Abstract
Background:
Epidermal growth factor receptor (EGFR)-mutant non-small-cell lung cancers (NSCLCs) have higher frequencies of bone metastases than those of wild type; however, the metastatic pattern and influence on clinical outcome remain unclear.
Objectives:
To analyze the association between bone metastatic sites and the clinical efficacy of the first-, second-, and third-generation EGFR-tyrosine kinase inhibitors (TKI), in these patients.
Design:
Retrospective multicenter cohort study.
Methods:
The clinical data of patients with advanced-NSCLC harboring EGFR mutation, who were treated by EGFR-TKIs as first-line treatment at five medical institutions (N = 411), were retrospectively assessed for bone metastatic sites, overall survival (OS), and progression-free survival (PFS).
Results:
Bone metastases were found in 41.1% of the patients at diagnosis, including 13.1%, 8.0%, and 20.0 for single, double, and multiple lesions (⩾3), respectively. The vertebra (76.3%) and pelvis (60.9%) were the most frequent metastatic sites. Femoral-, sternum-, and scapula-metastases were remarkably increased in the patients with multiple-bone metastases. In the EGFR-mutant NSCLC patient treated with osimertinib, both the OS and the PFS of the patients with femoral bone metastasis were significantly shorter than those of the patients without femoral bone metastasis (OS: not reached vs 12.1 months, p < 0.0001; and PFS: 17.2 vs 9.3 months, p < 0.0018). Furthermore, a multivariable Cox regression analysis, including several poor prognostic factors, such as L858R mutation and liver metastasis, demonstrated that femoral bone metastasis was a statistically independent predictor of OS.
Conclusion:
Femoral bone metastasis is associated with poor survival of EGFR-mutant NSCLC patients who were treated with EGFR-TKIs, including osimertinib, and is an independent prognostic factor of OS.
Keywords
Background
Non-small-cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer types, and the majority of patients with NSCLC are initially diagnosed at an advanced stage with various distant metastases, including bone metastases (30%–50%).1–3 Since bone metastases commonly cause pain and/or neuropathy, skeletal-related events (SREs) frequently affect the patient’s mental health and quality of life. In particular, multiple-bone metastases (MB) at the time of diagnosis, which decrease survival time, are a poor prognostic factor of patients with NSCLC. 4 Among NSCLC harboring oncogenic driver alterations, epidermal growth factor receptor (EGFR)-mutant NSCLC, which is found in 40%–50% of Asian patients and 10%–15% of Western patients, tend to develop bone metastases. 5 Although several previous clinical trials and retrospective studies showed that patients with EGFR-mutant NSCLC had higher frequencies of bone metastases than those of patients with wild type and the incidence of bone metastasis at diagnosis was approximately 40%–60% in patients with EGFR-mutant NSCLC,6–9 no detailed study has investigated the bone metastatic pattern and influence on the clinical outcomes.
In recent decades, EGFR-tyrosine kinase inhibitors (TKIs), including gefitinib, erlotinib, afatinib, dacomitinib, and osimertinib, have dramatically improved the clinical outcomes of patients with advanced-stage EGFR-mutant NSCLC, including those with bone metastases.10–14 The molecular mechanisms of EGFR-TKIs that suppress bone metastasis were previously investigated, and first-generation EGFR-TKIs, gefitinib, demonstrated the ability to block epidermal growth factor (EGF) signaling in bone marrow stromal cells, resulting in inhibition of osteoclast differentiation, activation and recruitment.5,15 Furthermore, some clinical studies demonstrated that EGFR-TKIs had a preventive effect on the development of SREs, and treatment with EGFR-TKIs was associated with good prognosis in NSCLC patients with bone metastases when compared with cytotoxic chemotherapy. 16 On the other hand, SREs persist in more than 30% of patients with EGFR-mutated NSCLC even after EGFR-TKI treatment,7,17,18 indicating that part of the bone metastasis will be poorly controlled even with EGFR-TKI therapy. Other therapeutic options, such as radiation therapy and bone-modifying agents, are available for controlling bone metastatic lesions. Therefore, identifying poorly controlled bone metastatic sites is critical in the clinical practice of EGFR-mutant NSCLC.
