Abstract
Background:
The esophageal mixed neuroendocrine–non-neuroendocrine neoplasm (MiNEN) is an extremely rare but poor prognosis tumor.
Objectives:
This retrospective study aimed to analyze the clinical characteristics of MiNEN and to investigate postoperative survival and prognostic factors.
Design:
This retrospective study analyzed 69 patients diagnosed with esophageal MiNEN at two major esophageal cancer centers in China from January 2000 to December 2021.
Methods:
We assessed demographic data, tumor characteristics, treatment modalities, and survival outcomes. Statistical analyses included Kaplan–Meier survival curves and Cox regression models to evaluate prognostic factors.
Results:
The most common histological types were combinations of small-cell carcinoma and squamous carcinoma (91.3%). The correct diagnostic rate of preoperative pathologic biopsy was only 4.3%. The median overall survival (OS) was 24.0 months, and disease-free survival (DFS) was 16.6 months. The 1-, 3-, and 5-year survival rates were 84.1%, 34.8%, and 25.3%, respectively. A peak period of recurrence or metastasis occurs in the first year after surgery, and regional lymph node recurrence is the main route of postoperative recurrence or metastasis. Tumor size, T-stage, N-stage, and tumor, lymph node, metastasis (TNM) stage were significant prognostic factors. Subgroup analyses showed that in patients with limited-stage MiNEN in stages I–III, the postoperative adjuvant treatment modality failed to improve OS and DFS compared with surgery alone. Postoperative adjuvant therapy also failed to prolong OS and DFS in patients with lymph node-positive MiNEN. No significant survival benefits were observed with different surgical techniques or adjuvant chemotherapy regimens.
Conclusion:
Esophageal MiNEN has aggressive behavior and a poor prognosis. In China, the pathologic type of esophageal MiNEN may be dominated by a combination of small-cell carcinoma and squamous carcinoma. Early-stage disease significantly correlated with improved survival outcomes. Current treatment protocols, similar to those for other esophageal cancers, show limited efficacy in improving patient survival.
Introduction
According to recent data from the International Agency for Research on Cancer, esophageal cancer accounted for approximately 511,000 new cases and 445,000 deaths globally in 2022, ranking it as the 11th most common and the 7th most lethal of the 33 assessed cancer types. 1 Particularly in China, esophageal cancer represents the fourth leading cause of cancer-related mortality,2,3 with a dismal 5-year overall survival (OS) rate of just 27.9%. 2 Squamous carcinoma and adenocarcinoma are two common pathologic types of esophageal cancer, 4 with the former being more common in China. 5
Esophageal mixed neuroendocrine–non-neuroendocrine neoplasms (MiNENs) are a notably rare and aggressive variant of esophageal cancer, characterized by poor prognosis.6,7 In 2017, the World Health Organization (WHO) defined MiNENs as tumors composed of over 30% neuroendocrine and non-neuroendocrine elements. 8 These tumors are primarily found in the gastrointestinal tract and pancreas, and are exceptionally uncommon, with the European Registry of Rare Cancers reporting an incidence of less than 0.01 per 100,000 individuals annually. 9 Within this category, esophageal MiNENs comprise about 15.9%–17.0% of all digestive system MiNEN occurrences.10,11
Owing to the scarcity of esophageal MiNEN, large-scale studies focusing on treatment strategies, long-term survival, and prognostic factors are lacking. The current approaches to diagnosis and treatment generally align with those established for other forms of esophageal cancer, though specific challenges associated with MiNEN require more targeted investigation. To date, most of the research on esophageal MiNEN has been limited to isolated case reports, and no consensus on treatment protocols has been achieved. This retrospective study aims to elucidate the clinicopathological characteristics, survival outcomes, and prognostic factors associated with esophageal MiNEN, providing insights that may inform future therapeutic strategies.
Methods
Patients and clinical data
This retrospective study evaluated 69 patients diagnosed with esophageal MiNEN who were treated from January 2000 to December 2021. The patients were treated at two leading esophageal cancer centers in China: Cancer Hospital of Chinese Academy of Medical Sciences/Beijing Union Medical College and the First Affiliated Hospital of Anhui Medical University. The inclusion criteria were patients who underwent therapeutic surgery and had a histopathological confirmation of MiNEN. The exclusion criteria were patients diagnosed with other types of mixed tumors that did not meet the WHO 2017 diagnostic standards, those who declined follow-up, and those with incomplete medical records.
Demographic data, tumor staging, and treatment records were systematically collected through each hospital’s medical record system. MiNEN staging was conducted using the 8th edition of the American Joint Committee on Cancer/Union for International Cancer Control tumor, lymph node, metastasis (TNM) staging system specifically for esophageal cancer. 12 The pathological diagnoses were independently reviewed by two pathologists specializing in neuroendocrine tumors, including histopathological slides and pathology reports. To ensure data integrity, three auditors double-checked the data daily, with random verification occurring twice weekly. The findings were then replicated and reviewed to confirm consistency.
