Abstract
Background:
There are currently limited real-world data on the effectiveness and safety of first-line pembrolizumab combined with chemotherapy in patients with advanced/recurrent or metastatic esophageal squamous cell carcinoma (ESCC) in China. This study was conducted to address this knowledge gap.
Methods:
This multicenter retrospective cohort study was conducted at 17 hospitals in China and included adults (⩾18 years) with stage IV primary ESCC, or recurring 6 months after radical radiotherapy/surgery-based combination therapy, who had received first-line pembrolizumab plus chemotherapy. Data were collected from electronic medical records. Endpoints included objective response rate (ORR), disease control rate (DCR) progression-free survival (PFS), overall survival (OS), and safety. Subgroup analyses were conducted to identify patient characteristics and treatment patterns associated with treatment response.
Results:
In total, 202 patients who had received treatment from 2018 to 2023 were included: 125 (61.9%) newly diagnosed and 77 (38.1%) with recurrence, 181 (89.1%) were male. Pembrolizumab was most commonly combined with paclitaxel + platinum (69.8%) or fluorouracil + platinum (19.3%). After a median follow-up of 22.6 months (95% confidence interval (CI) 20.1–25.4), the ORR and DCR were 60.9% and 87.6% and the median PFS and OS were 10.8 months (95% CI 9.1–13.5) and 17.3 months (95% CI 14.9–19.9), respectively. OS was similar in patients with treatment-naïve and recurrent disease. Among the combination chemotherapy regimens, paclitaxel + platinum was associated with the longest median OS (18.2 months, 95% CI 16.1–22.5). Favorable survival outcomes were observed in patients with oligometastases. No new safety signals were observed.
Conclusion:
These real-world data indicate that the first-line treatment with pembrolizumab plus chemotherapy is effective and safe in Chinese patients with advanced ESCC and show that paclitaxel + platinum is the most commonly used and most effective partner chemotherapy in China.
Background
Esophageal cancer is the seventh most common cancer globally and accounts for around 5.5% of cancer-related deaths worldwide. 1 China currently has the highest incidence of new cases of esophageal cancer of any country, contributing to over 50% of the global total. 2 In China, esophageal squamous cell carcinoma (ESCC) comprises up to 85.8% of new cases of esophageal cancer, 3 in contrast to only 30%–40% in Western countries, in which adenocarcinoma is generally the most common subtype. 4
Prior to the approval of immunotherapy for use in patients with esophageal cancer, the first-line treatment of advanced or metastatic esophageal cancer was based on chemotherapy with fluoropyrimidine plus platinum, with the addition of the HER2 inhibitor trastuzumab for patients with HER2+ adenocarcinoma, and other targeted therapies for patients with specific mutations or genetic alterations. 5 However, first-line chemotherapy is associated with poor survival outcomes in this patient population. 6 Immunotherapy for advanced or metastatic esophageal cancer was first approved based on the results of the Phase III KEYNOTE-590 trial, which showed the benefit of adding pembrolizumab, a programmed death-1 (PD-1) inhibitor, to fluoropyrimidine and cisplatin for the first-line treatment of locally advanced or metastatic esophageal cancer. 7 Subsequently, phase III trials of multiple other PD-1 inhibitors have shown a survival benefit of combined treatment with immunotherapy and chemotherapy in patients with ESCC in first-line treatment,8–14 and is recommended by the current National Comprehensive Cancer Network, European Society of Medical Oncology (ESMO), and Chinese Society of Clinical Oncology guidelines as a first-line standard of care for patients with advanced esophageal cancer.5,15
Despite the success of pembrolizumab in the treatment of esophageal cancer, the clinical application of this drug in China is associated with multiple challenges. First, although the pivotal KEYNOTE-590 trial of first-line pembrolizumab included about 53% Asian patients, the data do not fully reflect the specific characteristics of Chinese patients and Chinese clinical practice. For example, almost all (>85%) esophageal cancer in China is ESCC,3,16–18 and the KEYNOTE-590 trial included 27% of patients with adenocarcinoma histology. In addition, the KEYNOTE-590 study used 5-fluorouracil and cisplatin as a chemotherapy backbone. However, in China, different chemotherapy drugs are more commonly paired with immunotherapies versus Western countries, with higher use of chemoradiotherapy combined with immunotherapy, a general preference for paclitaxel plus platinum therapy and wider use of S1 (tegafur–gimeracil–oteracil potassium),17,18 though the approval for using pembrolizumab in the first-line setting in China was also based on the schedule used in the KEYNOTE-590 trial. Paclitaxel is generally preferred in Asia as it is better tolerated in Asian populations. 19 Furthermore, patients with esophageal cancer in Asia versus Western countries include a higher ratio of females, 20 as well as differential risk factors for developing esophageal cancer, disease stages at diagnosis, molecular epidemiology, and sensitivity to treatment. 21 Second, although pembrolizumab plus chemotherapy extended the median overall survival (OS) of 106 patients in the Chinese subgroup by 2.5 months (10.5 vs 8.0) compared to chemotherapy in the KEYNOTE-590 trial, the median OS still had a slight difference compared to the entire global population (9.8 vs 12.4 months), 22 and was also obviously lower than other clinical trials conducted in the Chinese population though cross-trial comparisons should be made with caution.11–15 Subsequently, no large sample clinical trial was carried out in the Chinese population using pembrolizumab as the first-line treatment for ESCC, and there is also a lack of real-world data on the use of pembrolizumab in China, which would provide valuable insights to inform clinical practice and supplement the data from global clinical trials.
