined as the time from the first dose of GCN to the first radiographic assessment of objective progression or death from any cause. OS was defined as the time from the first dose of GCN to death from any cause.
Clinical data were extracted according to the protocol—data were obtained from the medical records of each institution using a pre-specified case report form. Hypomagnesemia induced by GCN therapy, a characteristic adverse reaction to anti-EGFR antibodies, was prespecified as an adverse event of special interest. The following factors were also specified in advance: baseline serum Mg concentration, number of GCN doses, number of necitumumab doses, presence or absence of prophylactic Mg administration, onset date and outcome of GCN-induced hypomagnesemia, worst grade of hypomagnesemia, with or without Mg administration as treatment for necitumumab-induced hypomagnesemia, presence or absence of symptoms related to hypomagnesemia, and GCN treatment course. Toxicity was assessed based on clinical and laboratory events in accordance with the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. As our study was retrospective cohort one, there was no masking at any point in the data collection and synthesis stages of this study.
In terms of the outcomes related to hypomagnesemia induced by GCN therapy, “improvement of serum Mg concentration” was defined as an improvement of at least one grade from the worst CTCAE grade but still below the normal Mg concentration, while “recovery of serum Mg concentration” was defined as complete recovery to normal Mg concentration. Patients with low serum Mg concentrations at baseline and an unchanged CTCAE grade were defined as having hypomagnesemia induced by GCN therapy in that CTCAE grade.
Statistical analysis
The intention-to-treat population comprised all patients enrolled in the NINJA study. The full analysis set (FAS) comprised patients who received GCN therapy, except for one patient with inadequate administration due to infusion-related reactions. The subset of the FAS population comprised patients with a baseline Mg concentration measurement who received all three drugs in the first cycle of GCN therapy—the evaluation of hypomagnesemia induced by GCN therapy and the survival analysis were performed in this population.
The age cutoff threshold was aligned with those in the SQUIRE and JFCM Trials.
PFS and OS were estimated using the Kaplan–Meier method to obtain the median survival time. We calculated 95% confidence intervals (CIs) using the Brookmeyer–Crowley method. The log-rank test was used to compare groups.
In addition, a landmark analysis was performed. Based on previous reports that demonstrated an association between cetuximab and an early reduction in Mg concentration in patients previously treated with cytotoxic chemotherapy, we set the landmark time point at Day 21.12,13
Univariate and multivariate analyses were performed using Cox proportional hazards regression models, adjusting for the following potential prognostic variables that were deemed clinically relevant: age (<70 vs ⩾70 years), sex (female vs male), Eastern Cooperative Oncology Group performance status (0 vs 1–2), estimated glomerular filtration rate (eGFR; <60 vs ⩾60 mL/min/1.73 m2), history of platinum-agent administration prior to GCN therapy (no vs yes), and hypomagnesemia in fewer than 21 days after administration of the first dose of GCN (no vs yes).14 –16
Fisher’s exact test was used to compare the incidence between the two groups, as appropriate. Spearman’s rank correlation coefficient was used to evaluate the association between the number of administrated necitumumab doses and the severity of hypomagnesemia induced by GCN therapy.
The statistical significance level was set at 5%. All analyses were performed using EZR Version 1.61 (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a graphical user interface for R software (R Foundation for Statistical Computing, Vienna, Austria). 17
Ethics
This study was approved by the Ethics Review Board of Teikyo University (approval no. 22-002, approval date August 2, 2022; UMIN000048656). It was conducted in accordance with the ethical principles of the Declaration of Helsinki, the Ethical Guidelines for Medical and Biological Research Involving Human Subjects, and the study protocol. An opt-out method was used to provide explanations to the patients and obtain their informed consent. The study content was disclosed using the method prescribed by each participating institution. The reporting of this study conforms to the STROBE guidelines, and the completed checklist is provided in Table S1. 18
Results
Patient selection
A total of 93 patients from 35 institutions in Japan were enrolled. Among them, one patient with inadequate treatment administration due to grade 4 infusion-related reactions was excluded, resulting in 92 patients being included in the FAS. Among them, 17 patients without a baseline Mg concentration measurement were excluded, resulting in 75 patients being included in the FAS subset (Figure S1).