Follow-up data were obtained through telephone interviews and outpatient clinic visits. The primary endpoints of the study were OS and disease-free survival (DFS), defined respectively as the duration from the commencement of surgical treatment to death by any cause, and from surgical treatment to the occurrence of recurrence, metastasis, or death by any cause.
All procedures were in accordance with the ethical standards of the institutional and/or national research committee and with the Helsinki Declaration of Helsinki (1964) and its later amendments or comparable ethical standards. This retrospective, non-interventional study has anonymized patients and the requirement for informed consent was waived based on its retrospective nature. Our retrospective study conforms to the Reporting of Observational Studies in Epidemiology statement 13 (Table S1).
Treatment methods
All 69 patients underwent curative surgery intended to fully resect the MiNEN. Preoperative examinations indicated evident distant metastatic disease in none of the patients. Thirty-four (49.3%) patients underwent Sweet, 17 (24.6%) patients underwent Ivor-Lewis, 16 (23.2%) patients underwent McKeown, and 2 (2.9%) patients underwent endoscopic submucosal dissection. Only one patient received preoperative neoadjuvant chemotherapy, consisting of two cycles of paclitaxel plus cisplatin. A total of 27 patients underwent surgery alone. Forty-one patients received surgery combined with adjuvant therapy, of which 16 patients received surgery combined with chemoradiotherapy, 24 patients received surgery combined with chemotherapy, and 1 patient received surgery combined with radiotherapy. The primary adjuvant chemotherapy regimens included paclitaxel plus a platinum-based agent, etoposide plus a platinum-based agent, and 5-fluorouracil (5-FU) plus a platinum-based agent, and 14, 13, and 8 patients received the above chemotherapy regimens, respectively, administered over 1–10 cycles depending on the patient’s postoperative recovery and tumor response.
Statistical analysis
Statistical analyses were carried out using SPSS version 22.0 (IBM Inc., Chicago, IL, USA). Descriptive statistics and frequency analysis were used to characterize the clinical data. Survival analyses, including OS and DFS, were performed using Kaplan–Meier curves, and differences between groups were evaluated using the log-rank test. To determine the influence of clinicopathological features on survival, both univariate and multivariate Cox regression analyses were employed. A p-value of less than 0.05 was considered statistically significant.
Results
Demographic information and tumor characteristics
