Abstract
Patients with genitourinary (GU) malignancies have seen the development of multiple life-prolonging treatments in the past decade. As patients and clinicians consider their treatment options along the cancer journey, time spent with healthcare contact, or “time toxicity,” has emerged as a new outcome measure that comprehensively considers time receiving cancer care, including planned visits for evaluation and treatment as well as unplanned urgent care addressing complications. Despite its rising study across cancer populations, there has been a surprising lack of work evaluating time toxicity in patients with GU cancers. This narrative review aims to summarize the available studies on time toxicity in cancer, with a deeper dive into the methodology, strengths and limitations, and future directions of the field. A dedicated section focused on scenarios and best practices to measure and collect data on time toxicity can serve to spark interest in evaluating this novel health outcome on GU cancer survivors. Ultimately, time toxicity is a relevant patient-centered metric that can be incorporated into clinical trial design and routine clinical care to influence clinical decision-making.
Keywords
Introduction
Patients with genitourinary (GU) malignancies, including cancers of the prostate, bladder, kidney, and testis, comprise a large cohort of cancer survivors worldwide with heterogeneous prognosis and risk of morbidity and mortality, often including exposure to multiple lines of therapy. 1 While the up-front time-intensive treatment of some GU malignancies, such as testicular cancer, can result in the majority of patients experiencing prolonged survival after curative intent treatment, patients suffering from other GU cancers often develop advanced disease requiring chronic treatments over years. These different intensities of treatment, and the time that is devoted to ongoing care over time, can influence treatment decisions, especially when considered in the context of a finite period of remaining time for individuals whose cancer is not curable. In this setting, the notion of time spent with healthcare contact, also known as “time toxicity,” has emerged as a novel patient-centered outcome measure. Crucially, time toxicity includes the time spent receiving cancer-directed treatment (including clinic visits for evaluation and treatment, ancillary testing such as laboratory or imaging studies), as well as time spent addressing complications of the disease or treatment (including outpatient and inpatient care). This time spent on healthcare activities can significantly reduce the value of the time gained in survival. 2 Assessments of time toxicity may be particularly relevant for patients with advanced malignancies who are facing treatment choices, as their time may be more limited than for other patients, but it can be studied at any point along the cancer journey. Indeed, patients and their caregivers at any point in the care continuum can end up investing a substantial amount of their day in receiving cancer care, often at the cost of time otherwise spent at work or fulfilling personal activities.3,4 Although the first studies of time toxicity predominantly focused on patients with advanced solid malignancies, to date there has been a surprising gap in research examining time spent with healthcare contact in patients with GU cancers.
GU cancers offer an excellent opportunity to study the impact of time toxicity, especially since patients have seen the development of multiple life-prolonging treatments in the past decade that may be used in the same setting, but may have different impacts from a time toxicity perspective and hence influence treatment decision-making. For example, the recently approved combination of enfortumab vedotin and pembrolizumab has transformed the standard of care for patients with advanced bladder cancer, but the indefinite duration of the treatment plan and potential side effects, such as neuropathy, may require additional visits and supportive measures over time. Whether patients choose to pursue treatment with these medications indefinitely until progression versus pausing at a point of acceptable disease control is something that patients may consider and compare with the option to receive a more consolidated treatment course of chemoimmunotherapy.5,6 Similarly, the approval of 177Lutetium-PSMA-617 in metastatic castrate-resistant prostate cancer (mCRPC) may offer prolonged disease control to a subset of patients, but at the expense of increased laboratory and imaging monitoring, in addition to the need to travel to tertiary care cancers to receive this therapy, and a required period of separation from loved ones for the days immediately following treatment.7,8
The purpose of this review is to summarize the rapidly growing study of time toxicity in cancer, with a particular emphasis on its potential applications in supporting shared decision-making among patients with GU malignancies.
