Abstract
Bladder cancer was the 10th most commonly diagnosed cancer worldwide in 2020. Extracellular vesicles (EVs) are nano-sized membranous structures secreted by all types of cells into the extracellular space. EVs can transport proteins, lipids, or nucleic acids to specific target cells. What brings more attention and potential implications is the fact that cancer cells secrete more EVs than non-malignant cells. EVs are widely studied for their role in cancer development. This publication summarizes the impact of EVs secreted by urinary bladder cancer cells on urinary bladder cancer development and metastasis. EVs isolated from urinary bladder cancer cells affect other lower-grade cancer cells or normal cells by inducing different metabolic pathways (transforming growth factor β/Smads pathway; phosphoinositide 3-kinase/Akt pathway) that promote epithelial–mesenchymal transition. The cargo carried by EVs can also induce angiogenesis, another critical element in the development of bladder cancer, and modulate the immune system response in a tumor-beneficial manner. In summary, the transfer of substances produced by tumor cells via EVs to the environment influences many stages of tumor progression. An in-depth understanding of the role EVs play in the development of urinary bladder cancer is crucial for the development of future anticancer therapies.
Introduction
Urinary bladder cancer
In 2020, urinary bladder cancer was the sixth most common cancer and the ninth leading cause of cancer deaths among men. Urinary bladder cancer is more common in men than women (440,864 new cases in men and 132,414 new cases in women in 2020). Incidence and mortality rates in 2020 were 9.5 and 3.3 per 100,000 among men and 2.4 and 0.9 per 100,000 among women, respectively. A total of 573,278 new cases and 212,536 deaths from bladder cancer were reported in 2020. In conclusion, regardless of gender, bladder cancer was the 10th most commonly diagnosed cancer worldwide in 2020. 1 More than 70% of newly diagnosed cases are non-muscle invasive bladder cancer (NMIBC). However, approximately 25%–30% of bladder cancers infiltrate the bladder muscle layer (MIBC), in such cases, the treatment of choice is radical cystectomy, which requires urine diversion after bladder removal. 2 The 5-year relative survival for patients diagnosed with stage I bladder cancer is 80% but drops to as low as 14% for those diagnosed with stage IV bladder cancer. In summary, the 5-year relative survival for all bladder cancer stages is 77%. 3
Mechanisms of bladder cancer development
There are many complex mechanisms for the origin and development of bladder cancer, the details of which are not fully understood. 4 However, several mechanisms have been clarified, and two pathways for bladder cancer have been distinguished: the invasive pathway and the noninvasive papillary pathway. 5 Bladder cancer develops when hyperplastic urothelium begins to grow toward the bladder lumen. Such growth might be caused by genetic mutations in proto-oncogenes such as fibroblast growth factor receptor-3 (FGFR3) and the viral Harvey Rat sarcoma virus (HRAS).6,7 Mutations in FGFR3 are found in 75% of papillary tumors, while mutations in HRAS are found in 30% of cases. 8 Invasive urothelial carcinomas originate from one of two mechanisms: severe dysplasia or carcinoma in situ. They often result from the inactivation of tumor suppressor pathways such as TP53, RB1, or PTEN. In contrast, mutations in phosphoinositide 3-kinase (PI3K), TSC1, PTCH, CDKN2A, and DBC1 are associated with invasive and noninvasive bladder cancer types.4,8
Processes associated with bladder cancer
Epithelial–mesenchymal transition (EMT) is a term that refers to the process of reprogramming epithelial cells. Among the most significant changes during EMT are decreased expression of surface E-cadherin, loss of expression of basal polarity genes and tight junction molecules, and increased expression of vimentin and fibronectin. 9 Loss of E-cadherin expression facilitates β-catenin release, associated with reduced anchoring to the actin cytoskeleton and poor cell mechanical stability. These changes lead to increased tumor metastasis. In turn, the interaction of N-cadherin with fibroblast growth factor receptor-1 (FGFR1) increases the expression of matrix metalloprotease 9 (MMP-9), promoting tumor invasion through the PI3K/protein kinase B (Akt) pathway. Another important protein specific to the EMT process is vimentin. It is recognized as a biomarker of this process. Among other things, it is responsible for the migration or squeezing through the narrow spaces of tumor cells by protecting them from mechanical stress. 10 In addition, three transcription factors can be distinguished that are associated with the EMT process: TWIST, SNAIL, and ZEB. Proteins of the TWIST family are basic helix–loop–helix transcription factors. Phosphorylation of TWIST1 by p38 MAPK prevents its degradation. Detection of TWIST1 protein may indicate an ongoing EMT process. 11 Proteins of the SNAIL family are zinc-finger transcription factors that are responsible, among other things, for inhibiting E-cadherin transcription. 12 ZEB transcription factors may affect the repression of epithelial genes and transcription of mesenchymal genes. Their expression is often associated with the expression of TWIST and SNAIL proteins. 13 Angiogenesis is another crucial process occurring during cancer development. The development of cancerous blood vessels is caused by abnormal levels of growth factors secreted by cancer cells, these factors can be transported via EVs. 14 Evasion of cancer cells by the immune system is a hallmark of cancer. 15 Tumor extracellular vesicles (EVs) are an important factor in promoting this mechanism. 16
Extracellular vesicles
EVs are nano-sized membranous structures secreted by all types of cells into the extracellular space. EVs serve as a mediator of intercellular communication by regulating the exchange of genetic material or proteins between cells. Based on the biogenesis, release pathways, size, content, and function, EVs can be distinguished into three main subtypes: exosomes, microvesicles, and apoptotic bodies17,18 (Figure 1). EVs can transport proteins, lipids, or nucleic acids to specific target cells. What brings more attention and potential implications is the fact that cancer cells secrete more EVs than non-malignant cells. 19

Classification of extracellular vesicles according to their size and formation mechanism.