The patient selection flowchart is shown in Figure 1. Our retrospective analysis included 69 patients diagnosed with esophageal MiNEN, selected from two prominent esophageal cancer centers in China. As illustrated in Table 1, the cohort consisted predominantly of males (81.2%, n = 56) compared to females (18.8%, n = 13), resulting in a male-to-female ratio of 4.3:1. The mean age of the patients was 63.7 ± 8.6 years, with a range from 43 to 82 years. Notably, a significant proportion of the cohort had a history of smoking (60.9%, n = 42) and alcohol consumption (46.4%, n = 32). In addition, 9 (13%) patients reported a family history of malignancy, and 13 (18.8%) patients had other chronic diseases such as hypertension, diabetes mellitus, stroke, and epilepsy.
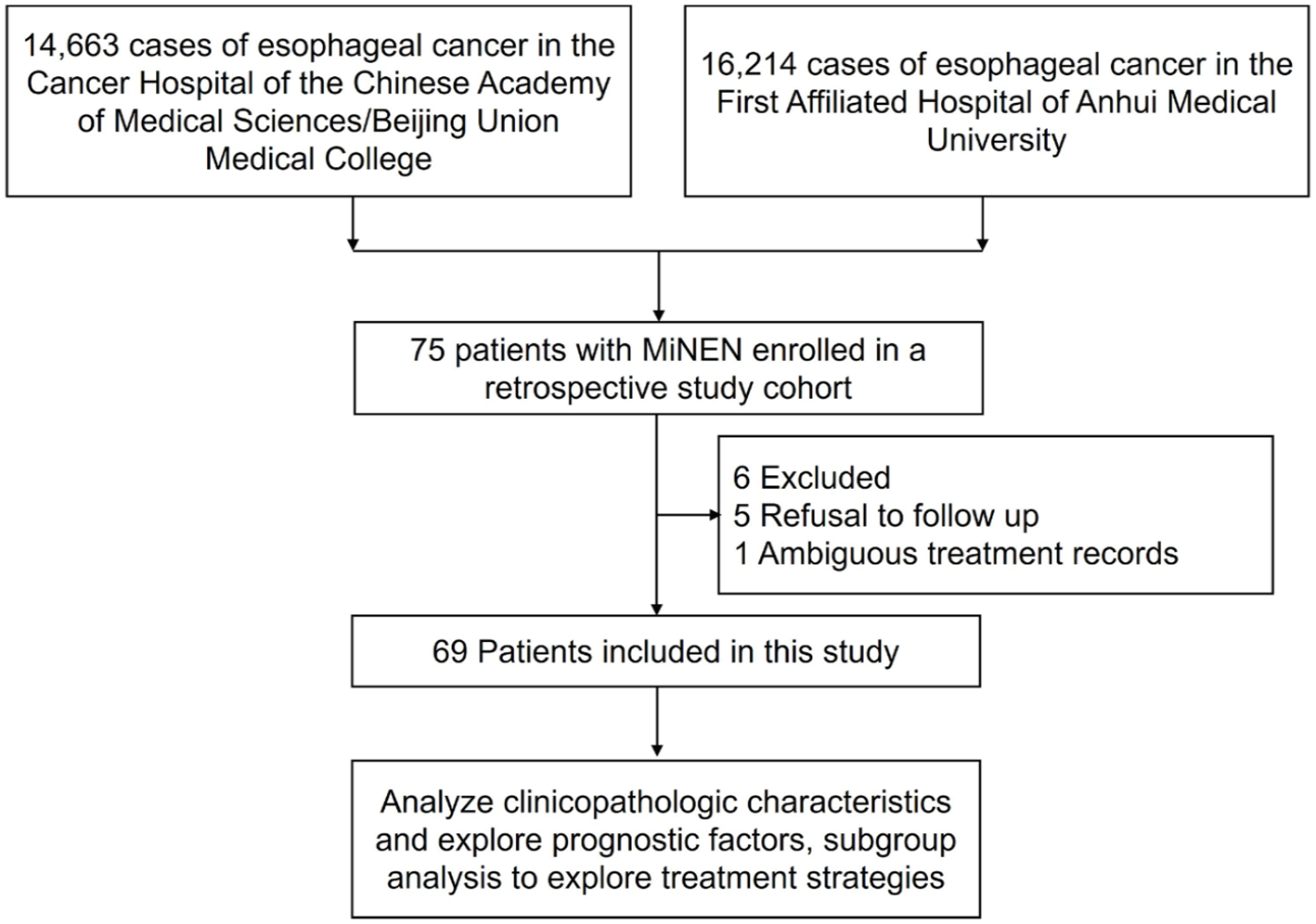
Patient selection flowchart.
Univariate analysis of survival in 69 patients with MiNEN.

BMI, body mass index; DFS, disease-free survival; ESCC, esophageal squamous cell carcinoma; LN, lymph node; MiNEN, mixed neuroendocrine–non-neuroendocrine neoplasm; OS, overall survival; PSCCE, primary small-cell carcinoma of the esophagus; TNM, tumor, lymph node, metastasis.
Endoscopic biopsy was completed preoperatively in all patients and its diagnostic accuracy was only 4.3%. The tumors are predominantly located in the mid-thoracic esophagus (59.4%), followed by the lower thoracic esophagus (34.8%), and the least in the upper thoracic esophagus (5.8%). Tumor sizes varied, with a majority (60.9%) measuring more than 3 cm in diameter. Histologically, the predominant post-surgery findings were a combination of small-cell carcinoma and squamous carcinoma, observed in 91.3% of cases (n = 63). A smaller fraction (8.7%, n = 6) exhibited a combination of small-cell carcinoma and adenocarcinoma.
Survival and recurrence
Follow-up information is available for all patients. Comprehensive follow-up data revealed that as of April 1, 2024, 18.8% of patients (n = 13) were alive, while 81.2% (n = 56) had succumbed to the disease. The median follow-up time for this cohort was 111.6 months (range: 63.0–160.2 months). The median OS was calculated at 24.0 months, with survival rates at 1, 3, and 5 years marked at 84.1%, 34.8%, and 25.3%, respectively, as shown in Figure 2(a). The median DFS was observed at 16.6 months, with DFS rates at 1, 3, and 5 years being 56.5%, 28.8%, and 19.4% (Figure 2(b)). Significant statistical associations were identified between both OS and DFS with tumor size, T-stage, N-stage, and TNM stage (Figure 3). Specifically, tumors ⩽3 cm were associated with better OS and DFS compared to those >3 cm (p = 0.031 and p = 0.024, Figure 3(a) and (e), respectively). In addition, earlier T-stages were associated with longer OS and DFS (p = 0.001 and p = 0.001, Figure 3(b) and (f), respectively). Earlier N-stages were associated with longer OS and DFS (p = 0.001 and p < 0.001, Figure 3(c) and (g), respectively). Not surprisingly, the same results were seen in the TNM stages, with longer OS and DFS the earlier the TNM stages (p < 0.001 and p < 0.001, Figure 3(d) and (h), respectively).

