Abstract
Background:
A newly generic microspheres, sustained-release formulation of triptorelin acetate 3.75 mg has been developed.
Objectives:
To evaluate the efficacy, pharmacokinetics, and safety of triptorelin 1-month formulation in Chinese patients with prostate cancer.
Design:
An open-label, multicenter clinical trial with one arm testing a 1-month sustained-release triptorelin formulation in prostate cancer patients.
Methods:
Patients with prostate cancer received three consecutive 28-day injections of triptorelin acetate. The primary endpoint was the proportion of successful patients over the total number of evaluable patients. Treatment success was defined as testosterone suppression below the clinical castration level (i.e., <0.5 ng/mL) at day 28 and maintenance of clinical castration until study completion (day 84). The frequency of patients with testosterone concentrations <0.2 ng/mL was also studied.
Results:
The study included 125 patients. All 125 patients received at least one dose of the study drug and 122 completed the study. The successful patient proportion among the evaluable patients was 97.6% (122/125; 95% CI, 92.7–99.2). 95.1% (116/122) achieved testosterone concentrations <0.2 ng/mL. The pharmacokinetic profile of triptorelin during the first 3 months of treatment, evaluated in a subset of the study population (n = 11), showed sustained release of triptorelin from the formulation. Values for AUC0−τ calculated from day 0 to 28, and day 56 to 84 were 134.42 (28.76), and 154.72 (21.86) h*ng/mL, respectively. The most common treatment-related adverse events were increased alanine aminotransferase (18.4%), increased aspartate aminotransferase (16.0%), and hot flashes (9.6%). Prolonged QT interval on electrocardiogram, erectile dysfunction, and decreased libido each occurred in ⩽4% of the patients. The frequently reported local adverse reaction was pain at the injection site, experienced by 2.4% (3/125) of the patients.
Conclusion:
3.75-mg Triptorelin acetate microspheres for injection were effective in achieving and maintaining testosterone suppression and were well tolerated in patients with prostate cancer.
Trial registration:
chictr.org.cn (ChiCTR2000033188).
Keywords
Introduction
Prostate cancer (PCa) is the second most frequent cancer and the fifth leading cause of cancer death among men in 2020. 1 The age-standardized rate or PCa incidence in China was 10.2 per 100,000 in 2020.1,2 Due to its vast population, China accounts for 8.2% of new PCa cases and 13.6% of PCa-related deaths globally, 1 which highlights the urgent need for greater attention to PCa in China.
Since the 1940s, suppression of gonadal production of testosterone via androgen deprivation therapy (ADT) has been the backbone of the management of advanced prostate cancer.3,4 Approximately 90% of PCa respond to initial androgen deprivation, providing patients with a quality-of-life benefit. 5 Medical castration, utilizing gonadotropin-releasing hormone (GnRH) agonists or antagonists, has become the preferred approach over surgical castration due to its reversibility and comparable efficacy.6,7 GnRH agonists, such as triptorelin, have been widely used since the 1980s for ADT. Over the last three decades, GnRH antagonists have been developed, with degarelix as a depot injection being the only one approved for the treatment of metastatic prostate cancer to date. It offers a more rapid onset of action and is associated with a lower incidence of cardiovascular side effects compared to agonists. 8 However, the clinical use of degarelix is limited due to the requirement for monthly injections and an incidence of injection-site reactions approaching 40%. 9
Triptorelin, like other GnRH agonists, has a short elimination half-life (t1/2 = 5 h). 10 The peptide was recently incorporated into microgranules of the biocompatible and biodegradable copolymer poly (lactide-co-glycolide) (PLGA) to create sustained-release compositions.11,12 Sustained-release 1-, 3- and 6-month formulations of triptorelin, administered intramuscularly or subcutaneously, have been developed by adjusting the molar ratio of lactic acid to glycolic acid, providing patients with greater flexibility and convenience. Sustained-release formulations of triptorelin are also utilized in the treatment of endometriosis, uterine fibroids, precocious puberty, female infertility, and sexual deviation.5,13,14 However, the high costs and technical challenges of these formulations disproportionately affect the accessibility and affordability of such treatments in developing countries. To give the target population another option, Livzo Pharmaceutical Group Inc. developed an original sustained-release formulation of triptorelin acetate. They strategically selected PLGA 5050 2.5 A as the carrier due to its superior drug-loading capacity and its established track record as a dependable and adaptable biomaterial, ensuring the efficacy and reliability of the drug delivery system. Pharmacokinetic studies in healthy volunteers have demonstrated that the modified triptorelin formulation has optimal release properties and castration effects.
