Abstract
Third-generation epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) have emerged as the mainstay of treatment for advanced EGFR-mutant advanced non-small cell lung cancer (NSCLC), effectively overcoming the problems of acquired threonine-to-methionine (T790M) mutations associated with the first- or second-generation TKIs. Evidence from several studies suggests that these agents, including osimertinib and aumolertinib, also show potential benefits in T790M-negative or unknown populations, particularly those with brain metastases, where the high permeability of the blood–brain barrier allows effective control of intracranial lesions. Despite the encouraging results, further high-quality research, including prospective trials, is warranted to fully elucidate the efficacy profiles of these third-generation TKIs in T790M-negative or unknown NSCLC patients after first- or second-line TKI failure. The present expert consensus highlights the evolving role of third-generation EGFR-TKIs in overcoming therapeutic resistance and optimizing patient outcomes.
Plain language summary
Newer medicines called third-generation EGFR inhibitors have become key treatments for a type of advanced lung cancer that has specific genetic changes. These drugs, such as osimertinib and aumolertinib, solve a problem encountered with older drugs: resistance caused by a mutation known as T790M. They’re especially promising for patients whose cancer has spread to the brain, because they can pass through the brain’s protective barrier and help control tumors there. While current evidence suggests these new drugs can help even when the T790M mutation isn’t present or hasn’t been tested for, more research is needed to fully understand how well they work in these situations. After other treatments have stopped being effective, using these advanced medicines may offer new hope by tackling treatment resistance and improving patients’ chances. This expert agreement outlines how these newer drugs are changing the way we overcome therapy challenges and enhance patient care.
Keywords
Introduction
Non-small cell lung cancer (NSCLC) patients with epidermal growth factor receptor (EGFR) exon 19 deletions or L858R mutations represent a distinct subset of malignancies that initially respond well to first-, second-, or third-generation EGFR tyrosine kinase inhibitors (EGFR-TKIs). 1 Despite the establishment of third-generation EGFR-TKIs as the preferred first-line therapy for advanced EGFR-mutant NSCLC, first- or second-generation EGFR-TKIs, such as gefitinib, erlotinib, icotinib, afatinib, and dacomitinib, remain relevant for a subset of patients. Their appeal lies in reduced costs and consistently demonstrated improved outcomes, with objective response rates (ORRs) ranging from 60% to 75% and progression-free survival (PFS) extending to approximately 1 year, exceeding those achieved with chemotherapy regimens. 2 However, the early emergence of therapeutic resistance and disease progression in patients previously treated with first- or second-line EGFR-TKIs remains a challenge, particularly with the frequent acquisition of a threonine-to-methionine substitution at exon 20 position 790 (i.e., the T790M mutation), with a prevalence of 66%–74%. 3 The advent of third-generation EGFR-TKIs, such as osimertinib, aumolertinib, and furmonertinib, has revolutionized the management of these patients after progression on prior EGFR-TKI therapy, offering new hope for sustained clinical benefit.4–6
Despite the recognized efficacy of third-generation EGFR-TKIs in targeting T790M-positive tumors, a significant proportion of patients do not develop this mutation or present with an indeterminate T790M status due to limitations in biopsy accessibility and detection methods. Consequently, these patients typically face a dearth of subsequent targeted therapies and have poorer prognoses.7–9 Recent scientific inquiry, including a systematic review and meta-analysis, 10 as well as individual clinical studies,11,12 has demonstrated the potential broader application of third-generation EGFR-TKIs beyond only T790M-positive cases.
Notably, osimertinib has shown consistent improvements in overall survival (OS), PFS, and ORR across a range of T790M statuses. Furthermore, intriguing results indicate that it can effectively treat brain metastases or leptomeningeal metastases regardless of T790M mutation status.10,12 Similarly, aumolertinib, which has been shown to have enhanced penetration of the blood–brain barrier (BBB), 13 has also been investigated for its potential to address EGFR T790M-negative or unknown NSCLC with brain metastases following resistance to previous generation EGFR-TKIs. 11
Therefore, these studies provide the basis for a comprehensive discussion of understanding and consensus on the efficacy and safety of third-generation EGFR-TKIs, including osimertinib and aumolertinib, for the treatment of patients with advanced NSCLC with EGFR mutations of various T790M mutation statuses that are resistant to early generations EGFR-TKI regimens. The purpose of the present expert consensus is to synthesize and evaluate the latest available research findings and to outline clinical treatment decisions for third-generation EGFR-TKIs in the context of T790M-negative or no definitive testing for T790M mutations following resistance to first- or second-generation EGFR-TKIs.
Methods and consensus development process
The development of this expert consensus was predicated on a rigorous and systematic approach to ensure the highest standards of scientific rigor and clinical relevance. Our methodology encompassed a comprehensive literature review, meticulous evaluation of included studies, and a structured consensus-building exercise.
Literature review
A systematic search of electronic databases, including PubMed, EMBASE, and Cochrane Library, was conducted to identify studies published from January 2010 to March 2024, focusing on third-generation EGFR-TKIs in the context of T790M mutation status and advanced NSCLC. Search terms included but were not limited to “third-generation EGFR-TKIs,” “osimertinib,” “aumolertinib,” “T790M mutation,” “non-small cell lung cancer,” and “brain metastases.” Two independent reviewers screened titles, abstracts, and full texts for eligibility, resolving disagreements through discussion or consultation with a third party.
Expert panel composition
The consensus was formulated by the Specialized Committee on Lung Cancer Cell Targeting and Immunotherapy of the Liaoning Society of Cell Biology, China. The panel comprised multidisciplinary experts in oncology, thoracic surgery, radiation oncology, and pharmacology, each with substantial experience in managing EGFR-mutated NSCLC. Members were selected based on their publication records, clinical expertise, and active engagement in relevant research.