OS and DFS in the study cohort: (a) OS and (b) DFS.
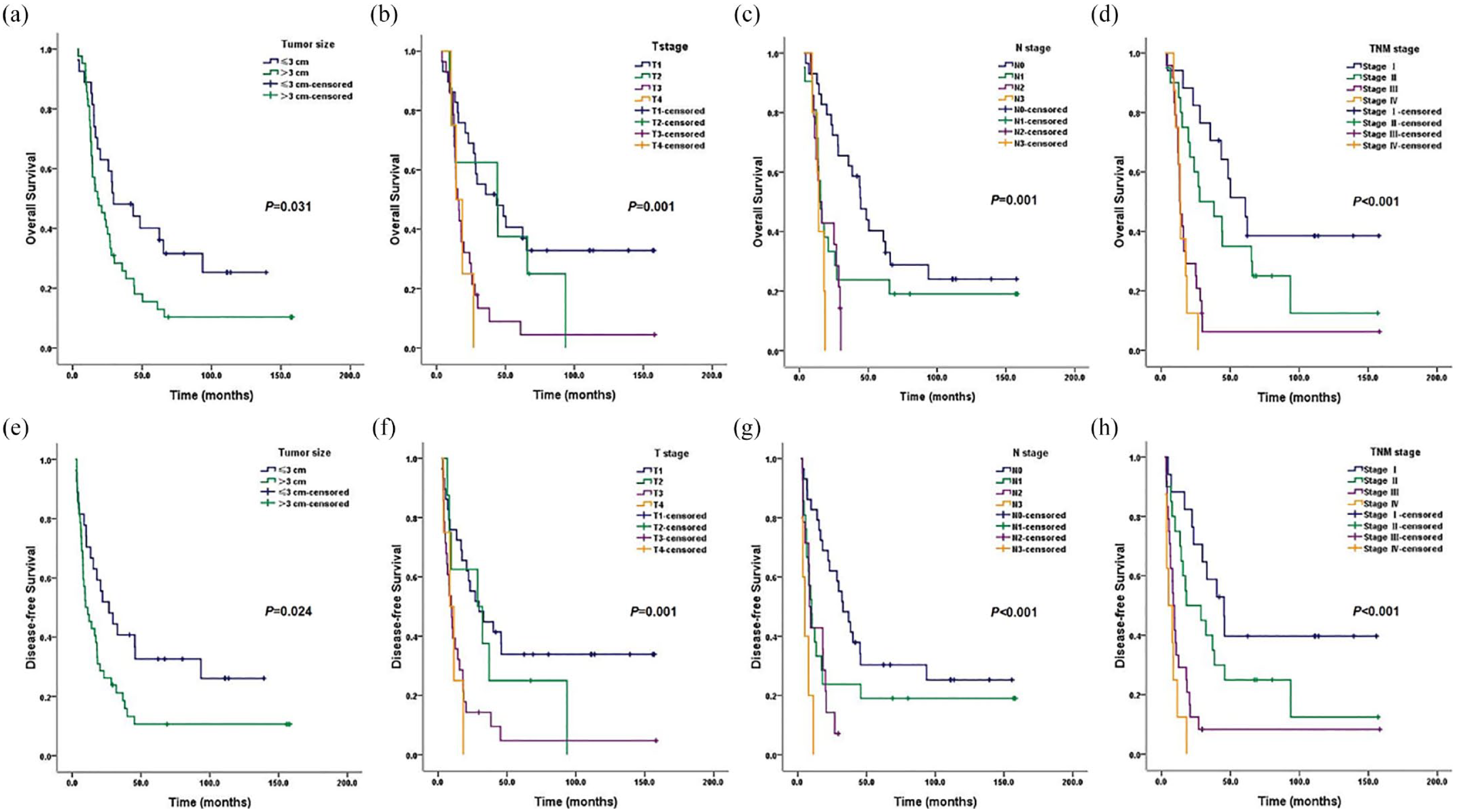
Kaplan–Meier survival curves. (a) OS by tumor size. (b) OS by T stage. (c) OS by N stage. (d) OS by TNM stage. (e) DFS by tumor size. (f) DFS by T stage. (g) DFS by N stage. (h) DFS by TNM stage. The p-value was calculated by the log-rank test.
Postoperative recurrence and metastatic status are shown in Figure 4. Postoperative recurrence or metastasis was documented in 72.5% of patients (n = 50), with over half of these cases (52.0%, n = 26) occurring within the first year following surgery, as detailed in Figure 4(a). The primary sites for these recurrences were the regional lymph nodes, with 74.0% (n = 37) of patients experiencing recurrence in this location (Figure 4(b)).