Methods
We performed a targeted search of peer-reviewed publications in publicly available journal databases (MEDLINE, Embase) for the period from 4/2016 up to 07/2024, using keywords such as “time toxicity,” “contact days,” “home time,” “healthcare contact,” with specific focus on queries related to GU malignancies (i.e., “prostate cancer,” “kidney cancer,” “bladder cancer,” “testicular cancer,” and related cancer specific terms) to complete a narrative review. We identified 22 relevant studies that were included in this review. Since the majority of studies on time toxicity have equated it with time spent with healthcare contact, these two terms will be used interchangeably. However, we recognize that there are many ways time may be indirectly dedicated to cancer care and its complications but not qualify as healthcare contact, as will be discussed in this review.
Quantifying time toxicity
While clinical trials offer reliable information on the expected likelihood and length of cancer control and rates of adverse effects, there is little definitive information describing the time investment associated with receiving cancer therapies. At best, patients receiving treatments administered in clinical settings have predictable schedules for the days that require treatment or associated supportive care. However, these a priori estimates rarely quantify the need for monitoring with laboratory studies and imaging, travel time to and from healthcare settings, or the likelihood of additional healthcare interactions related to complications from cancer or its treatment which can force patients to seek urgent medical care. 2 In addition, patients who live a significant distance away from healthcare institutions may need to dedicate more time to receive the same care than those who reside in urban areas. 9
The earliest proposed method for evaluating the time spent in versus out of healthcare settings by cancer patients receiving therapy was the Quality-adjusted Time Without Symptoms or Toxicity (Q-TWiST) measure. As the name suggests, QTWiST characterizes the time that patients spend with and without symptoms from cancer and/or treatment by incorporating adverse event rates and survival data from clinical trials. 10 It was conceived in the late 1980s to supplement traditional clinical outcomes, laying the groundwork for the current interest in time toxicity. Q-TWiST rapidly gained popularity across a variety of cancer and non-cancer settings, including in GU populations.11,12 However, it mostly remains a research methodology that requires an equation using utility coefficients to determine the quality time tradeoffs, limiting its accessibility by unfamiliar clinicians and patients.
In the development of time toxicity as an outcome, a number of mechanisms have been proposed for best measuring time spent with healthcare contact. The most pragmatic, intuitive, and widely adopted method involves separating days with and without physical healthcare contact, labeling them as “contact days” or “home days,” respectively.2,4,13 Any physical encounters with the health system, such as ambulatory clinic visits, nursing-administered treatments, ancillary testing, or inpatient level of care are considered whole days away from home and thus “time toxic.” Even though the rapid rise and establishment of telehealth as a type of healthcare encounter could prompt its inclusion in this model, arguably the time commitment for patients is drastically reduced when visits and other discussions occur through virtual methods of communication. Thus, currently, only physical encounters are included in this binary contact/home day model. 4 While most studies have abstracted contact days from electronic medical record systems or trial protocols, other studies were able to quantify healthcare contact based on medical insurance claims.14,15 Some studies have proposed variations of the contact/home day model, either reporting home days (i.e., the inverse of contact days) 16 or sharing the proportion of contact days with respect to overall survival time.17 –19 The adoption of contact days as the preferred method for quantifying time toxicity can be ascribed to its simplicity to quantify and interpret. Some limitations of the contact days method include the risk of overestimating the time commitment for patients (e.g., not every patient encounter involves multiple hours of the day), or conversely, underestimating healthcare contact due to confounders (e.g., decreased appointments due to limited access to care).
An innovative study suggested an alternate mixed methods approach for quantifying healthcare contact. 20 In this study conducted in patients with metastatic breast cancer, time spent receiving cancer-related healthcare was established using data derived from direct observation of patients in the clinic, patient surveys, and administrative claims. Ultimately, cancer care-related time was calculated using an equation that combined time spent traveling to and from outpatient appointments, services received during appointments, ancillary testing, inpatient care, and healthcare-related activities outside of the clinic. While this method likely offers the most comprehensive and realistic quantification of the time investment required by patients receiving cancer care, the multiple steps required through this method can limit its clinical implementation. A subsequent study sought to also directly observe time spent on ambulatory care by incorporating real-time location badges to track time patients spent in the clinic, together with estimates of travel times, to obtain a total “home to home,” but ultimately provided a narrower scope of time toxicity than other studies 21 (Table 1).