Biogenesis of EVs
Exosomes are a subtype of EVs in the 30–250 nm diameter range. 11 They are formed by inward budding of the limiting membrane of early endosomes, maturing during this process into multivesicular bodies (MVBs).19,20 MVBs then fuse with the plasma cell membrane, releasing exosomes into the extracellular space. 21 Microvesicles are a subtype of EVs ranging from 100 nm to 1 μm in diameter. They are formed by direct outward budding and cleavage of the cell’s plasma membrane.17,18,22 Apoptotic bodies are a subtype of EVs with the largest size ranging from 50 to 5000 nm in diameter. They are released by cells undergoing apoptosis into the extracellular space. They are formed as a result of the separation of the cell’s plasma membrane from the cytoskeleton due to increased hydrostatic pressure after cell contraction.23,24
Impact of EVs on cancer development
EVs, especially exosomes, are widely studied for their role in cancer development. The cargo they carry, such as RNA, DNA, proteins, metabolites, or lipids can affect the properties of target cells. After reaching the target cell, they can directly interact with extracellular receptors, be overtaken by direct fusion with the plasma membrane, or undergo internalization. 25 The direct interaction occurs through the direct binding of cell surface receptors to transmembrane ligands on the surface of the exosome and results in the generation of a signaling cascade and activation of the target cell. 25 Using this mechanism, EVs released from dendritic cells or derived from cord blood interact to activate T lymphocytes.24 –26 Exosomes can also fuse with the plasma membrane and release their contents directly into the cytosol of target cells. This fusion is mediated by the SNAREs and Rab families of proteins, as well as adhesion molecules, lipid raft-like domains, and integrins present on their surface.26 –28 However, EVs are primarily internalized by the target cell. We can distinguish clathrin-mediated endocytosis, lipid raft-associated membrane invagination, caveolin-dependent endocytosis, phagocytosis, or micropinocytosis.29 –33 The first example of their impact is the autocrine model. Vesicular cargo can affect the EV-releasing cells’ properties, thus promoting tumor growth. 34 Paracrine mechanisms are another example of their interaction with target cells. Tumor cells, through their secretion, can affect not only themselves but also neighboring tumor cells. EVs can activate receptors or alter miRNA or RNA expression in neighboring tumor cells causing, for example, an increase in their migratory, invasive, or proliferation potential.35 –37 The molecules they carry are not only transported between cancer cells. They can also transfer information between cancer cells and other types of cells, such as stromal cells. EVs from cancer cells can affect the stromal cells, causing the creation of a pro-cancer microenvironment38 –40 and can affect endothelial cells to promote angiogenesis resulting in increased tumor growth.41 –43 Another aspect of tumor growth that they also influence is their contribution to drug resistance. EVs can help cancer cells excrete cytotoxic drugs.44,45 Moreover, it was found that EVs secreted by drug-resistant cells can induce drug resistance in drug-sensitive target cells.46 –48 They also have a major impact on cancer metastasis. They can modulate the extracellular matrix to promote cell invasion49,50 and promote EMT of target cells, which is an essential process for tumor invasion and metastasis.51,52 EVs can also modulate the immune response by inhibiting T and NK cell activation, suppressing the activity of dendritic cells (DCs) and increasing the expansion of myeloid-derived suppressor cells and stimulating macrophage polarization toward the tumor-promoting M2 phenotype.53 –61
Materials and methods
To present the research situation regarding the impact of EVs on the development and metastasis of bladder cancer, publications from 2013 to April 2021 were selected. The PubMed database was selected to search for articles. Keyword combinations were selected for the search: ‘urinary bladder’ + ‘cancer’ + ‘development’/‘metastasis’ + ‘exosomes’/‘extracellular vesicles’. A total of 78 articles were found. A total of 56 of them were excluded during abstract screening as they were review articles or were not connected with the subject of this study. In addition, 11 studies were disqualified for having a different source of EVs than bladder cancer cells and no in vitro cell studies (Figure 2).