Consensus building
The consensus process entailed a series of meetings and online consultations where panel members critically appraised the available evidence. A modified Delphi method was adopted to achieve consensus, involving iterative rounds of anonymous voting and discussion until a pre-defined agreement threshold was reached. Each statement was evaluated based on its clinical significance, evidence quality, and applicability.
Recommendation grading criteria
Recommendations were classified into three grades according to the strength of supporting evidence and level of clinical certainty.
Grade A: Strongly supported by high-quality evidence, reflecting a high degree of clinical certainty.
Grade B: Moderately supported by evidence, indicating a moderate degree of clinical certainty.
Grade C: Weakly supported by evidence, yet considered viable options under particular circumstances.
Overview of third-generation EGFR-TKIs: focusing on osimertinib and aumolertinib
Third-generation EGFR-TKIs represented by osimertinib and aumolertinib have emerged as pivotal advancements in the targeted therapy of NSCLC carrying EGFR mutations. These agents have been designed to overcome the common resistance mechanisms to first- or second-generation EGFR-TKIs, particularly the emergence of the T790M mutation, while also improving their pharmacological properties to address metastases of the central nervous system (CNS). 14
Molecular structure and mechanism of action
Osimertinib is a selective irreversible inhibitor, distinguished by its covalent binding to the EGFR ATP binding site. This binding specifically targets activating sensitizing mutations (exon 19 deletions and the L858R point mutation) and the resistance mutation T790M. Its chemical structure features an optimized quinazoline core with a pyrimidine substituent that allows for high specificity and potency. 15
Aumolertinib, another innovative third-generation EGFR-TKI, has a unique cyclopropyl moiety in its molecular architecture. This structural element facilitates its enhanced affinity and selectivity for the mutated EGFR kinases, thereby overcoming T790M-mediated resistance while sparing/preserving wild-type EGFR to a greater extent than early generations of EGFR-TKIs.16–18
BBB penetration
Both osimertinib and aumolertinib demonstrate an enhanced capacity to penetrate the BBB in comparison to their predecessors. Osimertinib’s high bioavailability and lipophilic nature permit the attainment of meaningful drug concentrations in the cerebrospinal fluid (CSF), which translates into notable clinical benefits in patients with CNS metastases.10,19–21 APOLLO 21 and BLOOM 19 studies report that osimertinib achieves BBB penetrations of up to 31.7% and 16%, respectively, which is significantly higher than the 2.8%–5.1% for erlotinib, 1%–3% for gefitinib, and 0.7% for afatinib.
Aumolertinib also exhibits superior BBB penetration due to its cyclopropyl component, rendering it a compelling therapeutic candidate for patients with brain metastases. Despite the paucity of direct comparisons between osimertinib and aumolertinib in terms of BBB crossing, preclinical and clinical studies indicate that aumolertinib also has a significant advantage over earlier generations in reaching therapeutic levels within the CNS. 22
Advantages in treating CNS metastases
One of the key advantages of third-generation EGFR-TKIs is their ability to effectively manage CNS involvement, a common and challenging complication in advanced NSCLC. A number of studies have demonstrated that patients with brain metastases who are treated with osimertinib experience improved responses. Similarly, emerging data suggest that aumolertinib may also confer benefits in this setting, although further evidence is required to confirm its precise role compared to osimertinib.23,24
In summary, both osimertinib and aumolertinib embody the evolution of targeted therapy in NSCLC. They possess distinctive molecular structures that facilitate potent and specific inhibition of EGFR mutations, coupled with a higher capability to cross the BBB. This dual functionality renders them invaluable tools in combating CNS metastases, a crucial issue that was inadequately addressed by earlier generations of EGFR-TKIs. As the field of oncology continues to evolve, these agents are poised to redefine the standard of care for EGFR-mutant NSCLC patients with brain metastases, offering hope for improved survival and quality of life.
Regulatory approvals and clinical settings for third-generation EGFR-TKIs
Clinically, third-generation EGFR-TKIs, including osimertinib, aumolertinib, and furmonertinib, have been recommended as first-line treatment for patients with EGFR-mutated NSCLC due to their superior efficacy compared to first-generation EGFR-TKIs.1,2 They are also typically used in the second-line setting after disease progression on first- or second-generation EGFR-TKIs, specifically to overcome the secondary resistance caused by the acquired T790M mutation.4–6 Both the National Medical Products Administration in China and the US Food and Drug Administration have approved third-generation TKIs above indications. In China, the use of third-generation EGFR-TKIs generally follows international and national guidelines. Therefore, the clinical setting for these drugs varies depending on the patient’s treatment history and specific mutation profile. However, variations in the timing and sequencing of treatment may occur based on local availability, healthcare policies, and the evolving evidence base.
The state and challenges of T790M mutation testing
Mutation testing for T790M in EGFR is a fundamental aspect of the management of NSCLC patients with acquired resistance to first- or second-generation EGFR-TKIs. The diversity of T790M mutation detection methods, the accessibility of biological samples for molecular testing, along with their inherent limitations, contribute to a significant proportion of patients having an unknown T790M status.25,26
Tissue biopsy: Spatial and temporal heterogeneity, and branched evolution
The traditional tissue biopsy remains the gold standard for detecting the T790M mutation due to its high sensitivity and specificity. It offers more comprehensive genetic profiling, revealing additional resistance mechanisms or gene rearrangements that are crucial for guiding therapeutic decisions. This process involves the surgical removal or needle biopsy/aspiration of tumor tissue from a resistant lesion, followed by molecular analysis using techniques such as polymerase chain reaction (PCR) or next-generation sequencing (NGS).
