Abstract
Background:
Sorafenib is a first-line treatment option for patients with hepatocellular carcinoma (HCC). However, the impact of sorafenib resistance type on patient survival prediction and choice of second-line treatment regimen is unknown.
Objectives:
This study aims to explore the factors predicting resistance in patients with HCC receiving sorafenib, the impact of resistance on survival, and the optimal second-line treatment regimen.
Design:
This was a retrospective cohort study.
Methods:
We recruited all patients with advanced HCC who received first-line sorafenib from January 2019 to January 2023 in two medical centers in China. They were divided into primary and secondary resistance groups according to tumor progression within 3 months. Resistance was the primary outcome of this study. The secondary outcomes were progression-free survival (PFS) and overall survival (OS).
Results:
A total of 424 patients met the inclusion criteria, including 165 patients (38.9%) in the primary group and 259 patients (61.1%) in the secondary group. The independent risk factors for primary resistance were alpha-fetoprotein (AFP) > 400 ng/mL and alanine aminotransferase (ALT) > 40 U/L. Patients in the primary group had significantly shorter median OS than those in the secondary group (9.0 months vs 23.0 months,
Conclusion:
Sorafenib has a high incidence of primary resistance and short survival in patients who develop primary resistance. AFP and ALT are influential factors in primary resistance, and it is valuable to use these two metrics to guide the use of sorafenib. As second-line therapy, a TKI plus PD-1 inhibitor regimen should be preferentially recommended.
Keywords
Introduction
Hepatocellular carcinoma (HCC) accounts for 80% of all liver cancers, one of the leading cancers worldwide. 1 Significant temporal and geographic differences exist in the etiology of HCC. For example, in China, the country with the highest burden of HCC worldwide, the hepatitis B virus (HBV) is the most common etiologic agent. By contrast, the main etiologic agent in the United States is nonalcoholic steatohepatitis. 2 In addition, HCC is associated with age, sex, and genetic predisposition via complex oncogenic pathways. 3 The complex biological processes involved and the insidious nature of HCC result in late diagnoses for almost half of cases, often already accompanied by local or distant metastases. 4
For patients with HCC who do not respond to surgical intervention, systemic therapy has become the cornerstone of treatment. The recognized first-line options for systemic therapy in patients with advanced HCC include sorafenib, lenvatinib, and atezolizumab plus bevacizumab (A + T) regimens. However, the efficacy of these treatment regimens is limited with 11–19 months of overall survival (OS).5–7 This is because its efficacy is mainly limited by drug resistance, low tolerability, and even serious adverse events.
Resistance is an inevitable problem in antitumor therapy. A lot of research has been done on resistance mechanisms, but clinical translation is still lacking. 8 Resistance can be broadly categorized into primary and secondary resistance. 9 Primary resistance is said to occur when a medication is inherently ineffective in a patient. However, secondary resistance is the occurrence of resistance after a period of clinical benefit. Theoretically, the prognoses in the two cases are different. 9 If we group patients differently according to their drug resistance status, can we better personalize their treatment? Could we better predict patient prognosis?
Sequential therapy is the best way to prolong patient survival; however, there is currently no standard order of treatment for backline regimens. In 2023, the American Association for the Study of Liver Diseases (AASLD) recommended five options as second-line treatment after failure of sorafenib therapy. 10 Determining the optimal second-line treatment is complicated by clinical trial designs, etiological variability, complications, and patient quality of life. Previous studies have explored relevant markers and adverse effects that affect patient prognosis; however, there has been no significant breakthrough in improving patient survival.11,12 One question is whether we should categorize patients based on sorafenib resistance and then select favorable patients and explore the best second-line treatment options.
Considering the heterogeneity of HCC patients, the focus of HCC research is not only on drug development but also on individualization of therapy. Previously, drug resistance was not regarded as a factor affecting efficacy, and the impact of resistance on the choice of a second-line regimen was not considered. Therefore, this study aims to explore the factors predicting resistance in patients with HCC receiving sorafenib, the impact of resistance on survival, and the optimal second-line treatment regimen.
