Abstract
Objective
To evaluate the feasibility, safety, and efficacy of high-intensity focused ultrasound (HIFU) and microwave ablation (MWA) for the treatment of small liver metastatic tumors.
Methods
Fifty-eight patients with small liver metastatic tumors who underwent HIFU (n = 28) or MWA (n = 30) at Suining Central Hospital between January 2016 and December 2021 were retrospectively evaluated. Demographic and clinical characteristics were compared between the two groups.
Results
Operation times were longer and hospitalization costs were lower in the HIFU group than in the MWA group. Postoperative hospitalization times, tumor ablation rates, and clinical response and control rates did not differ significantly between the two groups 1 month after surgery. Rates of postoperative complications such as fever, liver dysfunction, injury, pain, and biliary leakage did not differ between the two groups. The 1- and 3-year cumulative survival rates were 96.4% and 52.4%, respectively, after HIFU and 93.3% and 51.4%, respectively, after MWA, which did not represent significant differences.
Conclusions
HIFU is a safe and feasible method of treating small liver metastatic tumors. Compared with MWA, HIFU was associated with lower hospitalization costs, reduced trauma, and fewer postoperative complications, making it a promising new local ablative treatment option for liver metastatic tumors.
Keywords
Introduction
Liver metastatic tumors are a common type liver cancer, the incidence of which has been markedly increasing. 1 Although surgical resection is the treatment of choice, only 10% to 15% of patients who present with liver metastatic tumors are suitable for surgical resection. Local ablation methods have achieved good therapeutic effects in patients with both primary and metastatic liver tumors, especially those with liver tumors ≤3 cm in diameter. 2 Although microwave ablation (MWA) is one of the most widely used local ablation methods, it has several drawbacks, including risks of severe bleeding and biliary leakage due to liver injury, burns, and tumor cell metastasis during passage of the needle. High intensity focused ultrasound (HIFU) is a non-invasive tumor ablation technique that can also be used to treat liver tumors. This study compared the clinical efficacy and safety of HIFU and MWA for the treatment of small liver metastatic tumors, providing evidence for the application of HIFU in the treatment of liver metastatic tumors.
Patients and methods
This retrospective study included patients with small liver metastatic tumors, as determined by imaging and/or pathological examination, who underwent HIFU or MWA at Suining Central Hospital between January 2016 and December 2021. The choice of ablation method was based on patient preferences and economic situations. Patients were included if they had undergone radical resection of the primary malignant tumor, with the tumor confirmed as malignant by postoperative pathology; if liver metastatic tumor was confirmed by imaging or pathology after removal of the primary lesion, with no extrahepatic lesions being present, and no other treatment received; and if they had Child–Pugh grade A or B liver function. Patients with a single lesion were included if the diameter was ≤5 cm, whereas patients with multiple lesions were included if they had ≤3 lesions with a maximum diameter of ≤3 cm. Other inclusion criteria included unwillingness to undergo surgery or other contraindications to surgery and expected survival time >3 months. Patients were excluded if they had multiple metastases throughout the body, primary hepatic malignancy, liver metastatic tumor accompanied by portal vein carcinoma thrombus, a coagulation dysfunction that was difficult to correct, or serious heart, lung, or renal dysfunction. The study protocol was approved by the Ethics Committee of Suining Central Hospital (approval number: LLSLH20210051). Written informed consent was obtained from all patients or their relatives for publication of this research and any accompanying images. All patient details have been de-identified. The reporting of this study conforms to STROBE guidelines. 3
For patients who underwent HIFU, general anesthesia was induced, and the patients were placed in the right or prone position, depending on the location of the tumor. Tumor location, number, size, and shape were assessed by ultrasound examination (Figure 1), and lesions were irradiated by applying point and line scans for 1 to 3 s at 2- to 6-s intervals. The initial ablation power was 300 W, increasing by 50 W until the gray level of the tumor tissue changed (Figure 2), up to a maximum power of 400 W. HIFU treatment was deemed complete when the entire tumor was covered by a gray mass, when the overall echo of the tumor had significantly increased, or when the ablation range of the tumor tissue at its outer edge was >1 cm.

