Abstract
Background:
Epidermal growth factor receptor-mutated (EGFR+) non-small-cell lung cancer (NSCLC) patients failing tyrosine kinase inhibitors (TKI) can benefit from next-line targeted therapies, but implementation is challenging.
Methods:
EGFR+ NSCLC patients treated with first/second-generation (1G/2G) TKI at our institution with a last follow-up after osimertinib approval (February 2016), were analyzed retrospectively, and the results compared with published data under osimertinib.
Results:
A total of 207 patients received erlotinib (37%), gefitinib (16%) or afatinib (47%). The median age was 66 years, with a predominance of female (70%), never/light-smokers (69%). T790M testing was performed in 174/202 progressive cases (86%), positive in 93/174 (53%), and followed by osimertinib in 87/93 (94%). Among the 135 deceased patients, 94 (70%) received subsequent systemic treatment (43% chemotherapy, 39% osimertinib), while 30% died without, either before (4%) or after progression, due to rapid clinical deterioration (22%), patient refusal of further therapy (2%), or severe competing illness (2%). Lack of subsequent treatment was significantly (4.5x,
Conclusion:
Osimertinib after 1G/2G TKI failure prolongs survival, but approximately 15% and 30% of patients forego molecular retesting and subsequent treatment, respectively, mainly due to rapid clinical deterioration. This is an important remediable obstacle to sequential TKI treatment for EGFR+ NSCLC. It pertains also to other actionable resistance mechanisms emerging under 1G/2G inhibitors or osimertinib, whose rate for lack of next-line therapy is similar (approximately 35% in the FLAURA/AURA3 trials), and highlights the need for closer monitoring alongside broader profiling of TKI-treated EGFR+ NSCLC in the future.
Introduction
Epidermal growth factor receptor (
Patients and methods
Study population and study endpoints
This retrospective study included all non-consecutive stage IV NSCLC patients with activating
Data collection and statistical analysis
Histological diagnosis of NSCLC was performed at the Institute of Pathology Heidelberg on tissue specimens according to the criteria of the current World Health Organization (WHO) Classification (2015) for lung cancer.
12
Molecular profiling of tissue and liquid biopsies was performed using DNA-based next-generation sequencing (NGS) with a laboratory turnaround time <10 working days, as described previously.13,14 Clinical data were systematically collected from the patients’ records with a cutoff on 30 June 2020. Since all patients were treated in-house, there were no missing data regarding the study endpoints (molecular retesting, subsequent treatment, survival) or loss-to-follow-up cases. The progression date under 1G/2G TKI was verified by the investigators with review of radiologic images, that is, chest/abdomen computed tomography (CT) and brain magnetic resonance imaging (MRI)-based restaging every 6–12 weeks, without formal RECIST re-evaluation, as several studies have demonstrated very good agreement between real-world and RECIST-based assessments.15,16 OS was calculated from start of treatment for stage IV disease. Follow-up time was calculated by the reverse Kaplan–Meier method.
17
Time-to-next-treatment (TNT) was calculated from the start of 1G/2G EGFR TKI until initiation of next-line therapy or death. Survival data were analyzed according to Kaplan–Meier and compared between patient groups with the logrank test. Numerical data were analyzed with the Student’s
Ethics
This study was approved by the ethics committee of Heidelberg University (S-145/2017 and S-469/2017). Since this was a non-interventional, retrospective study, informed consent was obtained whenever possible, but its need for every participant was waived by the ethics committee.