Materials and methods
Study design and participants
This retrospective study was conducted at the Affiliated Hospital of Guizhou Medical University and the Fifth Medical Center of the General Hospital of the People’s Liberation Army in China. We screened all patients with HCC who visited both hospitals between January 2019 and January 2023. The inclusion criteria were as follows: (1) HCC diagnosed by pathology or by two radiologic imaging techniques; (2) Child-Pugh class A or B; (3) at least one measurable tumor lesion as defined by the Response Evaluation Criteria in Solid Tumors, version 1.1 (RECIST 1.1); and (4) at least one imaging examination within 3 months of starting treatment. The exclusion criteria were as follows: (1) coexistence of other primary tumors; (2) nonadherence to sorafenib; (3) surgical treatment; (4) non-HBV/hepatitis C virus (HCV) infection; (5) unacceptable toxicity; and (6) incomplete follow-up data. All patients provided informed consent before enrollment. This study was approved by the Ethics Committee and complied with the 1975 Declaration of Helsinki. Patients were instructed to use the recommended medication dosage, 400 mg of oral sorafenib, twice daily. Based on the follow-up results, all patients who met the inclusion criteria were divided into either the primary resistance group (primary group) or the secondary resistance group (secondary group). The participants’ baseline characteristics were collected at two timepoints: initiation of sorafenib and initiation of second-line treatment after sorafenib treatment failure. The reporting of this study conforms to Strengthening the reporting of observational studies in epidemiology (STROBE) (Supplemental Table 1). 13
Related definitions and study endpoints
Resistance was the primary endpoint of this work. Primary resistance was defined as tumor progression within 3 months of initiation of sorafenib. Tumor progression, as defined by RECIST 1.1, is disease progression (PD) that occurs in a patient during the follow-up period. Secondary resistance was defined as tumor progression occurring more than 3 months after initiation of sorafenib but not within 3 months. The secondary outcomes of this study were OS and progression-free survival (PFS). OS was defined as the time interval from the initiation of treatment to either death from any cause or end of the study, whichever came first. PFS was defined as the time from the initial dose to the first radiologically confirmed tumor progression or death from any cause. Radiological response was recorded using dynamic computed tomography or magnetic resonance imaging at baseline and every 8–12 weeks after treatment initiation.
Statistical analysis
Categorical data were expressed as frequency with proportion and were analyzed using the chi-square test or Fisher’s exact test. The Kaplan–Meier method was employed to calculate PFS and OS and to plot the curve. The log-rank test was used to compare the cumulative survival rates of the two groups, and the risk curve was drawn. Logistic regression and Cox proportional hazards models were used to explore the factors influencing drug resistance and prognosis. Variables with
Results
Patient characteristics
From January 2019 to January 2023, 506 patients with unresectable HCC treated with sorafenib were screened. Among them, 82 patients were excluded, and the remaining 424 patients were included in the study. Of these, 165 (38.9%) were in the primary group and 259 (61.1%) were in the secondary group. At the time of data cutoff (November 2023), the median follow-up duration was 21.5 (95% CI: 20.1–22.9) months. During the follow-up period, 192 patients received second-line treatment, including 86 in the primary and 106 in the secondary groups. In the primary group, 36 patients received a combination of a tyrosine kinase inhibitor (TKI) and a programmed cell death protein-1 (PD-1) inhibitor, while 50 received only a TKI. In the secondary group, 51 patients received the combination therapy, while 55 received only TKI (Figure 1). The specific medications used in the TKI group (

Patients flow chart.
Baseline characteristics of the study population and primary resistance to sorafenib: univariate analysis.