Summary of outcome measures evaluating time spent with healthcare contact.

Q-TWiST, Quality-adjusted Time Without Symptoms or Toxicity; QOL, quality of life.
The ideal way of determining time spent engaging with healthcare likely involves a combination of measuring contact versus home days, with a more in-depth characterization of time involvement at each clinical encounter. It is foreseeable that the rise of tracking technology and computer-driven algorithms may refine how healthcare contact is collected and computed. Many healthcare systems are using real-time tracking services on patients and providers to improve care delivery, with the added benefit that the information gained can be used to study time toxicity.2,21 Personal devices equipped with tracking technology, such as smartphones or watches, could provide real-world evidence of the time spent in different health settings outside of clinic visits, including picking up medications at pharmacies or receiving physical therapy at home. In this regard, the aid of patients in capturing time toxicity outside of clinical settings will prove invaluable to derive precise measurements. A sufficiently comprehensive yet intuitive method of assessment is needed by researchers and clinicians, with the objective of precisely quantifying time estimates that can be considered in clinical decision-making (Figure 1).
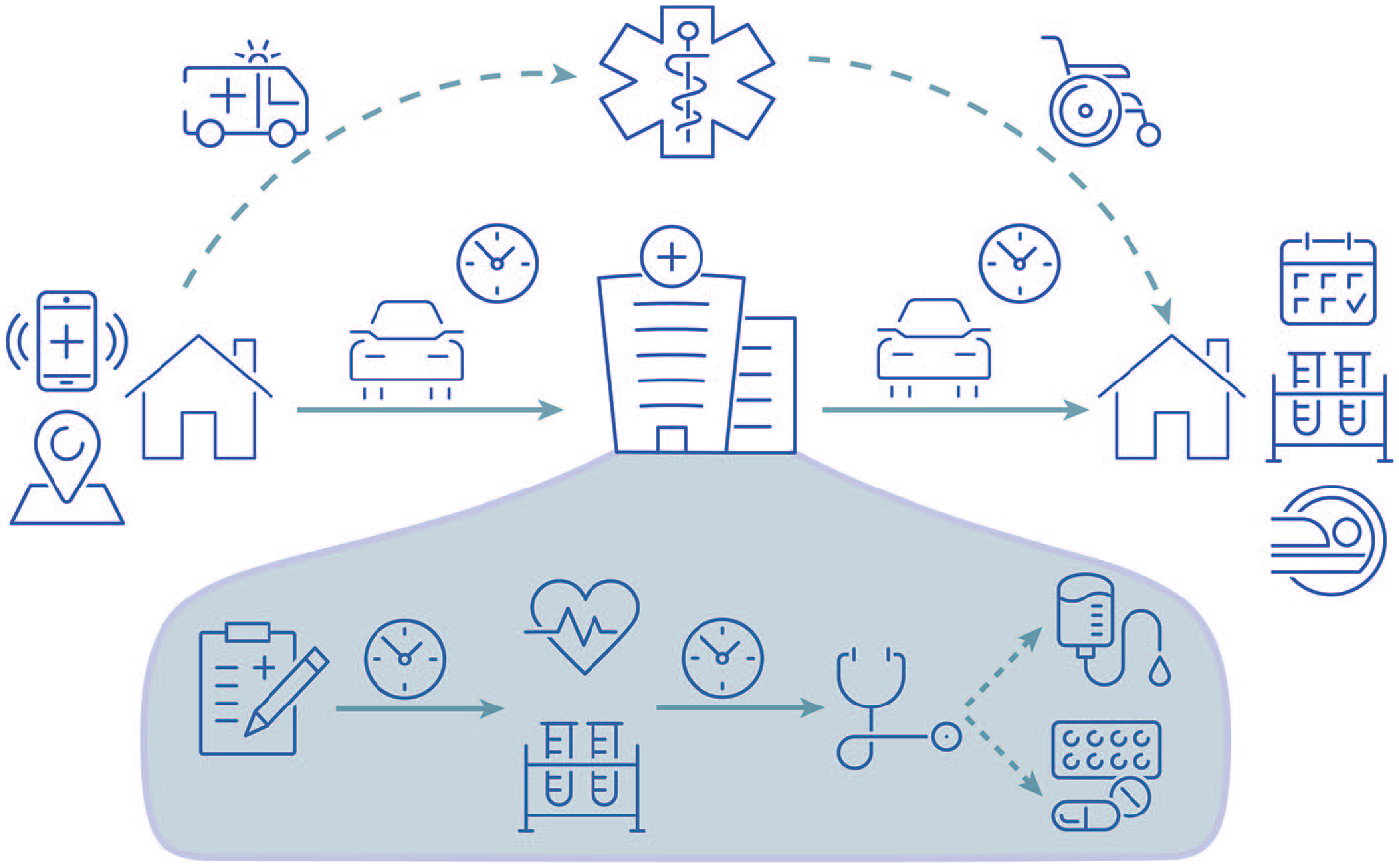
Healthcare contact continuum—traditionally covering “home-to-home” time including commuting; services during the ambulatory appointment such as check-in, vitals and lab work, clinician evaluation, and therapies administered; as well as follow-up testing before the next clinic visit; but also the potential need for urgent evaluation and subsequent recovery time. The incorporation of tracking services and partnerships with patients could yield more reliable estimates of time toxicity in future studies.
Time toxicity in oncology populations
