Abstract
Objective
To uncover the clinical significance of galectin-3 in the evolution of urinary bladder cancer by defining galectin-3 expression and examining the relationship between its expression in a group of urothelial carcinomas versus normal tissues along with clinicopathological factors.
Methods
This retrospective study included histopathological reports and archival blocks and slides of all patients with urinary bladder cancer treated at King Abdulaziz University Hospital (Jeddah, Saudi Arabia). An anti-galectin-3 monoclonal antibody was used for immunohistochemical staining of tissue microarray slides comprising 128 cases of urothelium carcinoma and 24 specimens of normal bladder mucosa.
Results
Galectin-3 was downregulated during transformation, with positive expression found in 50 (39%) urinary bladder neoplasms, of which 33 (66%) showed weak immunostaining. All positively-stained malignant tumor and normal bladder mucosa samples showed cytoplasmic staining; a few samples also showed nuclear staining. No correlation was noted between galectin-3 and histotype, grade, stage, muscularis propria invasion, lymph node invasion, vascular invasion, or metastasis. A Cox proportional hazards model and Kaplan–Meier survival curves did not show differences in survival on the basis of galectin-3 expression.
Conclusion
Galectin-3 is down-regulated in bladder cancer but is not a helpful marker for the diagnosis or prognosis of urinary bladder cancer.
Keywords
Introduction
Galectin-3 is a member of a special family of glycoproteins called lectins. Lectins are distinguished by their specificity for binding carbohydrate sequences (β-galactosides).1–2 Galectin-3 is a 31-kDa protein that is encoded by the LGALS3 gene located on chromosome 14.1–2 Galectin-3 protein is expressed in several human organs and tissues as well as by some malignant tissues. Galectin-3 functions in several subcellular locations including the nucleus, cytoplasm, and cellular membranes. It also mediates cellular communications with components of the extracellular matrix.3–6 Galectin-3 has several functions because it is expressed in different locations and can attach to a variety of ligands.3,4,7–9 It also takes part in the transformation process and carcinogenesis. Several studies have shown that galectin-3 plays significant roles in a collection of neoplasm-associated biological activities including apoptosis, differentiation, adhesion, proliferation, migration, angiogenesis, invasion, and metastasis.5–11
Previous studies have reported galectin-3 expression in several neoplasms and have attempted to estimate its diagnostic utility as a predictive factor for malignant progression. However, there remains a significant debate because galectin-3 has been found to be both over- and under-expressed in cancerous tissues compared with non-malignant tissues. Several studies have also suggested that increased galectin-3 expression indicates poor prognosis while others have concluded otherwise. 12 Increased galectin-3 expression has been found in mammary gland cancer, 13 stomach cancer, 14 liver cancer, 15 skin cancer, 16 cervical cancer, 17 tongue cancer, 18 and thyroid neoplasms. 19 In different reports, galectin-3 was found to be under-expressed in various neoplasms such as head and neck cancer, 20 prostatic carcinoma, 21 mammary gland neoplasms, 22 gastric cancer, 23 colorectal cancer, 24 and cervical cancer. 25 These discrepancies suggest that the engagement of galectin-3 in the transformation process is tumor-specific. However, there is inadequate information regarding the clinical value of galectin-3 immunostaining in bladder carcinoma.
Urinary bladder tumors are the thirteenth leading cause of cancer deaths worldwide. 26 Urinary bladder carcinoma is the sixth most frequent tumor, with approximately 81,400 newly registered neoplasms and approximately 17,980 bladder cancer deaths in the USA in 2019. 27 Despite the substantial progresses in cancer management, bladder neoplasms remain a huge challenge because of their high frequency of recurrence, with up to 70% of newly registered tumors recurring within 5 years, often developing into destructive, muscle-invasive, and metastatic tumors.28–29 Managing urinary bladder carcinoma greatly depends on the clinicopathological factors of patients, e.g., TNM stage and tumor grade are prognostic indicators. Still, these clinicopathological features are insufficient to forecast patients’ outcomes and generate considerable discrepancies within similar grades or stages. This is essentially linked to the heterogeneity of urinary bladder tumors. 30 Therefore, there is an urgent need to discover better markers for diagnosis, treatment response, risk stratification, and clinical management.31–32 Most current markers are not reasonably sensitive or specific, which has increased interest in markers than can are specific for malignant cells and the neoplasm milieu to assist in prognosis and diagnosis and give indications for therapy.
Therefore, this report defines galectin-3 expression and examines associations between its expression and clinical factors to determine the clinical importance of galectin-3 in the development of urothelial carcinoma. Moreover, this report assessed whether galectin-3 expression could be a useful biomarker and/or predictive factor for survival in urinary bladder carcinoma patients.
