Abstract
Background:
While targeted therapies have transformed the treatment landscape of oncogene-addicted non-small cell lung cancer (NSCLC), the influence of sex on treatment outcomes remains insufficiently understood.
Objectives:
This systematic review aimed to investigate the impact of sex on clinical outcomes in patients with NSCLC harboring driver fusions treated with targeted therapies enrolled in clinical trials.
Data sources and methods:
A comprehensive literature search was conducted using PubMed, Embase, and relevant conference abstracts to identify phase III randomized and early clinical trials that reported sex-specific data, including progression-free survival (PFS), overall survival (OS), overall response rate, and adverse events (AEs), in patients with fusion-positive NSCLC treated with tyrosine kinase inhibitors (TKIs).
Results:
This review involved 10 studies reporting PFS data and 3 studies with OS data, focusing on first-line treatments for ALK fusion (9 studies) and RET fusion-positive (1 study) NSCLC. Pooled analysis of hazard ratios (HRs) for PFS and OS in ALK inhibitors trials revealed no significant differences in survival outcomes based on sex. Additionally, none of the studies provided data on sex-based differences in response rates or toxicities, highlighting a significant knowledge gap regarding the impact of sex on secondary outcomes in targeted therapy.
Conclusion:
This review found no significant sex-related differences in survival outcomes among patients treated with ALK inhibitors. However, the lack of data on sex-specific response and toxicity emphasizes the need for future research to better understand the role of sex in modulating treatment outcomes and treatment decisions with TKIs.
Plain language summary
Introduction
Recent advances have identified critical driver alterations in non-small cell lung cancer (NSCLC), which play a significant role in the activation of cancer pathways, downstream signaling, and tumor proliferation. 1 These alterations, prevalent in lung adenocarcinoma, are found in about 60% of cases in Western populations, with higher frequencies among non-smokers, East Asians, and younger individuals. 1 Discoveries of these genetic aberrations have enabled the development of targeted therapies, such as tyrosine kinase inhibitors (TKIs), making biomarker testing a standard practice for treatment guidance.2,3
Common actionable mutations include EGFR and KRAS mutations, BRAF mutations, MET exon 14 skipping mutations, and ERBB2 mutations. Although less frequent, gene fusions involving ALK, ROS1, RET, and NTRK are also significant and targetable.4 –6 ALK fusions are present in up to 5% of NSCLC cases, notably in younger, non-smoking patients. Previous studies found no significant differences in the frequency of ALK fusions between men and women, suggesting a roughly equal distribution or slight variations depending on specific study populations.7,8 However, other sources, including some newer analyses and broader data sets, indicate a trend of slightly higher prevalence among women. This inconsistency underscores the complexity of demographic factors influencing ALK fusions and highlights that conclusions may vary depending on specific study cohorts and methods used. 9 The primary ALK fusion partner is EML4, accounting for 90%–95% of cases, although other partners like KIF5B and TFG are also identified. 9 ROS1 fusions, involving partner genes such as CD74, occur in 1%–2% of cases and are more common in younger, non-smoking patients, with no consistent sex prevalence reported across studies. Recent studies reported a higher frequency of ROS1 fusions in males.10 –14 NTRK1/3 fusions are rare in NSCLC (around 0.2% of cases) and do not appear to be influenced by sex or smoking history.15,16 RET fusions, found in 1%–2% of cases, are more common in non-smokers, and males, with adenocarcinoma histology, and frequent fusion partners are KIF5B and CCD6. 17
Targeted therapies, including TKIs, have been developed to inhibit fusion proteins, improving outcomes for patients with fusion-positive NSCLC.2,18 Current guidelines recommend testing for ALK, ROS1, NTRK1/3, and RET fusions in all patients with newly diagnosed advanced non-squamous NSCLC.6,19,20