Results
Evaluable study patients
Overall, 240 stage IV NSCLC patients with 1G/2G TKI treatment for activating
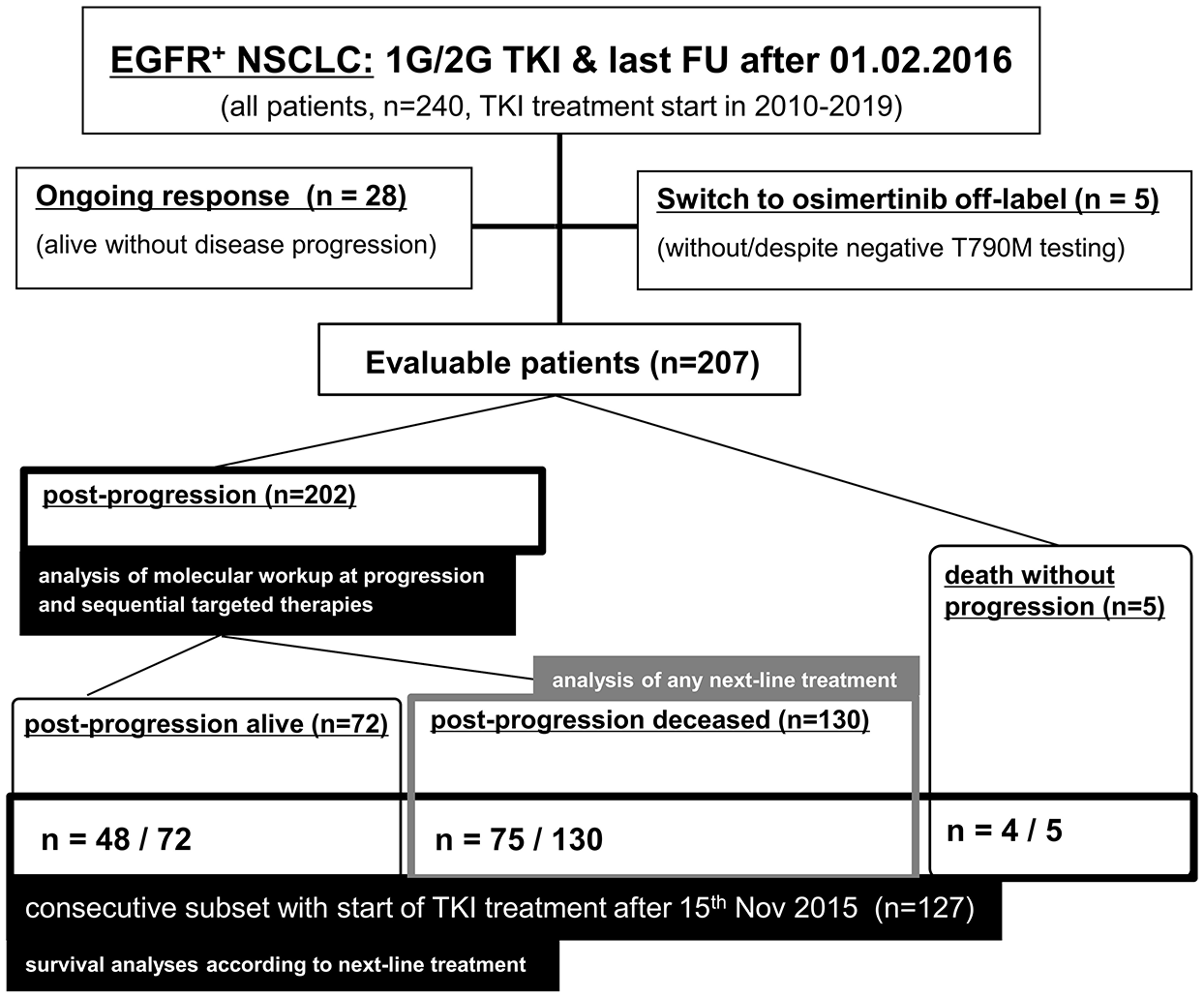
Flow diagram of the entire study population and subsets used to calculate each endpoint.

Molecular testing, T790M positivity and next-line osimertinib after failure of 1G/2G-generation EGFR inhibitors in the entire study population. (a) Rate of T790M testing, T790M positivity and next-line osimertinib administration among patients with documented radiologic disease progression under 1G/2G EGFR TKI (

Systemic treatment after failure of 1G/2G EGFR inhibitors in the subset of deceased patients. (a) Rate for implementation of any subsequent treatment, subsequent CHT, and subsequent osimertinib after failure of 1G/2G EGFR inhibitors. Error bars indicate 95% CI. (b) Association between administration of any next-line therapy and performance of T790M testing (chi-square
Characteristics of study patients.