This open-label, multicenter clinical study was performed to investigate the efficacy, pharmacokinetic properties, and safety profile of triptorelin acetate 3.75-mg in suppressing testosterone levels in patients with prostate cancer. Primary efficacy criteria were the proportion of patients achieving castrate serum testosterone levels by day 28 and the proportion of patients maintaining castration from month 2 until the end of month 3 (day 84), where castration was defined according to the standard and recognized serum testosterone level of <1.7 nmol/L (<0.5 ng/mL).15,16
Patients and methods
Study design and patients
This was an open-label, non-comparative, multicenter study with triptorelin acetate 3.75 mg given at an interval of 4 weeks to patients with locally advanced and metastatic advanced prostate cancer (Clinical Trial Registration, ChiCTR2000033188 at www.chictr.org.cn). Thirty-three centers in China participated in the study (Supplemental Table S1). Patients were enrolled in the study between April 6, 2020 and January 20, 2021.
Male subjects, 18 years or older, with histologically proven prostate cancer (locally advanced or metastatic disease), were included. Other key eligibility criteria included serum testosterone >1.5 ng/mL at screening; Eastern Cooperative Oncology Group (ECOG) performance status score of 0–1; serum prostate-specific antigen (PSA) ⩾4.0 ng/mL at screening; adequate end-organ function; a life expectancy of at least 6 months; and no known hypersensitivity to the active ingredient or excipients of the study formulation. The main exclusion criteria were hormonal treatment for prostate cancer within 6 months before the study started, use of finasteride or dutasteride within 1 month before the study started, known or suspected vertebral metastases with risk of spinal compression, and any use of medications known to affect the metabolism and/or secretion of androgenic hormones within 6 months before treatment start.
A signed informed consent that conformed to the Declaration of Helsinki and local laws was obtained from each patient before entry into the study. The reporting of this study conforms to the standard protocol items specified in the CONSORT Statement. 17
Investigational product
Triptorelin was presented as a freeze-dried powder in a single-dose vial and was reconstituted with 2 mL of solvent before injection.
Study procedures and assessments
Patients who met the eligibility criteria were given their first dose of triptorelin within 7 days of their screening visit. All patients were to receive three consecutive intramuscular investigational triptorelin injections on day 0, 28, and 56, followed by three consecutive marketed brand triptorelin injections since day 84. However, patients enrolled pharmacokinetic/pharmacodynamic (PK/PD) substudy would receive more course-marketed brand triptorelin injections starting on day 112. The flow chart of study design is shown in Figure 1.
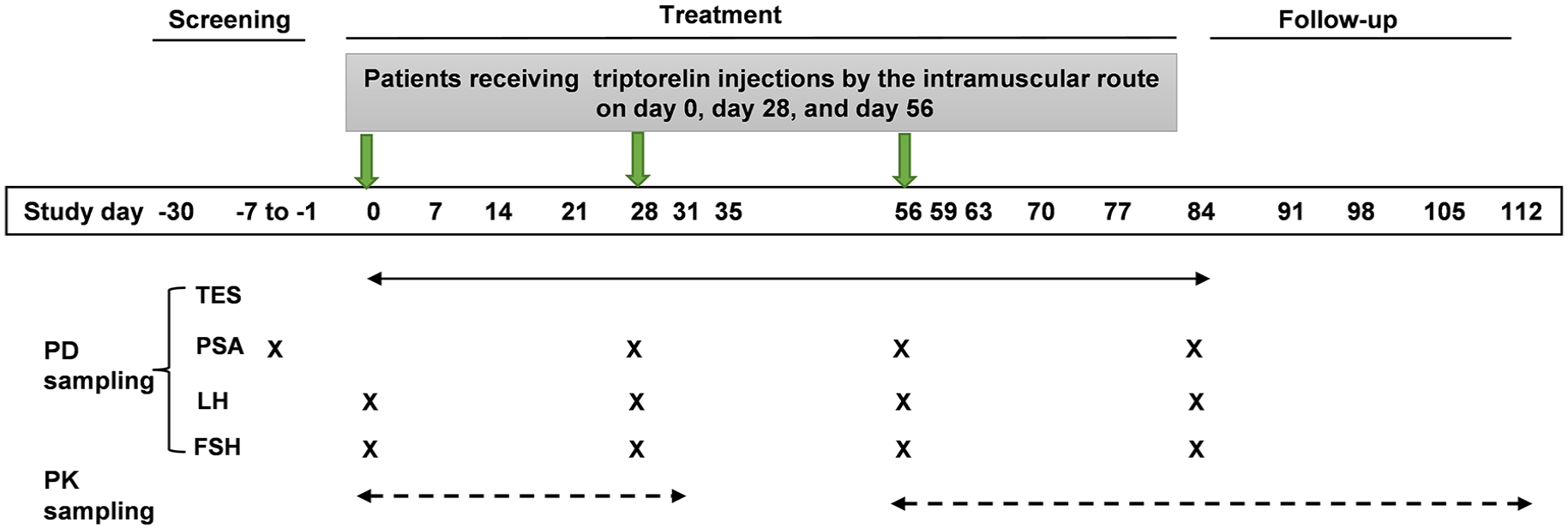
Flow chart of study design.