Crizotinib, the first developed TKI for ALK fusions, has been succeeded by second-generation TKIs like alectinib, brigatinib, and ensartinib, which offer longer progression-free survival (PFS) and better blood–brain barrier penetration.21 –24 Lorlatinib, a third-generation ALK TKI, shows promising survival benefits also in the first line. 25 For ROS1-positive NSCLC, crizotinib and entrectinib are approved as first-line treatments, with entrectinib preferred in case of brain involvement.26 –28 Similarly, ceritinib and lorlatinib have shown efficacy, with good central nervous system (CNS) activity.29,30 More recently, repotrectinib, targeting ROS1 and other kinases, has demonstrated high response rates and is currently under investigation. 31 Larotrectinib and entrectinib are effective treatments for NSCLC harboring NTRK fusions and have been approved by the U.S. Food and Drug Administration and the European Medicines Agency in this setting.32 –34 RET fusion-positive NSCLC is treated with selpercatinib and pralsetinib, which are now approved by regulatory agencies.35,36
Sex and gender influence NSCLC pathogenesis, diagnosis, and treatment.37,38 Men generally have higher lung cancer incidence and mortality rates than women. On the contrary, women are more likely to have actionable molecular alterations, such as EGFR mutations, particularly among never-smokers. 39 Sex differences also account for different responses to therapies and side effects, with women often showing higher chemotherapy response rates but also greater toxicities, including nausea and vomiting.40 –42 Besides different pharmacokinetic (PK) and pharmacodynamic (PD) profiles between men and women, sex-based differences in chemotherapy effectiveness could be explained by the impact of estrogen receptors, such as ERα and ERβ, in drug sensitivity and resistance.43 –45 Sex-related differences extend to targeted therapies and immunotherapy. Specifically, despite developing a stronger immune response after treatment with immune checkpoint inhibitors, increasing evidence suggests that women could benefit less from mono-immunotherapy compared to combination treatments.46 –49 Sex-based differences in the response to targeted therapies for fusion-positive NSCLC are not fully understood. Variability in drug metabolism and response could be influenced by PK and PD differences between sexes. 50 In addition, women’s physiological differences, such as gastrointestinal transit times and body composition, may also affect drug metabolism, potentially leading to variations in drug efficacy and toxicity. 51 In the case of EGFR-mutated NSCLC, women have been shown to gain a greater benefit in terms of PFS from first and second-generation TKIs as compared to men, though overall survival (OS) benefits are less clear. Data on sex-based differences in response to ALK, ROS1, NTRK, and RET inhibitors are limited and often come from exploratory analyses.52 –55
We conducted a systematic review to evaluate and synthesize available evidence on sex differences in treatment outcomes for patients with NSCLC harboring driver fusions. Our specific aims are to assess the impact of sex on survival outcomes in patients receiving targeted therapies and to investigate sex-specific differences in treatment-related adverse events (AEs) and quality of life (QoL). By addressing these objectives, this review seeks to contribute to the understanding of personalized cancer treatment and support the development of sex/gender-sensitive therapeutic strategies in NSCLC.
Methods
We conducted a systematic review to explore sex differences in outcomes and treatment effects of targeted therapies in patients with NSCLC harboring driver fusions. We searched different databases (i.e., PubMed, Embase) for clinical trials evaluating TKIs for the treatment of gene fusions-driven NSCLC published before May 2024. The reporting of this study conforms to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 statement (Supplemental Table 1). 56
Search strategy
A comprehensive search was conducted in PubMed and Embase in May 2024. We also included meeting abstracts from conferences such as the American Society of Clinical Oncology, the European Society for Medical Oncology, and the International Association for the Study of Lung Cancer. The search strategy combined the following terms: (1) NSCLC OR lung adenocarcinoma, (2) driver fusions (ALK OR ROS1 OR NTRK OR RET), and (3) TKIs (Supplemental Table 2).
Study selection criteria
We focused on randomized phase III trials involving adults (⩾18 years) with treatment-naïve advanced NSCLC harboring ALK, ROS1, NTRK, or RET gene fusions. Eligible trials compared a targeted TKI monotherapy to standard of care (another TKI or chemotherapy) and reported hazard ratios (HRs) for OS, PFS, or objective response rate (ORR). Only trials that provided sex-specific HRs were included. Exclusion criteria include studies with insufficient data, non-advanced/metastatic lung cancer, preclinical studies, case reports, letters, comments, and reviews. Duplicate reports from the same trial were reviewed, with only the most complete and updated data included. Only English-language full-text publications were considered.
