Abstract
Objective
To investigate the prevalence of mutations in domain V of
Methods
Domain V of 23S rRNA was sequenced in MP strains collected from children diagnosed with MPP in Nanjing. Clinical and laboratory data were obtained.
Results
Among the 276 MP strains, 255 (92.39%) harbored mutations, primarily A2063G in domain V of MP 23S rRNA. When children were stratified according to the presence or absence of mutations, no significant differences were found in sex, age, the MP DNA load at enrollment, lymphocyte counts, pulmonary complications, immunomodulator levels, fever duration, the duration of fever after macrolide therapy, and hospital stay. The prevalence of refractory MPP in the two groups was similar. Children with refractory MPP exhibited higher MP DNA loads than those with non-refractory MPP.
Conclusions
Despite the high prevalence of the A2063G mutation in domain V of MP 23S rRNA, mutations were not associated with the clinical characteristics of MPP. The MP DNA load significantly differed between refractory and non-refractory MPP.
Introduction
Because MP is intrinsically resistant to β-lactams and other antibiotics that target the cell wall, macrolides, fluoroquinolones, and tetracyclines are used to treat MP infection. Macrolides are the first-line drugs for children to avoid potential age-related side effects associated with other therapies (fluoroquinolones can negatively influence the growth of bones and articular cartilage, and tetracyclines may cause enamel hypoplasia and exert adverse effects on and bones).5,6 Unfortunately, in recent years, the frequency of refractory MP pneumonia (MPP) that does not respond to macrolides has increased steadily around the world.2,3
Drug inactivation, active efflux, and modification of the target sites by methylation or mutation are the main mechanisms underlying macrolide resistance. Macrolide resistance involves mutations in domains II and V of MP 23S rRNA and the MP ribosomal proteins L4 and L22. 9 However, no solid evidence supporting the participation of such in macrolide resistance in MP strains has been reported.7,8
Indeed, genetic mutations in domain V of MP 23S rRNA are believed to contribute to macrolide resistance,9–11 including point mutations of A2063, A2064, A2067, and C2617. Specifically, the A-to-G transition at position 2063 (A2063G) and A to G transition at position 2064 (A2064G) result in strong macrolide resistance,12,14 whereas mutations of A2067 and C2617 confer weaker resistance.2,6,14 The prevalence of macrolide-resistant MPP has increased rapidly since 2000, reaching 90% recently in Asia.6,15 The excessive use of macrolides might contribute to the emergence of mutations. The frequency of refractory MPP displaying no clinical or radiological response to macrolides is also increasing in China, and these cases may progress to severe or even fatal pneumonia.
Although studies have described the molecular epidemiology of MP infection,16,17 to our knowledge, few large-scale clinical studies have been conducted to characterize the clinical significance of MP mutations and explore the clinical relevance of mutant MP strains. Therefore, we sequenced 23S rRNA domain V in 276 clinically acquired MP strains and performed data analysis. This study explored the genotypes of clinical MP strains, determined the prevalence of mutations, and evaluated the correlation between domain V mutations and the clinical characteristics of MPP, especially refractory MPP.
Materials and methods
Study design and population
In this prospective observational trial, pediatric patients diagnosed with MPP from March 1, 2014 to March 1, 2016 in the Department of Respiratory of Children's Hospital of Nanjing Medical University were enrolled. MPP was diagnosed according to symptoms at admission, including fever, cough, sputum production, abnormal breathing sounds, and radiographic pulmonary abnormalities when MP infection was present as an etiology. Acute MP infection in this study was confirmed by both PCR and serological positivity. Serological positivity was defined as an IgM-specific anti-MP antibody titer of at least 1:160 or an at least 4-fold higher IgM-specific anti-MP antibody titer in the convalescent phase than in the acute phage. Nasopharyngeal swabs were collected from patients with lower respiratory tract infection on admission. Patients with immunosuppressive illness, chronic lung disease, or other systematic diseases and those who received macrolides for more than 3 days before admission or during convalescence were excluded. The flowchart of patient enrollment is presented in Figure 1.