PK sampling
A total of 14 patients had additional blood samples taken for triptorelin PK analysis. Blood samples for triptorelin analysis were collected to EDTA-K2 anticoagulant Tube with 50 μL aprotinin solution on day 0 prior to dose administration, and 0.5, 1, 1.5, 2, 3, 4, 6, 8, and 12 h post first dosing; and 24 h (day 1), 36 h, 48 h (day 2), 72 h (day 3), 96 h (day 4), 120 h (day 5), 144 h (day 6), 168 h (day 7), 336 h (day 14), 504 h (day 21), 672 h (day 28, predose second injection), day 31 (72 h post second dosing), day 56 (30 min before the third injection), and 0.5, 1, 1.5, 2, 3, 4, 6, 8, and 12 h post third dosing; and 24 h (day 1), 36 h, 48 h (day 2), 72 h (day 3), 96 h (day 4), 120 h (day 5), 144 h (day 6), 168 h (day 7), 336 h (day 14), 504 h (day 21), 672 h (day 28), day 35, day 42, day 49, day 56 post third dosing. Centrifuge conditions: 1600g, for 10 min at 4°C. The plasma was stored at −70°C ± 10° C and was detected by liquid chromatography with mass spectrometry (LC-MS/MS) in the central lab.
PD sampling
Blood samples were prospectively collected from each patient at baseline and during efficacy assessments. Blood samples for testosterone analysis were drawn on day 0 (30 min before the first triptorelin injection), day 28 (30 min before the second injection), day 31, day 35, day 56 (30 min before the second injection), day 59, day 63, and day 84 (predose). Blood samples for PSA were collected on day −7, day 28 (predose), day 56 (predose), and day 84. Blood samples for luteinizing hormone (LH) and follicle-stimulating hormone (FSH) were collected on day 0 (predose of the first injection), day 28 (predose), day 56 (predose), and day 84. Blood samples for efficacy assessments were collected in polyethylene tubes, kept at room temperature for 30 min, and then centrifuged at 1200g for 10 min to obtain serum. PD samples were frozen and stored at −70°C or below before batch transport to the central lab for analysis.
Patients eligible for the PK/PD substudy provided additional blood samples for testosterone analysis. Samples were collected at 0 h (30 min before the first injection), 24 h (day 1), 48 h (day 2), 72 h (day 3), 96 h (day 4), 120 h (day 5), 144 h (day 6), 168 h (day 7), 336 h (day 14), 504 h (day 21), 672 h (day 28, predose of second injection), day 31 (72 h post second dosing), day 35 (168 h post second dosing), day 56 (30 min before the third injection), and 3, 7, 14, 21, 28, 35, 42, 49, and 56 days post the third dosing. Serum levels of testosterone were determined by LC-MS/MS.
Outcomes
The primary efficacy endpoints were the percentage of subjects with serum testosterone <0.5 ng/mL (castration level) by day 28 ± 1 (i.e., within 28 days following the first injection of investigational product) and the percentage of subjects with testosterone suppression (<0.5 ng/mL) from day 28 through day 84 at all monthly assessments. Insufficient suppression of testosterone was defined as suppression that did not occur by day 28 or the occurrence of a testosterone level greater than or equal to 0.5 ng/mL at any point of monthly assessments between day 28 and 84.
Secondary endpoints included the proportion of subjects exhibiting castrated levels (<0.5 ng/mL) at 3 and 7 days after the second and third administration. The efficacy was further analyzed by examining the percentage of subjects with serum testosterone <0.2 ng/mL on day 28 and the cumulative probability of testosterone <0.2 ng/mL from day 28 to 84. Secondary endpoints also included the change from baseline in serum PSA values, and the proportion of subjects whose degree and PSA decreased by more than 90% from the baseline value. In addition, serum LH and FSH levels (time course change) were evaluated.
The PK behavior of triptorelin was evaluated by full PK profiles from serum triptorelin concentrations in subjects.
Safety
Treatment-emergent AE frequencies in patients who had received at least one triptorelin injection were summarized by body system and preferred term within the body system, along with severity and relationship to treatment. Local tolerance was summarized using absolute and relative frequencies. Laboratory examinations, vital signs, 12-lead electrocardiogram results, and physical examinations were recorded and summarized with appropriate descriptive statistics.
Statistical analyses
Sample size was calculated to demonstrate achievement and maintenance of castrate levels of testosterone in 96% of patients with a two-sided 95% confidence lower limit of 90% (with a drop-out rate of 20%). The populations for analysis were the full analysis set (FAS), per-protocol set (PPS), safety analysis set (SS), and pharmacokinetic analysis set (PKAS). The FAS was defined as subjects diagnosed with prostate cancer in whom the study drug was administered and at least one efficacy variable was evaluated after administration. The PPS is defined as subjects in the FAS, excluding those with any of the following significant protocol violations regarding study method and concomitant therapy: Violation of the inclusion or exclusion criteria and critical violation of the protocol that could affect the efficacy evaluation. Efficacy will be analyzed in FAS and PPS, and FAS is the main analysis set. The SS was defined as subjects who received the study drug. The PKAS was defined as subjects who received the study drug and from whom a sample for drug concentration measurement was collected at one or more time points.