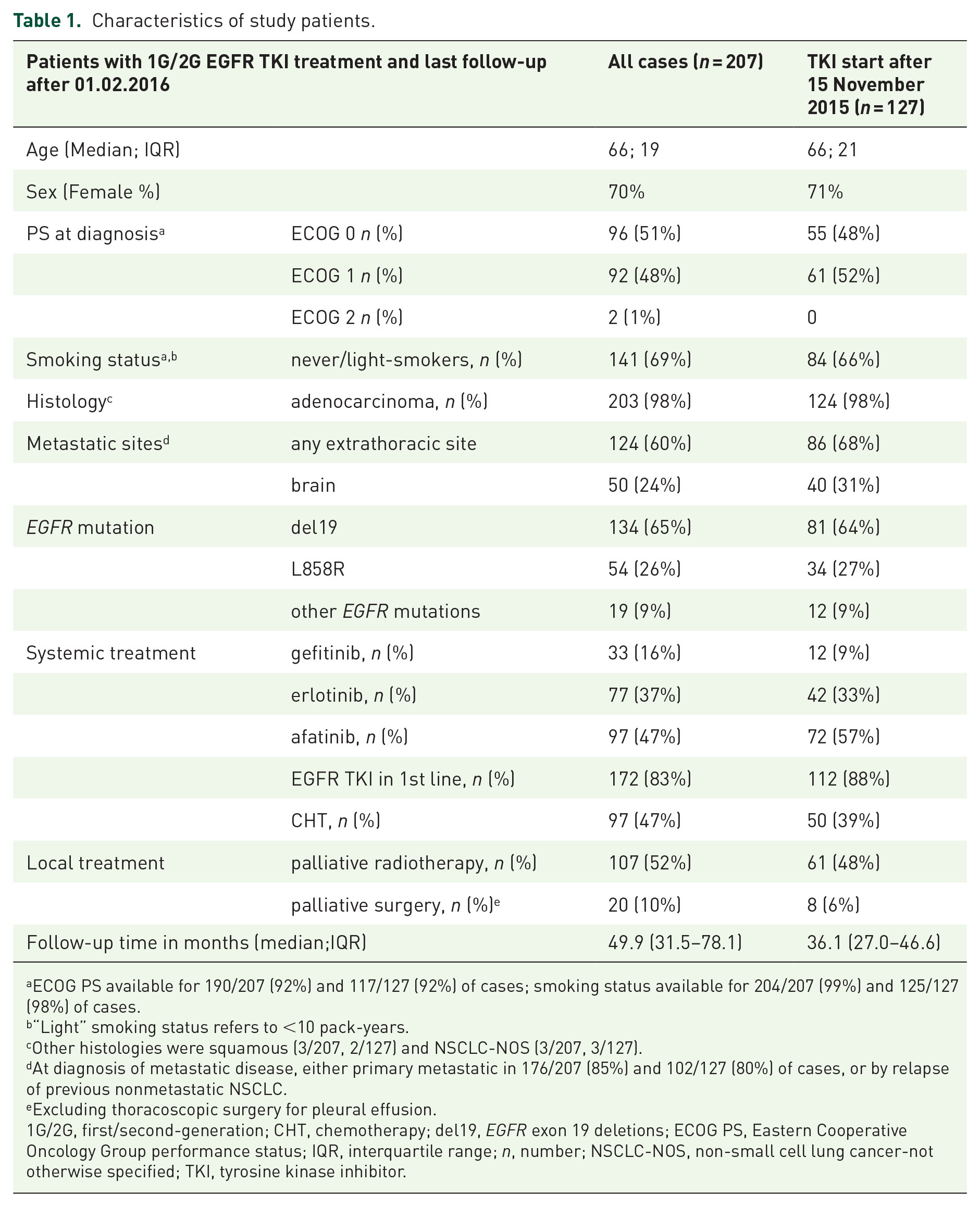
ECOG PS available for 190/207 (92%) and 117/127 (92%) of cases; smoking status available for 204/207 (99%) and 125/127 (98%) of cases.
“Light” smoking status refers to <10 pack-years.
Other histologies were squamous (3/207, 2/127) and NSCLC-NOS (3/207, 3/127).
At diagnosis of metastatic disease, either primary metastatic in 176/207 (85%) and 102/127 (80%) of cases, or by relapse of previous nonmetastatic NSCLC.