The equipment used for high-intensity focused ultrasound (HIFU): JC Focused Ultrasound Tumor Treatment System (Chongqing Hifu Medical Technology Co., LTD., Chongqing, China). (a) The HIFU tumor ablation system; (b) real-time monitoring during HIFU and (c) intraoperative treatment.
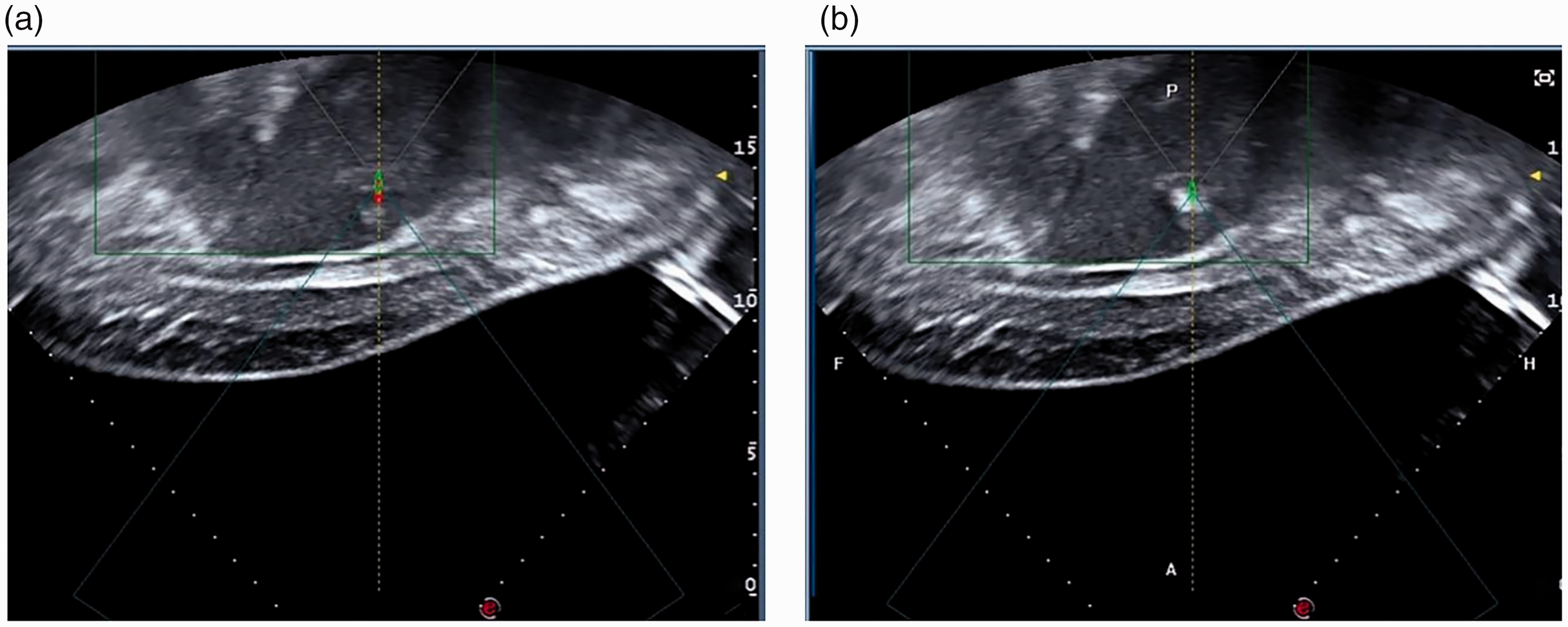
Ultrasound images of tumors. (a) Before ablation, tumors were slightly hyperechoic, with a hypoechoic halo seen around them and (b) There was strong echo in the target area during ablation.