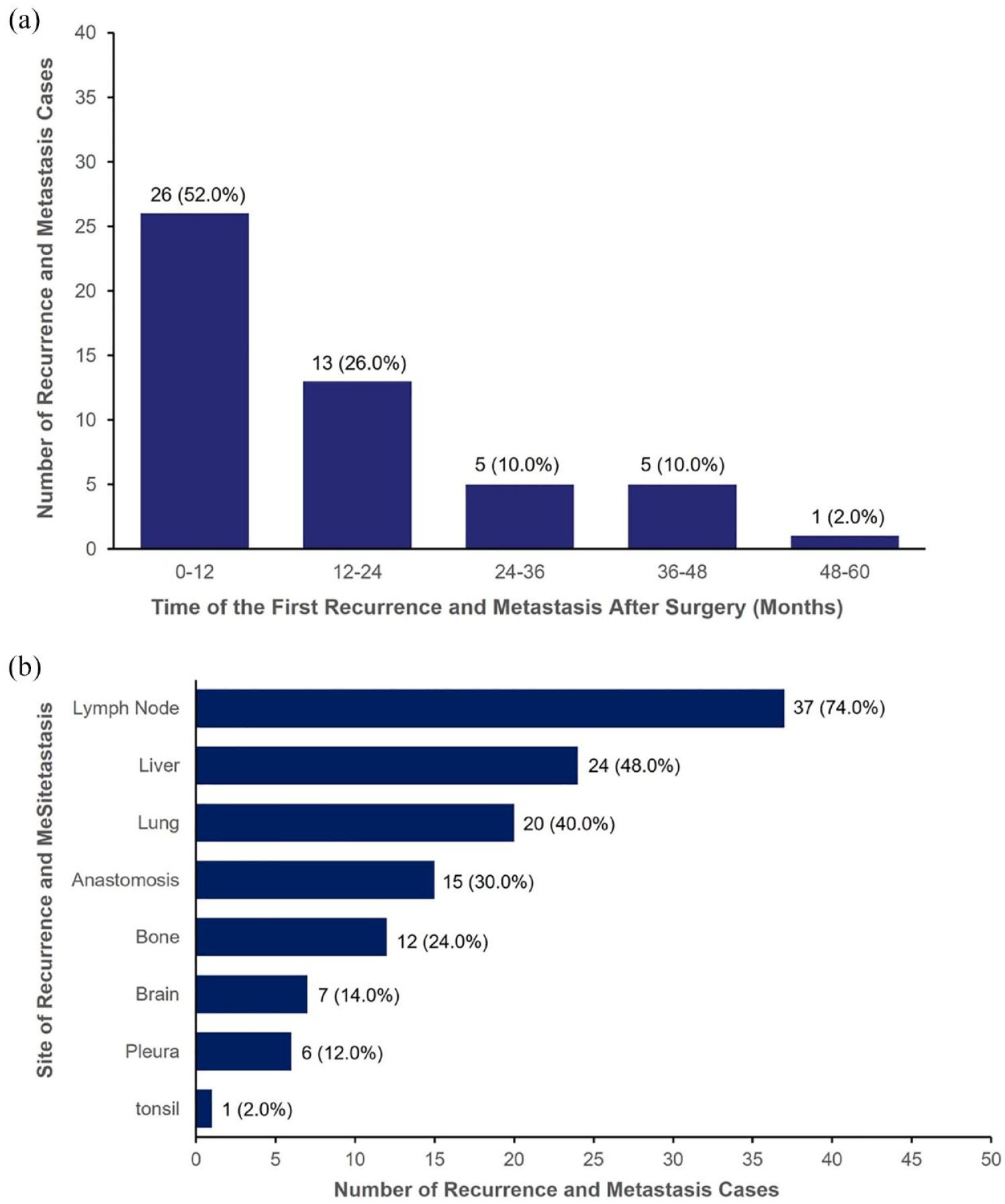
Time to first recurrence or metastasis and site of recurrence or metastasis in the study cohort. (a) Time of the first recurrence and metastasis after surgery and (b) number of cases of recurrence or metastatic sites.