Literature screening flow diagram.
The effect of EVs on the growth and development of bladder cancer
EVs secreted by tumor cells of higher malignancy affect tumor cells of lower malignancy by promoting their migratory, proliferative, and invasive potential. Yoshida et al. 62 studied the effect of EVs secreted by bladder cancer cells on the development and metastatic capacity of the tumor. They focused on the CRK protein, overexpression of which has been detected in various human cancers, where it induced the EMT.63 –66 A high-grade bladder cancer cell line UM-UC-3 was selected for the study: UM-UC-3. EVs were isolated from medium conditioned from UM-UC-3 cells and CRK-silenced UM-UC-3 cells. The effectiveness of the isolation was confirmed only by labeling the exosome-specific marker CD63. Unfortunately, the cited study did not analyze the protein and qualitative and quantitative analyses of the EVs used. Mass spectrometry analysis confirmed that ErbB2, integrin α2, ErbB3, and FAK proteins are downregulated in EVs isolated from CRK-silenced UM-UC-3 cells. These are potential effector proteins in the development and metastasis of bladder cancer. The effect of EVs secreted by UM-UC-3 cells (grade: CIS) on 5637 cells (grade: G2) was evaluated. It was observed that the proliferation and invasiveness of 5637 cells significantly increased while treated with wild-type UM-UC-3 released EVs, compared to a sample of EVs released by CRK-silenced cells. In addition, increased expression levels of ErbB2 and ErbB3 were observed in 5637 cells treated with EVs from UM-UC-3. 62 High levels of ErbB2 expression in bladder cancer patients correlated with an increased propensity for metastasis and a poor prognosis. 67 To further determine the effects of EVs on tumor angiogenesis, the effects of EVs secreted by UM-UC-3 cells on human umbilical vein endothelial cells (HUVECs) were assessed. The proliferation and chemotactic capacity of HUVECs treated with EVs increased significantly. In addition, the expression levels of ErbB2, ErbB3, and CRK-II and the phosphorylation levels of FAK and AKT increased in the target cells. It was also discovered that the level of ErbB2 protein contained in invasive exosomes derived from bladder cancer cells is CRK dependent. The exosomes tested may enhance cancer cell proliferation and invasion and activate FAK and PI3K/AKT signaling. 62 This effect on angiogenesis is crucial for cancer cells, as they require oxygen and nutrients for survival and proliferation. 68 Xue et al. 69 focused on two key factors influencing bladder cancer development: hypoxic conditions and oncogenic lncRNA urothelial cancer-associated 1 (UCA1). Long non-coding RNAs (lncRNAs) play an important role in cancer progression. 70 Hypoxia is considered one of the most fundamental stresses of the solid tumor microenvironment, which negatively affects tumor expansion. 71 However, tumor cells transform the surrounding microenvironment in response to hypoxia for survival and optimal growth and subsequently promote invasion. 72 UCA1, on the other hand, is an oncogenic lncRNA that is overexpressed in bladder cancer tissues. 73 It is responsible for bladder cancer cell proliferation by affecting several different metabolic pathways: cAMP response element-binding protein (CREB), chromatin remodeling factor (BRG1), PI3K, protein kinase B (AKT), and Wnt pathways.74 –76 It has been shown that lncRNA-UCA1 can promote migration and invasion of bladder cancer cells and EMT. 77 It then evaluated the effects of normoxic and hypoxic exosomes secreted by 5637 cells (grade: G2) on the proliferation, migration, and invasion of UMUC2 bladder cancer cells (grade: CIS). EVs were isolated from conditioned media from 5637 cells cultured under normoxic or hypoxic conditions. The results of the characterization of EVs showed that both hypoxic and normoxic populations of isolated EVs were mainly exosomes. 69 Both normoxic and hypoxic exosomes increased the viability, motility, and migratory ability of low-grade cells. However, hypoxic exosomes had a higher effect than normoxic exosomes. Subsequently, the presence of lncRNA-UCA1 was confirmed in both hypoxic and normoxic exosomes. To confirm whether exosomal lncRNA-UCA1 is an effector of UMUC2 cell changes, its expression was suppressed in hypoxic 5637 cells. Knockdown of lncRNA-UCA1 in hypoxic exosomes led to reduced proliferation, mobility, and invasiveness of UMUC2 cells. In addition, exosomal lncRNA-UCA1 was observed to promote tumor progression through EMT. These results confirm that hypoxia increases the transport of exosomal lncRNA-UCA1 into bladder cancer cells. Furthermore, lncRNA-UCA1 itself increases the proliferation of migration and invasion of bladder cancer cells, which is associated with tumor growth and progression. 