Data extraction
Two independent reviewers (R.L. and D.M.) screened titles and abstracts of retrieved records and then the full texts of potentially eligible papers, with discrepancies adjudicated by a third reviewer (P.A.). Detailed data extraction and risk of bias assessment were carried out by two independent reviewers (R.L. and D.M.), with discrepancies adjudicated by a third reviewer (P.A.). Extracted data included: (1) Basic details such as year of publication, author, and patient sex; (2) Treatment and prognosis data including PFS, OS, ORR, and safety. We assessed the methodological quality of studies (to ascertain the risk of bias) using the five-point Jadad ranking system. This system assesses the quality of randomization and double-blinding, and the flow of patients (withdrawals and dropouts). A controlled trial could receive a Jadad score of between 0 (poor methodological quality) and 5 (optimal methodological quality).
Statistical analysis
For each trial, we extracted the HRs and 95% confidence intervals (CI) for OS and PFS. We calculated overall HR for each endpoint (OS and PFS) and assessed heterogeneity (defined by p < 0.1 due to the low test power). We used a random-effects model where heterogeneity was present and generated a Forest plot for each sex and survival endpoint.
Results
Baseline characteristics of included studies
Our initial literature search identified 224 relevant citations from electronic databases. After removing duplicates, 197 studies remained. Of these, 187 studies were excluded after abstract review (for not fulfilling the inclusion criteria). Nine of these studies focused on ALK inhibitors, while one studied a RET inhibitor (Figure 1). Randomized treatment allocation sequences were generated in all trials. None of the trials was double-blinded. Jadad scores for each trial are listed in the Supplemental Material. The mean score was 3. No trial received a low-quality score (i.e., Jadad score of 1–2). All the included studies had a low risk of reporting bias, attrition bias, and other bias (Supplemental Table 3).
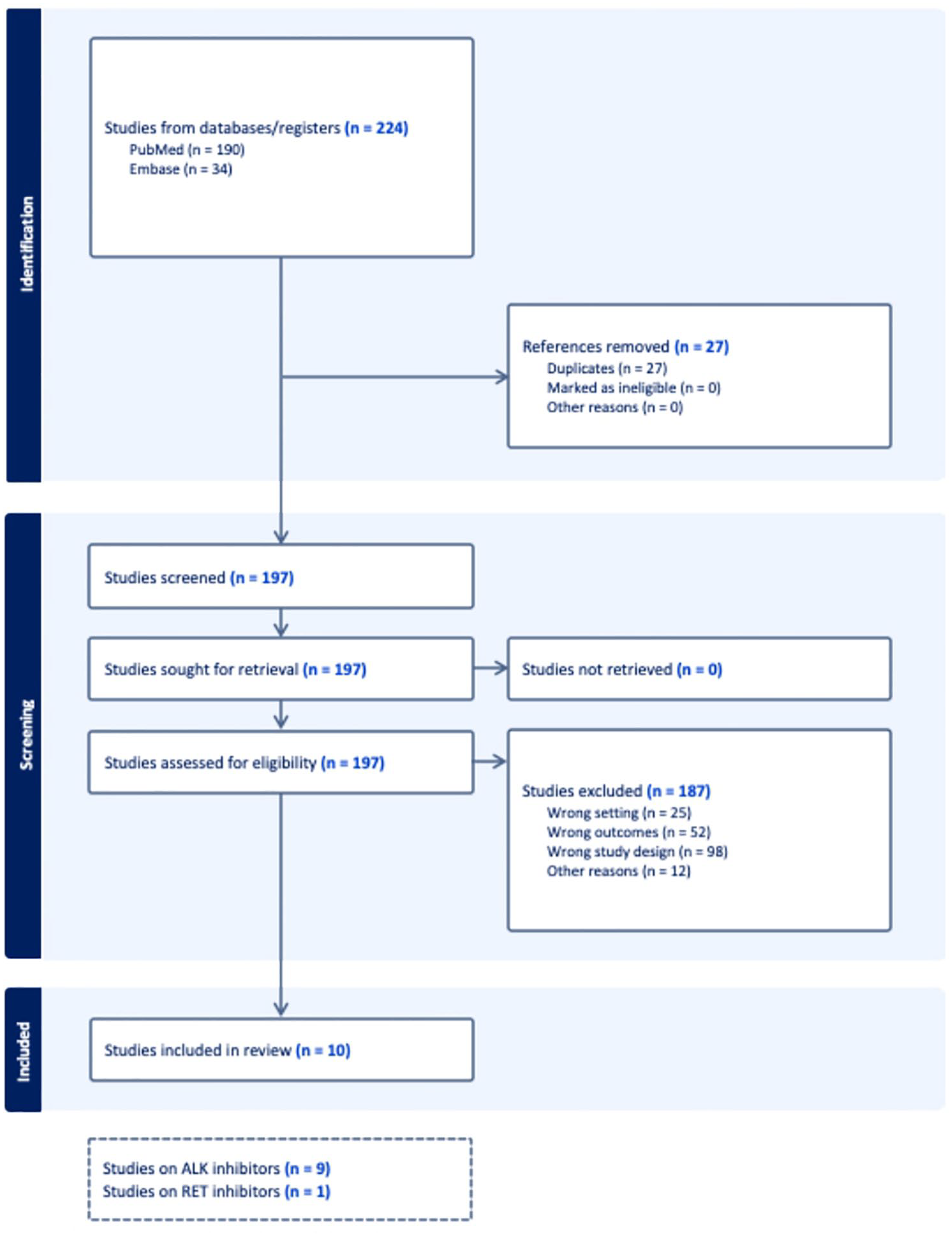
Study selection flowchart.
Sex differences in clinical outcomes with ALK inhibitors
We included nine phase III randomized trials evaluating ALK inhibitors. These trials comprised: two comparing crizotinib with platinum-based chemotherapy (PROFILE-1014 and PROFILE-1029), three comparing alectinib with crizotinib (ALEX, J-ALEX, and ALESIA), and one each comparing brigatinib with crizotinib, ceritinib with chemotherapy, ensartinib with crizotinib, and lorlatinib with crizotinib (ALTA-1L, ASCEND-4, eXalt3, and CROWN, respectively).21,24,57 –63 The main characteristics and outcomes of these trials are summarized in Table 1. Sex distribution for phase I and II clinical trials on ALK inhibitors is detailed in Supplemental Table 4.30,64 –71