Prognostic factor analysis
Further analysis explored the relationship between various clinicopathological factors and patient prognosis. Univariate analysis confirmed the prognostic importance of tumor size, T-stage, N-stage, and TNM stage (Table 1). Tumor size (median OS and DFS for ⩽3 vs >3 cm: 29.4 vs 17.9 months and 26.9 vs 9.8 months; p = 0.033 and p = 0.026), T-stage (median OS and DFS for T1, T2, T3, and T4: 43.6 vs 44.1 vs 15.2 vs 14.2 months and 29.7 vs 28.6 vs 9.5 vs 8.5 months; p < 0.001 for both), N-stage (median OS and DFS for N0, N1, N2, and N3: 44.4 vs 15.3 vs 14.2 vs 13.9 months and 32.9 vs 10.2 vs 8.6 vs 5.1 months; p < 0.001 for both), and TNM stage (median OS and DFS for stages I, II, III, and IV: 61.0 vs 27.9 vs 13.5 vs 13.9 months and 45.3 vs 18.1 vs 8.6 vs 5.1 months; p < 0.001 for both) were all significant predictors. No other factors such as sex, age, body mass index, smoking history, alcohol consumption, months between onset and treatment, family history of cancer, other chronic diseases, tumor location, macroscopic tumor type, and number of lymph nodes dissected were related to prognosis.
Multivariate Cox analysis identified tumor size, T-stage, N-stage, and TNM stage as prognostic predictors for esophageal MiNEN (Table 2). Before multivariate analysis, we evaluated the collinearity of all included variables, tumor size, T stage, N stage, and TNM stage. The results showed collinearity among the T stage, N stage, and TNM stage in Table S2. Therefore, we analyzed these variables including tumor size, T stage, N stage, and TNM stage, separately. Each factor with covariates such as sex, age, smoking history, alcohol consumption, and family history of cancer was included together by the multivariate analysis model. The results show that early T-stage (p < 0.001), early N-stage (p < 0.001), and early TNM stage (p < 0.001) were significantly associated with improved OS. Similarly, tumor size ⩽3 cm (p = 0.043), early T-stage (p < 0.001), early N-stage (p < 0.001), and early TNM stage (p < 0.001) were related to improved DFS.
Multivariate analysis of survival in 69 patients with MiNEN.

The model adjusted for sex, age, smoking history, alcohol consumption, and family history of cancer.
CI, confidence interval; DFS, disease-free survival; HR, hazard ratio; MiNEN, mixed neuroendocrine–non-neuroendocrine neoplasm; OS, overall survival; TNM, tumor, lymph node, metastasis.
Treatment impact on survival
Subgroup analyses focused on the influence of various treatment strategies on patient survival (Figure 5). This analysis revealed no significant extension in OS or DFS with postoperative adjuvant therapy compared to surgery alone in patients with localized disease (stages I–III; p = 0.420 and p = 0.523, Figure 5(a) and (b)). Likewise, for patients with positive lymph nodes, adjuvant therapy did not confer a survival advantage (p = 0.363 and p = 0.276, Figure 5(c) and (d)). Examination of different surgical approaches—Sweet, Ivor-Lewis, and McKeown—revealed no statistically significant differences in median OS and DFS among these methods (p = 0.611 and p = 0.592, Figure 5(e) and (f)). Furthermore, the primary chemotherapy regimens—paclitaxel plus platinum, etoposide plus platinum, and 5-FU plus platinum—also showed no notable differences in survival outcomes (p = 0.784 and p = 0.601, Figure 5(g) and (h)).

Kaplan–Meier survival curves. (a) Among the subgroup of patients with limited-stage (stage I–III) esophageal MiNEN, OS of patients who received surgery alone (S alone) or surgery plus adjuvant therapy (S + ADT), (b) among the subgroup of patients with limited-stage (stage I–III) esophageal MiNEN, DFS of patients who received surgery alone (S alone) or surgery plus adjuvant therapy (S + ADT), (c) among the subgroup of esophageal MiNEN patients with lymph node metastases, OS of patients who received surgery alone or surgery plus adjuvant therapy, (d) among the subgroup of esophageal MiNEN patients with lymph node metastases, DFS of patients who received surgery alone or surgery plus adjuvant therapy, (e) OS by different surgical procedures, (f) DFS by different surgical procedures, (g) OS by different adjuvant chemotherapy regimens, and (h) DFS by different adjuvant chemotherapy regimens. The p-value was calculated by the log-rank test.
Discussion
The results of this retrospective study suggest that esophageal MiNEN predominantly affects male patients and in China, the pathological types of the disease are mainly composed of a combination of small cell carcinoma and squamous carcinoma. Due to the limitations of pathological biopsy, the preoperative pathological diagnosis accuracy of esophageal MiNEN is low. The median OS and DFS for this group of patients were 24.0 and 16.6 months, respectively. The 1-, 3-, and 5-year OS rates were 84.1%, 34.8%, and 25.3%, respectively. Recurrence or metastasis often occurs within the first year after surgery, primarily in regional lymph nodes. Tumor size, T-stage, N-stage, and TNM stage were significantly associated with the prognosis of esophageal MiNEN. Postoperative adjuvant therapy did not extend survival for patients with localized or lymph node-positive MiNEN. Different surgical approaches or different adjuvant chemotherapy regimens did not significantly affect OS and DFS.