69 Huang et al. 78 evaluated the effects of EVs isolated from T24 (grade: G3) and J82 (grade: G3) cells on TSGH-8301 (grade: G2) cells. Based on the analysis of the EVs obtained, it was confirmed that the isolation was successful and that the exosomes constitute a population. Exosomes secreted by T24 and J82 cells significantly increased the viability of TSGH-8301 cells, especially J82 exosomes, which showed a stronger effect after 24 h of treatment. Furthermore, exosomes secreted by T24 and J82 cells also significantly promoted the migratory, invasive, and clonogenicity abilities of TSGH-8301 cells. Next, the effect of exosomes on the expression of EMT-associated molecules’ was examined. In TSGH-8301 target cells, exosomes secreted by T24 and J82 cells significantly increased protein expression levels of EMT effectors Snail, Slug, and Zeb2. In addition, exosomes secreted by T24 cells increased protein expression levels of Twist. The next stage of the study was to compare markers of epithelial-like or mesenchymal-like properties. It was observed that after the effect of exosomes secreted by T24 and J82 cells, the epithelial marker E-cadherin was significantly decreased, and the mesenchymal markers N-cadherin, α-SMA, fibronectin, and the interstitial indicator vimentin were increased. Exosomes secreted by T24 and J82 cells also increased the expression levels of metalloproteinases MMP-2 and MMP-9 in target cells. The three major upstream signaling cascades of the EMT process were then analyzed, including β-catenin/TCF4, Notch/HES1, and transforming growth factor β (TGF-β)/SMAD signaling. Exosomes secreted by T24 or J82 cells increased protein expression levels of β-catenin, TCF4, Notch1, Notch4, HES1, and Smad2/3. However, TCF4 was elevated after treatment with T24 exosomes but was not altered by treatment with J82 exosomes. These results suggest the presence of EMT regulators in the exosomes derived from the highly malignant bladder cancer cells, which led to the induction of the EMT process in TSGH-8301 target cells. One of the causes of the changes described above in TSGH-8301 cells are LINC00960 and LINC02470, which are transported by exosomes derived from T24 and J82 cells. The differences between the effects of T24 and J82 exosomes may be due to the reduced amount of LINC02470 transported in J82 exosomes compared to T24 exosomes. To confirm these findings, the expression of LINC00960 and LINC02470 was reduced in T24 cells. It was noted that exosomes isolated from these cells showed significantly reduced inductive effects on the viability, migration, invasion, and clonogenicity of target cells. In addition, levels of Snail, Slug, vimentin, MMP-2, MMP-9, N-cadherin, α-SMA, and fibronectin were significantly reduced, and levels of E-cadherin were elevated. These data suggest that exosomes isolated from T24 cells with decreased expression of LINC00960 and LINC02470 reduced the expression level of EMT-related molecules in TSGH-8301 target cells compared to exosomes secreted by T24 cells. In conclusion, they have shown that exosomes derived from high-grade bladder cancer cells promoted the malignant features in the low-grade bladder cancer cells. 78 Yang et al. 79 isolated EVs from T24 (grade: G3) bladder cancer cells and then evaluated their effects on 5637 (grade: G2) and T24 cells to assess a model of the paracrine and autocrine effects of EVs, respectively. The tested exosomes significantly uplifted the viability of 5637 and T24 cells in a time- and dose-dependent manner. Viability increased by 76.52% and 73.26%, for 5637 and T24 cells, respectively. In addition, the T24-derived exosomes inhibited apoptosis of the target cells. Exosomes affected 5637 cells by increasing Bcl-2 mRNA and protein levels, decreasing Bax mRNA and protein levels, and downregulating caspase-3 expression. Exosomes affected T24 cells by increasing the Bcl-2 mRNA and protein levels, increasing expression levels of cyclin D1, increasing expression levels of phosphorylated Akt and ERK1/2, and decreasing the levels of Bax mRNA and protein. These results indicate that activation of Akt and ERK pathways may be involved in the exosome-stimulated proliferation of bladder cancer cells. 79 A summary of the effects of EVs secreted by bladder cancer cells on bladder cancer cells of other types is illustrated in Table 1.
Effect of extracellular vesicles secreted by bladder cancer cells on bladder cancer cells of other types.