The efficacy of the treatment was assessed in accordance with the FDA-recommended Kaplan–Meier analysis method and the FDA criteria for GnRH analog approval. The FDA standards specify that the lower limit of the 95% confidence interval for the estimated percentage of patients maintaining castrate testosterone levels should be greater than 90%. 18 Treatment success was defined as plasma testosterone concentrations <0.5 ng/mL on day 28 and maintenance of castration levels at all monthly assessments until day 84, with no missing data at the monthly assessments. Sensitivity analyses for the primary endpoint included participants who withdrew from the trial permanently or had two or more consecutive missing test results, irrespective of their testosterone levels before and after the missing tests. In addition, the percentage of subjects achieving testosterone suppression (<0.2 ng/mL) was analyzed using the same Kaplan–Meier approach. The exact Clopper-Pearson method was used to calculate the 95% CI for the incidence rates of subjects exhibiting castrated levels (<0.5 ng/mL) at 3 and 7 days after the second and third administration and the proportion of subjects with a ⩾90% reduction in PSA from day 28 through the end of the treatment period. The percentage change in serum LH and FSH from pretreatment to day 28, 56, and 84 was reported using descriptive statistics. Triptorelin pharmacokinetics were reported as descriptive statistics. The frequency of AEs and adverse drug-related events (ADRs) was tabulated according to System Organ Class and Preferred Term of MedDRA.
All statistical analyses, including pharmacokinetic parameter calculations, were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA) and WinNonlin version 8.1 (Pharsight, Mountain View, CA, USA) (pharmacokinetics).
Results
Patients characteristics
The disposition of patients is shown in Figure 2. In total, 179 patients were screened, and 125 patients were enrolled. Three patients discontinued treatment within 28 days for the following reasons: Two for withdrawal of consent and one for other reasons. The baseline characteristics of the patients are shown in Table 1. There were 122 subjects who entered to FAS analysis and 122 subjects fitted in the per-PPS analysis.
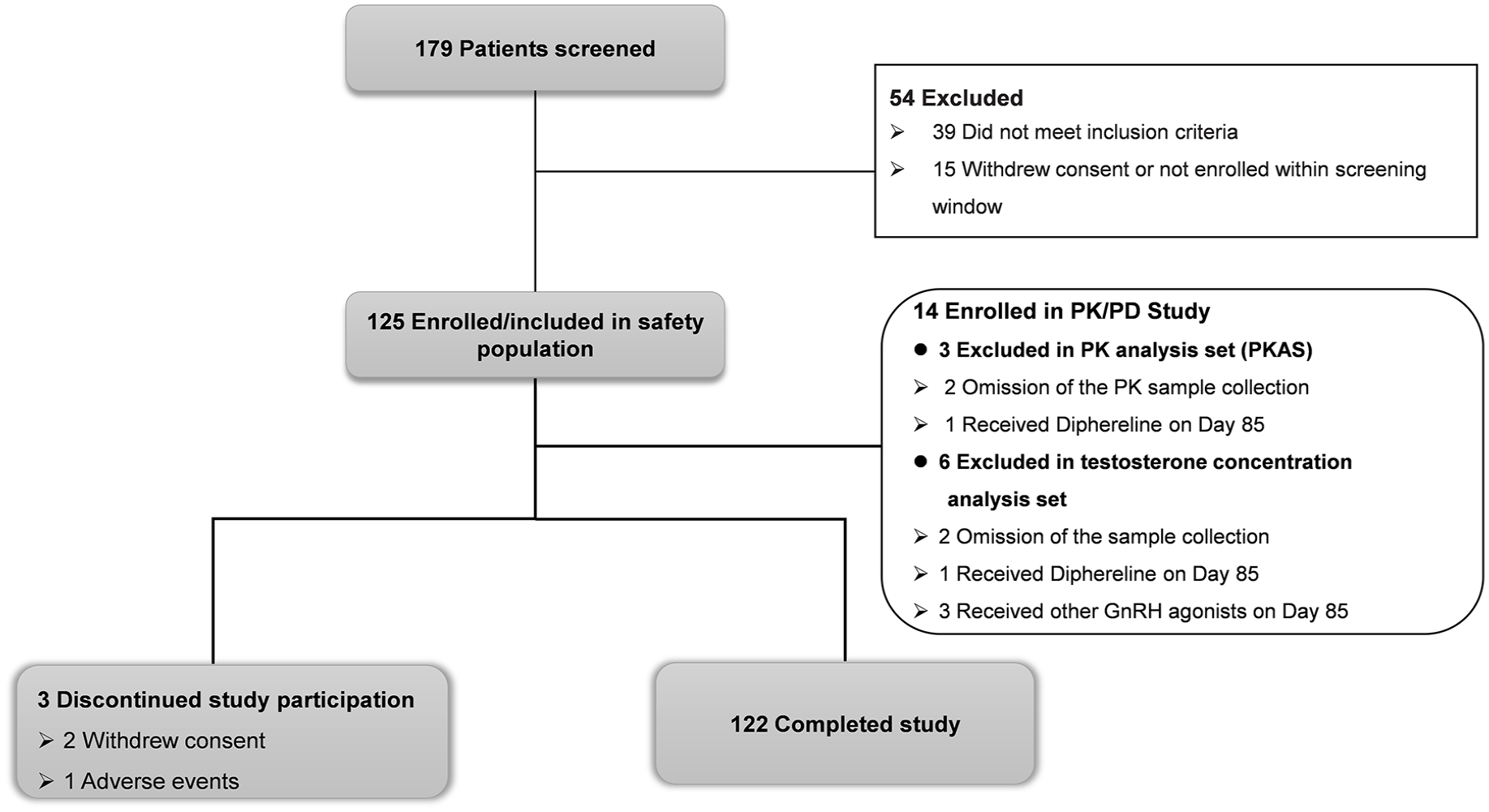
Subject disposition. Flow of subjects through the trial.