Therefore, this retrospective study was conducted to investigate the real-world effectiveness and safety of pembrolizumab in combination with chemotherapy as per clinical practice, in particular paclitaxel plus platinum which is a commonly used combination in China, in the first-line treatment of patients with advanced/recurrent or metastatic ESCC in China.
Methods
Study design
This multicenter retrospective cohort study was conducted at 17 hospitals in China. The study was conducted following the ethical principles described in the Declaration of Helsinki and following good clinical practice. The study was registered at ClinicalTrials.gov (NCT05142709).
Patients
The study included adults (⩾18 years of age) with stage IVB (TxNxM1, UICC/AJCC 8th edition) primary ESCC (metastasis to the supraclavicular, neck, hilar, and abdominal lymph nodes below the abdominal trunk or organ metastasis) or recurring 6 months after radical radiotherapy/surgery-based combination therapy (local recurrence, distant metastasis) and who had received first-line treatment with pembrolizumab combined with chemotherapy from 2018 to 2022.
Procedures
Data were collected from electronic medical records by the investigators and variables of interest were extracted from chart reviews. Data on patient sex were self-reported and selected from male/female. Endpoints evaluated included objective response rate (ORR), disease control rate (DCR), progression-free survival (PFS), OS, and the incidence of adverse events (AEs). In addition, subgroup analyses of ORR, PFS, and OS were conducted to identify patient characteristics and treatment patterns associated with treatment response.
Statistics
This study had no formal sample size calculation. All statistical analyses were exploratory and were conducted using SAS v9.4 (SAS Institute Inc.). Treatment responses were evaluated using the Response Evaluation Criteria in Solid Tumors v1.1. PFS was defined as the time from initiation of pembrolizumab-containing therapy until disease progression or death from any cause. OS was defined as the time from initiation of pembrolizumab-containing therapy until death from any cause. Time-to-event endpoints were evaluated using the Kaplan–Meier method. AEs were categorized and graded using the Common Terminology Criteria for Adverse Events v5.0. Missing data were not imputed.
For the subgroup analysis of ORR, univariable logistic regression was used to calculate odds ratios and 95% confidence intervals (CIs) and for the PFS and OS subgroup analyses a Cox regression model was utilized to calculate hazard ratios (HRs) with 95% CIs. Variables included in the subgroup analyses were patient status (treatment naïve vs recurrent metastasis), age (⩽65 vs >65 years), sex (male, female), Eastern Co-operative Oncology Group (ECOG) performance status (PS), primary tumor site, pathological differentiation, presence of oligometastasis (⩽5 metastatic sites excluding the primary esophageal tumor and regional lymph nodes, and ⩽3 lesions in the same organ. Extra-regional nodes were categorized by location (left and right neck, left and right supraclavicular, bilateral axilla, retroperitoneum, mediastinal non-regional, pelvic cavity, and the same region is counted as one lesion), local therapy (patients receiving local intervention such as surgery or radiotherapy before progression from chemoimmunotherapy), and chemotherapy regimen.