Nevertheless, the practical detection rates for the T790M mutation can fluctuate between 30% and 60%, influenced by issues such as tumor heterogeneity and sampling bias. The potential complications and the inability to access recurrent or metastatic lesions safely can limit their applicability. Furthermore, sampling bias due to tumor heterogeneity may lead to false-negative results, especially when the resistant clone is not adequately represented in the sampled tissue.27–29
Brain metastases exhibit branched clonal evolution with unique genetic alterations differing from extracranial sites. The reported concordance rate of EGFR mutations between paired primary NSCLC and brain metastases varies from 36% to 100%. 30 In contrast, spatially separated brain metastases tend to share similar clonal architectures, providing evidence for divergent evolution leading to CNS seeding. 31 The emergence of unique clonal events within brain metastases may arise from acquired mutations that confer therapeutic resistance. 31 In one study involving 78 patients who underwent rebiopsy following TKI failure, 41% of extracranial lesions harbored T790M mutations, compared to only 17% of brain metastases. 32 The biological basis for the disparity in EGFR T790M mutations across different sites remains unclear, but one possible explanation is the differential drug exposure due to the BBB. 31
NGS can detect a wide range of mutations, including EGFR T790M, making it suitable for identifying T790M in tumor tissue. NGS enables the testing of multiple genes in a single assay, which is efficient and cost-effective. However, tissue samples must be of sufficient quality and quantity, and NGS requires specialized equipment and expertise for data analysis.33,34 Amplification Refractory Mutation System (ARMS) PCR provides high-specific detection of the T790M mutation, with results obtained relatively quickly. ARMS-PCR can be more cost-effective than NGS for targeted mutation detection, but it may not detect T790M if present at very low levels and can be affected by the presence of non-tumor DNA.35,36 Digital PCR (dPCR) is highly sensitive and can detect T790M in small amounts of cDNA in both tumor tissue and liquid biopsies, providing quantitative data on the level of the mutant allele. However, dPCR requires high-quality DNA for accurate quantification and can be more expensive than other methods. 37
Liquid biopsy: Diversity and complexity, and diagnostic challenges
Liquid biopsy represents a minimally invasive alternative for the detection of the T790M mutation. This is achieved primarily through the analysis of circulating tumor DNA (ctDNA) in peripheral blood, CSF, or pleural effusion and ascites. ctDNA carries genetic information released from dying cancer cells and can reveal the presence of T790M mutations. The sensitivity of liquid biopsy (through ctDNA analysis) for the detection of the T790M mutation is typically between 50% and 90%, with variations dependent on the specific detection method employed and the patient’s clinical characteristics.38,39 With regard to specificity, liquid biopsy typically demonstrates a high level, often exceeding 90%. 40
Plasma genotyping is increasingly being adopted, with assays such as the PCR-based EGFR mutation test demonstrating a high degree of concordance with tissue genotyping for common EGFR mutations. However, the sensitivity of liquid biopsy varies depending on the technique, tumor burden, and ctDNA release dynamics, which can result in false negatives, particularly in patients with low tumor burden or isolated CNS metastases.41,42 It is important to note that in instances where liquid biopsy yields a negative result despite strong clinical suspicion of a T790M mutation, tissue biopsy remains essential due to the potential for false negatives resulting from low ctDNA concentrations.
CSF biopsy is often the preferred method for patients with stable primary tumors but evidence of CNS progression, particularly leptomeningeal metastases, which are common in individuals with EGFR mutations. CSF analysis is critical for investigating resistance mechanisms and guiding treatment decisions. Notably, the EGFR T790M mutation is detected less frequently in CNS lesions or in CSF-circulating free DNA from patients with CNS metastases compared to those with extracranial relapses. This disparity may be due to the limited penetration of TKIs into the CSF and the spatial and temporal heterogeneity of the T790M mutation, suggesting that additional mechanisms may underlie resistance to TKIs in brain/leptomeningeal metastases.43,44
CSF analysis, though less commonly used, can provide valuable insights into the T790M status within the CNS compartment. However, the quantity of ctDNA in CSF is usually much lower than in plasma, requiring highly sensitive methods to detect the mutation. Studies have shown that the prevalence of the T790M mutation detected in CSF is generally lower than in plasma, suggesting that plasma-based evaluations may not accurately reflect the mutational status within the CNS, and vice versa.45–48
Limitations and implications
Due to the technical and logistical challenges associated with this approach, around 50% of patients who are resistant to EGFR-TKIs undergo tissue re-biopsy, while 20%–50% rely on liquid biopsy for the determination of their T790M status. Despite the advances in liquid biopsy technology, its sensitivity and reproducibility for detecting the T790M mutation remain a subject of ongoing research and improvement. This variability results in a considerable number of patients with an uncertain or indeterminate T790M status, which complicates the decision-making process for subsequent therapy, including the administration of third-generation EGFR-TKIs, such as osimertinib and aumolertinib.49–52
In conclusion, the current state of T790M mutation testing is characterized by a diverse array of methods, each with its own set of strengths and weaknesses. The persistent challenges surrounding tissue accessibility, sensitivity issues in liquid biopsy, and inter-method discordances necessitate careful consideration of the optimal testing strategy for each patient. These limitations contribute to the large pool of patients with unknown T790M status, highlighting the urgent need for more accurate, reliable, and accessible diagnostic tools to guide personalized treatment decisions in the era of precision medicine.