Patient baseline characteristics (full analysis population).

ECOG, Eastern Cooperative Oncology Group; FAS, full analysis set; FSH, follicle-stimulating hormone; LH, luteinizing hormone; PSA, prostate-specific antigen.
Efficacy
At day 28, testosterone levels were less than 0.5 ng/mL in 97.6% of the recruited participants (122/125), with three individuals withdrawing within the first 28 days. From day 28 to 84, castration levels were maintained in 96.0% of the patients (120/125). This statistic accounts for two additional patients who exhibited an “acute-on-chronic” increase in testosterone levels exceeding 0.5 ng/mL, one on day 31 and the other on days 59–63.
Primary endpoint
The primary efficacy analysis population was the FAS population. One hundred twenty-two patients (122/122, 100%) achieved serum testosterone level <0.5 ng/mL on day 28. The cumulative probability of serum testosterone <0.5 ng/mL from day 28 to 84 (FAS, n = 122) was 100% (122/122; 95% CI, 100–100). On FDA request, 17 a more restricted analysis was performed that included not only patients with non-castrate testosterone levels but also patients with missing testosterone values between day 28 and 84 as failures. In the present study, only three patients had at least one missing testosterone value, and they all dropped out within 28 days. In other words, 97.6% of evaluable patients had castrate testosterone concentrations. The exact binomial test was fully satisfied (i.e., the null hypothesis was rejected at p < 0.001), and the two-sided exact 95% CI was 92.7–99.2, thus confirming the achievement of the primary endpoint.
Secondary endpoints
i. Only two subjects (2/122, 1.6%) exhibited post-suppression elevations of serum testosterone to > 0.5 ng/mL after reaching castrate level of testosterone on day 28. One was on day 31, and the other was on days 59–63. These transient elevations of serum testosterone levels were observed following the second administration on day 28 and the third administration on day 56. Both subjects had their serum testosterone levels below the castrate levels by the end of the study (day 84).
ii. Of 122 subjects who achieved the castrated level serum testosterone (<0.5 ng/mL), 116 subjects (116/122, 95.1%) achieved serum testosterone level <0.2 ng/mL on day 28. On day 56 and 84, all subjects who completed the study (122/122) achieved castrated serum testosterone levels <0.2 ng/mL. The cumulative probability of serum testosterone <0.2 ng/mL from day 28 to 84 was 95.1% (95% CI: 89.4–97.8; FAS, n = 122).
iii. Regarding the pharmacodynamic assessments, the mean-time course change in serum PSA levels from baseline to day 84 are shown in Figure 3(a). For FAS, the median percent change in serum PSA level from baseline (day −7 to −1) to day 28, 56, and 84 was −75.4%, −94.4%, and −96.5%, respectively. Percentage of subjects whose PSA decreased by more than 90% from baseline was shown in Table 2.
iv. The mean-time course change in serum LH and FSH levels from baseline to day 85 is shown in Figure 3(b) and (c), respectively.