Flowchart illustrating the inclusion process of the study population.
Clinical samples and DNA preparation
MP serology
IgM-specific anti-MP antibody was detected in 2 mL of acute-phase (on admission) and convalescent-phase (on discharge) patient serum using the Diagnostic Kit for Measurement of Antibodies to Mycoplasma pneumoniae (Passive Particle Agglutination) (SERODIA-MYCO II, Fujirebio, Inc. Malvern, PA, USA) in accordance with the manufacturer’s instructions and as described previously. 18
Detection of seven common viruses
Nasopharyngeal swabs were used to test for respiratory syncytial virus, influenza A virus, influenza B virus, adenovirus, and parainfluenza virus 1–3 using the D3 Ultra DFA Respiratory Virus Screening & ID Kit (Immunofluorescence Assay) (Diagnostic Hybrids, Inc. Athens, OH, USA) in accordance with the manufacturer’s instructions.
Fluorescence quantitative amplification of MP DNA
The MP DNA load was assayed in nasopharyngeal swab samples using an MP DNA PCR kit (ACON Biotech Co., Ltd., Hangzhou, Zhejiang, China). Specific primers and TaqMan probes were designed according to the conserved gene fragment of MP. In brief, according to the manufacturer’s instructions, nasopharyngeal swab samples were shaken for 30 s and centrifuged at 15,000 ×
DNA sequences of MP 23S rRNA domain V
All nasopharyngeal swab samples were mixed with four volumes of 0.9% normal saline, incubated overnight at 4°C, and centrifuged at 20,375 ×
Treatment, clinical data, and diagnosis of refractory MPP
According to the Expert Consensus on Diagnosis and Treatment of Mycoplasma Pneumoniae Pneumonia in Children (in Chinese), 21 macrolide therapy was the first-line treatment for MPP. The macrolide therapeutic strategies included intravenous 15 mg/kg erythromycin every 12 hours for 10 to 14 days or oral 10 mg/kg azithromycin once daily for 3 days, which could be repeated after 4 days. The course of macrolide therapy could be extended in severe cases. The second-line treatments of MPP included systemic glucocorticoid steroids and intravenous immunoglobulin. Systemic glucocorticoid steroids (intravenous 1–2 mg/kg methylprednisolone once daily for 3–5 days) and/or intravenous immunoglobulin (1 g/kg once daily for 1–2 days) were considered in patients with acute episodes or severe MPP. According to the clinical manifestations, 21 drugs including expectorants, cough medicine, bronchodilators, and inhaled corticosteroids were selectively used. Interventional therapeutic fiber bronchoscopy, thoracocentesis, and oxygen therapy were used as needed.
Clinical data, including sex, age, fever, length of hospitalization, radiological findings (atelectasis or hydrothorax), immune modulator use (systemic glucocorticoid steroids and/or intravenous immunoglobulin), MP DNA load, and laboratory data, were collected from medical records.
Refractory MPP was defined as prolonged high-grade fever (>38.5°C), worsening cough, and/or increasing infiltrates on chest radiographs after the administration of macrolide antibiotics for 7 days or longer.21,22
Ethics approval and informed consent
The study protocol conformed to the Ethical Review for Biomedical Research Involving Humans by the National Health and Family Planning Commission of the People’s Republic of China. In addition, the study protocol was approved by the Ethics Committee of the Children’s Hospital of Nanjing Medical University (approval numbers: ETYY2013030 and 201506002-1). Informed consent was obtained from at least one guardian of each patient before enrollment.