Patient characteristics
The baseline patient characteristics of the FAS subset are shown in Table 1. The median age was 68 years, with 46 patients aged <70 years. Sixty-five patients were men and 51 had an Eastern Cooperative Oncology Group performance status score of 1 or 2. A total of 5, 36, and 34 patients had postoperative recurrence, Stage III, and Stage IV disease, respectively. Sixty-two patients (82.7%) had an eGFR ⩾60 mL/min/1.73 m2, and 67 (89.3%) had a history of platinum-based chemotherapy. The median Mg concentration at baseline was 1.9 mg/dL (range: 1.4–2.5), with 61 patients (81.3%) having a concentration within the normal range and 14 (18.7%) having a low concentration that corresponded to grade 1 hypomagnesemia. The median follow-up period was 9.0 months (range: 1.3–29.7 months). During the follow-up period, the outcome events of OS and PFS were observed in 41 and 68 patients, respectively.
Baseline patient characteristics.

ECOG, Eastern Cooperative Oncology Group; eGFR, estimated glomerular filtration rate; FAS, full analysis set; GCN, necitumumab combined with gemcitabine and cisplatin; Mg, magnesium; PD-L1, programmed death-ligand 1.
Clinical course of GCN therapy
The clinical course of GCN therapy in the FAS subset is shown in Table 2. A median of 6 (range: 1–8) necitumumab doses were administered during the GCN induction phase. A total of 85.2% and 100% of patients with a normal and low serum Mg concentration at baseline, respectively, received prophylactic Mg at the induction phase. Of the 75 patients in the FAS subset, 37 proceeded to the necitumumab maintenance phase. Among them, 15 patients received prophylactic Mg.
Clinical course of GCN therapy.
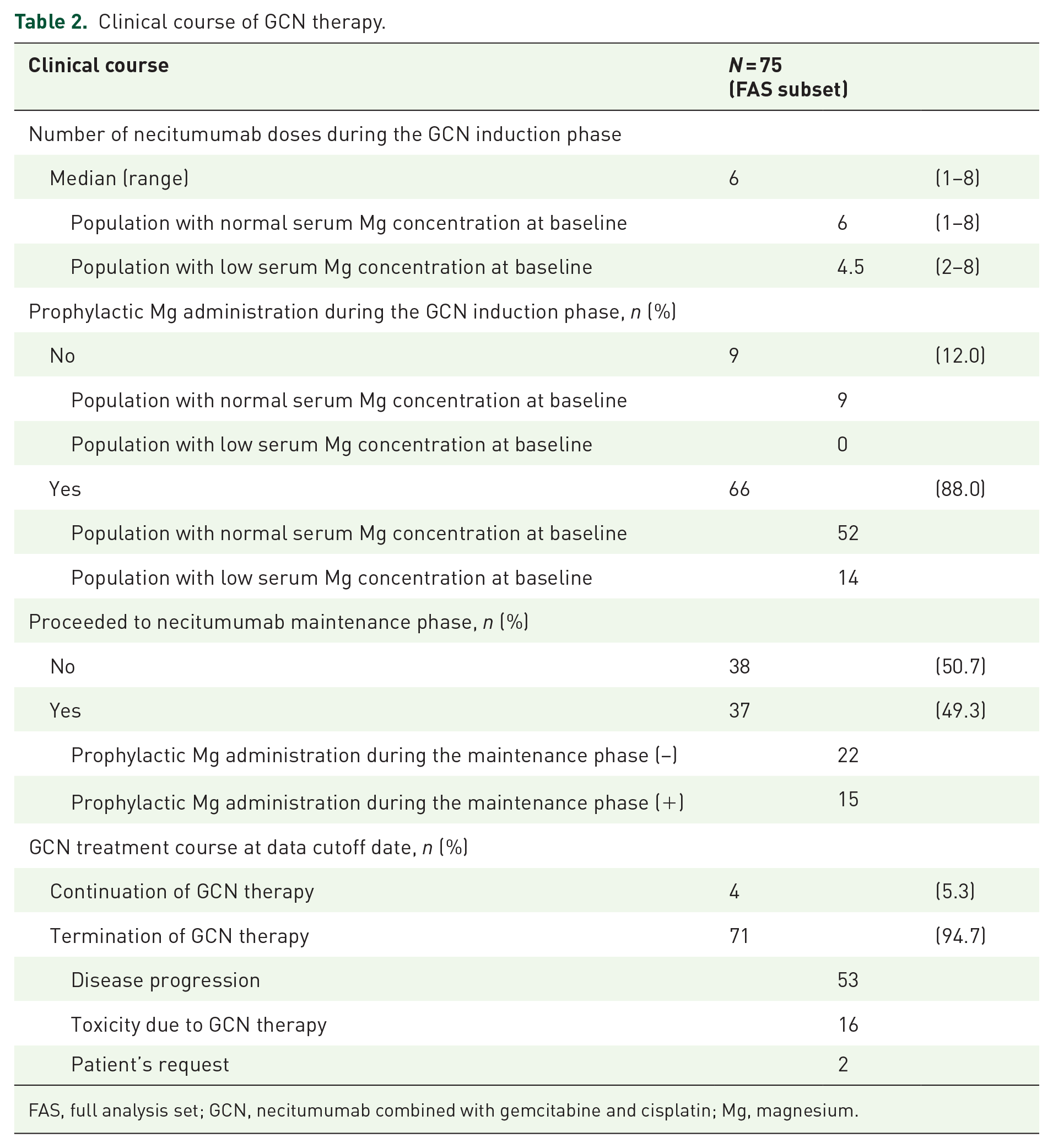
FAS, full analysis set; GCN, necitumumab combined with gemcitabine and cisplatin; Mg, magnesium.