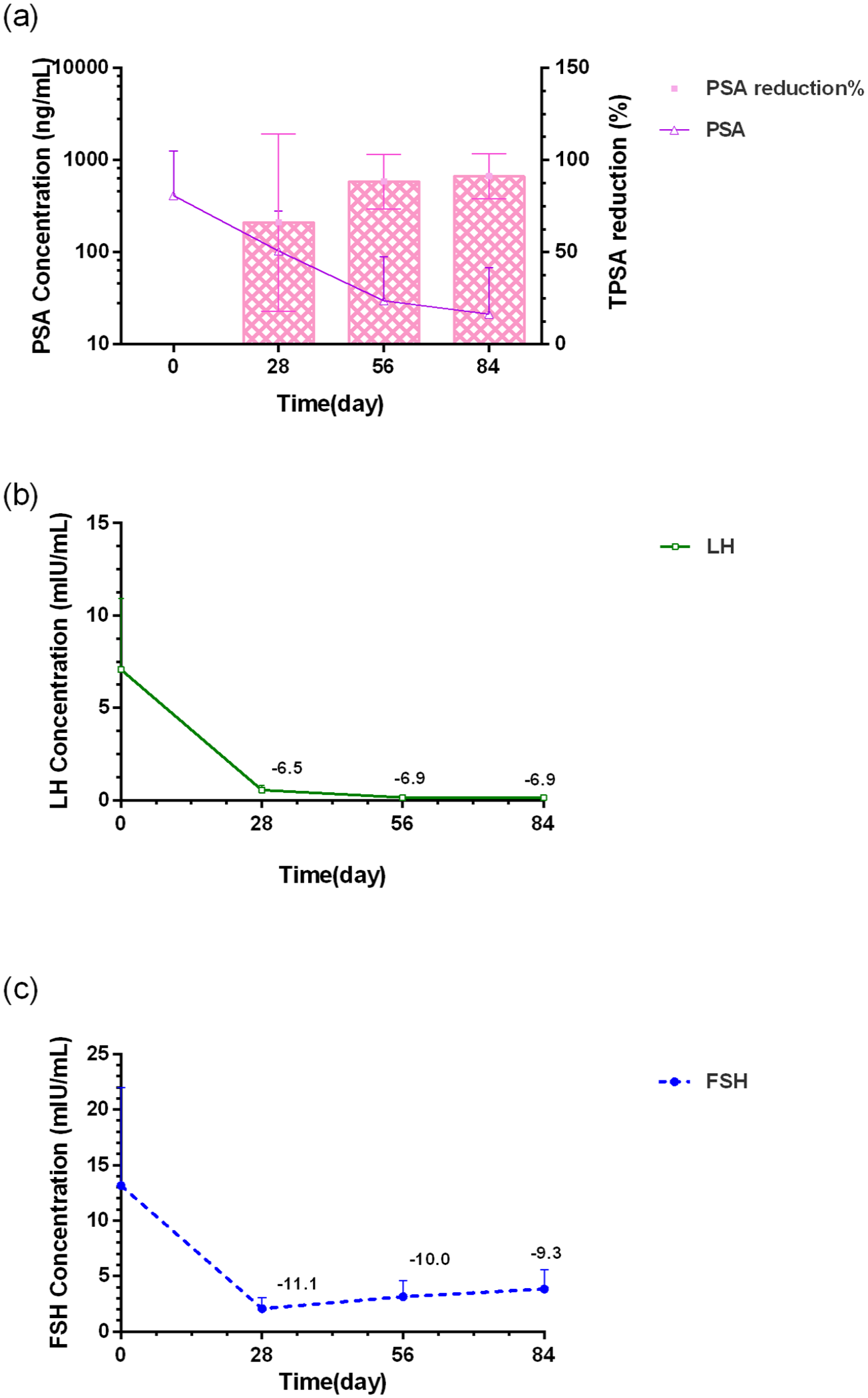
Median time course change of (a) PSA and median percent change of PSA from baseline (day −7 to −1) to day 84 (FAS). Median time course change in (b) serum LH and (c) FSH levels from baseline (day 0) to day 84 (FAS).
The proportion of subjects whose PSA decreased by more than 90% from baseline value.

CI, confidence interval; PSA, prostate-specific antigen.
Pharmacokinetic/pharmacodynamic
In the PK/PD analysis set, eight patients had sufficient testosterone measurements for analysis. Two patients were excluded due to omission of the sample collection and four received Diphereline® or other GnRH agonists on day 85. Mean testosterone levels are depicted in Figure 4(a) and (b). The time to treatment failure (serum testosterone >0.5 ng/mL) was observed from day 105, which was 49 days after the third dosing. This indicated that the modified triptorelin 3.75 mg formulation was effective in inducing and maintaining castration levels for over 6 to 7 weeks following administration.