For patients who underwent MWA, following induction of local or intravenous anesthesia, tumor location and the optimal puncture site(s) were determined by preoperative contrast-enhanced ultrasound. A microwave puncture needle (Figure 3) was then slowly inserted into the tumor through the skin, such that the emission site of the needle covered the bottom of the tumor. To treat patients with a single tumor ≤3 cm in diameter, a single microwave ablation needle was inserted into the center of the tumor; to treat patients with a single tumor of between 3 and 5 cm in diameter, the needle was used repeatedly. To treat patients with two or three tumors, two or three microwave ablation needles were inserted, one per tumor. Depending on the specific situation of each tumor, the needle could be inserted from different points as appropriate and with an ablation power of between 50 and 70 W applied for 5 to 8 minutes at each point. This was performed until tissue coagulation at the outer edge of each tumor was >1 cm. The needle path was routinely ablated when the needle was withdrawn, and the effect of ablation was assessed by contrast-enhanced ultrasound.

The equipment used for microwave ablation (MWA): ECO-100A1 microwave therapeutic instrument, microwave ablation needle (Nanjing Yigao Microwave System Engineering Co., LTD., Nanjing, China). (a) The microwave therapeutic apparatus; (b) a microwave ablation needle and (c) intraoperative MWA treatment.
Factors recorded for each patient included demographic and clinical characteristics before and after ablation, cost of hospitalization, operation time, postoperative hospital stay, tumor ablation rate, clinical response rate, clinical control rate, changes in tumor markers, and the incidence of intra- and post-operative complications.
All patients were followed-up at 1 month after ablation by HIFU or MWA with upper abdominal computed tomography (CT) or magnetic resonance imaging (MRI) (Figure 4) and measurements of serum tumor markers such as carcinoembryonic antigen (CEA) and alpha fetoprotein (AFP), as well as markers of liver and kidney function. Response to treatment was evaluated by the Modified Response Evaluation Criteria in Solid Tumors (mRECIST) criteria. Tumor ablation rate was calculated as the number of completely ablated lesions divided by the total number of lesions; clinical response rate was defined as the number of patients who achieved complete response (CR) and partial response (PR) divided by the total number of patients; and clinical control rate was defined as the number of patients who achieved CR, PR, and stable disease (SD) divided by the total number of patients. Survival rates of the two groups were also statistically analyzed.

Computed tomography images after high-intensity focused ultrasound (HIFU). Note: The patient was a 64-year-old woman with liver metastasis (approximately 2 cm) after lung cancer surgery. (a1, a2) Preoperative enhancement around the metastases in the arteriovenous phase; (b1, b2) no enhancement was observed in the metastases at 1 month after HIFU; (c1, c2) no enhancement was observed in the metastases 3 months after HIFU.
Aspartate transaminase (AST), alanine transaminase (ALT), and serum creatinine concentrations were measured before and after ablation. Postoperative pain in the surgical area was assessed using a 0 to 10 visual analogue scale, with 0, 1 to 3, 4 to 6, and 7 to 10 indicating no, mild, moderate, and severe pain, respectively. Other parameters evaluated included fever, liver dysfunction (transaminase >40 U/L within 24 hours after surgery), biliary leakage, gastrointestinal perforation, and other complications. Complications were graded by the complication classification scale of the International Association of Radiotherapy (ISR), with Grade A indicating that no treatment was required, with no adverse results; Grade B indicating a requirement for some treatment, with no adverse consequences, including overnight observation; Grade C indicating a requirement for medical treatment, with hospital stay of <48 hours; Grade D indicating a requirement for extensive treatment, improved nursing level, and extended hospital stay >48 hours; Grade E indicating permanent sequelae; and Grade F indicating death.