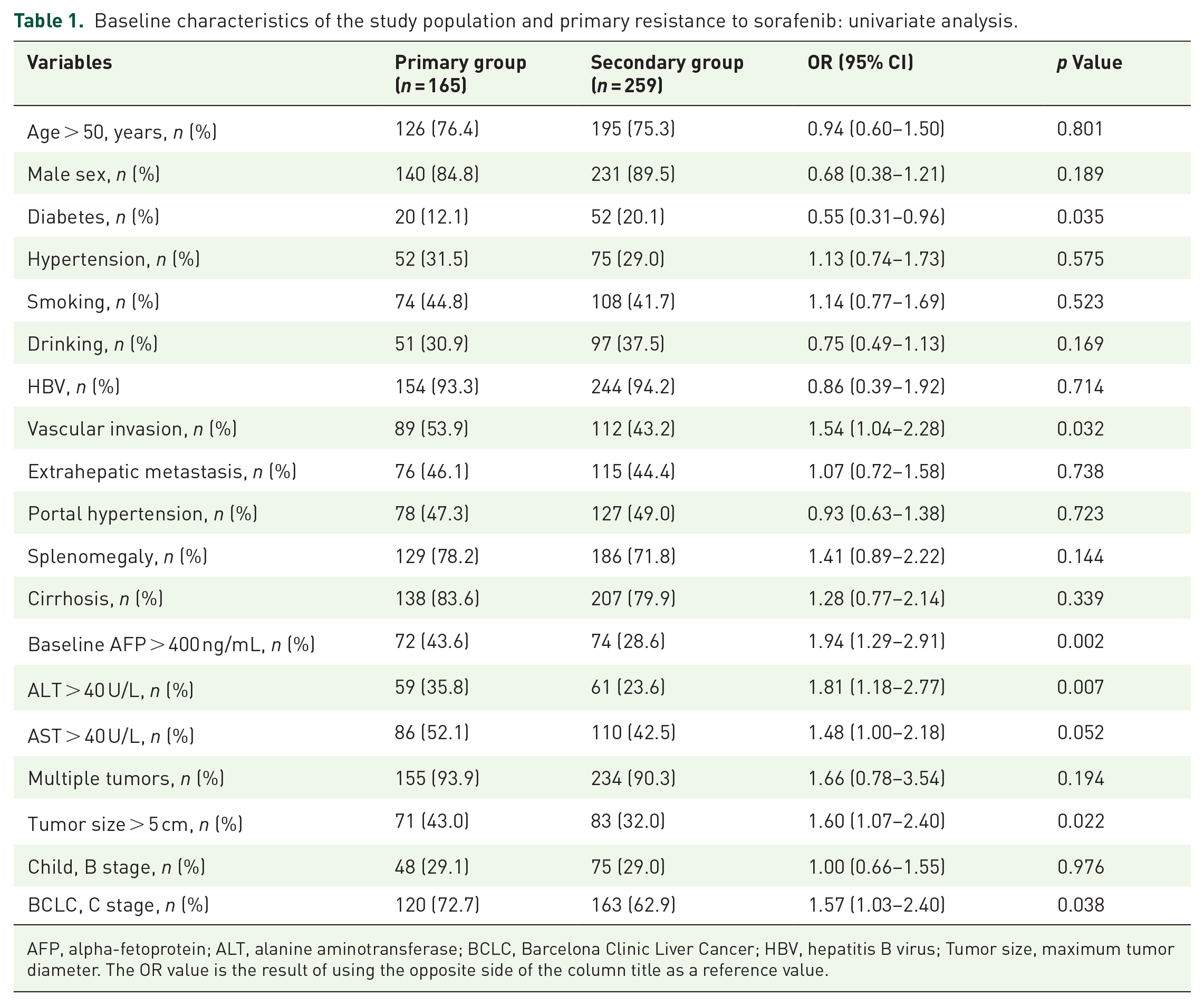
AFP, alpha-fetoprotein; ALT, alanine aminotransferase; BCLC, Barcelona Clinic Liver Cancer; HBV, hepatitis B virus; Tumor size, maximum tumor diameter. The OR value is the result of using the opposite side of the column title as a reference value.