Statistical analyses
Statistical analysis was performed using IBM SPSS statistics 20.0 (IBM Corp., Armonk, NY, USA). For continuous variables, Student’s
Results
Patient demographics, clinical signs, symptoms, and treatment
In total, 276 patients, including 137 boys (49.64%) and 139 girls (50.36%), were enrolled during the study period. The mean patient age was 4.93 years. All patients were treated with macrolide therapy, and 63 patients (22.83%) received immunomodulators (63 received systemic glucocorticoid steroids and 4 received intravenous immunoglobulin). Pulmonary complications were observed in 57 patients (20.65%), including atelectasis, pleurisy, and both atelectasis and pleural effusion in 31, 17, and 9 patients, respectively. Then, 37 patients (13.4%) underwent fiber bronchoscopy for atelectasis, 6 (2.17%) received thoracentesis for massive pleural effusion, and 32 (11.59%) received oxygen therapy. No patient was transferred to the intensive care unit or treated with mechanical ventilation. Thirty-eight patients (13.77%) with wheezing were treated with inhaled corticosteroids and bronchodilators. Sixty-two patients (22.46%) were treated with macrolides combined with cephalosporins on admission. Subsequently, within 4 days, cephalosporins were discontinued immediately after the absence of other etiological infection was confirmed. The duration of cephalosporins use in these 62 patients ranged from 1 to 4 days (mean, 2.06 days).
Domain V of 23S rRNA in MP strains
Among the 276 MP strains, 255 (92.39%) were found to harbor the A2063G mutation compared with the sequence of the M129 strain, whereas no mutations were detected in the remaining 21 strains (7.61%). One A2063G mutant MP strain contained an additional G insertion between positions 2586 and 2587. Another A2063G mutant strain had three other mutations, including T insertion between positions 2589 and 2590, G-to-C transition at position 2601, and T insertion between positions 2612 and 2613 (Table 1). Mutations at positions 2064, 2067, and 2617 were not detected in any clinical samples.
Mutations in domain V of

Characteristics of clinical MP strains
Pediatric patients were stratified into two groups based on the presence or absence of mutations in domain V of MP 23S rRNA. The mean age of the 255 patients (124 boys and 131 girls) with mutant MP strains was 4.84 years, compared with 6.07 years for patients without mutant strains. There were no significant between-group differences in the age and sex distribution, as well as in the frequency of