p-Values <0.05 were considered significant, using two-sided tests. Qualitative variables were summarized as the number and proportion of patients and continuous variables as mean ± standard deviation or median (min, max). The reporting of this study conforms to the ESMO Guidance for Reporting Oncology real-world evidence. 23
Results
Patient characteristics
Of the 248 patients screened for inclusion, 21 did not receive combination chemotherapy, 4 had received second-line treatment, and 21 did not have sufficient treatment data or follow-up data. Therefore, in total, 202 patients were included in this analysis; the majority (89.6%) were male, with a mean age of 62.7 years (Table 1). Of these patients, 125 (61.9%) had newly diagnosed cancer and 77 (38.1%) had experienced recurrence after radical therapy. Most patients had an ECOG PS of 1 (80.7%), and a similar proportion of treatment-naïve patients had primary tumors located in the mid-thoracic (44.0%) and lower-thoracic (40.0%) regions. Among all patients, the most common metastatic site was the lymph nodes (79.2%) and 48.0% of patients had oligometastatic disease (⩽5 sites). For patients who had experienced locoregional recurrence (n = 45), recurrences in the regional lymph nodes (62.2%) and esophagus (17.8%) were the most prevalent. Before initiating treatment, most patients were receiving a semi-liquid (61.4%) or normal (32.2%) diet. Of the 202 patients, only 13 had PD-L1 testing results which precluded inclusion of PD-L1 expression in the efficacy subanalysis.
Patient demographics and baseline characteristics (n = 202).

Percentage calculations for locoregional recurrence sites are based on patients with locoregional recurrence.
ECOG, Eastern Co-operative Oncology Group; SD, standard deviation.
Baseline treatment characteristics
Patients had received a median of six cycles of pembrolizumab (Supplemental Appendix Table A1). Thirty-three patients (16.3%) were still on anti-PD-1 treatment. The most common chemotherapy regimens used in combination with pembrolizumab were paclitaxel + platinum (69.8%) and fluorouracil + platinum (19.3%) and a median of four cycles (range: 1–11 cycles) of chemotherapy was received. Fifty-one (25.2%) patients received less than four cycles of chemotherapy. A total of 23 (11.4%) patients discontinued systemic therapy due to treatment-related AEs and 99 (49.0%) discontinued due to progressive disease. The main reasons for chemoimmunotherapy discontinuation are reported in Table 2. Among 93 patients who received local treatment, the majority had been treated with radiotherapy (87.1%) and 8.6% had undergone surgery. In all, 14 patients (6.9%) received supportive care before treatment.
Summary of safety data.

AE, adverse event.
Treatment effectiveness
After a median follow-up time of 22.6 months (95% CI 20.1–25.4), the ORR and DCR were 60.9% and 87.6%, respectively, including 9 (4.5%) complete responses and 114 (56.4%) partial responses (Supplemental Appendix Table A2). The median PFS was 10.8 months, with 1- and 2-year PFS rates of 46.8% and 29.3%, respectively (Figure 1(a)). The median OS was 17.3 months and the 1- and 2-year OS rates were 65.4% and 29.6%, respectively (Figure 2(a)).

Kaplan–Meier curves of PFS in (a) all patients and (b) by chemotherapy regimen.