Normally distributed continuous data are expressed as mean ± standard deviation and were compared by the Student’s t test. Non-normally distributed continuous data are expressed as median (interquartile range) and were compared by the rank sum tests. Categorical data are expressed as number (%) and were compared by the χ2 test or Fisher's exact test, as appropriate. All statistical analyses were performed using SPSS 26.0 software (IBM, Armonk, NY, USA), with P < 0.05 defined as statistically significant.
Results
Among the 58 patients who underwent ablation during the study period, 28 underwent HIFU and 30 underwent MWA. The two groups were well matched, with no statistically significant differences between them (Table 1). The average hospitalization cost was significantly lower and operation times were significantly longer in the HIFU group compared with in the MWA group (Table 2). However, postoperative hospitalization times did not differ significantly between the two groups.
Baseline demographic and clinical characteristics of patients in the high-intensity focused ultrasound (HIFU) and microwave ablation (MWA) groups.

Note: *Primary tumors were located in the gastrointestinal system in 49 patients (26 with rectal cancer, 11 with colon cancer, 5 with gastric cancer, 4 with esophageal cancer, and 3 with pancreatic cancer), and at other sites in nine patients (three each with ovarian and breast cancer, two with lung cancer, and one with nasopharyngeal cancer).
BMI, body mass index; SD, standard deviation; ALT, alanine transaminase; AST, aspartate transaminase; CEA, carcinoembryonic antigen; AFP, alpha fetoprotein.
Related indices during hospitalization in the high-intensity focused ultrasound (HIFU) and microwave ablation (MWA) groups.

At 1 month after surgery, there were no significant difference in tumor ablation rates between the two groups (Table 3). Postoperative clinical response and clinical control rates did not differ significantly between patients who underwent HIFU and MWA (Table 4). Serum CEA and AFP concentrations before treatment did not differ significantly in the two groups. After treatment, concentrations of both were reduced, with CEA but not AFP being significantly decreased (Table 5).
Tumor ablation efficacy in the high-intensity focused ultrasound (HIFU) and microwave ablation (MWA) groups.
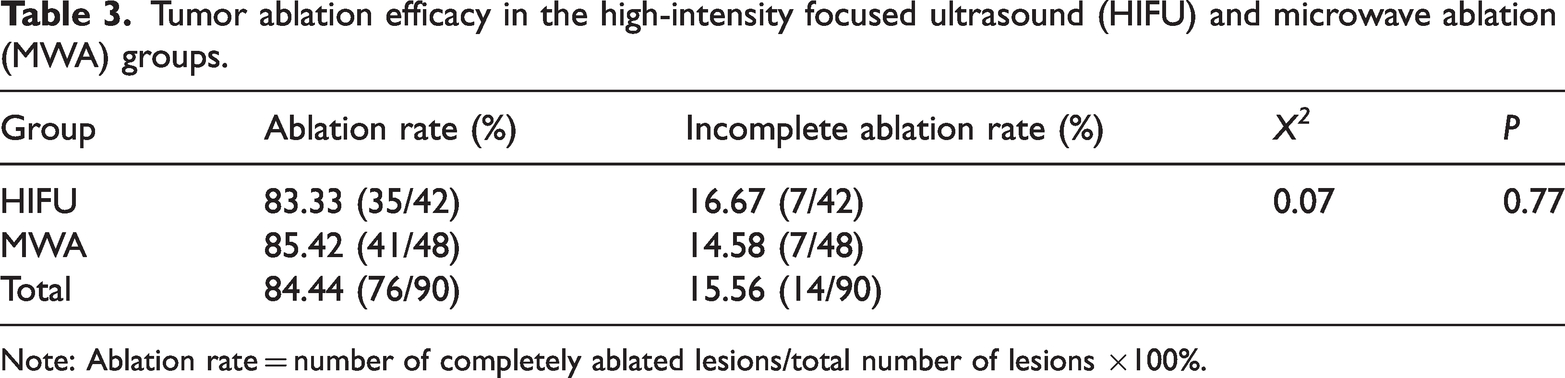
Note: Ablation rate = number of completely ablated lesions/total number of lesions ×100%.