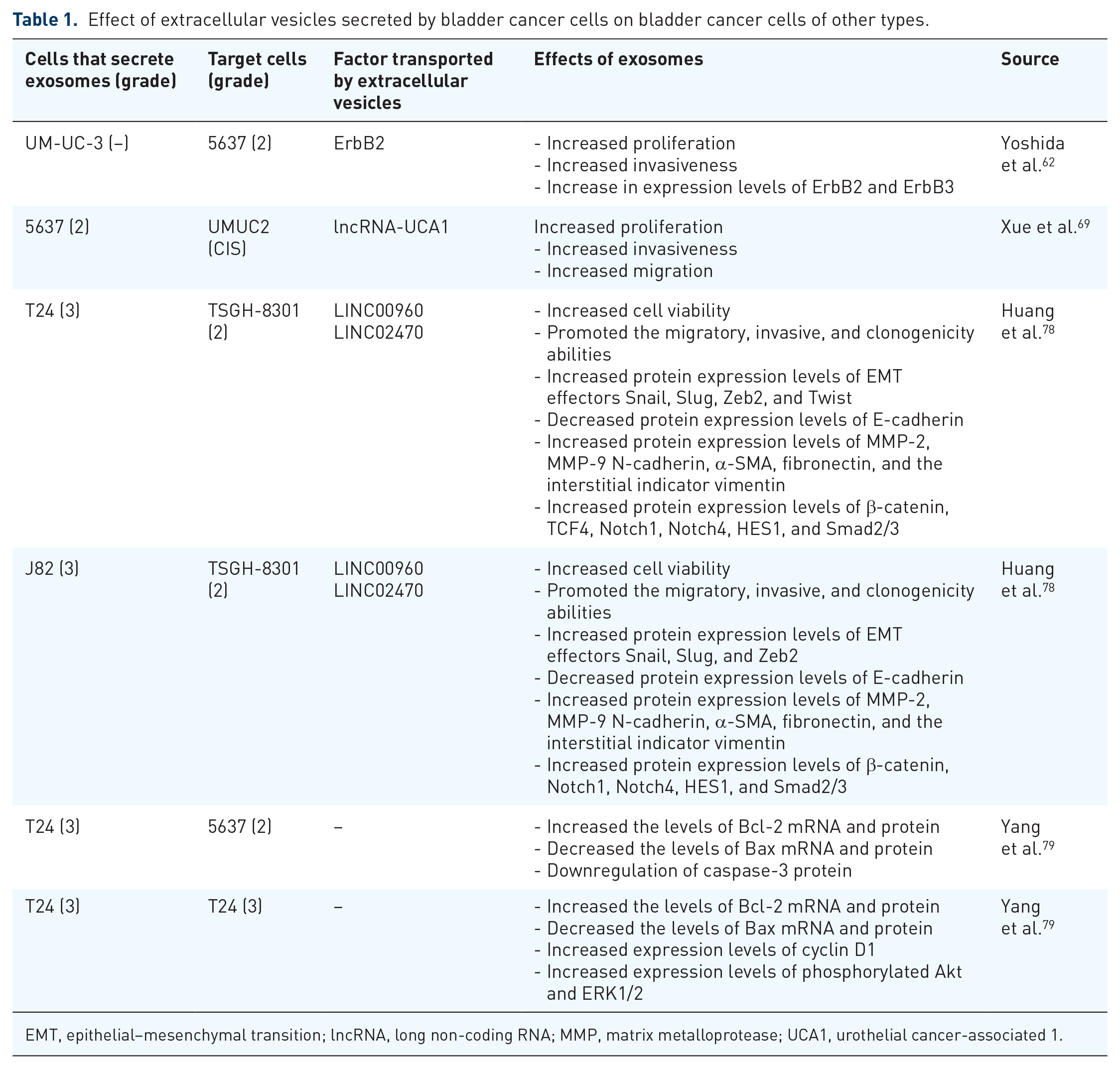
EMT, epithelial–mesenchymal transition; lncRNA, long non-coding RNA; MMP, matrix metalloprotease; UCA1, urothelial cancer-associated 1.