In 9 phase III trials, 691 female and 582 male patients were evaluated. In general, sex distribution was balanced across studies, although the PROFILE-1014, ALEX, J-ALEX, ASCEND-4, and CROWN studies included slightly more women (212 vs 131, 171 vs 132, 125 vs 82, 216 vs 160, and 175 vs 121, respectively; Table 1). Phase I/II trials also showed balanced sex distribution (Supplemental Table 4).
Characteristics and main outcomes of selected phase III randomized clinical trials of TKIs in patients with advanced fusion-positive NSCLC, with subgroup analysis data according to sex.

BID, bis in die; CI, confidence interval; FU, follow-up; HR, hazard ratio; NA, not available; OS, overall survival; PFS, progression free survival; QD, quaque die.
PFS was the primary endpoint for all the phase III randomized trials, with variability in assessment methods (investigator-assessed in two trials, independent review in five) and outcome reporting (median PFS in five trials, 1-year PFS rate in two). OS data were only available for three trials (PROFILE-1014, ALEX, J-ALEX).
For crizotinib, both PROFILE-1014 and PROFILE-1029 showed a trend toward improved PFS in women (PFS HR 0.45 vs 0.54 in men, and 0.371 vs 0.410 in men, respectively). However, PROFILE-1014 did not show statistically significant OS benefits for the experimental arm, complicating sex-based OS HR analysis.21,57
In the ALEX trial, alectinib demonstrated a greater PFS benefit for women compared to men (PFS HR 0.39 vs 0.61). Conversely, J-ALEX showed similar PFS outcomes for both genders (PFS HR 0.31 vs 0.35), while ALESIA indicated a greater benefit for men (PFS HR 0.35 vs 0.17).22,60,72 OS HR data from ALEX and J-ALEX did not show significant sex differences, although women appeared to benefit less than men. No sex-based PFS differences emerged from the ALTA-1L, eXalt3, and CROWN trials on brigatinib, ensartinib, and lorlatinib, respectively, with OS data not reported in these subgroups.25,24,73 Conversely, the ASCEND-4 trial, which included more women, showed a lesser PFS benefit for women compared to men (PFS HR 0.63 vs 0.41). 61
The pooled HRs for PFS and OS from the nine phase III trials are shown in Figure 2(a) and depicted as a bubble plot in Figure 2(b).