Materials and methods
This project was performed in the Laboratory of Pathology over a 2-year period, ending on 15 April 2022 and was approved by the Unit of Biomedical Ethics, Research Committee at King Abdulaziz University (Reference No. 77-21, 11 April 2021). Informed consent was waived by the Biomedical Ethics Committee as the research project used archived material. The practices followed were consistent with the 1975 Helsinki Declaration and its later amendments. Written informed consent is routinely obtained from all patients who undergo a surgical procedure at our institution for use of tissue samples in laboratory studies. This manuscript follows the guidelines of studies of diagnostic accuracy (STARD).
Cases with history of intravesical chemotherapy were excluded. Paraffin-embedded tissue blocks were cut into 4-µm slices, stained with hematoxylin and eosin (H&E), and reevaluated. Features of the neoplasm and patient clinicopathological information (sex and age, tumor histotype, stage, and metastasis) were gathered from the medical records of King Abdulaziz University Hospital. All normal bladder mucosa tissues were collected from patients who were biopsied for non-cancerous diseases. The World Health Organization recommendation was adapted for bladder cancer staging.
Tissue microarray (TMA) construction
The TMA was constructed as illustrated in an earlier paper. 33 All 128 bladder carcinoma and 24 control specimens were used. TMA blocks were cut into 4-µm sections and placed on aminosilane-coated slides.
Immunohistochemistry
Immunohistochemistry was performed using an autostainer (BenchMark ULTRA, Ventana, AZ, USA) in accordance with the manufacturer’s instructions. 34 A galectin-3 monoclonal antibody (Cell Marque, Rocklin, CA, USA; 1:100) was used in the immunohistochemical staining protocol. Staining was visualized by the ULTRAVIEW TM DAB system. A 4-µm-section of placenta specimen was used as a positive control. A slide treated with Tris buffer in place of galectin-3 antibody was used in every run of staining as a negative control. Cases that showed brown granular cytoplasmic staining in >5% of malignant cells were considered positive. Immunohistochemical results were analyzed by two pathologists. The average rate of cells with galectin-3 expression was estimated using a semiquantitative technique (three fields of view using a 40× lens). Our laboratory scoring system for IHC was applied for galectin-3 staining intensity using 1, 2, 3, and 0 for negative. Grades of staining are displayed as two groups: negative (0) and positive (1, 2 and 3).
Statistical analysis
All data were analyzed using SPSS v21 software (IBM, Armonk, NY, USA). All results are shown as numbers and percentages. Associations between clinicopathological data of bladder carcinoma patients and galectin-3 expression were investigated via the chi-squared and Fisher tests. A Cox proportional hazards model was used to determine if any clinicopathological factors had an important influence on survival. The assessment of survival distributions for different galectin-3 staining intensity scores was performed using a Kaplan–Meier survival curve. The significance level was set at p < 0.05.
Results
Paraffin-embedded tissues of 128 selected cases out of 211 total urinary bladder carcinomas representing all grades and stages were collected from the archives of the Pathology Department along with a control group (24 normal bladder samples). All 128 urinary bladder cancer cases (105 males and 23 females) were revised; clinicopathological parameters of the cases are displayed in Table 1. The median age was 62.4 years (range: 31–93 years). The most common type was urothelial carcinoma (82%) and less frequently squamous differentiation variant (13.3%) and squamous cell carcinoma (4.7%). We staged 118 cases of bladder cancer, and 116 were graded. The total number of deaths from bladder cancer was 38 (29.7%). Sixty-four cases showed muscularis propria invasion, 19 had vascular invasion, 22 had lymph node involvement, and 25 had distant metastases (Table 1). Of note, male patients showed considerably increased galectin-3 immunohistochemical staining.
Distribution of various clinicopathological variables according to galectin-3 expression in malignant urothelial carcinoma patients.

Galectin-3 was down-regulated during transformation, with positive expression found in 50 (39%) urinary bladder neoplasms, of which 33 (66%) showed weak immunostaining (Figure 1). Ninety percent of positive tumor cases showed immunostaining in <50% of malignant cells. In contrast, among the 24 normal bladder mucosa samples, 12 (50%) had weak to moderate galectin-3 expression. All positive staining in both malignant tumors and normal bladder mucosa was cytoplasmic, with a few samples also showing nuclear staining.