The effect of EVs on the environment of bladder cancer
Metastasis is a major cause of mortality in cancer patients and is caused by the extravasation of circulating tumor cells (CTCs) through the endothelium and the subsequent formation of tissue micrometastases. Exosomes can be transported into the interstitial space and spread throughout the body. Thus, they may be responsible for forming the pre-metastatic niche.80,81 Whitehead et al. 82 evaluated the effects of mechanical and innate immune interactions of exosomes isolated from T24 (grade: G3) and FL3 (grade: G3) bladder cancer cells and immortalized, non-malignant HCV-29 control urothelial cells on endothelial integrity. Obtained exosomes were evaluated using nanoparticle tracking analysis (NTA), western blot, and liquid quantitative nanomechanical mapping atomic force microscopy (AFM QNM) visualization. In NTA analysis, it was observed that exosomes from all cell lines showed similar sizes. Nanomechanical analysis showed a marked reduction in the stiffness of exosomes from malignant cells compared to exosomes from control cells. In addition, adhesion assessment showed a lower value for exosomes derived from malignant cells than those from control cells. The biological effect of the applied exosomes was examined by measuring the perturbation of the transendothelial electrical resistance (TEER) monolayer of HUVEC cells. In the case of exosomes derived from malignant cells, the formation of a HUVEC monolayer was observed, followed by its disruption, demonstrated by a decrease in TEER measurements. Moreover, exosomes from malignant cells disrupted the monolayer to a much greater extent than exosomes from control cells. The transport of exosomes through the intact endothelial monolayer was also evaluated. A significant increase in transport was observed for exosomes derived from malignant cells compared to exosomes derived from control cells. In addition, the level of complement activation under the influence of exosomes was checked. Three serum complement markers were used: anaphylatoxin, chemoattractant factor C5a, and soluble SC5b-9. The results showed that all exosomes induced complement activation. However, exosomes of malignant cell lines were more potent in complement activation than exosomes derived from non-malignant HCV-29 cells. These results suggest that mechanical properties of exosomes, such as reduced stiffness, might affect transport across biological membranes. These data suggest that malignant exosomes induce endothelial leakage, which may be a prerequisite for subsequent metastasis. 82 Lin et al. focused on the effect of exosomes secreted by T24 bladder cancer cells on the immune system. They evaluated their effects on human THP-1 acute monocytic leukemia cells differentiated into M0 macrophages. 83 Macrophages can be induced to exhibit an immunologic M1 phenotype that inhibits tumor growth or an M2 phenotype that promotes tumor growth and immunosuppression. 84 Similar to M2 macrophages, tumor-associated macrophages can promote metastasis by promoting angiogenesis, degrading the extracellular basal layer, and increasing cell motility. 85 It was observed that exosomes derived from T24 cells can promote M2 macrophage polarization. IL-10 and TGF-β are cytokine markers indicating macrophage polarization towards the M2 phenotype. The mRNA levels of IL-10 and TGF-β were significantly upregulated in exosome-treated macrophages compared with control macrophages. In addition, exosome-treated macrophages enhanced T24 cell migratory and invasive abilities. It was found that miR-21 transported by exosomes was a key inducer of macrophage polarization. It was found that miR-21 downregulates PTEN gene expression. In addition, the expression of p-AKT and p-STAT3 was higher in exosome-treated macrophages than in untreated macrophages. These data suggest that miR-21 directly downregulates PTEN and further enhances PI3K/AKT-induced STAT3 signaling activity. 83 Zheng et al., 86 based on analysis of urine samples from cancer patients, found that the exosomal lncRNA of BCYRN1 is correlated with bladder cancer lymph node metastasis. Subsequently, it was observed that the expression level of BCYRN1 was significantly increased in exosomes secreted by bladder cancer cells (T24: grade G3, 5637 grade: G2 and UM-UC-3) compared to the level in exosomes secreted by human normal bladder epithelial cells (SV-HUC-1). In addition, higher levels of BCYRN1 were found in exosomes secreted by bladder cancer cells compared to its intracellular levels. Isolated exosomes from bladder cancer cells dramatically increased the tube formation and migration of both human lymphatic endothelial cells (HLECs) and human dermal lymphatic endothelial cells (HDLECs). These results suggest that overexpression of exosomal BCYRN1 promotes bladder cancer lymphangiogenesis in vitro. Subsequently, exosomal BCYRN1 was confirmed to increase lymphangiogenesis and lymph node metastasis in mouse bladder cancer in vivo models. Studying the effector function of BCYRN1, it was observed to activate WNT5A transcription by binding to its promoter to form a DNA–RNA triplex structure and recruiting hnRNPA1 to increase its levels. WNT5A functions as an important glycoprotein ligand that activates the Wnt/β-catenin signaling pathway. 87 VEGF-C is a key inducer of tumor lymphangiogenesis. 88 Subsequent analyses confirmed that BCYRN1 activates the Wnt/β-catenin signaling pathway to promote VEGF-C secretion in bladder cancer. VEGFR3 is extensively involved in the sprouting of the lymphatic vascular networks and the spread of tumors by initiating the budding of endothelial cells. 89 Subsequent results confirmed that VEGFR3 expression was significantly upregulated in HLECs with tumor cell exosomes. In conclusion, the study’s results demonstrated a key role for exosomal BCYRN1 in lymphangiogenesis and metastasis of bladder cancer. 86 SUMOylation or small ubiquitin modifier (SUMO) binding is a key post-translational modification that regulates intracellular transport and signal transduction by mediating protein stability and subcellular localization. 