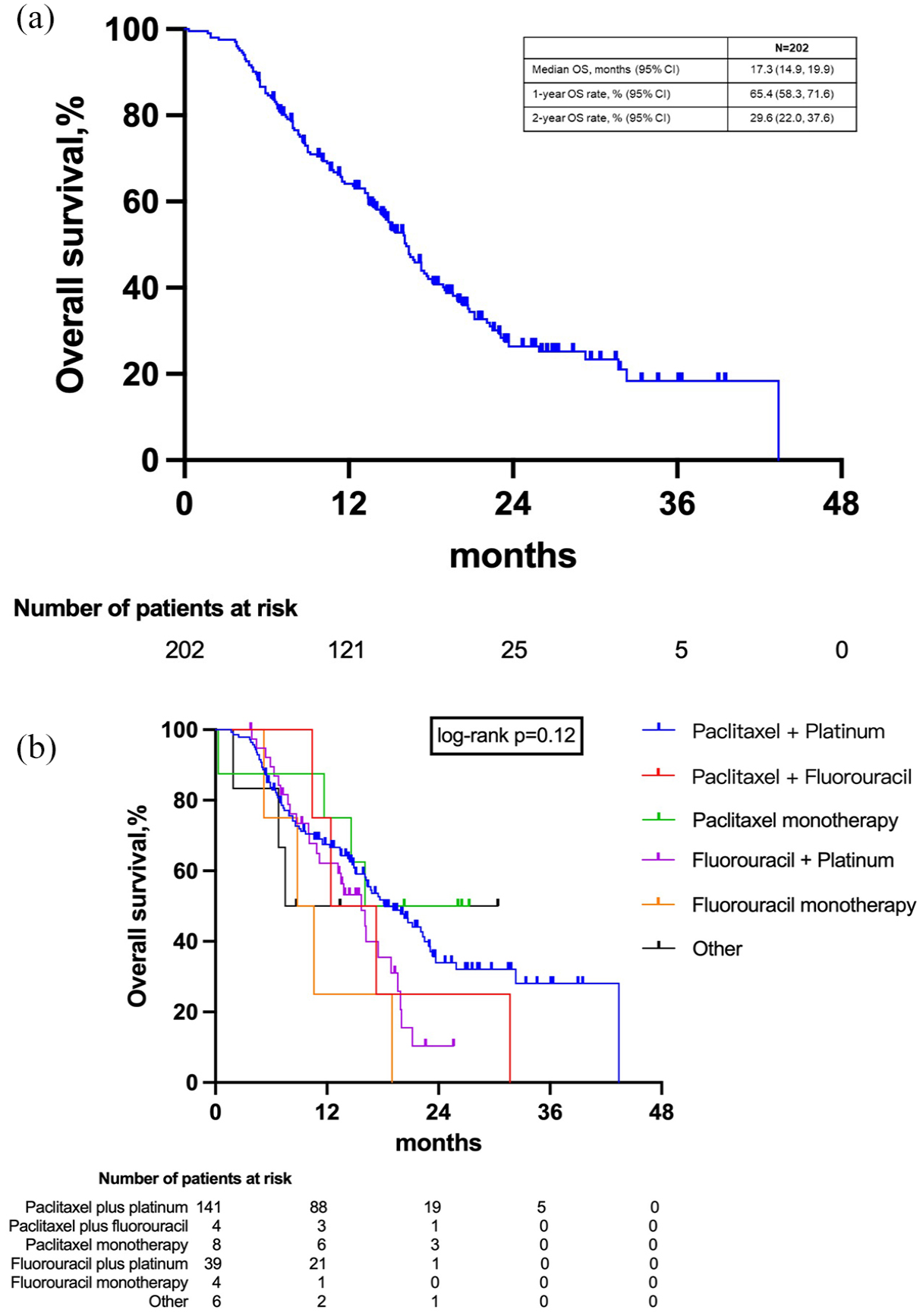
Kaplan–Meier curves of OS in (a) all patients and (b) by chemotherapy regimen.
Safety
The Grade ⩾3 AEs occurring in >1% of patients were neutrophil count decreased (5.0%), platelet count decreased (3.0%), anemia (2.0%), white blood cell count decreased (1.5%), pneumonia (1.0%), and acute kidney injury (1.0%). Immune-related Grade ⩾2 AEs were rare, occurring in only six patients, and included immune pneumonia, immune myocarditis, and immune thyroiditis.
Treatment effectiveness subgroup analyses
The subgroup analysis of ORR revealed broadly comparable outcomes following treatment with pembrolizumab combined with chemotherapy across most subgroups evaluated (Supplemental Appendix Table A3). The only statistically significant difference observed was in the ECOG PS subgroup, with higher ORRs in patients with an ECOG PS of 0 versus 1 or 2 (p = 0.035). Numerically higher ORRs were observed in patients with treatment-naïve disease versus recurrent metastasis (64.0 vs 55.8%), males versus females (63.0 vs 42.9%), patients with oligometastases versus polymetastases (65.0 vs 56.6%), and in patients who received local treatment versus those who did not (67.7 vs 55.0%), but these differences did not reach statistical significance.