The third-generation irreversible EGFR-TKI osimertinib demonstrated significantly longer progression-free survival (PFS) and overall survival (OS) than the comparator regimens of first-generation EGFR-TKIs in the double-blind phase III trial, FLAURA, resulting in osimertinib becoming a leading molecular targeted front-line therapy for patients with common EGFR mutations, such as exon 19 deletion and exon 21 L858R point mutation.14,19 We previously conducted a retrospective analysis of our real-world setting data to investigate the therapeutic efficacy of osimertinib in common EGFR-mutant NSCLC patients with various metastatic organs, and found that osimertinib provided a better clinical trend in the patients with bone metastasis when compared with those of other EGFR-TKIs. 20 However, the impact of various bone metastatic sites on the clinical outcome of osimertinib treatment remains unclear. Thus, we aimed to investigate the clinical data of the bone metastatic pattern at the time of diagnosis, which was collected from multiple institutions, and assess the influence on the treatment of EGFR-TKIs, including osimertinib, in patients with advanced-stage EGFR-mutant NSCLC.
Materials and methods
Study design
This retrospective cohort study was conducted with the approval of the ethical review committee of Nagoya University Hospital (approval number: 2018-017) and in accordance with the guidelines of the Declaration of Helsinki.21,22 We retrospectively reviewed the medical records of patients from five facilities, including the Nagoya University Hospital, Konan Kosei Hospital, Kariya Toyota General Hospital, Tosei General Hospital, and Japanese Red Cross Aichi Medical Center Nagoya Daiichi Hospital. The eligibility criteria of the enrolled patients in this study were previously described. 20 Briefly, (1) patients diagnosed with stage III/IV or recurrent non-squamous NSCLC, as confirmed by histological or cytological examination from January 2015 to December 2020; (2) patients presenting with a positive EGFR mutation; and (3) patients receiving first-generation EGFR-TKI (gefitinib or erlotinib), second-generation EGFR-TKI (afatinib), or third-generation EGFR-TKI (osimertinib) for first-line therapy (Supplemental Figure 1) were included. Clinical stages were assigned according to the eighth edition of the American Joint Committee on Cancer. Bone metastasis at the time of diagnosis was assessed by bone scintigraphy, contrast-enhanced computed tomography, contrast-enhanced magnetic resonance imaging, or positron emission tomography-computed tomography. Objective tumor responses (ORRs) were evaluated according to the Response Evaluation Criteria in Solid Tumors version 1.1. 23
Statistical analysis
PFS and OS were estimated using the Kaplan–Meier method and were defined as the time from the start of TKI therapy to disease progression or death, whichever was earlier, and the data were censored at the last follow-up date. Log-rank tests were implemented to analyze the differences in the PFS and OS between the patient groups. A Cox regression model was used to estimate the hazard ratio (HR) and 95% confidence interval (CI). Categorical data were compared using the one-way analysis of variance, Mann–Whitney U, Fisher’s exact, or Chi-square tests. Statistical analyses were performed using JMP software (Version 16), and the differences and correlations were considered statistically significant at p-value <0.05.
Results
Bone metastatic sites and pattern of patients with EGFR-mutated NSCLC
In our clinical data of advanced-stage EGFR-mutant NSCLC patients, bone metastatic sites of 411 eligible patients at the time of diagnosis were retrospectively assessed. In total, 169 patients (41.1%) had bone metastasis at diagnosis, and the number of patients per bone metastatic lesion was 54 (13.1%), 33 (8.0%), and 82 (20.0%) for single, double, and multiple lesions (⩾3; including triple- (N = 21, 5.1%), quadruple- (N = 19, 4.6%), quintuple- (N = 13, 3.2%), and more than sextuple lesions), respectively (N = 29, 7.1%) (Figure 1(a)). The patients of this cohort were divided into three groups based on the number of bone metastatic lesions as follows; non-bone metastasis (NB), single- or double-bone metastasis (SDB), and MB. No statistically significant differences were observed in the gender, smoking status, mutation status, and first-line EGFR-TKIs treatment between the three groups as shown in Table 1, while the MB group included relatively young patients, and statistically high ratio of poor performance status (PS), stage IV, and other distant organ metastases, such as brain, liver, and adrenal gland, as expected. Bone-modifying agents, such as denosumab and zoledronic acid, were used by 27 (31.0%) and by 21 (24.1%) patients in the SDB group, and by 30 (36.6%) and 27 patients (32.9%) in the MB group, respectively. The bone metastatic sites and patterns, including single-, double-, and multiple-bone metastases, are shown in Figure 1(b). Among the patients with bone metastases, the vertebra (N = 129, 76.3%), in particular the dorsal and/or lumbar vertebra (N = 127, 75.1%), was the most frequent metastatic site, and the pelvis (N = 103, 60.9%) was the second most frequent metastatic site. Interestingly, in the MB group, nearly all the cases had bone metastases in the vertebrae and/or pelvis (N = 81, 98.8%) (Figure 1(b) and (c)). Furthermore, the frequency of bone metastatic lesions in the femur (N = 31, 18.3%), scapula (N = 28, 16.6%), and sternum (N = 27, 16.0%) was remarkably increased in the MB group, and the proportion of patients with these metastatic sites tended to increase correlatively with the number of the bone metastatic site (Figure 1(d)).