90 It has been observed that SUMOylation plays an important role in the packaging of EVs by mediating the recognition of molecules by the endosomal sorting complex required for transport and facilitating their loading into the MVB.91,92 Based on an analysis of databases and cancer patient samples, Chen et al. 93 focused on the lncRNA ELNAT1. They observed ELNAT-1-high expression in both tumor tissues and EVs of cancer cells. UM-UC-3 and T24 bladder cancer cells were used for further studies. The effect of tumor EVs on lymphangiogenesis was then examined. The tube formation and migratory capacity of HLECs were markedly increased after incubation with EVs secreted by UM-UC-3 and T24 cells. The in vitro results were confirmed in a mouse animal model. The molecular mechanism responsible for the changes described above was then determined. It has been observed that exosomal ELNAT1 regulates UBC9 transcription by forming a triplex structure with the promoter sequence, inducing hnRNPA1-related modification of H3K4me3. It has been confirmed that UBC9 can catalyze SUMOylation of target proteins to regulate their interaction with biomolecules. 94 Subsequently, ELNAT1 was confirmed to be packaged into EVs secreted by bladder cancer cells by UBC9-induced SUMOylation of hnRNPA1. Subsequent analyses revealed that exosomal ELNAT1 promoted lymphangiogenesis through transcriptional regulation of SOX18 expression in HLECs. In conclusion, exosomal ELNAT1 has been shown to promote lymphangiogenesis and metastasis of bladder cancer in a SUMOylation-dependent manner. 93 Chen et al. 95 conducted another study analyzing the lncRNA LNMAT2. LNMAT2 expression positively correlated with bladder cancer metastasis in a group of 266 patients studied; moreover, LNMAT2 overexpression correlated with shorter overall survival and disease-free survival. The presence of LNMAT2 was observed in exosomes isolated from the urine of sick patients and in exosomes isolated from the conditioned medium of cancer cells (5367, UM-UC-3). In in vitro tests, it was observed that exosomes isolated from tumor cells promoted tube formation and migration of HLEC cells. These results suggest that exosomal LNMAT2 contributes to lymphangiogenesis in vitro. These results were also confirmed in vitro using a mouse animal model. The next step in the study was to find out the molecular mechanism responsible for the observed changes. It has been observed that LNMAT2 is packaged into exosomes in a hnRNPA2B1-dependent manner. 96 Studies indicated that VEGF-C produced by tumor cells contributes to lymphangiogenesis and metastasis of bladder cancer 97 ; however, no effect of LNMAT2 on VEGF-C protein expression was observed. Prospero homeobox 1 (PROX1) is crucial in the formation of the lymphatic vascular system and is responsible for the regulation of endothelial cell differentiation and induction of lymphatic endothelial cell budding. 98 In subsequent assays, it was observed that exosomal LNMAT2 forms a triplex with the PROX1 limpet promoter sequence and upregulates its transcript levels. The results of the cited study indicated a VEGF-C-independent mechanism for bladder cancer metastasis, in which exosomal LNMAT2 promotes lymphangiogenesis and metastasis through transcriptional regulation of PROX1 in HLECs. 95 Goulet et al. 99 hypothesized that the EVs of bladder cancer can affect and alter the function of fibroblasts surrounding the tumor. EVs were isolated from RT4 (grade: G1), T24 (grade: G3), and SW1710 (grade: G3) cells. The uptake of exosomes by normal fibroblasts was then examined. It was observed that exosomes derived from RT4, T24, and SW1710 cells internalized target cells and accumulated around the nuclei of normal fibroblasts. In addition, the exosomes tested promoted the proliferation of normal fibroblasts in in vitro culture. Cancer-associated fibroblasts (CAFs) are a key component of the tumor microenvironment with diverse functions.100 –102 CAFs are characterized by increased expression of α-SMA and FAP, and Galectin proteins.103,104 The EVs tested resulted in increased expression levels of α-SMA, FAP, and Galectin proteins. These results indicate the differentiation of normal fibroblasts into CAFs. TGF-β is involved in the differentiation of normal fibroblasts into CAFs, and TGF-β was found in exosomes isolated from RT4, T24, and SW1710 cell lines. In addition, TGF-β transported by EVs was observed to induce the differentiation of normal fibroblasts into CAFs by activating a signaling cascade involving phosphorylation of SMAD2 and 3. 99 Wu et al. 105 evaluated the effects of EVs secreted by TCCSUP (grade: G4) cells on non-malignant human SV-HUC urothelial cells. Loss of inhibition of proliferation by cell-to-cell contact is one of the hallmarks of cancer cells. It was observed that SV-HUC cells acquired resistance to inhibition of proliferation by cell-to-cell contact under the influence of EVs secreted by TCCSUP cells. Another hallmark of cancer is increased genomic instability, which was assessed by examining the expression levels of the antioxidant stress gene: SOD and the DDR genes GADD45A, GADD45B, GADD45G, and RAD50. 106 Their expression levels were elevated in cells treated with EVs. In addition, significantly higher levels of reactive oxygen species (ROS) were observed in cells treated with EVs. The level of DNA damage was then assessed by measuring histone H2AX phosphorylation. The nuclei of cells treated with EVs had significantly more γH2AX foci than untreated cells. These results suggest that cells exposed to tumor-derived EVs have increased genome instability. An increase in invasiveness characterized cells treated with tumor-derived EVs. These results are confirmed by elevated levels of N-cadherin mRNA expression and decreased levels of E-cadherin mRNA expression, associated with EMT in target cells. 105 The unfolded protein response of the endoplasmic reticulum (UPRER) pathway was responsible for these changes. Persistent over-activation of UPRER causes pathological changes and potentially leads to malignancy. 107 A summary of the effects of EVs secreted by bladder cancer cells on normal cells is illustrated in Table 2.