(a) Forest plot of comparison of PFS and OS HRs according to gender in phase III studies on ALK inhibitors. (b) Bubble plot for PFS and OS according to gender in phase III studies on ALK-inhibitors.
Data on differential response patterns (ORR, disease control rate, duration of response, clinical benefit rate), CNS activity, or toxicity profile according to sex were not reported in these trials. Notably, the phase I study on crizotinib reported a higher ORR in men compared to women (ORR 64.8% in men vs 56.9% in women). 64
Sex differences in clinical outcomes with other TKIs for the treatment of fusion-positive NSCLC
No phase III trials were available for ROS1 inhibitors at the time of the review. We reviewed 8 early-phase trials involving 576 patients with ROS1 fusion-positive NSCLC (Supplemental Table 4).26,27,29,30,74 –77
Of these, 321 were women and 255 were men. Most studies had balanced sex representation, with some showing a higher proportion of women (e.g., ALKA-371-001, STARTRK-1, and STARTRK-2). 27
Data on survival outcomes were limited, focusing mainly on ORR and toxicity. Crizotinib trials reported varying gender-related ORR outcomes: the phase I PROFILE-1001 study showed higher ORR in men (ORR 78.3% vs 66.7% in women), whereas the phase II NCT01945021 study indicated a superior ORR for women (ORR 78.1% vs 63% in men).26,74 CNS activity and toxicity data were not available.
Two early-phase trials assessed NTRK inhibitors larotrectinib and entrectinib in 71 patients, with 35 males and 36 females.32,78 Due to the small sample size, differential outcomes in survival, CNS activity, or toxicity were not available.
For RET fusion-positive NSCLC, a phase III trial compared the RET inhibitor selpercatinib with platinum-based chemo-immunotherapy, showing a positive PFS benefit (PFS HR 0.46; 95% CI 0.31–0.70). 36 The LIBRETTO-001 and ARROW trials evaluated selpercatinib and pralsetinib, respectively. The phase III trial of selpercatinib included an equal number of men and women (Table 1), while phase I/II trials had similar sex distribution (Supplemental Table 4).79,80 In the selpercatinib phase III trial, the PFS benefit was greater in men (PFS HR 0.386 vs 0.599 in women), with pending OS data. In the ARROW study, women had a slightly higher ORR with pralsetinib (ORR 75% vs 69% in men), with no differential data on survival, CNS activity, or toxicity reported.
Discussion
Despite increasing awareness, significant gaps remain in understanding how sex, gender, and related factors influence the presentation, diagnosis, and treatment of lung cancer. We conducted a review of major phase III randomized trials involving targeted therapies for patients with advanced NSCLC harboring driver fusions, such as ALK, ROS1, NTRK, and RET. Our analysis revealed that while some studies of ALK-positive populations suggested a trend toward better PFS in women, no statistically significant differences in PFS or OS were observed between sexes. Similarly, in RET fusion-positive NSCLC, no significant PFS or OS differences were found between men and women. However, a critical gap exists in data regarding how sex-based differences impact treatment tolerability and tumor response across these trials.
Sex differences in PKs are known to influence sex-specific AEs, yet the absence of sex-stratified PK data for numerous drugs in public records indicates that these differences are often overlooked. 38 The common practice of prescribing identical drug doses for both men and women may ignore PK variations, potentially leading to overmedication and higher rates of AEs in women. 41 No significant PK differences have been observed between men and women for several TKIs, including imatinib, vemurafenib, and dabrafenib, and available data on anti-ALK therapy, such as brigatinib, also show no PK differences based on age, sex, or BMI.81 –84 Nevertheless, women tend to experience more drug-related AEs. 85 This trend has already been documented in chemotherapy, where women exhibit lower drug elimination and higher toxicity for agents such as 5-fluorouracil, paclitaxel, doxorubicin, and platinum-based therapies. 86 Similar trends might apply to targeted therapies. One known sex difference that could influence TKI tolerability is QT interval duration; women are at a higher risk of developing ventricular arrhythmias, such as torsades de pointes, due to their longer QT intervals, a factor that occurs twice as often in women than in men. 87
This gap in safety data highlights the need for further research to better understand and prevent drug-related AEs. With impressive median PFS not reached after 5 years of follow-up with lorlatinib upfront in ALK-positive NSCLC, patients are on TKI therapy for extended periods, making the prevention of long-term AEs strongly relevant. 88 A logical next step would be to examine phase III trial data on tolerability by sex. Additionally, stratifying patients by sex in future analyses could also help assess potential differences in efficacy and tolerability in patients receiving TKIs.