(a) The numbers of EGFR-mutated NSCLC patients with bone metastases are 54, 33, and 82 for single, double, and multiple lesions (⩾3), respectively. (b) The bone metastatic sites and pattern of single or double (N = 87), and multiple lesions (N = 82). (c) The proportion of the patient’s number with each bone metastatic site based on the number of bone metastatic sites include 54, 33, 21, 19, 13, 11, 9, and 9 for single, double, triple, quadruple, quintuple, sextuple, septuple, and more than octuple lesions, respectively. (d) The proportion of the number of patients with bone metastatic sites of femur (N = 31), scapula (N = 28), and sternum (N = 27) based on the number of bone metastatic sites. The lines indicate approximation curves. Correlation coefficients and p-values were calculated using Pearson correlation tests.
Clinical characteristics of 411 NSCLC patients with EGFR mutations.

p-Values were calculated by one-way ANOVA, Fisher’s exact test, or Chi-square test.
Information was not available for eight cases.
Information was not available for three cases.
p-Values were calculated between SDB group and MB group.
EGFR, epidermal growth factor receptor; MB, multiple-bone metastases; NB, non-bone metastasis; NSCLC, non-small-cell lung cancer; PS, performance status; SDB, single- or double-bone metastasis; TKI, tyrosine kinase inhibitor.
To confirm that multiple-bone metastases at the time of diagnosis affect the prognosis in patients with EGFR mutations, we first performed univariate analyses of OS and PFS in the common EGFR-mutant NSCLC patients treated with the three generations of EGFR-TKIs. The median follow-up time was 22.4 (range, 0.2–94.6) months. The Kaplan–Meier analysis demonstrated that the OS of the patients in the MB group was significantly shorter than that of the patients in the NB or SDB group (median OS: MB vs NB- 25.4 vs 46.6 months, p = 0.0026, and MB vs SDB- 25.4 vs 41.2 months, p = 0.0223; Figure 2(a)). Furthermore, the PFS of the patients in the MB group was relatively shorter than that in the patients of the NB and SDB groups (median PFS: MB vs NB; 10.7 vs 14.0 months, p = 0.0096, and MB vs SDB; 10.7 vs 11.4 months, p = 0.1296; Figure 2(b)). The median overall survival (mOS) was significantly longer in patients with bone metastases treated with bone modifying agents; however, the median progression free survival (mPFS) was not significantly longer (median OS: 34.4 vs 28.1 months, p = 0.0431; median PFS: 12.7 vs 9.6 months, p = 0.3215; Supplemental Figure 2(A) and (B)). To further confirm that MB affects the clinical efficacy of EGFR-TKIs. In the patients treated with osimertinib, the MB group included a statistically higher ratio of poor PS and stage IV (Supplemental Table 1), and a similar tendency was observed in the patients treated with first-generation EGFR-TKIs (Supplemental Table 2). The Kaplan–Meier analysis revealed that the OS of the patients in the MB group, who were treated with osimertinib, was statistically significantly shorter than that in the patients of the NB or SDB group (median OS: MB vs NB, 18.0 months vs not reached, p = 0.0202; MB vs SDB, 18.0 months vs not reached, p = 0.0091; Figure 2(c)). Likewise, the PFS of the patients in the MB group was relatively shorter than that in the patients of the NB and SDB groups; however, the difference was not statistically significant (median PFS: MB vs NB, 14.2 vs 17.1 months, p = 0.1142; MB vs SDB, 14.2 vs 19.8 months, p = 0.0528; Figure 2(d)). In the patients treated with first-generation EGFR-TKIs, the OS and PFS of the patients in the MB group demonstrated shorter tendencies than those in the patients of NB and SDB groups; however, the difference was less significant compared with that in patients treated by osimertinib (median OS: MB vs NB, 25.4 vs 36.7 months, p = 0.1253; and MB vs SDB; 25.4 vs 38.2 months, p = 0.6563; median PFS: MB vs NB; 8.5 vs 12.2 months, p = 0.0125, and MB vs SDB, 8.5 vs 9.0 months, p = 0.5431; Supplemental Figure 2(A) and (B)). These results indicate that MB contributes to poor outcomes, especially in cases treated with osimertinib.