The association between choice of combination chemotherapy regimen and median PFS did not approach significance (p = 0.062); however, paclitaxel + platinum (12.5 months; 95% CI 9.8–15.0) and paclitaxel + fluorouracil (10.7 months; 95% CI 5.5, not analyzed) resulted in the highest median PFS values, while fluorouracil + platinum was associated with a higher risk of disease progression or death versus paclitaxel + platinum (HRPFS 1.65; 95% CI 1.08–2.51) (Figure 1(b)). Numerical trends toward higher median PFS were observed in patients with recurrent metastasis versus treatment naïve patients, those aged >65 versus ⩽65 years, and oligometastases versus polymetastases; however, these associations did not reach statistical significance (Table 3).
Subgroup analysis of PFS and OS.

Calculated using a univariate Cox regression model.
CI, confidence interval; ECOG, Eastern Co-operative Oncology Group; NA, not applicable; PFS, progression-free survival; OS, overall survival.
Numerical values of median OS were generally comparable for most of the subgroups evaluated (Table 3). Patients with oligometastasis had a longer median OS versus those without oligometastasis and this association was statistically significant (20.8 months vs 13.5 months; HR 0.54; 95% CI 0.37–0.77; p < 0.001). Notably, a similar median OS was achieved by patients who were treatment naïve and those with recurrent metastasis. Although the subgroup analysis by combination chemotherapy type did not reach statistical significance, the combination of pembrolizumab with paclitaxel + platinum was associated with the highest median OS value (18.2 months, 95% CI 16.1–22.5) and fluorouracil monotherapy (median OS: 9.7 months) was associated with a higher risk of death from any cause than paclitaxel + platinum (HROS 2.84; 95% CI 1.03–7.80) (Figure 2(b)).
Discussion
In this retrospective study, we investigated the real-world effectiveness and safety of pembrolizumab combined with chemotherapy, in particular paclitaxel plus platinum which is commonly used in China, for the first-line treatment of patients with advanced/recurrent or metastatic ESCC. To the authors’ knowledge, this is the largest multicenter, retrospective study of first-line treatment with pembrolizumab plus chemotherapy in patients with advanced/recurrent or metastatic ESCC conducted in China to date. Our data are also reported after a relatively long 2-year median follow-up time. The results confirm that the most commonly used partner chemotherapy regimen in China is paclitaxel + platinum (64.4%), which was used more frequently than the international guideline-recommended fluorouracil + platinum (13.9%). In general, pembrolizumab plus chemotherapy led to a high ORR (60.9%) and a median PFS and OS of 10.8 and 17.3 months, respectively, with a comparable median OS for patients with treatment-naïve and recurrent cancer. Subgroup analyses identified several patient groups who achieved a particular benefit from treatment, including those receiving paclitaxel + platinum chemotherapy, those with oligometastatic disease, and those receiving local therapy after response to immunotherapy. Importantly, our results also show that pembrolizumab plus chemotherapy is generally well tolerated in real-world practice; only 11% of patients discontinued treatment due to a treatment-related AE, and no new safety signals were observed.
The real-world data from this nationwide, multicenter study showed the effectiveness and safety of first-line pembrolizumab plus chemotherapy in patients with advanced esophageal cancer in China, with consistent results to those reported by the pivotal global phase III KEYNOTE-590 trial. 7 The median PFS and OS observed in the present study were comparable to or higher than those reported in KEYNOTE-590 (PFS: 10.8 vs 6.3 months; OS: 17.3 vs 12.6 months). In addition, the 2-year OS rate in the present study was similar to that reported in KEYNOTE-590 (29.6% and 29.0%). However, cross-trial comparisons should be made with caution due to differences in patient populations and treatment regimens. However, our findings suggest that the characteristics of Chinese patients and Chinese clinical practice do not impact the effectiveness of this treatment regimen. For example, in KEYNOTE-590, pembrolizumab was combined with fluoropyrimidine + platinum-based chemotherapy and the results from the present study show that the combination with paclitaxel + platinum, which represents the most commonly used combination in China and East Asia, does not negatively impact treatment effectiveness. There have been limited previous real-world studies of pembrolizumab in the first-line treatment of patients with advanced esophageal cancer in China. However, a small Chinese real-world study with 57 patients with different ESCC clinical stages reported ORRs of around 74.1% for patients who received pembrolizumab plus platinum and nab-paclitaxel, which is broadly comparable to the results of the present study (60.9%). 24 The effectiveness findings of the present study are also broadly comparable to the results of clinical trials of other immunotherapies conducted in Chinese patients with ESCC, which reported median PFS values of 5.7–9.0 months and median OS values of 15.3–17.0 months.10–14 In addition, a multicenter real-world study of camrelizumab plus chemotherapy in advanced esophageal cancer reported a first-line ORR of 54.2% and median PFS and OS of 10.1 and 17.5 months, respectively, which are similar to the findings of the present study. 25