The past 5 years have seen multiple studies evaluating time spent with healthcare contact in patients with cancer; below we have highlighted studies conducted in non-GU populations where time toxicity outcome measures were the primary endpoint, rather than part of a larger healthcare utilization analysis (Table 2).
Current literature on time toxicity in patients with non-GU cancers, ordered by ascending number of patients.

EMR, electronic medical record; GU, genitourinary; ORR, response rate; OS, overall survival; PDH, Proportion of home days; QOL, quality of life; RCTs, randomized clinical trials; SCT, stem cell transplant; VA, Veterans Affairs.
Several studies were able to retrospectively retrieve healthcare contacts using clinical trial protocols and available trial data.26,27,31 These post hoc analyses provide clinically applicable findings by comparing time toxicity between different treatment approaches. In one analysis, patients with advanced gastroesophageal cancer had significantly fewer contact days if treated with immunotherapy (median 28 days (interquartile range (IQR) 24, 41)) than if treated with chemotherapy (39 days (IQR 21, 51)) or chemoimmunotherapy (39 days (IQR 30, 52)). 31 Data derived from trials evaluating a later-line treatment compared to best supportive care can be similarly meaningful for patients with advanced cancer who are interested in the actual time spent at home if they were to forgo treatment. An evaluation of patients with metastatic colorectal cancer receiving cetuximab versus supportive care ultimately showed that patients had a similar number of home days whether or not they received treatment (median 140 days (IQR 75, 244) vs 121 days (70, 193), respectively). 26 All in all, it appears feasible to measure healthcare contact from existing published trials to obtain evidence describing time toxicity, with the expected limitations of post hoc analyses and the possibility that the generalizability of data from clinical trial populations may be limited when applied to real-world patients.