BBB and challenges of EGFR-TKIs in treating brain metastases
The role of the BBB in brain metastases and drug delivery
The BBB and the blood–CSF barrier are distinct anatomical and functional barriers in the brain. The blood–CSF barrier is more permeable than the BBB, allowing water-soluble substances to enter the CSF at a rate inversely proportional to their molecular weight. Importantly, drug distribution into the CSF does not necessarily reflect the permeability of the BBB. Intrathecal drug delivery to the CSF mimics a slow intravenous injection, with the drug subsequently distributed to the blood and the surface of the brain, but not penetrating deeply into the brain parenchyma beyond 1–2 mm from the CSF compartment.53,54
Preclinical studies indicate that osimertinib has higher brain exposure compared to other EGFR-TKIs. First-generation TKIs have limited CNS penetration (1.3% ± 7% for gefitinib and 4.4% ± 3.2% for erlotinib) and are less effective against the T790M mutation.55,56 In contrast, osimertinib has higher BBB penetration and a higher CNS ORR (91% vs 68% for gefitinib or erlotinib in the FLAURA study). 57
Challenges of EGFR-TKIs in treating brain metastases
Stewart 58 found that drug concentrations in brain tumors are typically higher than in CSF and normal brain tissue, suggesting that the BBB may not be as impermeable as previously thought. Response rates to chemotherapy in brain metastases are similar to those in other sites, particularly in treatment-naïve patients. In the FLAURA study, osimertinib showed similar efficacy in patients with and without brain metastases at baseline, with comparable response rates for intracranial and extracranial lesions. These findings challenge the traditional view of the BBB’s role in treating brain metastases. However, there is also evidence that the BBB remains important even when disrupted in brain and leptomeningeal metastases.31,59 These metastases exhibit a distinct molecular mutation profile, including a different frequency and abundance of resistant mutations such as T790M. 31
Osimertinib is more effective than first-generation TKIs in preventing new brain metastases. In the ADJUVANT study, gefitinib had a high rate of CNS recurrence (27.4% as the first site of recurrence and 40% estimated probability at 36 months). 60 In the ADAURA study, osimertinib reduced the risk of CNS relapse (2% at 36 months and 97% alive and CNS disease-free). 61 These findings suggest that the BBB may not significantly hinder osimertinib’s efficacy in preventing brain metastases, while it may play a more significant role in the efficacy of first-generation TKIs like gefitinib.
Even with third-generation EGFR-TKIs, the treatment and prevention of brain metastases remain a major challenge. Molecular heterogeneity, that is, the difference in molecular characteristics between brain metastatic tumor cells and primary tumor cells, can occur in up to 50% of patients. 62 Another related issue is that micrometastases (less than 1 mm) do not alter the BBB, thereby reducing the efficacy of anticancer drugs used in adjuvant therapy. 63 Restricted access to therapeutic agents in the normal brain is one of the main reasons for the increasing incidence of brain metastases, as the BBB creates a pharmacological sanctuary that protects tumor cells from being targeted. 64 For these reasons, a major challenge in the treatment of brain metastases from these tumors is the development of molecules with the ability to cross the BBB effectively.
Efficacy of osimertinib across different T790M mutation status
Osimertinib has demonstrated remarkable clinical efficacy in the treatment of patients with NSCLC who have developed various T790M mutation statuses following resistance to first- or second-generation EGFR-TKIs. A closer examination of pertinent research, including a systematic review and meta-analysis conducted by Yi et al., 10 provides insight into the nuanced performance of osimertinib across these different mutational landscapes.
The study by Yi and colleagues, which included a total of 1313 patients with EGFR-mutated NSCLC from 10 retrospective studies and one prospective study,19,65–75 demonstrated a clear distinction in the overall clinical benefits of osimertinib between patients with T790M-positive and T790M-negative tumors. T790M-positive patients exhibited superior outcomes in terms of OS, PFS, and ORR when treated with osimertinib compared to their T790M-negative counterparts. The hazard ratio (HR) for OS was 0.574 (p = 0.015), the HR for PFS was 0.476 (p = 0.017), and the relative risk ratio for ORR was 2.025 (p = 0.000). This finding is consistent with the rationale behind the design of osimertinib, which was developed to target the T790M mutation. However, a noteworthy observation emerges when analyzing the impact of osimertinib in the context of brain metastases. When focusing on patients with brain metastases, the study found no statistically significant differences in OS between T790M-positive and T790M-negative patients (HR = 0.75, p = 0.449), nor between T790M-positive and unknown patients (HR = 0.90, p-value not specified). This indicates that osimertinib confers clinical benefits to patients with brain metastases, irrespective of their T790M status.
This observation has significant clinical relevance, as brain metastases are a common and often devastating complication in NSCLC patients, and the BBB presents a formidable obstacle for many systemic therapies. The ability of osimertinib to effectively cross the BBB and exert antitumor effects on brain metastases, independent of the status of T790M, implies a broadened therapeutic window and underscores its potential as a key treatment option for patients with brain involvement, regardless of the status of their T790M mutations.
A single-arm, phase II study by Park et al. 12 evaluated the efficacy of 80 mg once daily osimertinib in patients with leptomeningeal metastases resistant to prior first- or second-generation EGFR-TKIs, demonstrating significant intracranial efficacy and survival benefits, with an ORR for leptomeningeal metastases of 51.6%, a disease control rate (DCR) of 81.3%, and a median OS of 15.6 months, supporting the consideration of osimertinib as a treatment option for patients with EGFR-mutated NSCLC and leptomeningeal metastases, regardless of T790M mutation status.
The evidence gathered from the study by Yi et al. and other related works supports the notion that osimertinib displays a robust clinical profile across different T790M mutation states, particularly in the context of brain metastases. Although its efficacy is indeed pronounced in T790M-positive patients, its action on brain metastases appears to transcend the status of the T790M mutation. This provides a promising avenue for improving outcomes in a patient population with otherwise limited therapeutic options.