To date, research on esophageal MiNEN has mostly been presented in the form of case reports. Due to the rarity of the disease, conducting prospective studies with large sample sizes is exceptionally challenging. Therefore, we collaborated with two major esophageal cancer diagnosis and treatment centers in China to retrospectively analyze the clinical pathological characteristics and prognostic factors of 69 cases of esophageal MiNEN and conducted a preliminary discussion on treatment methods.
In this group, the proportion of male patients was significantly higher than that of female patients, consistent with the gender distribution of esophageal squamous carcinoma, adenocarcinoma, and small-cell carcinoma,4,14 and which can also be seen in other sites of MiNEN. 15 This phenomenon might be related to the etiology of esophageal cancer, as historically, smoking and drinking are more prevalent among male patients. Over 60% of patients in this study were smokers, and the majority were older than 60 years, consistent with the findings of Frizziero et al. 9 The epidemiological characteristics of esophageal MiNEN seem to be similar to other esophageal malignancies. The esophageal MiNEN reported in some case report studies was predominantly a combination of neuroendocrine carcinoma and adenocarcinoma,16–18 whereas less than 10 cases of MiNEN consisting of neuroendocrine carcinoma and squamous carcinoma have been reported. 19 However, past studies state that esophageal MiNEN is mainly composed of a mixture of neuroendocrine and squamous carcinoma components.20,21 In our study, the combination of small-cell carcinoma and squamous carcinoma was the main pathological type, likely due to squamous carcinoma being prevalent in the esophagus. The combination of neuroendocrine carcinoma and adenocarcinoma is relatively rare, usually occurring at the distal esophagus and gastroesophageal junction, 20 possibly related to Barrett’s metaplasia. 22 Consistent with common esophageal cancer management patterns, patients with esophageal MiNEN typically undergo comprehensive endoscopic biopsy before surgery. However, biopsy samples have limitations in diagnosing MiNEN. Due to the limited amount of sample material, qualitatively and quantitatively differentiating various tumor components is difficult. Previous studies indicate 9 that endoscopic biopsy can only identify one-third of mixed tumors, but the diagnostic accuracy rate for MiNEN is unknown. In our study, the preoperative diagnostic accuracy rate for esophageal MiNEN was only 4.3%, with most patients preoperatively diagnosed merely as “malignant tumors” or squamous carcinoma. Given the limitations of pathological biopsy in diagnosis, we believe that the incidence of MiNEN is underestimated. Perhaps the emergence of liquid biopsy could soon bring hope for the preoperative diagnosis of esophageal MiNEN.
It is reported that the median survival time for esophageal MiNEN is 20–28 months, while for pure neuroendocrine carcinoma, it is only 8–15 months.23,24 Compared to pure neuroendocrine carcinoma, the prognosis for MiNEN is relatively better, possibly due to a lesser neuroendocrine component. 25 Therefore, studies suggest that the aggressive behavior of MiNEN is primarily driven by the neuroendocrine component. 9 In our group, the median survival time for MiNEN was 24 months, with a 5-year survival rate of only 25.3%. Over half of the patients experienced recurrence or metastasis within the first year after surgery. These results also confirm that esophageal MiNEN is a rapidly progressing and aggressive tumor. 10 Similar to other types of esophageal cancer, tumor burden remains an important prognostic factor for MiNEN.4,26 Our study shows that tumor size, T-stage, N-stage, and TNM stage are significantly related to survival. Patients with tumor sizes ⩽3 cm, earlier T-stage, N-stage, or TNM stage typically have better survival and prognosis. It should be noted that the staging of MiNEN in this study used the TNM staging for esophageal cancer, as there is currently no specific staging system for esophageal MiNEN. Therefore, to better understand the prognosis of esophageal MiNEN, it is necessary to explore and establish a specific staging system.