Kaplan–Meier plot of overall survival (a) and progression-free survival (b) in the patients treated using the three generations of EGFR-TKIs with non-bone, single/double-bone, and multiple-bone metastases, respectively, and in the patients treated by osimertinib with non-bone, single/double-bone, and multiple-bone metastases (c, d), respectively. p-Values were calculated using log-rank tests.
Femoral bone metastasis is associated with poor prognosis in patients treated with EGFR-TKIs
Thereafter, we analyzed which bone metastatic sites are most significantly associated with poor OS of patients with EGFR mutation who were treated with EGFR-TKIs. As shown in Figure 1(c) and (d), the bone metastatic site of the femur was notably increased in patients with MB. In the total 411 patients treated with the three generations of EGFR-TKIs, the OS of the patients with femoral bone metastasis was significantly shorter than that in patients without femoral bone metastasis (median OS: 20.1 vs 41.2 months, p = 0.0024; Figure 3(a)) and the PFS of the patients with femoral bone metastasis was relatively shorter than that in patients without femoral bone metastasis; however, the difference was not statistically significant (median PFS: 9.3 vs 13.1 months, p = 0.1416; Figure 3(b)). The majority of the patients were treated with first- and second-generation EGFR-TKIs and femoral metastasis was significantly associated with a poor prognosis.

Kaplan–Meier plot of overall survival (a) and progression-free survival (b) in the patients with femoral bone metastases treated using the three generations of EGFR-TKIs, respectively, and in the patients with femoral bone metastases treated using osimertinib (c, d), respectively. p-Values were calculated using log-rank tests.
Femoral bone metastasis: An independent prognostic factor of osimertinib treatment
In the 156 patients with EGFR mutations and treated with osimertinib, 13 patients (8.3%) had femoral bone metastasis at diagnosis, and the patient characteristics did not show statistically significant differences between patients with femoral bone metastasis and those without, except PS (Table 2). Bone-modifying agents were used in nine (69.2%) patients with femoral bone metastasis. Both the OS and PFS of the patients with femoral bone metastasis were significantly shorter than those in patients without femoral bone metastasis (median OS: 12.1 months vs not reached, p < 0.0001; median PFS: 9.3 vs 17.1 months, p < 0.0027; Figure 3(c) and (d)). Even in the MB group, the OS and PFS of the patients with femoral bone metastasis were significantly shorter than those in patients without metastases (median OS: 12.1 vs not reached months, p < 0.0001; median PFS: 5.8 vs 14.6 months, p = 0.0047; Supplemental Figure 3(A) and (B)). However, the patients with bone metastases at other sites, such as the scapula or sternum, showed less significant OS and PFS compared with those without those metastases (scapular: median OS, not reached vs not reached, p = 0.9433 and median PFS, 17.0 vs 13.3 months, p = 0.2475; sternum: median OS, 19.2 vs not reached months, p = 0.0316 and median PFS: 13.3 vs 17.0 months, p = 0.1514, Supplemental Figure 4(A)–(D)), suggesting that femoral bone metastasis could critically affect poor survival in EGFR-mutant NSCLC patients treated with osimertinib. The ORR and disease control rate (DCR) were 61.5% (8/13) and 84.6% (11/13), respectively, in the patients with femoral bone metastases, and 67.1% (96/143) and 83.2% (119/143), respectively, in those with no femoral bone metastases (Supplemental Table 3), indicating that femoral bone metastasis was not related to the clinical response of osimertinib.