Efficacy of aumolertinib in T790M-negative or unknown-status patients
Aumolertinib has drawn considerable interest for its potential use in the treatment of NSCLC patients with brain metastases who have developed resistance to first- or second-generation EGFR-TKIs and whose T790M mutation status is either negative or unknown. In a comprehensive effort led by Dr Han and his team, a retrospective multicenter study was conducted across multiple Chinese hospitals to illuminate the efficacy and safety profile of aumolertinib in this specific patient group where current clinical data is relatively scarce. 11
The distinctive chemical structure of aumolertinib, characterized by the inclusion of a cyclopropyl group, confers enhanced penetration through the BBB, a crucial feature when addressing brain metastases. Early clinical evidence from pivotal trials such as AENEAS and APOLLO indicated the potential efficacy of aumolertinib against intracranial lesions in NSCLC.5,76–78 However, the precise advantages of using aumolertinib in T790M-negative or indeterminate patients post-EGFR-TKI resistance remain unclear.
The study by Dr Han retrospectively evaluated the clinical outcomes of patients who had demonstrated resistance to previous generations of EGFR-TKIs and lacked a positive mutation status of T790M. These patients often face a lack of effective targeted therapies to follow and generally have a poor prognosis. The administration of aumolertinib to this cohort aimed to explore its viability as a therapeutic option. 11
The study yielded preliminary yet encouraging results, with the median PFS for patients treated with aumolertinib reaching 10.1 months. However, this result was not stratified based on T790M status. The analysis showed a remarkable DCR of 94.7% and a complete intracranial DCR of 100% within a subset of patients, indicating the potential of aumolertinib to effectively manage brain metastases. Despite these promising signs, the investigation did not provide explicit comparative numerical data between patients with T790M-negative or unknown status and those with T790M-positive mutations. 11
Despite the inherent limitations of retrospective designs and the modest sample size, the study contributed significant insights into the potential application of aumolertinib for T790M-negative or unknown status NSCLC patients with brain metastases. The high DCR and intracranial DCR suggest that aumolertinib may offer clinical benefits, evidenced by PFS and overall disease control, even in the absence of the T790M mutation. Furthermore, the drug was found to be well tolerated with manageable side effects, which were primarily mild adverse reactions such as rash and diarrhea.
These preliminary findings suggest the necessity for further robust investigation, ideally through larger-scale prospective trials, to confirm the initial observations and firmly establish aumolertinib as a reliable therapeutic strategy for this challenging patient demographic. The collective findings thus far indicate that aumolertinib may serve as an alternative therapeutic agent in the management of NSCLC patients with brain metastases and T790M-negative or unknown mutation status. This could expand the repertoire of effective targeted therapies for individuals who might otherwise have limited treatment options following the development of EGFR-TKI resistance.
Role of third-generation EGFR-TKIs in uncommon EGFR mutations
Uncommon EGFR mutations, including exon 20 insertions, G719X, L861Q, and S768I, account for approximately 10% of all EGFR mutations and typically respond less favorably to first-generation EGFR-TKIs compared to common mutations. 79 Afatinib shows activity against uncommon EGFR mutations, especially G719X, L861Q, and S768I, but has limited efficacy against T790M and exon 20 insertions. 80 In a retrospective study, patients with uncommon EGFR mutations had a lower incidence of the T790M mutation (27.1%) after TKI resistance compared to those with exon 19 deletions (55.2%) and L858R mutations (37.2%). Patients with uncommon mutations treated with subsequent osimertinib had significantly shorter median PFS (4.6 months) and OS (8.1 months) than those with exon 19 deletions (PFS: 11.6 months, OS: 35.4 months) and L858R (PFS: 12.1 months, OS: 24.9 months). 81
Several studies have evaluated the efficacy of third-generation EGFR-TKIs, including osimertinib, aumolertinib, and furmonertinib, in patients with uncommon EGFR mutations.82–85 Two phase II studies of osimertinib in these patients showed ORRs of 50% (KCSG-LU15-09; n = 37) and 55% (UNICORN; n = 40), with median PFS of 8.2 and 9.4 months, respectively.82,83 In a phase II trial of aumolertinib, the ORR was 30% with a DCR of 70%. 84 A recent meta-analysis of 331 osimertinib-treated patients with uncommon EGFR mutations reported an overall ORR of 49.5%, a DCR of 90%, a median PFS of 9.5 months, and a median OS of 24.5 months. 85
These findings highlight the potential therapeutic benefit of third-generation EGFR-TKIs in treating patients with uncommon EGFR mutations, although the benefit is generally less than that observed in patients with common mutations.
Integrating third-generation EGFR-TKIs with other therapeutic modalities
In the context of developing more sophisticated treatment strategies for specific patient populations, such as those with brain metastases in EGFR-mutated NSCLC, the integration of third-generation EGFR-TKIs such as osimertinib and aumolertinib with other modalities such as radiation therapy, surgery, antiangiogenic therapy, and chemotherapy warrants detailed consideration.
For patients presenting with brain metastases, a multifaceted approach may be essential to optimize outcomes. Although third-generation EGFR-TKIs have demonstrated strong CNS penetration and promising efficacy in controlling both systemic and intracranial lesions irrespective of the status of the T790M mutation, combination strategies could offer additional benefits. For instance, stereotactic radiosurgery or whole-brain radiation therapy could be used in conjunction with osimertinib to manage selected brain metastases, particularly those that are large or symptomatic, providing immediate local control before the targeted therapy takes effect.