At the data cutoff date for this analysis, 71 patients terminated GCN therapy owing to disease progression (53/71), GCN-induced toxicity (16/71), and patient request (2/71). Among the 16 patients who discontinued GCN because of toxicity, one discontinued treatment because of grade 2 hypomagnesemia.
Incidence of GCN-induced hypomagnesemia
The incidence of hypomagnesemia induced by GCN therapy in the FAS subset is shown in Figure 1. A total of 61.3%, 22.6%, and 4.0% of the patients experienced any grade, grade ⩾2, and grade ⩾3 hypomagnesemia, respectively. Among the FAS subset, 62.3%, 18.0%, and 3.3% of the patients with a normal serum Mg concentration at baseline experienced any grade, grade ⩾2, and grade ⩾3 hypomagnesemia, respectively, while 57.1%, 42.8%, and 7.1% of those with a low serum Mg concentration at baseline experienced any grade, grade ⩾2, and grade ⩾3 hypomagnesemia, respectively. The incidence of grade ⩾2 hypomagnesemia in patients with a low serum Mg concentration at baseline tended to be higher than that in patients with a normal Mg concentration at baseline (grade ⩾2: 42.8% vs 18.0%, p = 0.073).

Incidence of GCN-induced hypomagnesemia based on serum Mg concentration at baseline.
One patient in the group with a low serum Mg concentration at baseline discontinued GCN therapy due to grade 2 hypomagnesemia, while no patients in the group with a normal serum concentration at baseline discontinued therapy. The difference between the groups was not statistically significant (7.1% (1/14) vs 0% (0/61), p = 0.187; Table S2).
Correlation between worst severity of hypomagnesemia and number of administered necitumumab doses
In the 75 patients included in the FAS subset, the worst severity of hypomagnesemia during the induction phase of GCN therapy positively correlated with the number of administered necitumumab doses (Spearman’s rank correlation coefficient; r = 0.389, p < 0.001), as shown in Figure 2.

Correlation between the number of administered necitumumab doses and severity of hypomagnesemia during the induction phase of GCN therapy (FAS subset).