Clinical characteristics of common EGFR-mutant NSCLC patients treated with osimertinib.

p-Values were calculated by Mann–Whitney, Fisher’s exact test, or Chi-square test.
Information was not available for four cases.
EGFR, epidermal growth factor receptor; NSCLC, non-small-cell lung cancer; PS, performance status.
To further confirm the effect of femoral bone metastasis on the patient’s clinical outcome, we conducted a multivariable Cox regression analysis, including several poor prognostic factors of osimertinib, such as PS, L858R mutation, and liver metastasis. Among them, femoral bone metastasis was statistically significant for the OS (HR, 7.62; 95% CI, 2.22–26.12; p = 0.0012), but not for the PFS (HR 2.45; 95% CI, 0.97–6.29; p = 0.0619) (Table 3). These results indicate that femoral bone metastasis was markedly associated with the poor survival of EGFR-mutant NSCLC patients who were treated with osimertinib and could be considered an independent prognostic factor of OS.
Multivariable analysis of OS and PFS in the patient treated by osimertinib.
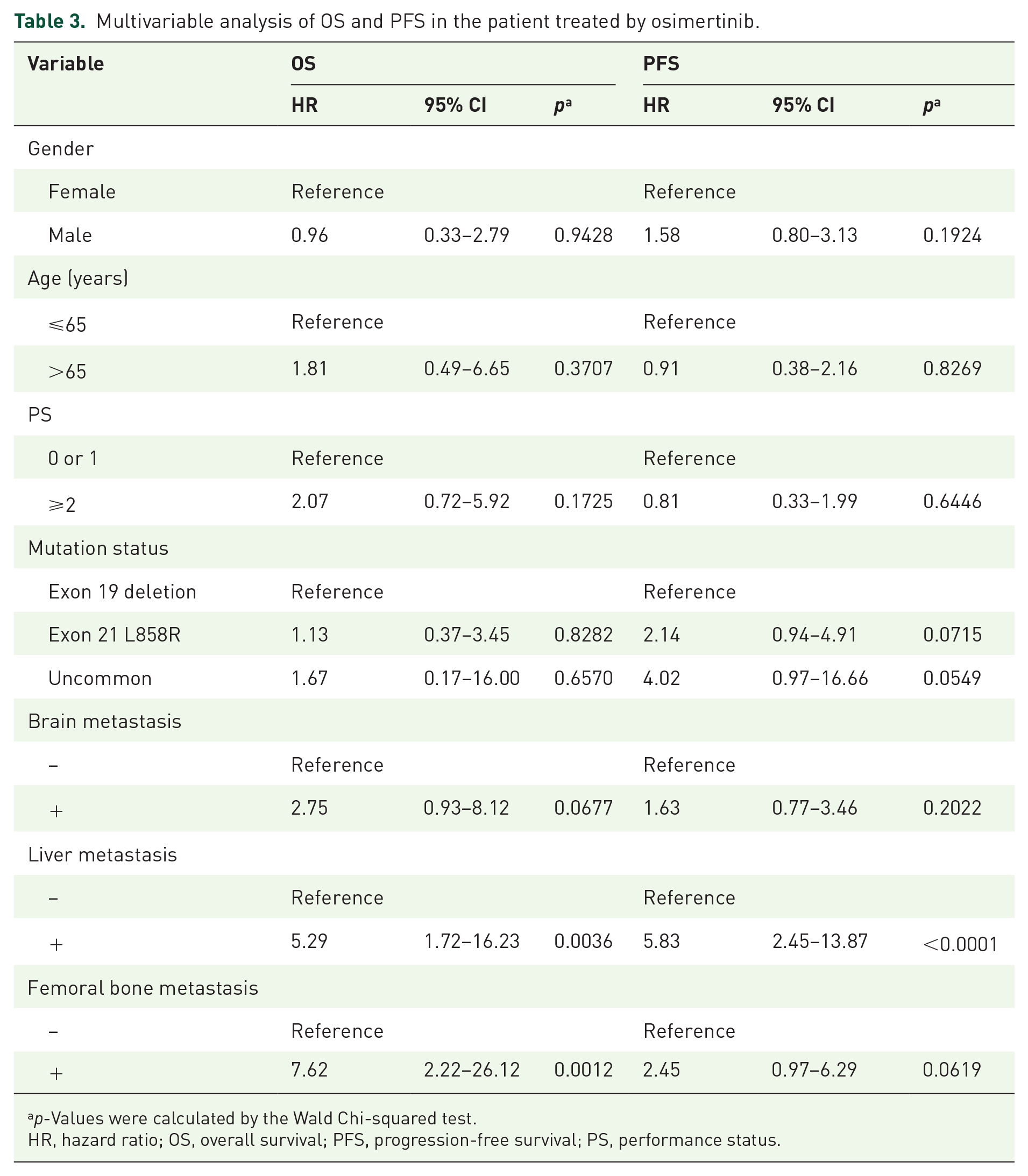
p-Values were calculated by the Wald Chi-squared test.