Comparative clinical efficacy of high-intensity focused ultrasound (HIFU) and microwave ablation (MWA).

CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease.
Effects of high-intensity focused ultrasound (HIFU) and microwave ablation (MWA) on serum tumor markers.

Note: *P < 0.05 compared with preoperative concentration, ^P > 0.05 compared between groups.
CEA, carcinoembryonic antigen; AFP, alpha fetoprotein.
There were 15 deaths in the HIFU group and 18 in the MWA group. The 1- and 3-year cumulative survival rates were 96.4% and 52.4%, respectively, after HIFU and 93.3% and 51.4%, respectively, for MWA; thus, there were no significant differences between the two groups (Figure 5).

Cumulative survival curves for patients treated with high-intensity focused ultrasound (HIFU) and microwave ablation (MWA).
Fever was observed in 15 (54%) patients in the HIFU group and 20 (67%) in the MWA group, although fever never exceeded 38.5°C (Table 6). Fever was relieved spontaneously or by physical cooling. Pain in the operative area was experienced by 13 (46%) patients in the HIFU group, including 9 with mild and 4 with moderate pain, and by 19 (63%) patients in the MWA group, including 13 with mild and 6 with moderate pain. Pain was generally relieved by treatment with analgesics. Liver dysfunction was observed in 17 (61%) and 22 (71%) patients, in the HIFU and MWA groups, respectively, with mean ALT concentrations of 53.71 ± 56.84 U/L and 143.18 ± 64.06 U/L, respectively, and mean AST concentrations of 146.63 ± 61.12 U/L and 157.82 ± 74.54 U/L, respectively (Table 7). Liver dysfunction was reduced by liver protective treatment. Rates of postoperative biliary leakage were 0% (0/28) and 7% (2/30) in the HIFU and MWA groups, respectively, with biliary leakage treated by insertion of an abdominal drainage tube. None of the above differences were statistically significant.
Postoperative complications in the high-intensity focused ultrasound (HIFU) and microwave ablation (MWA) groups.
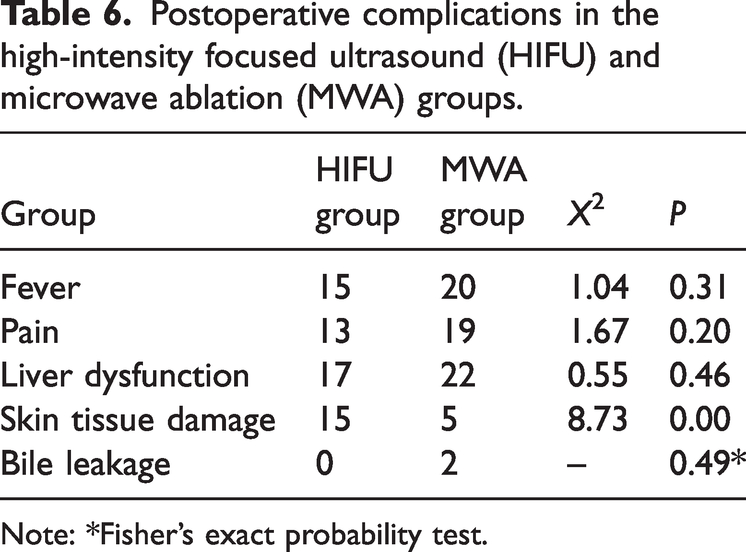
Note: *Fisher's exact probability test.
Postoperative liver dysfunction in the high-intensity focused ultrasound (HIFU) and microwave ablation (MWA) groups.

Note: *P > 0.05 compared with the HIFU group.
ALT, alanine transaminase; AST, aspartate transaminase.