Other studies assessed large population datasets to establish healthcare contact in patients with advanced cancer and compare time toxicity between therapeutic approaches.14,15,19,20 One study analyzed home time in patients with acute myeloid leukemia using a SEER-Medicare dataset and found a higher proportion of home time among patients who received hypomethylating agents when compared with patients treated with anthracycline-based chemotherapy (60.8% vs 51.9%). 19 A primary advantage of population-based analyses includes the potential for precise estimates of healthcare contact based on linked billing claims data from large populations of patients treated in real-world settings. Moreover, these large datasets allow for covariate analysis that can identify differences in time toxicity between groups of patients, as was noted in a study in patients with metastatic breast cancer from SEER-Medicare that revealed increased healthcare contact among patients receiving radiotherapy versus those who did not (RR 1.45 (95% confidence interval (CI): 1.37, 1.54)). 14 Despite the benefit of providing data in a real-world population, these analyses are limited by the lack of patient-level detail regarding treatment doses, and laboratory and radiology appointments, among other factors.
Finally, several studies measured healthcare contact in regional patient cohorts, often from a single institution.13,16 –18,21,22 –25,28 –30 The benefit of studying time toxicity in these smaller patient groups is the opportunity to obtain significant levels of detail into the types of healthcare contact that patients may face, and the ability to assess unstructured data on chart review. For example, in a study of patients enrolled in early-phase clinical trials at an Australian hospital, the median contact days were 29% of the survival time, with a stark difference between patients who stayed on therapy and completed over 10 cycles versus those who only completed 2 cycles (14% vs 35%). 18 A different study evaluating “home-to-home” time in patients with both solid tumors and hematologic malignancies receiving ambulatory treatment was able to determine that, out of 197 median minutes spent in total ambulatory time, about 50 of those minutes were lost in travel. 21 Finally, these studies can also provide comparisons of healthcare contact between treatments, as in a study of patients with acute myeloid leukemia who received first-line treatment and identified distinct differences in home time between low-, intermediate-, and high-intensity treatments (7.6 vs 5.5 vs 13.1 months, respectively). 16 The principal limitation of these types of analyses is their applicability to wider populations and a relatively high risk of unmeasured confounders. Nonetheless, the findings from these smaller studies can serve as a blueprint to identify trends and advance methods of analysis for future studies that reproduce and validate the results in larger cohorts.
Time toxicity in GU oncology populations
Compared to the literature reviewed above conducted in patients with solid and hematologic malignancies, there has been a relative paucity of studies evaluating time toxicity in patients with GU cancers. One dedicated study in this area examined healthcare contact in elderly patients with non-muscle invasive bladder cancer (NMIBC). 32 Using SEER-Medicare data of 93,395 patients with NMIBC over age 66, the study focused on the association between treatment burden, or contact days in the year after diagnosis, with comorbidities associated with geriatric conditions. The adjusted mean contact days in this cohort was 8.9 (95% CI: 8.6, 9.2), with higher healthcare contact seen in patients living in rural areas as well as for those with ⩾2 comorbidities. This study highlighted an overall low number of contact days in 12 months, but there was insufficient information on the type of contact days, or how these evolved over time (e.g., whether most of the healthcare contact occurred early after diagnosis but decreased afterward). Strengths included the large sample size and the covariate analysis that enabled the investigators to assess patient-related factors associated with time toxicity in the older adult NMIBC population.
Another study aimed to measure the patient-reported treatment burden associated with intravesical treatment in patients with NMIBC but did not directly quantify the time toxicity associated with these therapies. 33 Instead, it used a cross-sectional survey of bladder cancer survivors to determine the amount of time required by patients to attend the clinic to receive treatment, as well as the average time of receiving the treatment. The results revealed that 46% of patients drove for less than 30 min, 36% traveled between 30 and 60 min, and only 7.4% traveled for more than 2 h. In terms of the treatment duration, 44% of patients reported receiving treatment under 2 h, 38% reported 2–4 h, and 17.9% reported undergoing treatment for over 4 h. These findings help provide an early look into the time commitment of receiving outpatient intravesical treatment, but further investigation including less potentially subjective tracking of time spent with healthcare use may provide a more reliable estimate by reducing recall bias.