Factors associated with sorafenib resistance
Table 1 and Supplemental Table 2 show factors associated with sorafenib resistance. Univariate analysis showed that diabetes (
Sorafenib resistance and prognosis
To explore the impact of sorafenib resistance on the prognosis of HCC patients, we divided all patients (
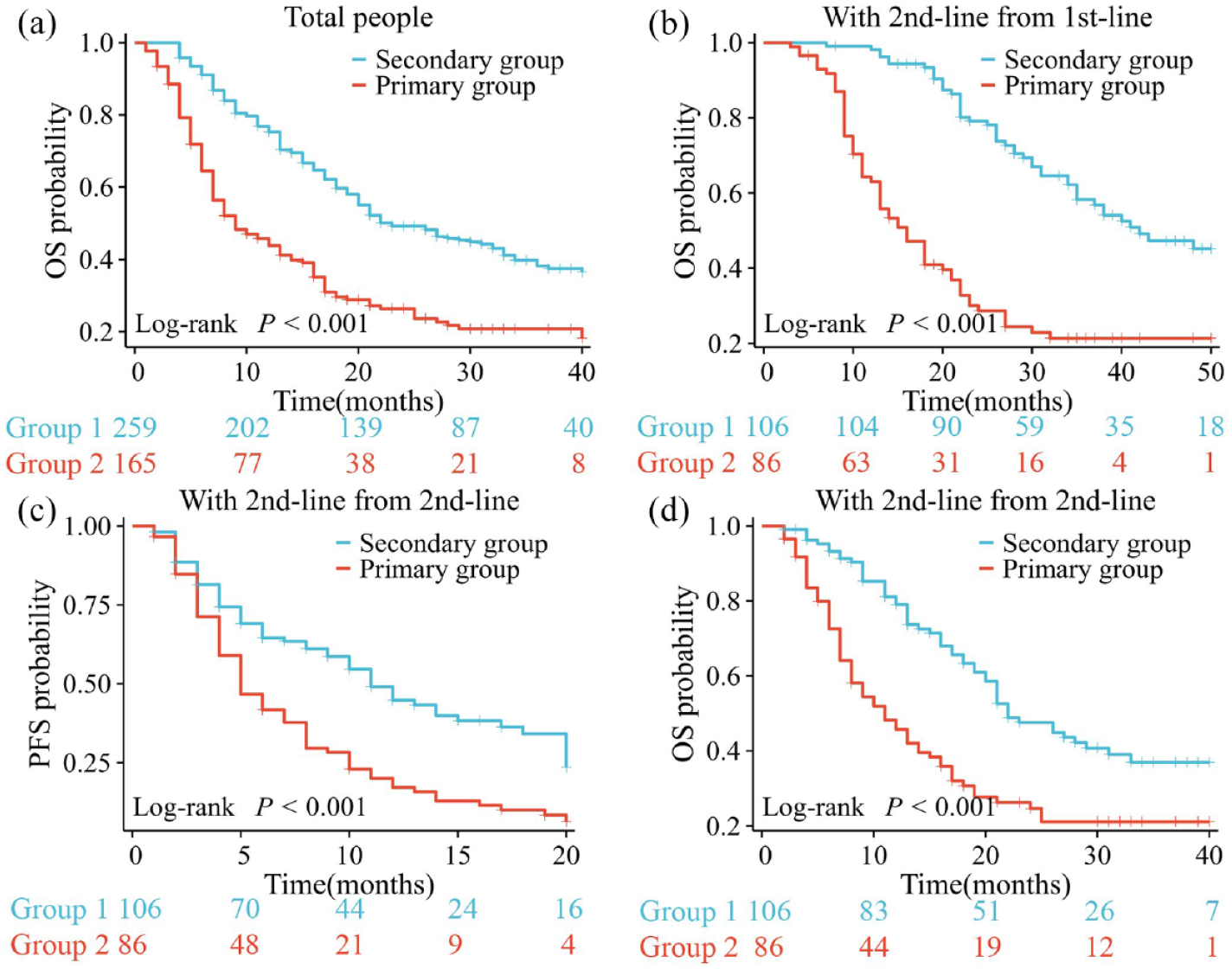
The K-M survival curve is used to calculate the overall survival (OS) of all patients (a), the OS of patients receiving second-line treatment with survival time calculated from the start of sorafenib treatment (b), and the progression-free survival (PFS) (c) and OS (d) of patients receiving second-line treatment with survival time calculated from the start of second-line treatment.
To further confirm whether the prognosis of the sorafenib primary group was poorer than that of the secondary group, we further analyzed it in subgroups according to the second-line treatment regimen. Patients in the primary group had significantly shorter median OS than those in the secondary group in all patients treated with TKI monotherapy (8.5 vs 21.0 months,

K-M survival curves were used to analyze overall survival (OS) after receiving different second-line treatment regimens, including TKI monotherapy (a), lenvatinib monotherapy (b), TKI plus PD-1 inhibitor therapy (c), and sorafenib plus sintilimab therapy (d).
To exclude the influence of other factors, we further performed univariate and multifactorial analyses of PFS and OS in all patients who received second-line therapy after sorafenib treatment failure. The results showed that PFS was shorter in multiple tumors (hazard ratio (HR) 5.48, 95% CI: 1.74–17.33,
Analysis of prognostic risk factors in patients with second-line treatment.

AFP, alpha-fetoprotein; ALT, alanine aminotransferase; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; HBV, hepatitis B virus; HR, hazard ratio; TKI, tyrosine kinase inhibitor; Tumor size, maximum tumor diameter. The HR value is the result of using the opposite side of the column title as a reference value. Bold text is to highlight the multifactorial analysis data.