Excluding thoracoscopic surgery for pleural effusion.
1G/2G, first/second-generation; CHT, chemotherapy; del19,

OS according to next-line treatment in the consecutive subset of EGFR+ NSCLC patients. Within the subset of consecutive patients with 1G/2G TKI start after 15 November 2015 (
Analysis of EGFR T790M testing, T790M positivity, and next-line osimertinib
Among patients with radiologic disease progression under 1G/2G EGFR TKI (
Analysis of subsequent treatment
Among deceased patients, subsequent treatment was offered to 70% (94/135) and consisted mainly of chemotherapy (58/94, 43% of 135 deceased patients) and osimertinib (53/94, 39% of 135, Figure 3a), while immunotherapy (5/94) and other TKI (4/94) were used less frequently. The 30% of patients (41/135) that died without any next-line therapy were significantly (4.5×) enriched among cases foregoing T790M testing (15% or 16/106 patients without next-line therapy among T790M-tested
Survival according to subsequent treatment
Among the consecutive patients failing TKI that had started after 15 November 2015 (
Discussion
Even though the superiority of osimertinib after failure of 1G/2G EGFR inhibitors was demonstrated by the AURA3 trial already in 2017, 5 practical implementation remains challenging with widely variable success rates in the literature. In contrast to large randomized clinical trials spanning over several countries, the present study analyzes an all-comers patient population under the homogenous, but real-world conditions of a single large academic institution with the aim of defining bottlenecks and priorities close to the circumstances of daily clinical practice.
A first finding is that main obstacles to sequential therapy with osimertinib in EGFR+ NSCLC are lack of T790M testing in approximately 15%, and T790M negativity in approximately 45% of progressive patients, while almost all (94%) T790M positive patients receive the drug (Figure 2a). Of these, the rate of T790M positivity for progressive patients is more-or-less similar across 1G/2G EGFR inhibitors and current analytical methods,19,20 with the 50–55% observed in our study corresponding well to the literature21,22; therefore, the main bottleneck appears to be initiation of T790M testing. Traditionally, the main issue here has been tissue availability, since many advanced lung cancer patients are not suitable or willing to undergo invasive procedures.
23
Meanwhile, this problem is largely solved by liquid biopsies, which are not only feasible for every patient, but also provide results earlier than tissue rebiopsies due to the faster sample collection.
24
Their marginally lower sensitivity of 75–80% compared with tissue testing is offset by their wider applicability, and their use together with conventional tumor rebiopsies maximizes yield (Figure 2d).25–27 However, the clinical impairment of many patients remains an important limitation: in our study, rapid clinical deterioration was the main reason for both lack of T790M testing (Figure 2b), and lack of any subsequent treatment (Figure 3d), which correlated (Figure 3b), and also prevented some T790M positive cases from receiving osimertinib (Figure 2c). Along the same lines, the mere performance of T790M testing was associated more strongly with OS than exposure to next-line osimertinib, type of

Implementation of sequential targeted therapies for metastatic EGFR+ NSCLC. Critical parameters, possible improvement strategies and feasibility limit for implementation of sequential targeted therapies in metastatic EGFR+ NSCLC. The maximum testing rate of 95% is taken from Supplemental Table S2 and excludes only patients with EGFR TKI discontinuation due to reasons precluding further treatment (i.e. “other reasons” or patient decision); the actual testing rate of 80–85% is taken from Figure 2a, Supplemental Figure S1a and the literature cited in the Discussion; the actual treatment rate for T790M positive patients with osimertinib of 90–95% is taken from Figure 2a and Supplemental Figure S1A; the maximum treatment rate for T790M positive patients of >95% additionally considers that 3/6 T790M positive patients foregoing osimertinib treatment suffered early death before the drug could be started (Figure 2c), which could potentially have been prevented by an earlier change in therapeutic strategy facilitated by closer patient monitoring; the x% rate of marker-positivity is mutation-specific, for example, approximately 55% for
The longer OS of T790M positive patients treated with next-line osimertinib in our real-world cohort (Figure 4) underlines the importance of sequential TKI administration for clinical outcome, as already demonstrated by the randomized AURA3 trial,
5
the international observational GioTag study,
40
and other smaller series.41,42 However, besides
The main limitations of our study are the retrospective, single-institution design, and small patient number. Specific strengths are the homogenous, standardized testing and treatment of our patients within the same academic institution, the dissection of the entire study population into those subsets that are most suitable for estimation of each endpoint (Figure 1), and the sensitivity analyses that demonstrated robustness of findings (Supplemental Figure S1). Whenever possible, we have compared our estimated parameters with the findings of other investigators or published data from clinical trials, and found good agreement, which suggests generalizability. The results presented provide a comprehensive picture of clinical implementation for sequential targeted therapies in EGFR+ NSCLC, define obstacles, prioritize potential measures to overcome them, and highlight the importance for patient survival in the real-world setting.