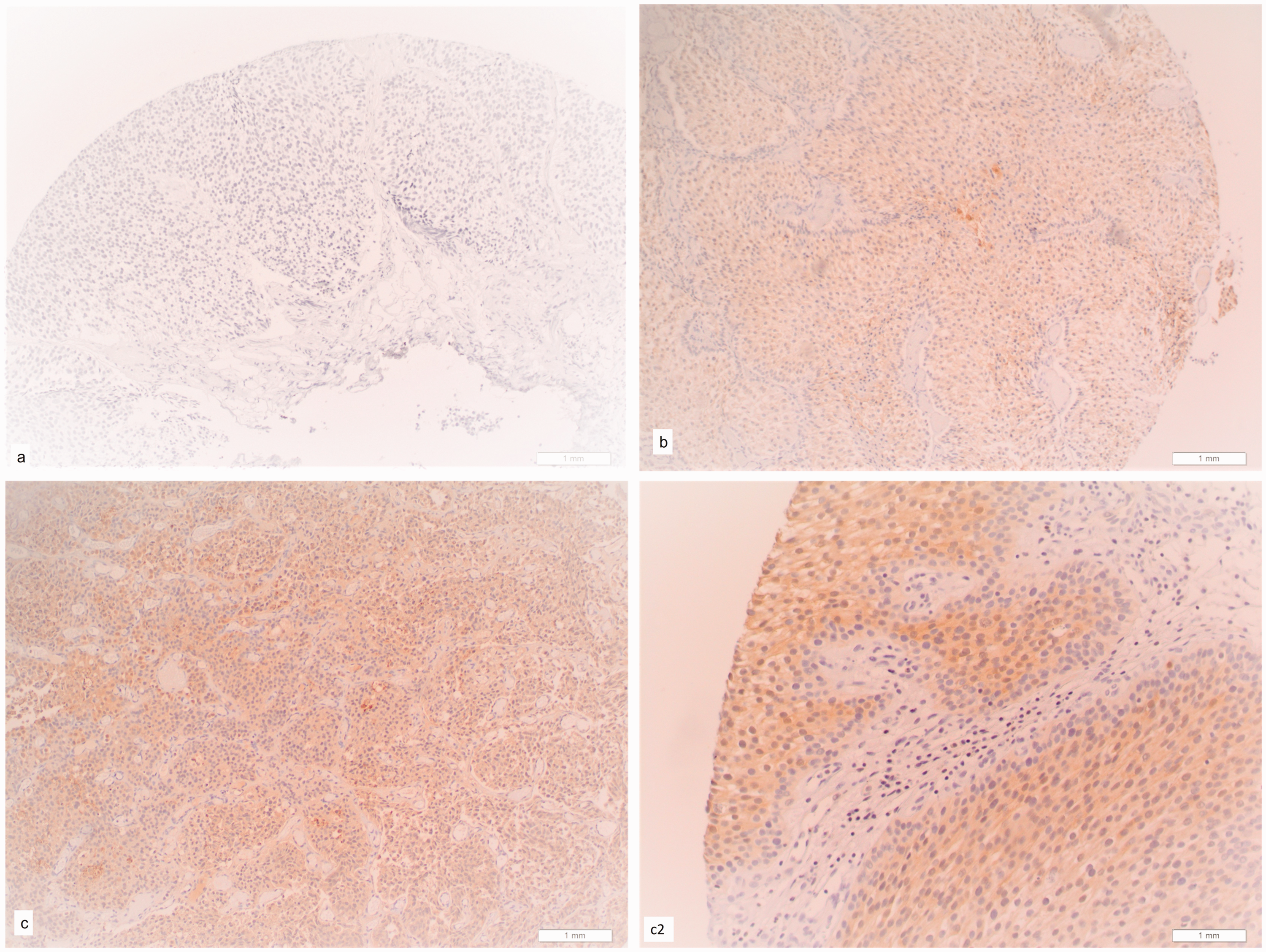
Galectin-3 immunohistochemistry staining patterns in bladder cancer. a, negative galectin-3 staining in bladder cancer (10× magnification); b, weak galectin-3 staining in malignant urothelium of bladder cancer (10× magnification); c, moderate galectin-3 staining in malignant urothelium of bladder cancer (10× magnification); c2, moderate galectin-3 staining in malignant urothelium of bladder cancer (20× magnification).
Regarding the histologic type of bladder cancer, 86% of positive cancer cases were urothelial carcinomas, but no important correlation was noted between galectin-3 and tumor histotype (p = 0.467). Galectin-3 immunohistochemical staining showed similar patterns of frequency, intensity, and distribution in both low- and high-grade tumors. Statically, galectin-3 was not associated with tumor grade in urinary bladder cancer (Table 1).