The sequence of treatments is of great importance in determining the most appropriate course of action. In certain cases, initial treatment with a third-generation EGFR-TKI may be preferable to shrink or stabilize brain metastases before considering surgical intervention, thereby potentially improving operability. Conversely, surgery followed by targeted therapy may be beneficial in instances where resection offers the possibility of complete removal of all detectable diseases, thus allowing better disease control with subsequent EGFR-TKI treatment.
Additionally, while many chemotherapeutic agents have limited ability to cross the BBB, certain studies indicate that the efficacy of chemotherapy in treating brain metastases is comparable to its effectiveness in other metastatic sites. 86 More recent findings suggest that combining chemotherapy with osimertinib can significantly improve outcomes in patients with brain metastases to a similar extent as in those without brain metastases.87,88
To enhance therapeutic outcomes and address resistance, researchers are investigating the potential of combining these highly potent TKIs with antiangiogenic agents, including large-molecule monoclonal antibodies (e.g., bevacizumab or ramucirumab) 89 and small-molecule multitargeted angiogenesis inhibitors (e.g., lenvatinib, vandetanib, anlotinib, or apatinib). This dual approach aims to address two critical pathways in the progression of NSCLC: directly inhibiting tumor cell proliferation through the EGFR pathway, and suppressing tumor vasculature development and maintenance through antiangiogenic mechanisms. Recent studies and clinical trials have investigated the potential benefits of these combinations. Nevertheless, no difference in PFS was observed in patients who progressed on prior EGFR-TKI therapy between osimertinib plus bevacizumab and osimertinib alone in two randomized phase II studies.90,91 Similarly, the integration of third-generation EGFR-TKIs with novel angiogenesis inhibitors, still in investigative stages, is being explored to address resistance mechanisms and optimize treatment outcomes.
Clinicians must consider several factors when deciding on the integration of third-generation EGFR-TKIs with other treatments, including the extent and location of brain metastases, patient fitness for combined treatments, and the potential for overlapping toxicities. Sequential or concurrent treatment algorithms can be tailored depending on the molecular profile of the tumor and the individual patient’s circumstances. Future research focusing on prospective clinical trials and biomarker-driven strategies will continue to refine these treatment paradigms, with the aim of providing the most personalized and effective therapeutic plan for the unique needs of each patient within the challenging landscape of resistance to EGFR-TKI.
Dose escalation with third-generation EGFR-TKIs
With regard to the topic of dose escalation with third-generation EGFR-TKIs, the current literature and conference reports indicate promising data. Studies have explored the possibility of increasing doses of osimertinib and aumolertinib in specific subsets of EGFR-mutated advanced NSCLC patients with varying T790M mutational statuses after resistance developed to previous EGFR-TKIs. 92
Although standard doses of osimertinib have been established, emerging evidence suggests that increased doses may be tolerated and potentially beneficial in selected patients with aggressive disease or when standard doses do not produce the desired response. However, formal studies directly comparing escalated versus standard dosing are limited, and the majority of findings remain anecdotal or observational. In certain clinical scenarios, such as rapid progression or insufficient disease control, case reports and small series have indicated that increased doses could lead to longer progression-free intervals or improved response rates. 93
Alternatively, aumolertinib’s notable ability to penetrate the BBB effectively and its demonstrated clinical efficacy in patients harboring the T790M mutation prompts inquiries into whether escalating the dosage could unlock further therapeutic advantages, particularly for those with unfavorable prognoses or treatment-resistant brain metastases with T790M-negative or unknown status. Although a limited number of studies have explored dose-escalation approaches with aumolertinib, preliminary findings suggest that this approach may be efficacious and well-tolerated.77,94 The ongoing ATTACK trial is evaluating the efficacy and safety of high-dose aumolertinib (165 mg once daily) versus osimertinib (80 mg once daily) in patients with EGFR T790M-positive NSCLC with brain metastases. 95
It is crucial to highlight that any decision to increase doses must be weighed against the potential for increased toxicity and the necessity for close patient monitoring. While there are encouraging indications suggesting the feasibility of higher doses in specific patient populations, further prospective, controlled studies are required to formally investigate the safety, efficacy, and optimal dosing regimens for third-generation EGFR-TKIs in the context of dose escalation, especially in relation to T790M status and the presence of brain metastases. This would assist in the refinement of treatment paradigms and provide guidance for personalized medicine approaches in the management of EGFR-mutated NSCLC.
Expert consensus and recommendations
A synthesis of multiple research studies and adherence to the principles of evidence-based medicine has yielded a comprehensive expert consensus on the strategic deployment of third-generation EGFR-TKIs for patients with varying T790M mutation statuses. In light of the accumulated data, experts have recommended tailored approaches for the treatment of patients who are unable to have their T790M status definitively determined or for those found to be T790M-negative. Table 1 provides a detailed account of the expert consensus.
Expert consensus recommendations for the use of third-generation EGFR-TKIs in EGFR-mutant advanced NSCLC with varying T790M status after resistance developed to prior treatment with first- or second-generation EGFR-TKIs.