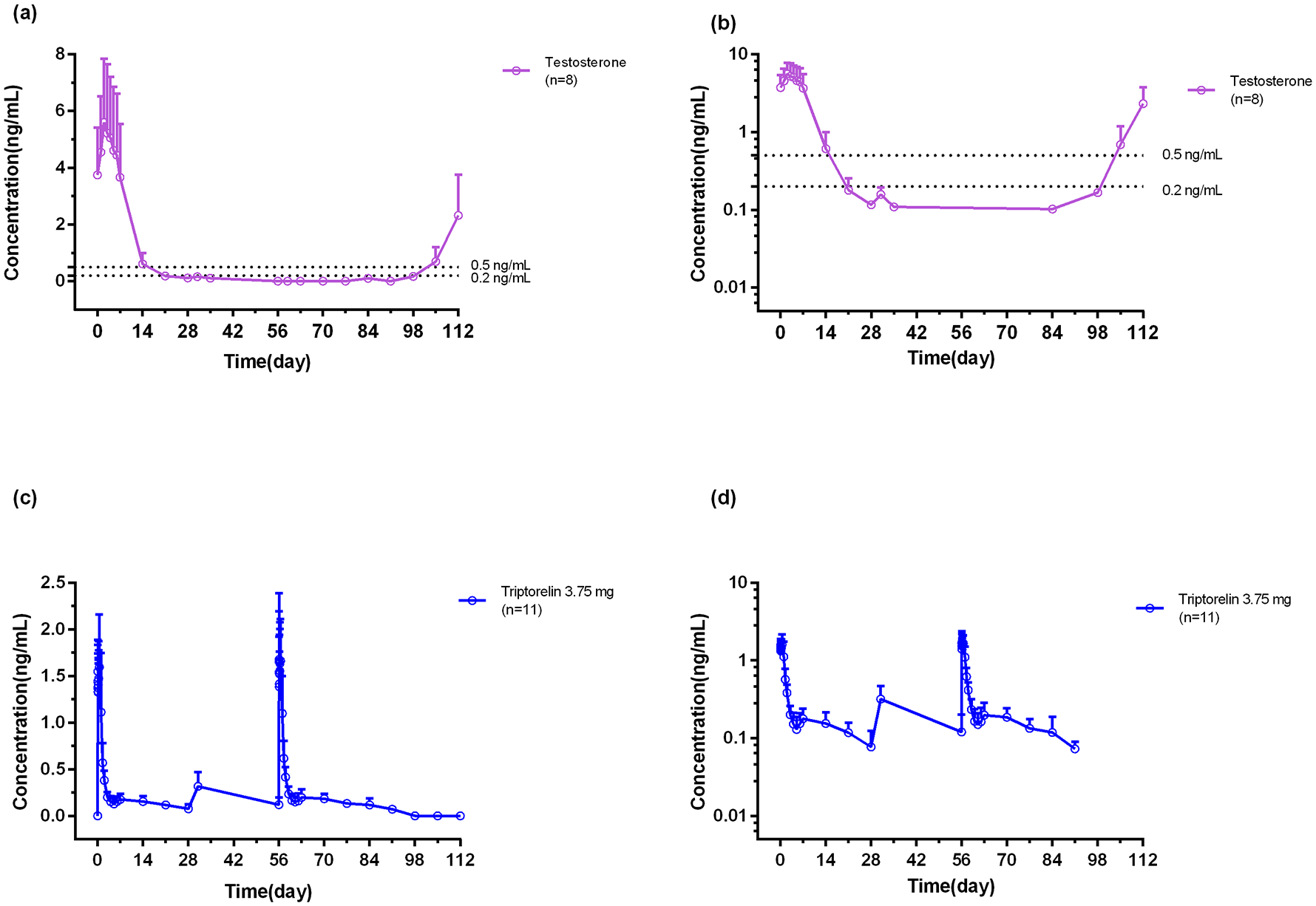
The linear (a) and semi-log (b) of mean concentrations of testosterone -time curve in serum. The linear (c) and semi-log (d) of mean concentrations of triptorelin-time curve in serum.
Eleven in 14 patients (78.6%) were included in the PKAS. Two patients were excluded due to omission of the PK samples collection and one received Diphereline on day 85. PK data showed an initial release of triptorelin after each injection with Cmax serum values of 1.85 ± 0.45 ng/mL at 7.9 h after the first injection and 2.07 ± 0.45 ng/mL at 3.1 h after the third injection (Figure 4(c) and (d)). Serum triptorelin then decreased slowly from day 4 to 28 (plateau phase). During the plateau phase mean serum triptorelin generally remained between 0.08 and 0.2 ng/mL with a 1-month mean value of 0.24 ± 0.07 ng/mL. Total bioavailability, AUC was 134.42 ± 28.76 ng*h/mL after the first dose and 154.72 ± 21.86 ng*h/mL after the third dose. Triptorelin release was 48% (63.3 ng*h/mL) during the first plateau phase and 43% (67.8 ng*h/mL) during the second plateau.
Safety
The overall incidence of AEs was 84.8% (106/125). Serious AEs occurred in 8.8% (11/125). The incidence of ADRs was 41.6% (52/125). The treatment-emergent AEs with an incidence of ⩾10% are shown in Table 3. The incidence of expected adverse reactions related to the pharmacological mechanism of the experimental drug: hot flush, erectile dysfunction, and decreased libido, were 9.6%, 3.2%, and 3.2%, respectively (Table 3). The incidence of injection-site pain was 2.4%. No serious drug-related adverse reactions occurred. There were no adverse reactions that led to withdrawal from the trial, termination of experimental medication, or death.