HR, hazard ratio; OS, overall survival; PFS, progression-free survival; PS, performance status.
Discussion
To our knowledge, this is the first report of bone metastasis pattern at the time of diagnosis in patients with advanced-stage EGFR-mutant NSCLC. In our cohort, EGFR-mutant NSCLC patients with bone metastases demonstrated a high incidence of vertebral and/or pelvis metastases, while the frequency of bone metastatic lesions in the femur, scapula, and sternum was remarkably increased in the MB group. Among the bone metastatic sites, femoral bone metastasis was critically associated with poor survival in the patients treated with osimertinib.
Biologically, oncogenic EGFR signaling increased vascular endothelial growth factor A (VEGF-A) expression, which promotes tumor angiogenesis,24,25 and the secreted VEGF-A contributes to distant metastases and formation of the tumor microenvironment.26,27 Moreover, EGFR signal activation results in the enhancement of various other immunosuppressive factors, such as transforming growth factor β and interleukin-6, which can contribute to metastatic niche formation including bone metastasis. 28 These molecular biological mechanisms are one of the reasons why bone metastases are more common in EGFR-mutant NSCLC. Similar to previous reports, our data demonstrated that more than 40% of EGFR-mutant NSCLC patients had bone metastases at the time of diagnosis, and MB was observed in approximately 50% of the patients with bone metastases. Several previous studies reported the vertebrae (40%–50%), ribs (20%–25%), and pelvis (15%–25%) as common sites of bone metastases in patients with NSCLC, including the EGFR wild type, and that bone metastases can frequently cause fractures in the proximal portion of long bones, including vertebrae, ribs, or femur.16,29 In addition, vertebral bone metastasis is also highly associated with sudden onset of back pain and neurologic symptoms. Upon comparison with previous reports, our data demonstrated that EGFR-mutant NSCLC patients demonstrated a higher incidence of vertebral bone metastasis (76.3%), while the presence or absence of vertebral bone metastases had a small effect on the clinical outcome of EGFR-TKIs treatment. In particular, osimertinib is known to have high transferability to tissues and a previous in vivo analysis demonstrated that osimertinib effectively regulated tumors in a mouse model of EGFR-mutant bone metastasis. 30 These results indicate that osimertinib monotherapy is sufficient for managing bone metastases in certain cases.