To the best of our knowledge, these two studies in NMIBC represent the only peer-reviewed published studies of time toxicity in GU populations currently reported. We recently presented preliminary results of a single-center retrospective study of healthcare contact in the last year of life of patients with mCRPC. 34 Contact days were abstracted from scheduled and unscheduled clinical visits and categorized based on type of healthcare contact (e.g., clinic, imaging, hospitalization). Patients with mCRPC had increased contact days in their last 3 months of life compared to the first 3 months of their last year of life (median 6 days (IQR 2, 17) vs 4 days (IQR 3, 6)). This difference was driven by a higher inpatient level of care (i.e., emergency room visits and hospitalizations) in the last 90 days of life. Interestingly, while there were more contact days associated with infusional treatments such as chemotherapy in the first 3 months of the last year of life, in the last 3 months, there were no appreciable differences in contact days between treatments. Lastly, although the population overall had an increase in healthcare contact in the last 3 months of life, a majority of patients enrolled in hospice before their death and had a reduction in healthcare contact days. The biggest decrease in healthcare contact was among the patients with earlier hospice enrollment.
Thus, there remain numerous unexplored scenarios in which we can investigate time toxicity specific to GU populations. Broadly speaking, data on healthcare contact are often considered most helpful in scenarios of clinical equipoise, such as when several therapies are available but there is no clear clinical benefit between them, or toward the end-of-life as treatment benefits diminish and toxicity increases. For instance, there are multiple similarly effective first-line therapies in advanced renal cell carcinoma when patients may receive infusional checkpoint inhibitors and oral tyrosine kinase inhibitors. Analyzing the healthcare contact associated with each of these combination treatments could inform decision-making for patients and providers considering these options. Given that the contact day model of measuring time toxicity is currently preferred, it would be feasible to extract data on planned and unplanned visit dates from the landmark registration trials for these treatments and obtain a preliminary sense of the contact days that patients could expect when receiving first-line treatment for advanced kidney cancer. Separately, knowledge of the healthcare contact between GnRH agonist and antagonist could be informative to patients who remain ambivalent over which therapy to choose in localized prostate cancer. Similarly, determining the expected healthcare contact between curative intent treatment modalities such as surgery or radiation plus systemic therapy can complement the robust quality of life data available for patients with localized prostate or bladder cancer. To answer this question, a prospective collection of days spent in the clinic or the hospital setting for at least 6 months after treatment initiation would be most helpful to account for updates in surgical and radiation techniques over the past decade, which have attempted to reduce the morbidity of localized prostate cancer treatment. Finally, patients considering adjuvant treatments in renal or bladder cancer could benefit from knowing the healthcare contact linked with immunotherapy as opposed to surveillance, and use that information in concert with expected cancer control outcomes to decide whether to proceed with adjuvant immunotherapy. While predictably any adjuvant treatment would be associated with increased healthcare contact compared to surveillance, the key would be to compare days spent off treatment in each arm at the time of disease recurrence since that would better quantify the time gained by patients by pursuing adjuvant immunotherapy. This list is by no means exhaustive but can help frame scenarios applicable to patients with GU cancers where information on expected healthcare contact could influence treatment decisions. From our work in mCRPC, we recognize that particular challenges with evaluating time toxicity in GU patients are reduced frequency of treatments (i.e., many treatments are given several weeks or even months apart) and lower disease morbidity (e.g., patients with prostate and kidney cancer generally develop complications over several weeks rather than acutely). These and other factors could lower the expected time toxicity in GU cancers compared to other malignancies but do not limit the validity or importance of the findings for patients making difficult decisions on their care.
Future directions
Still in its developing phase, there is ample room for refining and improving the way healthcare contact is studied. In our opinion, the three main areas that need dedicated attention are patient engagement, standardization of outcome measures, and best practices for incorporating findings into clinical care.