Second-line therapy and prognosis
To explore the influence of the choice of second-line treatment regimen on prognosis, we divided the patients receiving second-line therapy into primary (
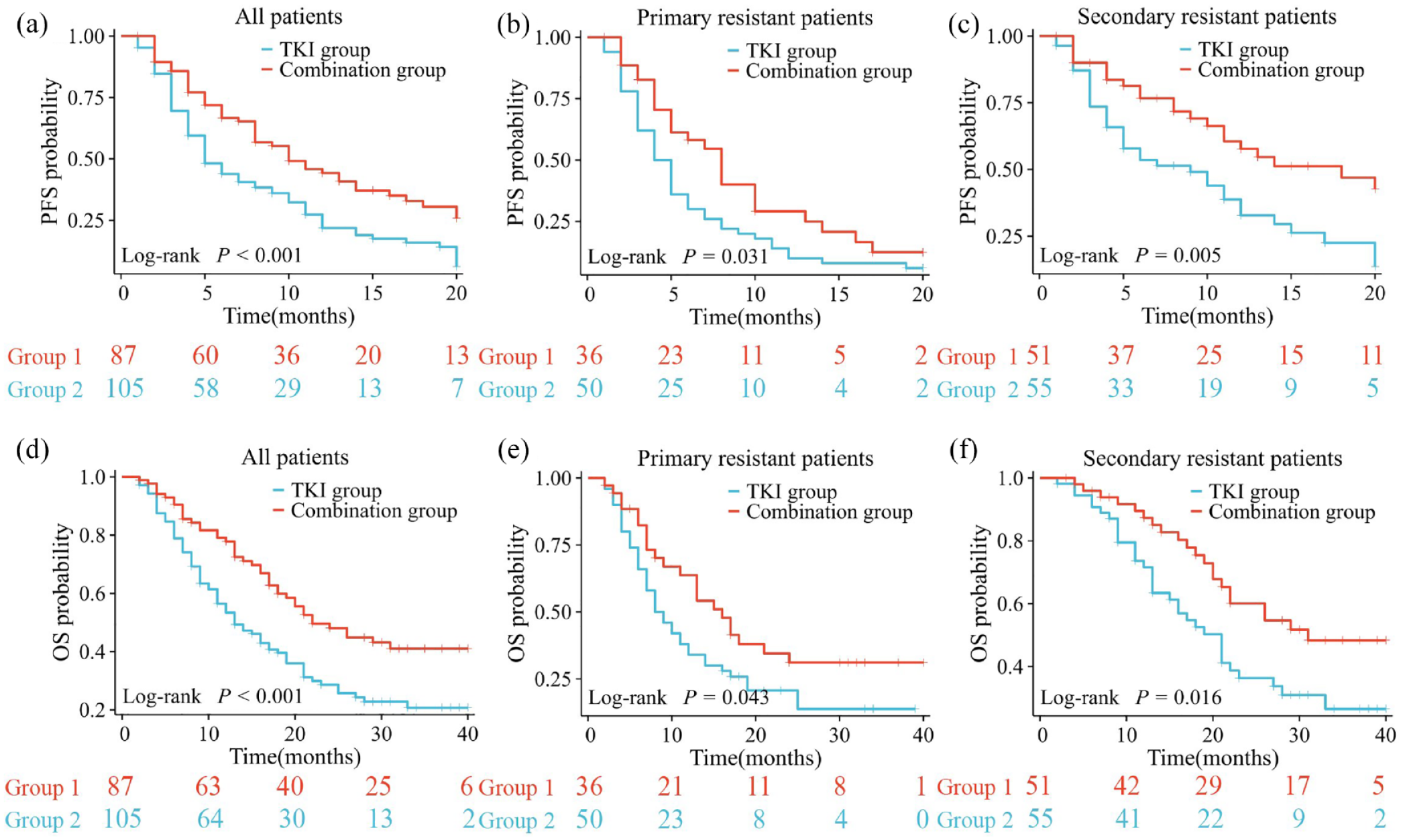
K-M survival curves were used to analyze progression-free survival (PFS) (a, b, c) and overall survival (OS) (d, e, f) between the TKI group and the combination group in three different subgroups of patients receiving second-line therapy, including all second-line regimens, primary-resistant patients, and secondary-resistant patients.