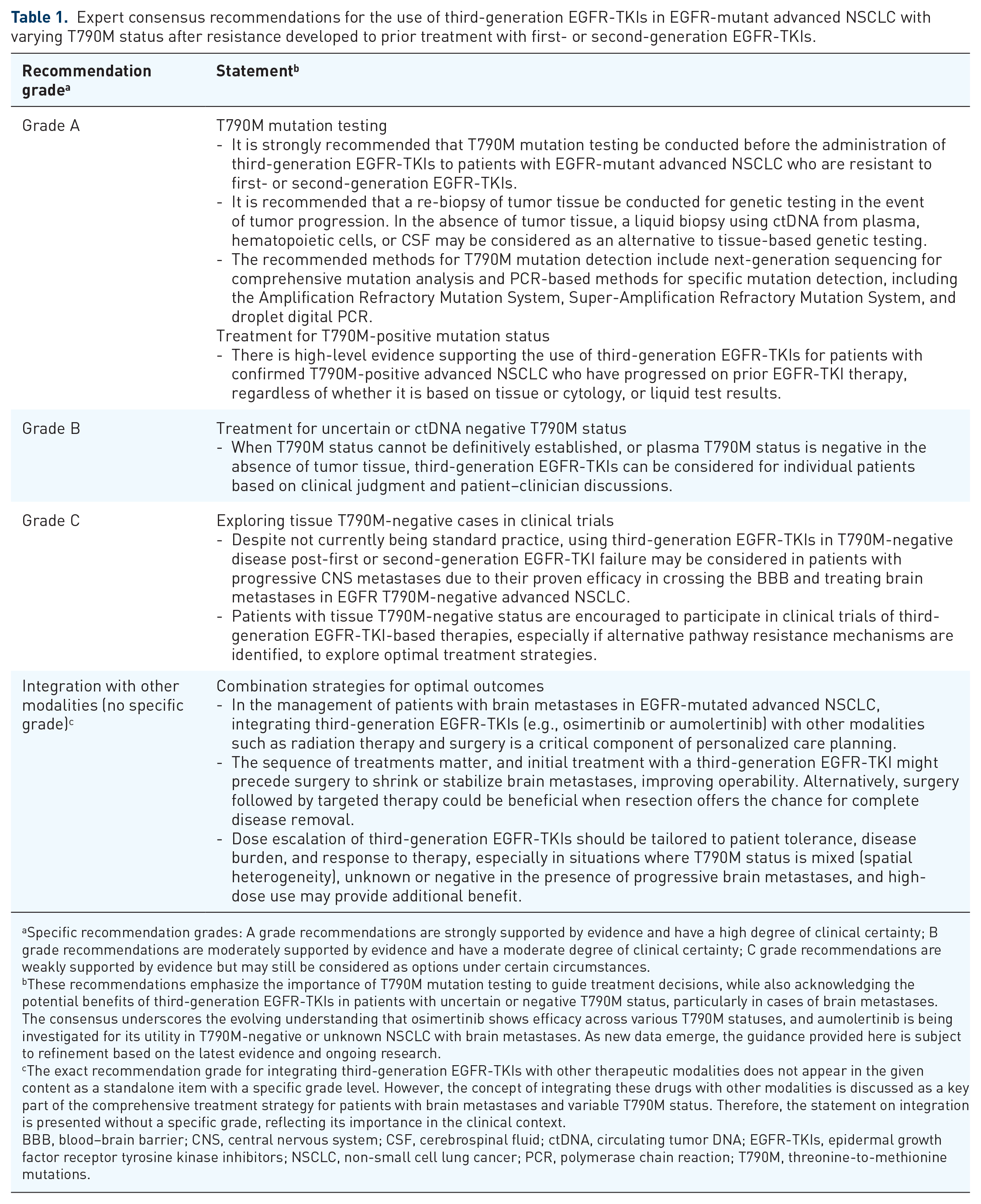
Specific recommendation grades: A grade recommendations are strongly supported by evidence and have a high degree of clinical certainty; B grade recommendations are moderately supported by evidence and have a moderate degree of clinical certainty; C grade recommendations are weakly supported by evidence but may still be considered as options under certain circumstances.
These recommendations emphasize the importance of T790M mutation testing to guide treatment decisions, while also acknowledging the potential benefits of third-generation EGFR-TKIs in patients with uncertain or negative T790M status, particularly in cases of brain metastases. The consensus underscores the evolving understanding that osimertinib shows efficacy across various T790M statuses, and aumolertinib is being investigated for its utility in T790M-negative or unknown NSCLC with brain metastases. As new data emerge, the guidance provided here is subject to refinement based on the latest evidence and ongoing research.
The exact recommendation grade for integrating third-generation EGFR-TKIs with other therapeutic modalities does not appear in the given content as a standalone item with a specific grade level. However, the concept of integrating these drugs with other modalities is discussed as a key part of the comprehensive treatment strategy for patients with brain metastases and variable T790M status. Therefore, the statement on integration is presented without a specific grade, reflecting its importance in the clinical context.
BBB, blood–brain barrier; CNS, central nervous system; CSF, cerebrospinal fluid; ctDNA, circulating tumor DNA; EGFR-TKIs, epidermal growth factor receptor tyrosine kinase inhibitors; NSCLC, non-small cell lung cancer; PCR, polymerase chain reaction; T790M, threonine-to-methionine mutations.
For patients in whom T790M mutation status cannot be confidently established or is confirmed as negative following resistance to first- or second-generation EGFR-TKIs, the consensus leans toward incorporating third-generation EGFR-TKIs comprising osimertinib and aumolertinib as a preferred second-line or subsequent therapeutic option. This recommendation is largely driven by their proven efficacy across various T790M mutation states, especially in the context of brain metastases where its ability to cross the BBB offers significant clinical advantages.
In the scenario of brain metastases, the collective evidence highlights the potential of third-generation EGFR-TKIs to confer benefits regardless of T790M status, with a trend toward equivalent or even favorable survival outcomes compared to traditional platinum-based chemotherapy. This makes third-generation EGFR-TKIs a compelling choice, especially given the historically poor prognosis associated with brain metastases and the limitations of conventional chemotherapy in effectively treating intracranial disease.
Furthermore, experts recognize the need to balance the benefits of third-generation EGFR-TKIs with considerations such as patient comorbidities, overall health status, and availability of other targeted therapies. They advocate for a multidisciplinary approach in making treatment decisions, ensuring that individual patient characteristics are taken into account alongside the latest clinical data.