Supplemental Material
sj-pdf-1-tam-10.1177_1758835921996509 – Supplemental material for Real-world implementation of sequential targeted therapies for EGFR-mutated lung cancer
Supplemental material, sj-pdf-1-tam-10.1177_1758835921996509 for Real-world implementation of sequential targeted therapies for EGFR-mutated lung cancer by Nikolaus Magios, Farastuk Bozorgmehr, Anna-Lena Volckmar, Daniel Kazdal, Martina Kirchner, Felix J. Herth, Claus-Peter Heussel, Florian Eichhorn, Michael Meister, Thomas Muley, Rami A. Elshafie, Jürgen R. Fischer, Martin Faehling, Mark Kriegsmann, Peter Schirmacher, Helge Bischoff, Albrecht Stenzinger, Michael Thomas and Petros Christopoulos in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank Petra Kettenring of the Clinical Trial Unit in the Thoraxklinik Heidelberg for assistance with the collection of patient data and samples.
Conflict of interest statement
FB reports personal fees from Novartis, MSD, Chugai Pharma, Roche, and AstraZeneca and research grants from AstraZeneca, BMS, and Roche.
ALV reports personal fees from AstraZeneca.
DK reports personal fees from AstraZeneca, personal fees from Bristol-Myers Squibb GmbH, personal fees from Pfizer Pharma GmbH, outside the submitted work.
FJH reports advisory board fees and honoraria from Lilly, Roche, AstraZeneca, Novartis, Boehringer, Chiesi, Teva, Pulmonx BTG, and Olympus as well as research funding from Lilly, Roche, AstraZeneca, Novartis, Boehringer, Chiesi, and Teva.
CPH reports consultation, lecture and other fees from Novartis, Basilea, Bayer, Grifols, Boehringer, Pierre Fabre, Covidien, Siemens, Chiesi, Intermune, MEDA Pharma, Bracco, Pfizer, MSD, Roche, Lilly, AstraZeneca, Schering-Plough, Essex, Gilead, MeVis, Fresenius, and Astellas as well as ownership of GSK stocks
TM reports research funding from Roche and patents with Roche.
JRF reports advisory board honoraria from Boehringer, Roche, Celgene, and AstraZeneca.
PS reports advisory board honoraria from Pfizer, Roche, Novartis, and AstraZeneca as well as speaker’s honoraria and research funding from Roche, AstraZeneca, and Novartis.
AS reports advisory board honoraria and/or speaker fees: Astra Zeneca, Bayer, Eli Lilly, Roche, BMS, Illumina, MSD, Novartis, Pfizer, Seattle Genetics, Takeda, and Thermo Fisher, and research grants from BMS, Bayer, and Chugai.
MT reports advisory board honoraria from Novartis, Lilly, BMS, MSD, Roche, Celgene, Takeda, AbbVie, Boehringer, speaker’s honoraria from Lilly, MSD, Takeda, research funding from AstraZeneca, BMS, Celgene, Novartis, Roche and travel grants from BMS, MSD, Novartis, Boehringer.
PC reports lecture/advisory board fees from AstraZeneca, Boehringer, Chugai, Novartis, Pfizer, Roche and Takeda, as well as research funding from AstraZeneca, Novartis, Roche, and Takeda.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by AstraZeneca and the German Center for Lung Research (DZL).
Data availability
Data supporting the results presented in this study are available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