Our results confirm previous reports suggesting that paclitaxel + platinum is the most common chemotherapy combined with immunotherapy in Chinese and East Asian patient populations.17–19 In addition, paclitaxel + platinum resulted in the longest median PFS and OS times of all chemotherapy regimens evaluated. Interestingly, a systematic review of data from East Asian patients with advanced ESCC receiving first-line chemotherapy concluded that there may be a trend toward improved efficacy with paclitaxel + platinum versus fluoropyrimidine + platinum in this patient population. 19 In addition, a recent meta-analysis of clinical trials of first-line therapy in patients with advanced ESCC also concluded that the combination of immunotherapy with paclitaxel + platinum may lead to longer PFS than fluoropyrimidine + platinum. 26 Chemotherapy increases tumor sensitivity to immunotherapy through multiple immunostimulatory mechanisms including the induction of immunogenic cell death and inhibiting immunosuppressive cells.27,28 Fluoropyrimidine and paclitaxel have differential modes of action and exert distinct immunomodulatory effects27,28 and it is possible that the longer PFS observed in the present study with paclitaxel + platinum versus fluoropyrimidine + platinum is due to these differences. However, the longer PFS with paclitaxel could also be explained by higher patient compliance to paclitaxel, which can be infused within 48–72 h compared with 120 h for fluoropyrimidine and may be the better-tolerated agent in Chinese and Asian patients. 19
Our study found that an ECOG PS of 0 was associated with a higher ORR versus a PS of 1 or 2, although ECOG PS was not significantly associated with PFS or OS outcomes. This is consistent with the results of the KEYNOTE-590 trial in which a similar OS and PFS benefit of pembrolizumab plus chemotherapy versus chemotherapy alone was reported for patients with ECOG PS or 0 and 1. 7 In addition, similar OS and PFS benefits of immunotherapy plus chemotherapy versus chemotherapy alone in patients with an ECOG PS of 0 or 1 were also reported in clinical trials of nivolumab, 9 sintilimab, 10 toripalimab, 12 and serplulimab. 29 Taken together, these findings highlight that immunotherapy combined with chemotherapy can be effective in patients with a range of ECOG PSs. Moreover, 11 patients with ECOG PS 2 were included in the analysis. We believe some patients with ECOG PS 2 may be able to tolerate PD-1 inhibitors with or without chemotherapy in clinical practice. However, we were unable to observe a significant difference in prognosis between patients with PS 2 and those with PS 0–1 from the analysis due to the small sample size of the subgroup.
The results of our subgroup analysis also suggest that patients with oligometastases achieve a higher median OS than those with polymetastases following treatment with pembrolizumab plus chemotherapy. Interestingly, previous studies suggest that patients with oligometastases derive a particular benefit from combined treatment with systemic therapy and local treatment compared with systemic or local therapy alone.30,31 In addition, a randomized, phase II study conducted in China reported that combined treatment with systemic therapy plus local treatment resulted in improved PFS versus systemic therapy alone in patients with ESCC and oligometastases (15.3 months vs 6.4 months). 32 Interestingly, among the patients receiving systemic therapy plus local treatment in this study, around 40% received immunotherapy, highlighting the potential value of combining immunotherapy and local treatment for patients with oligometastatic disease. Oligometastatic disease represents an intermediate stage before the development of systemically metastasized disease and therefore has the potential to respond to definitive treatment. 33 In this regard, the results of our study demonstrate the value of combining systemic and local therapy for patients with ESCC oligometastasis. In addition, our findings also highlight the different prognosis for patients with advanced ESCC with oligometastasis compared with those who have extensive metastasis. Taken together, our results suggest that careful stratification of patients with advanced ESCC and the selection of optimal combination treatment strategies are required to maximize patient benefit from treatment. However, phase III trials are required to confirm the benefits of this treatment approach.