Postoperative skin tissue injury was significantly more common the HIFU group than in the MWA group (P < 0.05). Fifteen patients (54%) in the HIFU group experienced postoperative skin tissue injury, including 12 with skin swelling, 2 with first degree erythema burns, and 1 with a second degree blister burn (Table 7). In comparison, five patients (17%) in the MWA group experienced postoperative skin tissue injury, including three with skin swelling and two with first degree erythema burns. Skin tissue injuries were cured by ice or symptomatic treatment.
Discussion
Although surgical resection of liver metastatic tumors is superior to local ablation, only between 10% and 15% of patients meet the clinical conditions for surgery.4,5 Surgery is contraindicated in most patients due to poor physical condition, adhesion after abdominal surgery, multiple intrahepatic metastases or metastases in certain locations, small postoperative residual liver volume, and metastases to lymph nodes and other organs. 6 Local ablation is more frequently performed and has become an important part of the comprehensive treatment of liver tumors. Guidelines formatted by the European Association for the Study of the Liver (EASL) 7 recommend local ablation for patients with small liver tumors, consisting of a single tumor ≤2 cm in diameter or ≤3 tumors ≤3 cm in maximum diameter. Similarly, guidelines of the American Association for the Study of Liver Diseases (AASLD) 8 recommend local ablation for patients with liver tumors <5 cm in diameter, and Chinese guidelines 9 recommend local ablation for patients with a single tumor ≤5 cm in diameter or ≤3 tumors ≤3 cm in maximum diameter without invasion of adjacent organs, bile ducts, blood vessels, or distant metastases.
Local ablation has been shown to significantly increase survival time and enhance the quality of life for patients with liver metastatic tumors, with fewer complications and shorter hospital stays compared with surgical treatment. 10 For example, HIFU ablation of 47 patients with liver tumors <3 cm in diameter resulted in a tumor ablation rate of 87.2%. 11 And in a retrospective study, Zhang et al. 12 compared the therapeutic efficacy of percutaneous RFA to MWA in liver tumor ≤ 5 cm in diameter by evaluating 155 patients. The tumor ablation rate was 83.4 % for RFA compared with 86.7 % for MWA. This study yielded similar results, with tumor ablation rates 1 month after HIFU and MWA of 83% and 85%, respectively, clinical response rates of 89% and 90%, respectively, and clinical control rates of 96% and 90%, respectively. The 1- and 3-year cumulative survival rates were 96.4% and 52.4% after HIFU, respectively, and 93.3% and 51.4%, respectively, for MWA. These results indicate that both local ablation techniques can effectively ablate small liver metastatic tumors and prolong overall survival.
Although mean hospitalization costs were significantly lower for HIFU than for MWA (P < 0.05), HIFU operation times were significantly longer (P < 0.05) and there were no significant differences in postoperative hospitalization stays. HIFU was associated with relatively fewer postoperative complications and did not require disposable high-value consumables such as disposable microwave ablation needles, which reduced costs. Because HIFU treatment is relatively longer, patient tolerance should be fully evaluated before surgery.
CEA is a surface protein antigen secreted by tumor cells that is significantly increased in most gastrointestinal malignancies. CEA has important value in tumor diagnostics and evaluations of treatment efficacy, postoperative recurrence, and metastasis.13,14 AFP is a marker of hepatocellular carcinoma that is produced in large quantities when hepatocytes are damaged or cancerous, making it important for the screening, diagnosis, and prognosis of primary hepatocellular carcinoma. 15 CEA can also be used to determine postoperative efficacy and prognosis in patients with liver metastatic tumors, and in combination with AFP, can be used to differentiate between patients with primary and metastatic liver tumors.16,17 Levels of CEA and AFP in patients with liver metastatic tumor are dependent on the type of primary tumor. CEA is mainly highly expressed in the sera of patients with primary gastrointestinal cancers, whereas AFP is highly expressed in the sera of patients with primary liver cancers.18,19 In this study, the preoperative CEA positivity rates in the HIFU and MWA groups were 57% and 67%, respectively, and the AFP positivity rates were 25% and 27%, respectively, differences that were not statistically significant. Similar to previous studies,18,19 the results of this study indicate that CEA levels are generally higher than normal in patients with liver metastases of primary gastrointestinal cancers, with or without AFP elevation. CEA concentrations were significantly reduced in both groups after ablation, whereas AFP concentrations were reduced, but not significantly. Following HIFU and MWA, CEA concentrations decreased by 81% and 70%, respectively, whereas AFP levels decreased by 57% and 75%, respectively. The reductions in both serum CEA and AFP concentrations after HIFU and MWA indicate that both procedures can result in good patient prognosis.