In summary, the evolving expert consensus supports the preferential use of third-generation EGFR-TKIs, such as osimertinib and aumolertinib, in the management of advanced EGFR-mutated NSCLC patients with ambiguous or negative T790M status, especially those with brain metastases, contingent on the integration of all available evidence and individual patient factors. Future research and continued surveillance of clinical outcomes will further refine these recommendations and enhance the precision with which oncologists can tailor treatment strategies to suit the unique circumstances of each patient.
Limitations and prospects
In light of the current state of research on third-generation EGFR-TKIs, it is crucial to recognize existing limitations and discuss future avenues for progress. One of the main limitations highlighted concerns the modest sample sizes of numerous studies investigating the efficacy of third-generation EGFR-TKIs in patients with negative T790M or unknown status. Smaller samples can introduce bias, reduce statistical power, and hinder the generalizability of results, necessitating larger and more representative cohorts to derive definitive conclusions.
Another significant limitation stems from the predominantly retrospective design of many studies. Retrospective analyses inherently carry a higher risk of selection bias and are less able to establish causal relationships compared to randomized controlled trials. Therefore, there is a pressing need for well-designed, prospective, multicenter clinical trials to confirm the precise efficacy and safety profiles of third-generation EGFR-TKIs in T790M-negative or unidentified patients.
Furthermore, optimizing the T790M mutation detection methods is paramount. This could involve advances in liquid biopsy techniques, including ctDNA analysis, to improve the accuracy and accessibility of the T790M mutation test, allowing clinicians to maximize the potential benefits of third-generation EGFR-TKIs.
Conclusion
Despite the multitude of challenges faced, osimertinib has proven its efficacy and feasibility across a spectrum of T790M mutation statuses in clinical practice. The drug has demonstrated its ability to prolong survival and improve outcomes in patients, whether they harbor the T790M mutation or not, especially in those with brain metastases where its ability to cross the BBB is crucial. On the other hand, aumolertinib, while still accumulating a body of evidence, has begun to demonstrate its potential as a viable therapeutic option for T790M-negative or unknown-status patients with EGFR-mutated NSCLC. Preliminary data from retrospective studies and smaller clinical trials suggest its effectiveness in managing disease progression, especially in the context of brain metastases. However, more comprehensive and robust data are needed to support these observations. It will be necessary to conduct well-designed, prospective clinical trials to confirm the precise efficacy and safety profiles of third-generation EGFR-TKIs in T790M-negative or unidentified patients.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to the Specialized Committee on Lung Cancer Cell Targeting and Immunotherapy, Liaoning Society of Cell Biology, for their support and contributions. We especially thank the following members: Cui Xiujie, Dai Zhaoxia, Han Yun, Jin Bo, Li Xiaohan, Liu Yuhui, Man Li, Wang Ling, Wang Zhenghua, Xu Long, Zhang Lingyun, Zhang Xuhong, Bai Jinghui, Chen Tiejun, Chen Qiang, Cui Wei, Deng Jian, Dong Yuyang, Fang Fengqi, Gao Lijie, He Lijie, Huang Bo, Huo Wei, Li Ailin, Li Wanya, Liu Xiaomei, Liu Yongye, Lyu Jinfang, Mou Yanshu, Qu Yanli, Ren Yi, Sun Nan, Wang Gang, Wang Liang, Zhang Jian, Zhao Yi, Zheng Jiahe, Zhou Caiyun, Geng Yan, Wang Yanru, Guan Chang, Bai Nan, Ma Wenbo, Wang Ying, Xu Ying, Song Shuxi, Liu Junling, Liu Xingchi, Li Bo, Sun Ying, Ren Xue, Wang Yongjie, Cao Jing, Sun Yuening, Huang Fusheng, Xu Chengtao, Lyu Li, Pan Xinyan, Sun Zhuo, Jiang Jianing, Cui Saiqiong, Shan Xiu, Li Ying, Tian Lili, Wang Aman, Dong Yan, Cai Xin, Gao Ping, Wang Yiqian, Zhang Chunxia, Liu Xin, Song Jincheng, Li Dan, Lyu Jinyan, Chen Lei, Xie Fang, Yang Hang, Yang Jiashuo, Li Meng, Yang Yi, Piao Jinhua, Zhao Wei, Li Jing, Guan Runzhi, Chen Liyan, Shi Wei, Chen Qu, Jiao Lixin, Tong Yuna, Han Qiuyue, Wang Xiaoyu, Wang Ying, Han Yujia, Wang Xiaozhou, Zhou Zhicheng, Sun Xin, Yuan Meng, Zhu Libo, Duan Hongyan, He Yijiang, Kong Donghui, Zhang Xiaoning, Cai Cunwei, Chen Shuchen, Cheng Liangliang, Liu Cheng, Hao Yuanyuan, Liu Zhe, Liu Shiguang, Zhang Yu, Zhao Yongsheng, Jin E, Shen Longhai, Chen Can, Ding Chao, Jin Jiajun, Liu Ting, Tong Xin, Qin Yimin, Kong Qingdong, Sun Jing, Li Zhengjun, Liu Chang, Wang Shina, Liu Cong, Wang Shuai, Zhao Jun, Han Qiang, Wang Shuo, Lyu Ling, Men Wanfu, Wang Yan, Yang Zichang, Luo Ying, Ren Kaiming, Tang Juan, Zhang Shuling, Zhao Jianzhu, Sun Li, Zhang Xiangyan, Wang Yudong, Li Peng, Mao Yuqiang, Cao Wei, Sun Cheng, Gao Yajie, Li Xiaoling, Sun Liangxin, Wang Wei, Zheng Zhendong.