Outcomes of GCN-induced hypomagnesemia
Among the FAS subset, 46 patients experienced hypomagnesemia of any grade induced by GCN therapy. The median time to hypomagnesemia was 37.0 days (range: 8–162) and 14.5 days (range: 4–99) in patients with a normal (n = 38) and low (n = 8) Mg concentration at baseline. A total of 54.3% of the patients recovered from hypomagnesemia; the median time to recovery was 112.0 days (range: 18–337; Table S3).
Regarding the clinical symptoms associated with hypomagnesemia, one patient experienced nausea due to grade 1 hypomagnesemia. No severe symptoms, such as tetany, epilepsy, or fatal arrhythmias, such as torsades de pointes, were observed.
Survival analysis according to hypomagnesemia
The association between hypomagnesemia at any point during the entire study period and survival is shown in Figure 3. The median PFS (Figure 3(a)) in patients with and without hypomagnesemia was 4.4 and 3.0 months, respectively. There was no significant difference between the groups (p = 0.337). Similarly, the median OS (Figure 3(b)) in these two groups of patients was 13.9 and 13.4 months, respectively. This difference was also not statistically significant (p = 0.868).

Kaplan–Meier curves of PFS (a) and OS (b) based on the presence of GCN therapy-induced hypomagnesemia.
Landmark analysis based on hypomagnesemia at day 21 after the first GCN dose
Clinical characteristics
Twelve patients developed GCN-induced hypomagnesemia in fewer than 21 days. No baseline characteristics predicted the development of this treatment-related adverse event (Table S4).
Survival analysis
In the landmark analysis of the association between hypomagnesemia induced by GCN therapy in fewer than 21 days after the first dose and survival, patients with an early reduction in serum Mg concentration had significantly unfavorable PFS when compared with those without such a reduction (median PFS: 4.1 vs 4.4 months, p = 0.048, 6-month PFS rate: 0% vs 37.4%; Figure 4(a)). For OS, a similar tendency, although not statistically significant, was observed between these two groups (median OS: 9.7 vs 15.7 months, p = 0.062, 12-month OS rate: 19.4% vs 61.5%; Figure 4(b)).

Kaplan–Meier curves of PFS (a) and OS (b) based on the presence of GCN therapy-induced hypomagnesemia at Day 21 after the first dose.
Multivariate analysis
Multivariate analysis showed that hypomagnesemia in fewer than 21 days after the first dose of GCN therapy was an independent prognostic factor of both PFS (hazard ratio (HR) 2.46, 95% CI 1.20–5.05, p = 0.014) and OS (HR 2.78, 95% CI 1.17–6.59, p = 0.021; Tables S5 and S6).
Discussion
In this subanalysis of the NINJA study, we evaluated the incidence of hypomagnesemia according to the baseline serum Mg concentration, the correlation between the severity of hypomagnesemia and the number of necitumumab doses administered during the induction phase of GCN therapy, and the association between hypomagnesemia and survival in patients receiving second-line or later GCN therapy.
Our findings suggest that second-line or later GCN therapy may be tolerable for patients with LSqCC regardless of their baseline serum Mg concentration. However, an early reduction in serum Mg concentration during GCN treatment may be a poor prognostic factor.
In our subanalysis, the incidence of grade ⩾2 hypomagnesemia in patients with a low serum Mg concentration at baseline tended to be higher than that in patients with a normal Mg concentration. Kimura et al. 19 reported that one of the risk factors for grade 3 or 4 cetuximab-induced hypomagnesemia in patients with colon cancer was a baseline serum Mg concentration <1.8 mg/dL. Our results are consistent with those of their retrospective study. Furthermore, our study showed that the severity of hypomagnesemia correlated with the number of administered necitumumab doses, which is consistent with previous reports using other anti-EGFR antibodies. 20 However, in our subanalysis, GCN therapy discontinuation due to hypomagnesemia was observed in only one patient with a low serum Mg concentration at baseline, and no hypomagnesemia-related symptoms were noted. Thus, in terms of hypomagnesemia, second-line or later GCN therapy may be well tolerated regardless of the patient’s serum Mg concentration at baseline.