We performed further subgroup analyses to compare prognosis among the specific TKI and combination regimens. Due to the limited number of cases, we categorized the TKI regimens into the lenvatinib group and the other TKI group, and the TKI combined PD-1 inhibitors regimens into sorafenib plus PD-1 inhibitors and other TKI plus PD-1 inhibitors. No statistical difference in OS was found between the lenvatinib group and the “other TKI” group, either in all patients (
Discussion
To the best of our knowledge, this is the first multicenter retrospective study to analyze and compare the prognosis of patients with advanced HCC in different subgroups based on sorafenib resistance. The results of our study showed a significantly worse prognosis in patients with primary resistance. Independent risk factors for primary resistance were AFP > 400ng/mL and ALT > 40 U/L. We also found that patients who continued to receive TKI plus PD-1 inhibitors as a second line after sorafenib resistance had a better prognosis than those who received TKI monotherapy.
For more than 15 years, sorafenib has been the recommended first-line treatment for patients with HCC. It prolongs patient survival by targeting vascular endothelial growth factor receptors to inhibit tumor angiogenesis. 5 However, resistance to sorafenib is inevitable. Prospective clinical studies have shown that the disease control rate of patients treated with sorafenib is only 43%, and the PFS is approximately 4 months, indicating that it has no clinical benefit in more than 50% of patients. 5 Our study classified patients into primary and secondary resistance based on whether tumor progression occurred within or after 3 months of sorafenib initiation. We found that primary resistance occurred in 39.9% of patients. With improvements in medical technology and healthcare services, the percentage of patients without clinical benefit is reducing; however, there has been very little progress.
Primary resistance occurs due to the genetic heterogeneity of tumor cells, leading to their insensitivity to therapeutic drugs. Hence, primary resistance factors are present before the start of treatment. 14 On the other hand, secondary resistance is caused by drug insensitivity during treatment owing to mutations in tumor cells; hence, the production of secondary resistance factors takes time. 15 The mechanisms of primary resistance mainly include epithelial growth factor receptor (EGFR) overexpression 16 and accumulation of cancer stem cells. 17 Secondary resistance is mainly related to PI3K/AKT/mTOR signaling pathway 18 and Ras/Raf/MEK/ERK pathways. 19 Although a variety of factors and pathways related to sorafenib resistance mechanisms have been identified, they have not yet been utilized in the clinic due to limitations in trial technology and clinical value. Theoretically, the survival of patients with primary resistance is severely limited, which is consistent with our results. Furthermore, this is also consistent with our previously published correlation between primary resistance to levatinib and prognosis. 20 In our study, the median OS of patients with primary resistance was 14 months shorter than that of patients with secondary resistance. We also found that analysis by resistance subgroups is of great value for predicting patient prognosis. Although we have demonstrated a large difference in patient prognosis between primary and secondary resistance; however, it is extremely urgent to further explore the differences between the two resistance types at a later stage.
In terms of the mechanism of resistance, assuming that sorafenib resistance-related markers are tested prior to treatment, it would be possible to avoid wasting the patient’s treatment time due to primary resistance and prolong the patient’s survival using lenvatinib or A + T as the first-line treatment option.6,7 The EGFR molecule, a key molecule in primary or secondary resistance to sorafenib, is not only a potential predictor of resistance but also holds promise for improved prognosis with anti-EGFR therapy. 21 From the results of previous studies, c-Jun N-terminal kinase is also one of the markers for predicting sorafenib resistance 22 ; however, there are numerous obstacles to its clinical use in terms of cost, testing time, sensitivity, and specificity in predicting resistance. Our study identified AFP > 400 ng/mL and ALT > 40 U/L as independent risk factors for primary resistance to sorafenib based on clinical indicators. The results showed an AUC value of 0.57 for both indicators, with optimal cutoff values of 371.2 ng/mL (AFP) and 41.2 U/L (ALT). Although they have limited value in predicting sorafenib resistance, these two indicators may guide the choice of first-line treatment of HCC.