Adding the patient’s perspective to existing measures must represent a priority to researchers of time toxicity. Healthcare contact was developed to serve as a useful resource for patients who are contemplating treatment options along their cancer trajectory. Therefore, the relative importance of time spent receiving cancer care to patients must be weighed against other valuable clinical outcomes, such as survival, time until subsequent therapy, or quality of life. From the outset, there has been an emphasis on sharing time toxicity results in a neutral, objective way to guide, but not exert undue influence on, patients’ decisions. 2 Indeed, some experts suggest steering away from the term “time toxicity” because it carries a negative implication and that may not necessarily be the case for certain patients. 4 For example, a treatment that portends significant healthcare contact in the short run may be considered worthwhile to patients if that treatment significantly lengthens time alive in the long run. Likewise, treatment may have increased healthcare contact compared to supportive care but prove to have a tremendous positive impact on quality of life. As such, studies specifically focusing on the subjective value of time toxicity by patients are needed. The use of structured interviews or questionnaires developed with patient advocates could help validate healthcare contact as a critical patient outcome.3,35 For instance, a recent qualitative analysis of patients’ attitudes and preferences regarding cancer treatments with different progression and overall survival benefits revealed several themes. About half of the study participants spontaneously brought up time spent receiving care as a factor in deciding treatments. Many of those patients not only acknowledged the toll associated with frequent visits but also the lengthy time that may be spent in each of these encounters. Importantly, there was a strong sense that time spent with healthcare contact eroded the psychosocial well-being of patients and their caregivers, as well as their overall quality of life. 36 Upcoming studies could build on these findings and assess patients’ responses to the expected time toxicity associated with particular treatments to further determine the role of time toxicity in decision-making.
As previously mentioned, there are ongoing efforts to refine healthcare contact as an outcome measure.20,21 While the contact/home days model remains the preferred method, the use of tracking services could enhance the current measurements, particularly around travel time as well as time spent in ambulatory-related care outside of the scheduled clinic appointments. Reporting and incorporating measured time into current models will present its challenges, not only methodologically in terms of deriving practical and comprehensible results for patients but also issues around generalizability to other health settings or regions. Nonetheless, patients deserve the most accurate estimations of healthcare contact possible to make well-informed decisions. Much like the development of validated quality-of-life instruments, time toxicity measurements will require consensus on definitions, analysis, and reporting.
While laudable and important to move the field forward, the majority of studies assessing time toxicity are from older patient cohorts that may not reflect current patterns of care.19,26 Furthermore, studies that derived time toxicity measurements from clinical trial populations carry the risk of both underestimating healthcare contact (given participants in trials are generally in good performance status with minimal comorbidities) but also overestimating it (patients usually require frequent visits as part of protocols). As such, prospectively measuring healthcare contact in both real-world clinical settings and clinical trials will allow us to differentiate between types of healthcare contact (e.g., planned vs unplanned, treatment or cancer related) and improve the generalizability of the findings. We advocate for the incorporation of prospectively assessed healthcare contact measures to be included as additional objectives in future clinical trial protocols, as this will produce time toxicity estimates with a high level of validity for the treatments assessed in the trial.
Finally, as healthcare contact is more commonly utilized in clinical decision-making, clinicians will be tasked with interpreting and sharing time toxicity findings with patients. In the same way that we simultaneously consider efficacy and toxicity data from clinical trials, we clinicians will have to incorporate healthcare contact considerations in the context of patient preferences and values. These discussions will benefit from data gathered using standardized methods and will undoubtedly evolve over time as the field matures.
Conclusion
The growing interest in assessments of time spent with healthcare contact, or time toxicity, as an important outcome measure in cancer research overall contrasts with a relative scarcity of available data for patients with GU malignancies, where the only available published work on time toxicity focused on patients with NMIBC but not in other stages of bladder cancer or other GU malignancies. Upcoming work in this field should shed light on the most relevant, reproducible, and actionable methods for measuring time spent with healthcare contact, as well as determining its relative importance to patients among more established outcomes like survival or quality of life. Overall, time toxicity is poised to become a relevant patient-centered metric, intuitive to patients and clinicians alike, that can be readily incorporated into clinical trial design as well as routine clinical care, and ultimately inform treatment decisions along the patient’s cancer journey.