Regarding the association between hypomagnesemia induced by GCN therapy and survival in our study, patients with hypomagnesemia that occurred fewer than 21 days after administration of the first dose of GCN had a significantly unfavorable PFS compared with patients without hypomagnesemia. The same tendency was observed for OS. Based on the multivariate analysis, this result is robust.
In colon cancer, a subanalysis of two randomized phase III trials using cetuximab monotherapy suggested that an early reduction in serum Mg concentration caused by cetuximab was associated with a poor prognosis. In the NCIC CTG/AGITG CO.17 trial, Vickers et al. demonstrated that patients who developed cetuximab-induced hypomagnesemia within 28 days of treatment initiation had worse survival outcomes than those without this condition (median OS: 3.7 vs 8.0 months for grade ⩾1 vs 0, respectively; HR 1.60, 95% CI 1.10–2.31). Similarly, patients with a ⩾20% reduction in serum Mg concentration from baseline within 28 days also had worse survival outcomes than those with a <20% reduction in serum Mg concentration (HR 2.01, 95% CI 1.28–3.16). 10 Furthermore, in the ASPECCT trial, in which the median number of administered cetuximab doses was 14, Price et al. demonstrated that patients who developed cetuximab-induced hypomagnesemia within 35 days of treatment initiation had worse survival outcomes than those without this treatment-related adverse event (median OS: 8.1 vs 10.5 months; HR 1.67, 95% CI 1.08–2.56). Similarly, patients with a ⩾20% reduction in serum Mg concentration from baseline within 35 days also had worse survival outcomes than those with a <20% reduction in serum Mg concentration (median OS: 7.3 vs 10.8 months; HR 2.16, 95% CI 1.52–3.08). 11 The results of our NINJA study are consistent with those two previous trials. However, in the ASPECCT trial, the patients with hypomagnesemia at any point during the trial had longer median survival time versus those without hypomagnesemia (median OS: 12.6 vs 9.3 months for grade ⩾1 vs 0, respectively; HR 0.71, 95% CI 0.56–0.90, median PFS: 6.6 vs 3.2 months for grade ⩾1 vs 0, respectively; HR 0.54, 95% CI 0.43–0.69), while no significant difference was observed between these two groups in NINJA study. This discrepancy might be attributed to the different impact of the patients on OS who developed hypomagnesemia in the late treatment phase between the two studies. Furthermore, based on the previous reports in colon cancer, as well as the NINJA study, the number of administered anti-EGFR antibody doses correlates with the reduction of serum Mg concentration, the patients who could receive anti-EGFR antibodies more frequently are more likely to develop hypomagnesemia while being able to achieve longer PFS and OS. Thus, in our NINJA study with a median number of administered necitumumab doses during a follow-up period of 7, the magnitude of survival benefit might have been attenuated by the small number of administered necitumumab doses.
Although our study results that the early serum Mg reduction is associated with a poor prognosis are consistent with those of previous reports in colon cancer, no clear biological explanation has been identified; thus, the relationship between an early reduction in serum Mg concentration and cancer progression remains unclear.
Ionized Mg has several important functions in the human body, including intracellular signaling, serving as a cofactor for protein and DNA synthesis, DNA duplication and repair, apoptosis, the inhibition of chemical carcinogenesis, the reduction of inflammation and oxidative stress, phosphorylation, cardiovascular tone, neuromuscular excitability, and bone formation. However, the role of Mg in oncogenesis is controversial.21,22 In vitro, Mg deficiency has been suggested to lead to a decrease in tumor growth depending on the upregulation of cell cycle inhibitory proteins such as p21 and p27 and the downregulation of cell cycle promoting proteins such as cyclin D and E. 23 However, in murine models, Mg deficiency upregulates cytokines such as tumor necrosis factor-α, interleukin-1, and interleukin-6, contributing to genetic instability and an invasive and metastatic phenotype.24 –26
Further basic research and prospective clinical trials are required to confirm the association between anti-EGFR antibody-related hypomagnesemia and patient prognosis. However, the association between a poor prognosis and an early reduction in serum Mg concentration caused by GCN therapy in patients with LSqCC is a new insight in clinical settings, and our results suggest that early evaluation of hypomagnesemia may be useful for predicting the prognosis of patients receiving GCN therapy.