We previously reported that liver metastasis in addition to L858R mutation was an independent poor prognostic factor for osimertinib treatment, 20 and the present analysis also identified femoral metastasis as an additional independent poor prognostic factor for OS, even when the multivariate analysis was conducted including poor effectors plus PS and age. Although osimertinib showed high clinical efficacy for EGFR-mutant NSCLC patients in the SDB group, the presence of femoral metastases had a statistically significant impact on the OS of the patients treated with osimertinib rather than the PFS of those patients. One possible reason for this result is that while osimertinib has a high antitumor effect on bone metastases, tumor re-growth is inevitable over time, and chemotherapy after second-line treatment was less effective for femoral bone metastases, which may have led to a negative effect on the OS. In general, femoral metastases, not limited to lung cancer, are more common in cases of MB and have a greater impact on the OS because of the frequency of fractures. 31 Femoral metastases also tend to cause severe pain, and complete fractures have a particularly strong impact on clinical outcomes. Surgery may be an option in addition to radiotherapy for local control; however, previous retrospective studies have reported that the effect of surgery on the OS is extremely limited.32,33 Recently, immune checkpoint inhibitors (ICIs) are also available as treatment options after the second line; however, they are less effective in patients with bone metastases, 34 and ICI monotherapy has also been found to be less effective than docetaxel in NSCLC patients with EGFR mutations. 35 In KRAS-mutated NSCLC, KEAP1 and SMARCA4 are reported to be more common in bone metastasis cases and contribute to shorter OS. In the presence of KEAP1 and SMARCA4, the addition of ICI may not be effective because of the immunosuppressive microenvironment. The tumor microenvironment in cases of EGFR-mutated NSCLC harboring bone metastases has not been studied but is frequent and warrants further investigation. These previous results and the present analysis suggest that the antitumor effects of EGFR-TKIs monotherapy, including osimertinib, and various regimens used from the second line onward are limited for NSCLC patients with EGFR mutations who have femoral bone metastases. Limitations of this analysis include the lack of data on the presence of SRE due to retrospective data collection and the limited number of patients with femoral metastases, which makes the efficacy of radiotherapy and bone-modifying drugs uncertain. However, our results indicate that EGFR-mutant NSCLC patients with femoral metastases at diagnosis have a poor prognosis, even when they are treated with osimertinib. Recently, a phase III trial FLAURA2 demonstrated that first-line treatment of osimertinib combined with platinum-pemetrexed resulted in a significantly longer PFS than osimertinib monotherapy for patients with EGFR-mutated advanced NSCLC. Our results suggest that patients with MB, especially those with femoral metastasis were less effective for osimertinib monotherapy, and they might be good candidates for osimertinib plus chemotherapy. On the other hand, despite the lack of evidence that chemotherapy is effective for brain metastasis, mPFS in patients with brain metastases was better in the chemotherapy combination group than in the osimertinib monotherapy group. In TracerX, metastatic disseminated subclones have been reported to have subclonal expansion within the primary tumor. 36 Thus, the addition of cytotoxic chemotherapy may show efficacy against a variety of subclones in cases with distant metastases due to tumor heterogeneity. However, the efficacy of osimertinib in combination with chemotherapy for bone metastases needs to be further verified owing to the difference in tissue migration of the drug between brain metastases and bone metastases. Furthermore, combined treatment, including radiotherapy and bone-modifying agents in addition to EGFR-TKIs, and the development of new therapeutic strategies are warranted for these patients.
Conclusion
In patients with advanced-NSCLC harboring EGFR mutation, vertebrae and/or pelvic bone metastases were the most frequent metastatic sites, and the frequency of femoral, scapula, and sternum metastases was significantly increased in the cases with MB. Furthermore, we found that even after treatment with EGFR-TKIs, including osimertinib, patients with MB, especially femoral metastases, had an extremely poor outcome. These results indicate that new therapeutic strategies are urgently needed for patients with femoral bone metastasis upon diagnosis.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241303090 – Supplemental material for Femoral bone metastasis is a poor prognostic factor in EGFR-TKIs-treated patients with EGFR-mutated non-small-cell lung cancer: a retrospective, multicenter cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359241303090 for Femoral bone metastasis is a poor prognostic factor in EGFR-TKIs-treated patients with EGFR-mutated non-small-cell lung cancer: a retrospective, multicenter cohort study by Ichidai Tanaka, Kazumi Hori, Junji Koyama, Soei Gen, Masahiro Morise, Yuta Kodama, Akira Matsui, Ayako Miyazawa, Tetsunari Hase, Yoshitaka Hibino, Toshihiko Yokoyama, Tomoki Kimura, Norio Yoshida, Mitsuo Sato and Makoto Ishii in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359241303090 – Supplemental material for Femoral bone metastasis is a poor prognostic factor in EGFR-TKIs-treated patients with EGFR-mutated non-small-cell lung cancer: a retrospective, multicenter cohort study
Supplemental material, sj-pdf-2-tam-10.1177_17588359241303090 for Femoral bone metastasis is a poor prognostic factor in EGFR-TKIs-treated patients with EGFR-mutated non-small-cell lung cancer: a retrospective, multicenter cohort study by Ichidai Tanaka, Kazumi Hori, Junji Koyama, Soei Gen, Masahiro Morise, Yuta Kodama, Akira Matsui, Ayako Miyazawa, Tetsunari Hase, Yoshitaka Hibino, Toshihiko Yokoyama, Tomoki Kimura, Norio Yoshida, Mitsuo Sato and Makoto Ishii in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