The efficacy of palliative treatment for advanced esophageal cancer is limited, the proportion of patients experiencing local recurrence is high and late oligometastasis is common. Therefore, local treatments such as surgery and radiotherapy remain important elements of treatment in the advanced setting and are valuable for improving symptoms, quality of life, and patient survival rates. Retrospective studies have shown that a subset of esophageal cancer patients with non-visceral or non-bone metastases can achieve good survival outcomes following local treatment 34 and highly selected patients with metastasis can achieve good outcomes following surgical resection. 35 A recent meta-analysis also concluded that surgery with curative intent can lead to high 3-year survival rates in patients with metastatic esophageal cancer. 36 In the present study, around half of the patients received local therapy combined with immunotherapy, of whom 86.1% (n = 93) received radiotherapy. Radiotherapy is commonly used in the treatment of patients with ESCC across various stages, from perioperative adjuvant or definitive therapy to palliative use. 37 A beneficial effect of radiotherapy combined with oral nutrition support has been shown in patients with esophageal cancer and metastases, including improvement of symptoms and quality of life. 38 Radiotherapy may also improve local disease control in patients with local recurrence, but the feasibility of using radiotherapy in patients who have previously received radiotherapy and experienced recurrence requires further study. 39 There is also evidence suggesting radiotherapy combined with immunotherapy provides a synergistic improvement in efficacy in patients with advanced or metastatic ESCC.40,41 In particular, a phase II single-arm trial reported that low-dose radiotherapy plus camrelizumab and irinotecan was associated with favorable survival outcomes in patients with advanced ESCC who had progressed on first-line immunotherapy plus chemotherapy. 42 In addition, an increase in PD-L1 expression has been observed in patients with locally advanced esophageal cancer after receipt of neoadjuvant chemoradiotherapy. 43 Taken together, the available evidence suggests that combining immunotherapy with radiotherapy is a promising treatment approach for ESCC but requires further investigation.
This study had several limitations that should be mentioned. First, due to the inherent nature of retrospective analyses, there were missing data, and the number of patients in certain subgroups was too small to allow robust analysis, including PD-L1 expression and tumor history, resulting in the inability to analyze the impact of PD-L1 expression levels on the treatment efficacy and outcomes. Similarly, some AEs were possibly overlooked as they were entirely collected from medical records, which also makes it impossible to compare the safety between the patients using a paclitaxel-based regimen and those with the fluoropyrimidine-based regimen, though our previous trial in patients with local advanced ESCC has found that the toxicity profiles of the two chemotherapy regimens are quite different but there was no significant difference in the incidence of acute grade 3 or higher AE. 44 Second, there was a possible risk of treatment selection bias as the treatment assignment was not randomized. More prospective trials are needed for external validation. However, our real-world data could provide important insights into clinical practice and real-world patient care.
Conclusion
The results of this multicenter, retrospective study provide real-world evidence of the effectiveness and safety of first-line treatment with pembrolizumab plus chemotherapy in Chinese patients with advanced esophageal cancer. In addition, the results showed that paclitaxel + platinum is the most common partner chemotherapy used with pembrolizumab in China and was associated with the longest median PFS and OS times among the chemotherapy combinations analyzed. Our results suggest that specific patient subgroups may achieve particularly good survival outcomes following treatment with pembrolizumab plus chemotherapy, including those with oligometastases. These preliminary findings require validation in a larger cohort of patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241297092 – Supplemental material for Effectiveness and safety of first-line pembrolizumab plus chemotherapy in patients with advanced/recurrent or metastatic esophageal squamous cell carcinoma in China: a real-world multicenter study
Supplemental material, sj-docx-1-tam-10.1177_17588359241297092 for Effectiveness and safety of first-line pembrolizumab plus chemotherapy in patients with advanced/recurrent or metastatic esophageal squamous cell carcinoma in China: a real-world multicenter study by Yu Lin, Wenbin Shen, Jinjun Ye, Honglei Luo, Xizhi Zhang, Yuanji Xu, Qin Lin, Wenyang Liu, Yingying Zhang, Yujin Xu, Wei Jiang, Lina Zhao, Anwen Liu, Lei Wu, Hong Ge, Conghua Xie, Kuaile Zhao, Junqiang Chen, Luhua Wang and Qi Liu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
All authors had access to all relevant aggregated study data, participated in writing or reviewing the manuscript, and provided final approval for the decision to submit the manuscript for publication. The authors would like to offer their sincere thanks to Jing Qian and Danfeng Shi from MSD GMA China for their scientific support of this manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