Our study has certain limitations. First, it had a retrospective design without strict evaluation rules regarding the baseline serum Mg measurement and the timing of its measurement after GCN treatment compared to a prospective design study. Therefore, 17 patients were excluded from the ITT population, which may have limited the reliability of the results, and a strict conclusion cannot be drawn regarding the association between early reduction in Mg concentration induced by GCN therapy and survival. However, in Japan, a guide for the appropriate use of GCN in clinical settings exists; therefore, the results of our study might be replicated in the NESSIE study (jRCTs051200138), an ongoing phase II study using GCN therapy as second-line treatment. 27 Second, our study lacked information on patient comorbidities, such as frailty/sarcopenia, poor oral intake, dysphagia, or odynophagia, and those associated with the use of medications that affect Mg concentration. Loop diuretics, thiazide diuretics, and proton pump inhibitors are the major drugs that can cause hypomagnesemia. 28 Furthermore, Sato et al. 29 suggested that the use of antacids during treatment with anti-EGFR antibodies may promote hypomagnesemia. Thus, we could not clearly conclude whether the frequency of hypomagnesemia was solely due to GCN therapy. However, these findings suggest that second-line or later GCN therapy is tolerable in patients who are candidates for cisplatin-containing chemotherapy. Third, the dosage, dosing interval, and method of prophylactic Mg administration were not evaluated. Therefore, the significance of prophylactic Mg administration during GCN therapy could not be verified. Fourth, we could not clarify the mechanism of either the relationship between early serum Mg reduction and poor prognosis, or between hypomagnesemia in the early phase and poor efficacy of GCN treatment in our study. Further elucidation of this issue is warranted.
Conclusion
In conclusion, GCN as a second-line or later treatment may be tolerable and effective regardless of the patient’s baseline serum Mg concentrations. On the other hand, early reduction in serum Mg concentration, occurring in fewer than 21 days after the first dose of GCN, may be a poor prognostic factor. However, caution should be needed because our results lacked sufficient information for confounding variables other than those analyzed here that may influence the correlation between hypomagnesemia and survival, such as comorbidities, medications, and nutritional conditions. Further investigations are needed to confirm our study results.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251318850 – Supplemental material for Clinical impact of hypomagnesemia induced by necitumumab plus cisplatin and gemcitabine treatment in patients with advanced lung squamous cell carcinoma: a subanalysis of the NINJA study
Supplemental material, sj-docx-1-tam-10.1177_17588359251318850 for Clinical impact of hypomagnesemia induced by necitumumab plus cisplatin and gemcitabine treatment in patients with advanced lung squamous cell carcinoma: a subanalysis of the NINJA study by Shigeru Tanzawa, Hiroshige Yoshioka, Toshihiro Misumi, Eisaku Miyauchi, Kiichiro Ninomiya, Yasunori Murata, Masafumi Takeshita, Fumihiko Kinoshita, Takatoshi Fujishita, Shunichi Sugawara, Yosuke Kawashima, Kazuki Hashimoto, Masahide Mori, Akihiko Miyanaga, Anna Hayashi, Hisashi Tanaka, Ryoichi Honda, Masafumi Nojiri, Yuki Sato, Akito Hata, Nobuhisa Ishikawa, Toshiyuki Kozuki, Takahisa Kawamura, Go Saito, Teppei Yamaguchi, Kazuhiro Asada, Satoshi Tetsumoto, Hiroshi Tanaka, Satoshi Watanabe, Yukihiro Umeda, Kakuhiro Yamaguchi, Kazuya Nishii, Kosuke Tsuruno, Yuki Misumi, Hiroshi Kuraishi, Ken Yoshihara, Akira Nakao, Akihito Kubo, Toshihiko Yokoyama, Kana Watanabe and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