pulmonary complications, treatment performed (fiber bronchoscopy, thoracentesis, oxygen therapy, and immunomodulator treatment), MP DNA load at enrollment, duration of fever, duration of fever after macrolide therapy, and length of hospitalization. Although the lymphocyte count was similar between the groups, leukocyte and neutrophil counts were significantly higher in patients with mutant strains (both
Characteristics of clinical MP strains.
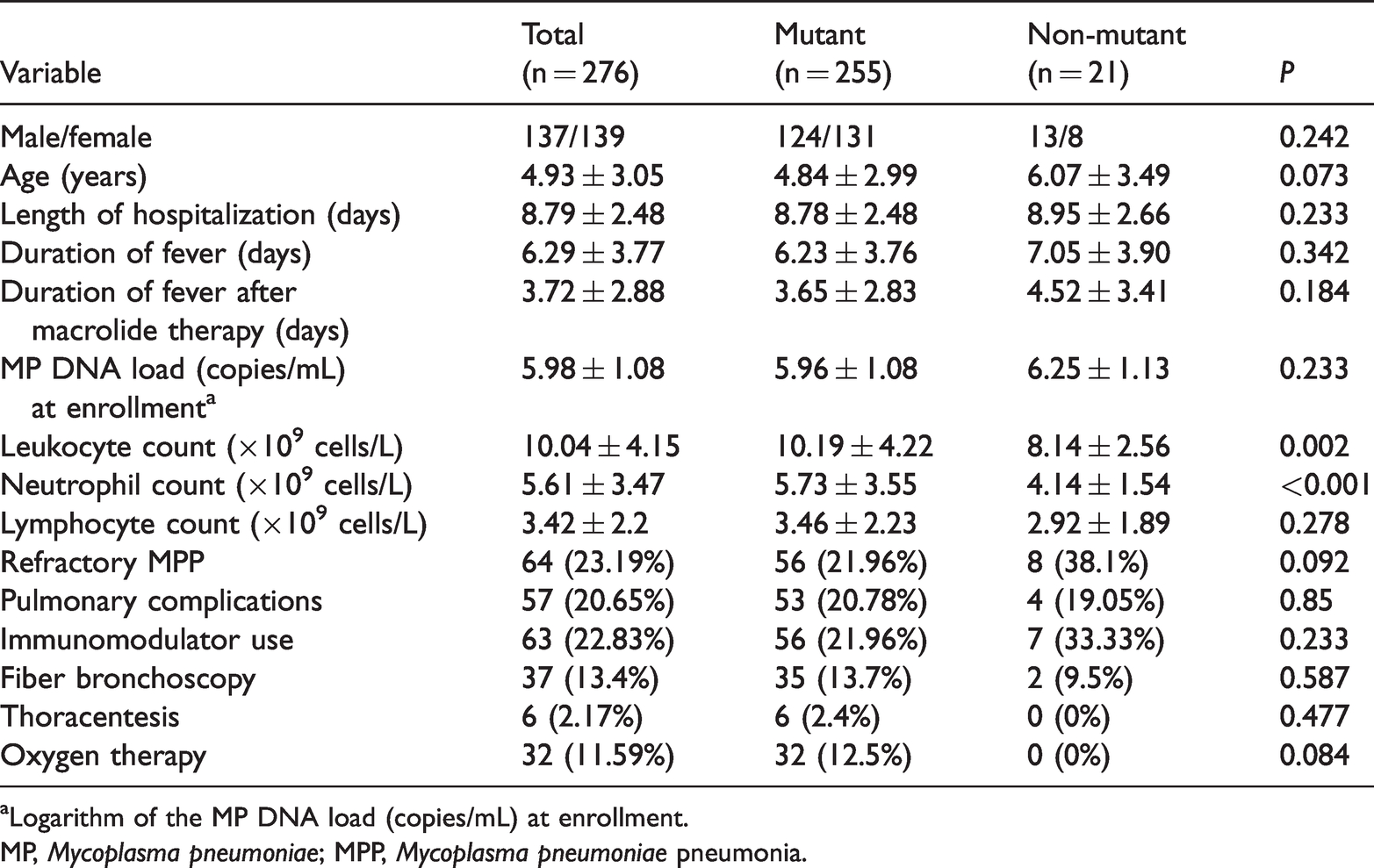
aLogarithm of the MP DNA load (copies/mL) at enrollment.
MP,
Based on the clinical and radiologic findings, 64 patients (29 boys and 35 girls) were diagnosed with refractory MPP, and the remaining 212 patients (108 boys and 104 girls) were diagnosed with non-refractory MPP. There was no statistical difference in sex between the two groups. However, the mean patient age and MP DNA load were significantly higher in the refractory MPP group (both
Characteristics of refractory Mycoplasma pneumoniae pneumonia.

aLogarithm of the MP DNA load (copies/mL) at enrollment.
MP,
Discussion
Although MP infection is typically self-limiting, the number of cases progressing to refractory, severe, and even life-threatening pneumonia has steadily increased in recent years.1,2 In addition, the long-term and extensive use of macrolides as the first-line therapeutics for children has fueled macrolide resistance. 5 Because macrolides inhibit protein synthesis by binding to domain V of MP 23S RNA, mutations of nucleotide 2063, 2064, 2067, or 2617 of domain V protect protein synthesis, thus resulting in resistance to macrolides and the survival of MP. This microbiological problem has been aggravated globally since 2000. The highest prevalence of macrolide-resistant MP was reported found in East Asia, including rates of 87.2% in Korea, 81.6% in Japan, and 90% to 100% in China.6,15 In the present study, 255 (92.39%) clinical MP strains from children with MPP were harbored the A2063G mutation, which is consistent with previous studies in China.20,24 Notably, although mutations at 2064, 2067, and 2617 were not detected, four novel mutations were found, including T insertions between positions 2589 and 2590 and positions 2612 and 2613, a G insertion between positions 2586 and 2587, and a G-to-C transition at position 2601 in domain V of MP 23S rRNA. The number of patients with A2063G and the novel mutations was insufficient to permit statistical analysis. Further studies are required to test whether these new mutations can result in macrolide resistance.