MWA is an invasive procedure with postoperative complications that include fever, intraoperative pain, liver dysfunction, and skin injury. Compared with radiofrequency ablation, MWA has several advantages including high ablation temperature, shorter operation times, and reduced heat precipitation effects and is now widely used for the ablation of liver tumors. 20 However, MWA also has several drawbacks. Liver damage during the puncture procedure can cause severe bleeding, bile leakage, and burns. Moreover, each needle passage carries a risk of seeding tumor cells, enhancing the likelihood of metastasis. Additionally, MWA is a high-risk treatment for patients with liver metastatic tumors who have a small amount of ascites, mild coagulation dysfunction, liver cirrhosis, and other contraindications to invasive procedures. Because of its noninvasive nature, HIFU can be a more appropriate choice for such patients. Frequent complications after HIFU include fever, pain at the operative site, skin and soft tissue burns at the operative site, and edema.21,22
All 58 patients in this study completed local ablation (either HIFU or MWA) with no statistically significant between-group differences in complications such as fever, intraoperative pain, and liver dysfunction. Two patients in the MWA group experienced biliary leakage, which was related to liver puncture, and was improved after placing an abdominal drainage tube. The rate of skin tissue injury after ablation was significantly higher in the HIFU group than in the MWA group; these injuries consisted mostly of skin tissue swelling, with no patients experiencing greater than second degree burns. The higher rate of skin tissue injury in the HIFU group may be due to the greater length of the procedure.
This study had several limitations, including its retrospective design and its inclusion of patients from a single-center. Moreover, the number of patients was relatively small and patients were followed-up for only 1 month, with only survival time collected and analyzed after treatment. Multicenter, prospective studies with large numbers of patients and longer follow-up times are needed to evaluate the long-term efficacy of HIFU in patients with liver metastatic tumors.
Conclusions
HIFU is a safe and feasible method of treating small liver metastatic tumors, as it can completely ablate tumor cells. Compared with MWA, HIFU was associated with lower hospitalization costs, less trauma, and no serious postoperative complications. Especially for tumors adjacent to large blood vessels, gallbladder, the diaphragm, and other difficult locations in the liver, HIFU is more advantageous than MWA. Therefore, HIFU is expected to be a new option for the local ablation of liver metastatic tumors.
Research Data
Research Data for Comparison of high-intensity focused ultrasound and microwave ablation for the treatment of small liver metastatic tumors
Research Data for Comparison of high-intensity focused ultrasound and microwave ablation for the treatment of small liver metastatic tumors by Junjie Wang, Lili Wan, Guohua Huang, Cheng Chen and Yakun Wu in Journal of International Medical Research
Footnotes
Author contributions
Cheng Chen and Yakun Wu designed the study and revised the manuscript; Guohua Huang was responsible for data collection; Junjie Wang and Lili Wan analyzed the data and wrote the first draft of this manuscript.
Data availability statement
The datasets generated during and/or analyzed during this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science & Technology Department of Sichuan Province (grant no. 2019YJ0704). The funder had no influence on the design of this trial or in the writing of the manuscript and presenting the study outcomes, and had no role in the collection, analysis, and interpretation of data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