Effects of extracellular vesicles secreted by bladder cancer cells on normal cells.

HDLEC, human dermal lymphatic endothelial cells; HLEC, human lymphatic endothelial cells; HUVEC, human umbilical vein endothelial cells; TGF-β, transforming growth factor β.
Discussion
In conclusion, depending on the transported cargo, EVs can differentially affect the development and metastasis of bladder cancer. The impact is evident at different stages of development. This interaction can affect neighboring tumor cells by promoting their proliferative potential, migratory, and invasive properties. EMT is the key process involved in tumorigenesis; the vast majority of bladder tumors undergo EMT during tumor progression 108 (Figure 3, Table 1). Another group of target cells is the cells surrounding the tumor. EVs from tumor cells can affect stroma cells, causing a pro-cancerogenic microenvironment or endothelial cells promoting angiogenesis, resulting in increased tumor growth. Angiogenesis is another crucial element in the development of bladder cancer. Tumors rapidly develop a new vascular network to promote a high proliferation rate of tumor cells 109 (Figure 4, Table 2). Another critical factor is the effect on the immune system, as the EVs can induce drug resistance or support tumor cells in shedding cytotoxic drugs. Tumor-derived EVs also have the effect of inhibiting T and NK cell activation, suppressing the activity of DCs. In addition, they increase the expansion of myeloid-derived suppressor cells and stimulate the polarization of macrophages towards the tumor-promoting M2 phase, which enhances the promotion of the development of the niche/tumor microenvironment (Figure 5, Table 2).110

The effect of cargo carried by EVs on the EMT process of target cells.

The effect of cargo carried by EVs on the angiogenesis process of target cells.

The effect of cargo carried by EVs on the immune response.
According to the International Society for Extracellular Vesicles guidelines for the characterization of EVs based on the presence of characteristic proteins, at least one protein from the following categories should be determined: trans-membrane or GPI-anchored proteins associated with the plasma membrane and/or endosomes; cytosolic proteins recovered in EVs and major components of unisolated EV structures. 110 Unfortunately, the EVs used were not always adequately characterized in the studies used. Four of the studies relied on protein characterization of EVs based on only one category of proteins, namely trans-membrane proteins or GPI-anchored proteins associated with the plasma membrane and/or endosomes.62,78,79,99 The following markers were analyzed: CD63 and CD9. Determination of protein markers from two categories: trans-membrane or GPI-anchored proteins associated with the plasma membrane and/or endosomes, and cytosolic proteins recovered in EVs were observed in five studies.69,86,93,95,105 The following markers were analyzed for group 1: CD63, CD9, and CD81, respectively; for group 2: TSG101, Hsp70, Hsp90, ALIX, and Flotillin. Only one study used all three categories of proteins: from Group 1: CD81; Group 2: Syntenin, TSG101; and Group 3: Calreticulin and VDAC1 (Figure 6). 83 Accurate characterization of isolated EVs is a crucial element when studying their molecular effects on different types of cells. We need to be sure that a given effect is caused by EVs and not by other co-isolated molecules. In future studies using EVs, greater awareness of the need to follow the guidelines of the International Society for Extracellular Vesicles is essential.

Proteomic characterization of used extracellular vesicles. Category I: Transmembrane or GPI-anchored proteins associated with the plasma membrane and/or endosomes, Category II: Cytosolic proteins recovered in EVs, Category III: Major components of non-EV co-isolated structures.
In summary, the transfer of substances produced by tumor cells via EVs to the environment influences many stages of tumor progression such as activation of proliferative and angiogenic pathways or modulation of the immune response. EVs secreted by tumor cells can promote the tumorigenesis of neighboring cells by transforming or affecting their proliferation. They can also beneficially modify the tumor’s growth environment by promoting inflammation or remodeling the extracellular matrix. EVs can therefore be used as a target for therapy. Moreover, since EVs accumulate in readily available body fluids, they may become a potential biomarker for bladder cancer. All of these facts provide non-negligible evidence of the involvement of EVs in bladder cancer progression and the necessity of further studies as both diagnostic and therapeutic targets.