Furthermore, it is important to consider the unique AEs experienced by women, both in clinical trials and real-world settings. Targeted therapies, including TKIs, can impact gonadal function, leading to infertility, hormonal imbalances, and emotional distress—especially in young female patients. 89 Pregnant women have largely been excluded from TKI clinical trials, resulting in limited safety data that relies on epidemiological studies, case reports, and animal models. These sources, while informative, are not easily generalizable and raise concerns about maternal and fetal outcomes. Preclinical studies suggest potential gonadotoxicity; for instance, EGFR TKIs such as afatinib, gefitinib, and osimertinib may reduce fertility in animal models.90,91
Gefitinib has also been associated with reduced testosterone and DHEA levels in patients with NSCLC. While similar concerns have been raised for RET inhibitors, there are no studies on the fertility effects of ALK and ROS1 TKIs. Given these uncertainties, female patients of reproductive age with fusion-positive NSCLC are generally advised to use effective contraception during treatment. However, considering the extended survival associated with TKIs—often with OS outcomes exceeding 5 years—patients with well-controlled disease may still inquire about the possibility of pregnancy. Comprehensive oncofertility counseling should be provided before initiating targeted therapies, with careful consideration of patient preferences and prognosis to balance maternal benefits and risks. We advocate for future clinical trials to include QoL factors and sex/gender-specific interventions, acknowledging women’s differences in poorer health outcomes. 92
This work has several limitations that should be acknowledged. While this review relies on published clinical trials, the potential for publication bias is inherent; however, the relatively small number of studies included in this analysis may limit the robustness of such assessments. Furthermore, confounding variables, such as age and comorbidities, that could vary between sexes, but were not adjusted for, may affect the interpretation of the results. Although formal sensitivity analyses were not conducted in this review due to the limited number of studies, future analyses incorporating more comprehensive data could involve excluding lower-quality studies or focusing on those with primary endpoints such as PFS or OS. Although this review was unable to conduct extensive subgroup analyses due to the paucity of sex-specific data, such analyses would be relevant for future research to determine whether outcomes for targeted therapies differ significantly between male and female patients. This underscores the importance of designing future trials with comprehensive data reporting on sex-specific outcomes.
Conclusion
In this systematic review, we did not observe significant differences in key treatment outcomes with TKIs based on sex in patients with fusion-positive (ALK, ROS1, NTRK, RET) NSCLC. However, our findings highlight a concerning gap in understanding the toxicity profiles according to sex. We strongly advocate for more preclinical and clinical research focused on sex-based differences in treatment responses and adverse effects with targeted therapies. Additionally, a comprehensive understanding of the clinical relevance of sex differences in drug treatment should be integrated into board certification processes for healthcare providers and emphasized in continuing medical education.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241306940 – Supplemental material for Sex differences in patients with Non-Small Cell Lung Cancer harboring driver fusions treated with tyrosine kinase inhibitors: a systematic review
Supplemental material, sj-docx-1-tam-10.1177_17588359241306940 for Sex differences in patients with Non-Small Cell Lung Cancer harboring driver fusions treated with tyrosine kinase inhibitors: a systematic review by Rita Leporati, Édouard Auclin, Daniel Morchón, Miquel Ferriol-Galmés, Juan Carlos Laguna, Teresa Gorria, Cristina Teixidó, Maria Aranzazu Amores, Paolo Ambrosini, Dolores Isla, Giuseppe Lo Russo and Laura Mezquita in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