Due to the rarity of esophageal MiNEN, no standard treatment model has been established for the disease. Basically, treatment methods follow those for esophageal cancer. 18 Surgery is considered the preferred treatment for nearly all resectable esophageal MiNEN. 9 Whether to perform adjuvant therapy after surgery for esophageal MiNEN is a current controversial topic. The potential benefits of adjuvant therapy after resection of localized MiNEN are not yet clear, although past retrospective studies have shown its effectiveness. 27 Previous findings indicate that perioperative treatment compared to surgery alone does not improve recurrence-free survival and OS for MiNEN patients. 10 Pommergaard et al. 11 conducted a survival analysis on 142 localized MiNEN patients, showing that perioperative chemotherapy is not associated with improved OS or progression-free survival. In this study, we conducted a subgroup analysis on esophageal MiNEN patients, showing that postoperative adjuvant therapy does not extend OS or DFS for localized patients. This result may be related to the fact that patients receiving adjuvant therapy tended to have a higher TNM stage, and the small sample size attenuated this distributional trend. Given the importance of lymph node status in the prognosis of esophageal cancer, we conducted a subgroup analysis on patients with positive lymph nodes. The results show that postoperative adjuvant therapy does not significantly extend OS or DFS for lymph node-positive patients. Lim et al. 6 believe that the choice of surgical approach for esophageal MiNEN is diverse and should be considered based on factors such as tumor location, lymph node metastasis status, and surgeon preference, with no specific surgical method demonstrating a survival advantage. In this study, we analyzed the three main surgical methods: Sweet, Ivor-Lewis, and McKeown, and found no significant advantage of any method for survival. The European Neuroendocrine Tumor Society recommends chemotherapy regimens for MiNEN based on neuroendocrine carcinoma, 28 but some scholars believe it should be based on the most aggressive part. 29 In this study, the main adjuvant chemotherapy regimens for esophageal MiNEN included paclitaxel plus platinum, etoposide plus platinum, and 5-FU plus platinum, but no specific adjuvant chemotherapy regimen extended OS or DFS. This might be related to the influence of other tumor components, as the chemotherapy regimens in this group were mainly aimed at a specific tumor component. Therefore, it is necessary to explore combination medications for the efficacy of esophageal MiNEN. Excitingly, significant progress has been made in targeted therapy and immunotherapy research for MiNEN.30,31
This study explores the clinical pathological characteristics, prognostic factors, and treatment methods of esophageal MiNEN in a relatively large sample size cohort, but there are some limitations worth acknowledging. First, as a retrospective study, the reliability of the data may not be as high as that of prospective experiments. Due to the rarity of this type of esophageal cancer, implementing multi-center prospective studies may be necessary. Second, the inability to obtain more pathological information from medical records may affect our discussion of prognostic factors, such as the Ki-67 index. Studies have shown that the Ki-67 proliferation index is a major prognostic factor for MiNEN, 32 which is worth further exploration in the future. Furthermore, given the retrospective design of this study, the exclusion of patients who refused follow-up could introduce a potential source of bias, as these individuals may have experienced worse outcomes. While this group represents a small fraction of the overall cohort, it is important to recognize the possible impact on survival and recurrence data. Future studies should aim to include data from these patients, to improve the overall quality of the data presented. Lastly, there were insufficient cases of neoadjuvant therapy in this study, with only one patient undergoing two cycles of paclitaxel plus platinum neoadjuvant chemotherapy before surgery. Therefore, the role of neoadjuvant therapy needs to be analyzed in future prospective studies.
Conclusion
Our retrospective study of esophageal MiNEN elucidates the aggressive nature and challenging prognosis of this rare cancer type. This type of tumor is prevalent in male patients over 60 years of age, and preoperative pathological diagnosis is difficult. In China, the pathologic type of esophageal MiNEN may be dominated by a combination of small-cell carcinoma and squamous carcinoma. Recurrence or metastasis occurs mainly in the first year after surgery and is dominated by regional lymph node metastasis. Tumor size, T-stage, N-stage, and TNM stage are prognostic predictors of esophageal MiNEN. Current adjuvant therapy regimens similar to other esophageal cancers have limited efficacy in improving patient survival, so further exploration of more effective regimens is needed.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241303066 – Supplemental material for Characterizing esophageal mixed neuroendocrine–non-neuroendocrine neoplasms: insights from a retrospective multicenter study of clinical outcomes and prognostic indicators
Supplemental material, sj-docx-1-tam-10.1177_17588359241303066 for Characterizing esophageal mixed neuroendocrine–non-neuroendocrine neoplasms: insights from a retrospective multicenter study of clinical outcomes and prognostic indicators by Qian Hong, Kaiming Wu, Chen Chen, Yan Dang, Qiuju Zhang, Xue Zhang, Liting Wang, Rui Han, Chenguang Zhao, Hang Yi, Fang Li, Renquan Zhang, Juwei Mu and Jiagen Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241303066 – Supplemental material for Characterizing esophageal mixed neuroendocrine–non-neuroendocrine neoplasms: insights from a retrospective multicenter study of clinical outcomes and prognostic indicators
Supplemental material, sj-docx-2-tam-10.1177_17588359241303066 for Characterizing esophageal mixed neuroendocrine–non-neuroendocrine neoplasms: insights from a retrospective multicenter study of clinical outcomes and prognostic indicators by Qian Hong, Kaiming Wu, Chen Chen, Yan Dang, Qiuju Zhang, Xue Zhang, Liting Wang, Rui Han, Chenguang Zhao, Hang Yi, Fang Li, Renquan Zhang, Juwei Mu and Jiagen Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