Previous studies mostly focused on the minimum inhibitory concentrations of macrolides
Some studies reported a longer fever duration after treatment in patients infected with mutant MP strains.26,27 In our study, the overall duration of fever and duration of fever after macrolide therapy were similar in patients with or without mutant MP strains. The disparity between our findings and previous results may be attributed to the different prevalence of mutant MP in the studies. Although high mutation rates of MP 23S rRNA domain V are found in China, Japan, and South Korea,6,15,20 the mutation rates vary greatly over time and across regions, even within the same country.26,28–32 Additionally, MP has a self-limiting nature, and the administration of macrolides is beneficial against both non-mutant and mutant MP infection. 33 The severity of pneumonia may be an important determinant of the clinical course in patients infected by mutant strains.
Refractory MPP is characterized by prolonged fever and severe pulmonary inflammatory responses. In this study, the rates of refractory MPP in pediatric patients infected with mutant and non-mutant MP were 21.96% and 38.1%, respectively, suggesting that the presence of mutant strains does not increase the prevalence of refractory MPP. The study results indicate that the clinical features of MPP may not be associated with macrolide-resistant mutations in domain V of MP 23S rRNA. The development of refractory MPP might partially be attributable to the immune reaction related to MP infection regardless of the mutations.
24
The direct invasion of MP into human bronchial or lung tissue stimulates the host inflammatory response in proportion to the strength and persistence of the infection. In the treatment of refractory MPP, immunomodulators such as corticosteroids or intravenous immunoglobulin are commonly added to reduce the immune reaction.
25
Therefore, compared with the investigation of genetic mutations in MP, it is probably more critical to explore other risk factors that may trigger refractory, severe, or even life-threatening pneumonia. According to previous research, MP membrane fractions, inflammatory factors, and goblet cell hyperplasia were risk factors for refractory or severe MPP. MP membrane fractions can enhance mucin production and neutrophil recruitment.
34
Additionally, MP can activate the production of cellular tumor necrosis factor-α, a cytokine that both triggers and maintains the inflammatory response.
35
Moreover, mucus cell hypersecretion and goblet cell hyperplasia have been observed in the airways of MP
Interestingly, in this study, the significantly different MP DNA load at enrollment between patients with refractory and non-refractory MPP 21 indicated that the MP DNA load is a risk factor of refractory MPP because of the direct injury caused by an increased MP load.23–25 Moreover, persistent stimulation by high loads of MP may induce a more intense host inflammatory response, contributing to refractory MPP in children.
Our study had some limitations. First, all samples were obtained from the same tertiary hospital over a period of 2 years. The results should be interpreted with caution because of the possibility of selection bias. Second, the sample size was relatively small. Third, the minimum inhibitory concentration, P1 genotype, and MLVA types were not measured in our clinical samples. Therefore, studies with more extensive surveillance are highly awaited.
Conclusions
The present study illustrated that clinical mutant MP strains were responsible for 92.39% of cases of pediatric MPP in Nanjing. The A2063G mutation was found in all mutant strains, and other identified mutations require further study. Moreover, the results confirmed that mutations in domain V of MP 23S rRNA were not associated with clinical characteristics, laboratory results, pulmonary complications, the efficacy of macrolide treatment, and the development of refractory MPP. In addition, we found that the MP DNA load and host immune response may drive the development of refractory MPP.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by grants from the Nanjing Health and Family Planning Commission (YKK18194) and Nanjing Medical University Science and Technique Development Foundation (njmuzd054).
Author contributions
C. Xu and H. Deng participated in the design of the study and the statistical analysis. H. Deng and H. Tang wrote and submitted the manuscript. J. Zhang, Y. Zhu, Q. Rong, Y. Quan, and C. Xu participated in sample preparation, gene magnification, and sequence alignment. D. Zhao participated in the design of the study. All authors have read and approved the final manuscript for submission.