Summary of adverse events (safety population).

AEs, adverse events; AESIs, adverse events of special interest related to the study drug; SAE, serious adverse event; SS, safety analysis set; TRAEs, treatment-related treatment-emergent adverse events.
Discussion
In this trial, a newly developed 1-month sustained-release formulation of microencapsulated triptorelin acetate 3.75 mg was effective in achieving and maintaining castrate testosterone levels in patients with prostate cancer. The proportion of successful patients out of the total number of patients was 97.6% (122/125), confirming the study’s primary endpoint. The efficacy results were compared with other marketed GnRH agonists, including triptorelin pamoate (93.3%–94.4%), 19 leuprolide acetate (93.4%–96.8%),20 –23 leuprolide mesylate (97.0%).24,25
Triptorelin was the most potent GnRH agonist, achieving the lowest mean testosterone levels and the highest rate of chemical castration at <0.1 ng/mL testosterone. In the present study, this product’s achievement rate of significant castration and maintenance cumulative castration (serum total testosterone 0.2 ng/mL) from day 28 was 95.1%, which was significantly higher than the achievement rates of comparable triptorelin marketed formulation (72%–83%) 26 and leuprolide acetate (57%). 27 In addition, only 1.6% (2/122) of this product’s cases involve the “acute-on-chronic” phenomenon, which is significantly lower than the 4%–10% range for comparable products. 28
The sustained suppression of testosterone with these triptorelin formulations leads to reductions in PSA levels. The triptorelin 3.75 mg 1-month formulation was shown to reduce median PSA levels from 100 ng/mL at baseline to 3.39 ng/mL at 84 days. Before the second triptorelin injection, the mean LH level was fully suppressed to only 0.56 mIU/mL, whereas before the first injection, it was 7.08 mIU/L. The above analysis illustrates that the new triptorelin product, which is a sustained-release formulation, is an efficient medication to decrease the testosterone level below castration levels.
The innovative 1-month formulation maintains relatively stable triptorelin serum concentrations after the short initial burst. Cmax was comparable after the first and third injections. Microspheres formulated with PLGA 5050 2.5 A and prepared using the solvent emulsion evaporation method may result in high encapsulation efficiency, low initial burst, and long-term sustained release.
Regarding the drug safety profile for this study, adverse effects generally reflected testosterone suppression caused by triptorelin administration. The occurrence rate of hot flashes of common castration-related adverse reactions of this product (3.75 mg) was only 9.6%, which was significantly lower than that of marketed 1-month triptorelin formulations TRELSTAR® (58.6%). 19 In our study, adverse reactions at the injection site (2.4%) were lower than that reported for the available 1-month formulation (3.75 mg TRELSTAR), for which local reactions were 3.6%. 19
The present study has some limitations. First, since this study only included Chinese patients, our findings cannot be generalized to other ethnic populations. Second, this study is an open-label, single-arm study design and no direct safety and efficacy comparisons to reference drugs can be done. Finally, the dosing duration was limited. Last but not least, longer sustained-release medications are currently being developed. By lowering the frequency of injections, these medications may be more convenient for both patients and doctors. While some patients (and physicians) prefer a shorter injection schedule to have more frequent visits. Therefore, the 1-month formulation remains a viable option for prostate cancer therapy.
Overall, the results of the present study reveal a safety profile consistent with the triptorelin safety profile established in previous clinical studies and reported for pharmacologic ADT used in the treatment of prostate cancer.
Conclusion
The results of this study indicate that the new modified 1-month triptoreline acetate 3.75-mg microspheres formulation is effective in achieving and maintaining testosterone concentration below castration levels in patients with prostate cancer and is well tolerated.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241307818 – Supplemental material for Effectiveness, pharmacokinetics, and safety of triptorelin acetate microspheres in patients with locally advanced and metastatic prostate cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359241307818 for Effectiveness, pharmacokinetics, and safety of triptorelin acetate microspheres in patients with locally advanced and metastatic prostate cancer by Guolan Wu, Feng Zhou, Haiping Wang, Kan Liu, Dexin Yu, Lianlian Fan, Yangyun Han, Xiaohong Ai, Youhan Cao, Xiaolin Wang, Sheng Wang, Chaohong He, Jitao Wu, Ji Wu, Youlei Wang, Yanqing Wang, Baiye Jin and Jianzhong Shentu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank the clinical site staff and for their contributions. The authors also would like to thank N. N. Xu and Z. J. Zhou for their assistance in quality control through the entire clinical trial. The authors thank Jiangsu Value Pharmaceutical Services Co., Ltd. and Guangzhou Kingmed Center For Clinical Laboratory Co., Ltd. for the bioanalytical analysis. The authors would also like to thank the contributions from the study sponsor Livzon Pharmaceutical Group Inc.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