In recent years, research on novel systemic therapeutic options for HCC has revolutionized landscapes, bringing exciting hope for prolonging patient survival; however, the search for the optimal option has burdened both physicians and patients. There are three FDA-approved first-line treatment regimens for HCC. Of these, A + T is the most efficacious but is not used in patients with a high risk of gastrointestinal bleeding or immunologic contraindications. 23 Excluded patients may be treated with sorafenib or lenvatinib.5,6 Compared with sorafenib, lenvatinib is superior in terms of PFS and disease control rates but worse in terms of hypertension and proteinuria adverse events. 6 In addition, the recommended first-line treatment options, include tremelimumab plus durvuluma (approved by AASLD and National Comprehensive Cancer Network (NSSN)), 24 sintilimab + IBI305 (approved by China) 25 and donafenib (approved by China). 26 Previous studies analyzed the choice of first-line treatment regimens in terms of efficacy, adverse events, economic conditions, geographical differentiation, mental burden, patients’ wishes, and physicians’ personal experiences. Our study further included drug resistance. When AFP and ALT levels exceeded the optimal cutoff values, patients had a higher incidence of primary resistance to sorafenib. In these cases, other first-line treatment regimens were preferentially recommended, such as the A + T or lenvatinib regimen.
From the latest relevant guidelines, sorafenib still plays an important role, and there is a big dilemma on how to choose the second line after receiving sorafenib resistance. The FDA-approved second-line regimens for HCC are in the categories of monotherapy (cabozantinib, regorafenib, pembrolizumab, or ramucirumab (AFP ⩾ 400 ng/mL)) and combination therapy (ipilimumab + nivolumab).
24
All treatment regimens above used a placebo in the control group; hence, they lacked reliable crossover trial comparisons. However, some medications were compared, such as regorafenib versus nivolumab,
27
regorafenib versus cabozantinibz
28
and cabozantinib versus ramucirumab
29
in retrospective studies but no positive results. To explore the optimal second-line treatment regimen, we compared the prognosis of patients who failed sorafenib treatment in a TKI monotherapy group versus a combination therapy group, and the results showed that the combination therapy had a very promising median PFS (6.0 vs 10.0 months,
This study has some limitations. First, this was a retrospective study, which may have generated information and selection biases. Second, we excluded patients without HBV or HCV infection, and those who switched medications due to adverse events; therefore, data related to patients with other etiologies were lacking. Third, the second-line sorafenib treatment regimens were analyzed primarily as TKI or TKI combined with PD-1 inhibitors. Therefore, data from a single-drug prospective study are needed to reach more robust conclusions regarding sorafenib resistance. Fourth, because we included patients from two centers, there may have been differences in care that could have introduced bias into the study results.
Conclusion
The incidence of primary resistance is high in patients with HCC being treated with sorafenib. AFP > 400 ng/mL and ALT > 40 U/L are independent risk factors for primary resistance, and both indicators are valuable in predicting primary sorafenib resistance and can be used to guide the choice of treatment regimen. Patients with secondary sorafenib resistance have a higher survival than those with primary resistance. Furthermore, TKIs combined with PD-1 inhibitors should be recommended as second-line treatment in HCC when resistance occurs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241299678 – Supplemental material for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC
Supplemental material, sj-docx-1-tam-10.1177_17588359241299678 for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC by Jin Lei, Hongyuan Dai, Ya Zhang, Guangling Ou, Zhi Peng Liang, Yinying Lu and Haiyang Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241299678 – Supplemental material for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC
Supplemental material, sj-docx-2-tam-10.1177_17588359241299678 for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC by Jin Lei, Hongyuan Dai, Ya Zhang, Guangling Ou, Zhi Peng Liang, Yinying Lu and Haiyang Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359241299678 – Supplemental material for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC
Supplemental material, sj-docx-3-tam-10.1177_17588359241299678 for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC by Jin Lei, Hongyuan Dai, Ya Zhang, Guangling Ou, Zhi Peng Liang, Yinying Lu and Haiyang Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-4-tam-10.1177_17588359241299678 – Supplemental material for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC
Supplemental material, sj-jpg-4-tam-10.1177_17588359241299678 for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC by Jin Lei, Hongyuan Dai, Ya Zhang, Guangling Ou, Zhi Peng Liang, Yinying Lu and Haiyang Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-5-tam-10.1177_17588359241299678 – Supplemental material for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC
Supplemental material, sj-jpg-5-tam-10.1177_17588359241299678 for Prognostic impact of primary versus secondary resistance to sorafenib in patients with HCC by Jin Lei, Hongyuan Dai, Ya Zhang, Guangling Ou, Zhi Peng Liang, Yinying Lu and Haiyang Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