Galectin-3 expression did not show a significant correlation with tumor stage or muscularis propria invasion. Most positive bladder tumors did not show lymph node involvement, blood vessel invasion, or metastasis (90%, 92%, and 80%, respectively). However, no substantial association was found between galectin-3 expression and invasion of lymph nodes, blood vessels, or distant organs.
Results of the Cox proportional hazards model were not significant. Thus, galectin-3, sex, age, histologic type, stage, grade, muscular invasion, lymph node involvement, vascular invasion, and metastasis could not adequately estimate survival. Kaplan–Meier curves also did not show differences in survival.
Discussion
Together histological type, grade, and tumor stage remain the most reliable prognostic features for urinary bladder cancer. Various tissue biomarkers have been examined for their diagnostic and prognostic importance. No urinary tumor marker examined to date offers satisfactory sensitivity and specificity. Improved predictive markers are needed to recognize indolent tumors from those that are possibly fatal.35–36
Intracellular galectin-3 expression has been found in different types of tumor cells, where it binds to its ligands. Elevated levels of galectin-3 have been correlated with cell proliferation and transformation, cellular adhesion processes, metastasis, and resistance to apoptosis.10,37 Subcellular localization appears to have a role in whether galectin-3 expression is involved in tumor development and death. Increased cytoplasmic galectin-3 expression has been correlated with tumor aggressiveness. 38 Yu et al 39 concluded that translocation of galectin-3 into perinuclear membranes leads to cytochrome c inhibition and the release of caspases from mitochondria.
The immunohistochemical expression of galectin-3 in urothelial tumors and its association with clinicopathological factors have rarely been investigated, only six studies were found in the literature to date. There is no consistency among the results of these studies including this work (Table 2). In 2007, Langbein et al. 40 reported that galectin-3 expression was not homogenous in urinary bladder tumors. Staining was weak or absent in most early-stage and low-grade tumors; in contrast, high-risk tumors showed strong cytoplasmic staining. Most muscle-invasive tumors showed intense cytoplasmic immunoreactivity. They also found a significant correlation between tumor stage and galectin-3 expression, but not with grade or other clinicopathological factors.
Association between galectin-3 expression and clinicopathological variables from immunohistochemical studies.

In 2008, Kramer et al. 41 analyzed galectin-3 expression in 162 urothelial carcinomas and found that galectin-3 expression was negatively correlated with tumor grade. Furthermore, galectin-3 expression could predict the duration of recurrence-free survival. Cytoplasmic expression of galectin-3 was prominent in tumor cells, with a small number of cases showing nuclear immunostaining. In 2010, Canesin et al. 42 observed enhanced cytoplasmic galectin-3 expression in urothelial cells of muscle-invasive tumors in contrast to non-muscle-invasive tumors. Galectin-3 expression showed statistically important correlations with tumor stage, grade, and progression into invasive disease. High galectin-3 expression was also detected in highly lethal tumors; decreased expression was found in cases with good survival. In 2014, Gendy et al. 43 reported increased galectin-3 expression in urothelial tumors compared with normal bladder mucosa. Galectin-3 expression was also considerably greater in muscle-invasive tumors compared with non-invasive tumors. Furthermore, galectin-3 expression is significantly linked with tumor grade. In 2016, Raspollini et al. 44 reported that galectin-3 was not correlated with survival outcomes. Later in 2018, Fang et al. 45 found that galectin-3 was upregulated in urothelial tumors, and survival analysis showed that galectin-3 expression was inversely correlated with overall survival.
Discrepancies among the results of previous studies looking for associations between galectin-3 expression and clinicopathologic factors and this work could be attributable to the various histological types of bladder tumors, cases with different stages of tumors development, antibody and/or technique sensitivity, difference among patients and/or sample size, and the debatable anti- or pro-apoptotic function of galactin-3 in each tumor as demonstrated by in vitro overexpression studies in different types of cell lines.46–47 Alternatively, non-specific engagement of galectin-3 in tumor progression is possible. This and earlier studies have all had some weak points, e.g., relatively low sample sizes and semiquantitative immunostaining. More comprehensive studies are needed to evaluate the roles of galectin-3 in the development, diagnosis, and prognosis of bladder tumors.
Conclusion
The results of this study suggest that galectin-3 is downregulated in bladder cancer evolution but is not a useful marker for the diagnosis and prognosis of urinary bladder cancer.
Footnotes
Author Contributions
Jaudah Al-Maghrabi: conception, performance of work, interpretation of data, and writing the article.
Mohamad Nidal Khabaz: conception, performance of work, interpretation of data, and writing the article.
Data availability statement
All authors have access to the data. All original research data are available upon request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University (Jeddah, Saudi Arabia) (grant number
