Abstract
Background:
Until recently, targeted therapies have failed to benefit patients with human epidermal growth factor receptor 2 (HER2)-low-expressing breast cancer (BC). Nevertheless, antibody–drug conjugates (ADCs) have reshaped their prognosis.
Objectives:
We performed a systematic review and meta-analysis to assess the effectiveness of ADCs in patients with HER2-low advanced/metastatic (a/m) BC.
Design:
This study is a systematic review and meta-analysis.
Data sources:
We searched PubMed, Embase, and Cochrane databases as well as the American Society of Clinical Oncology, European Society for Medical Oncology, and San Antonio Breast Cancer Symposium conference proceedings.
Methods:
Studies evaluating ADCs (trastuzumab deruxtecan (T-DXd), sacituzumab govitecan (SG), MRG002, and RC48-ADC) in patients with HER2-low a/mBC were included. We used R software (v.4.2.2) and random effects models for all analyses. Heterogeneity was assessed using the I2 test.
Results:
Overall, 14 studies were included (five real-world studies and nine clinical trials (CTs)), with 2883 HER2-low a/mBC patients: 808 received treatment of physician’s choice (TPC), and 2075 ADCs. Most were treated with T-DXd (n = 1691), followed by SG (n = 310), MRG002 (n = 56), and RC48-ADC (n = 18). Patients treated with T-DXd achieved a significantly higher objective response rate (ORR), disease control rate (DCR), and clinical benefit rate (CBR) than those receiving other ADCs. In the pooled analysis of four randomized CTs, ADCs statistically prolonged progression-free survival (n = 1828, hazard ratio (HR) 0.50, 95% confidence interval (CI) 0.36–0.68, I2 = 82%, p < 0.001) and overall survival (n = 1546, HR 0.70, 95% CI 0.57–0.86, I2 = 43%, p < 0.001) compared with TPC. Patients on ADCs also achieved a greater antitumor response than TPC, including better ORR (odds ratio (OR), 3.7, 95% CI 2.5–5.6, I2 = 59%, p < 0.001), DCR (OR, 2.7, 95% CI 2.1–3.5, I2 = 0%, p < 0.001), and CBR (OR, 3.6, 95% CI 2.6–5.2, I2 = 56%, p < 0.01).
Conclusion:
Our systematic review and meta-analysis confirms the efficacy of ADCs in HER2-low a/m BC patients over TPC. Future studies should focus on bringing ADCs into earlier lines of therapy in this population.
Trial registration:
This study was registered in PROSPERO (CRD42024452962).
Keywords
Introduction
Human epidermal growth factor receptor 2 (HER2) constitutes an important receptor within the transmembrane tyrosine kinase protein family. 1 Its crucial role in cellular proliferation and survival signaling pathways translates into its high oncogenic potential.1,2 Overexpression or amplification of HER2 is found in up to 20% of breast cancers (BCs). 3 Initially, the overexpression of HER2 in breast tumors was associated with a more aggressive phenotype and poor prognosis. 4 However, nearly three decades ago, HER2 was identified as an effective therapeutic target across several tumors. 4 The development of HER2-directed agents such as trastuzumab and pertuzumab transformed the HER2-positive BC treatment landscape and improved the survival rates of patients at all stages. 4
Precise determination of HER2 status is therefore essential to guide clinical decisions. 4 HER2-positivity by immunohistochemistry (IHC) is defined as complete staining of over 10% of the tumor cell membrane (translating to a score of 3+) or by a weak/moderate staining (<10% of tumor cell membrane—IHC 2+) and a positive in situ hybridization (ISH) test. 5 Historically, HER2-negative BC was characterized by IHC scores of 0, 1+, or 2+ and ISH negative. Although anti-HER2 treatments were not effective in HER2-negative BC patients, some studies suggest that patients with diverging HER2 status between local and central pathology tests could benefit from such therapies.6,7 This highlights the considerable variability in HER2-testing tools and the challenge in identifying patients eligible for anti-HER2 treatments.6,7
Over 50% of HER2-negative metastatic BC (mBC) patients express modest levels of HER2. 8 These tumors, referred to as HER2-low, are defined by an IHC 1+ or IHC 2+ and negative ISH. 9 In the past, HER2-low expression was not considered a prognostic marker as patients also failed to benefit from anti-HER2 agents. 10 In early-stage HER2-low BC, no significant differences were seen in recurrence and survival rates when adding trastuzumab to adjuvant chemotherapy. 11 In the metastatic setting, studies testing trastuzumab emtansine (T-DM1), the first anti-HER2 antibody–drug conjugate (ADC) approved for BC, reported limited efficacy and treatment resistance associated with HER2 heterogeneity.12–14 This further solidified the idea that only patients with HER2-positive tumors should be considered for HER2-targeted strategies. 12
More recently, the practice-changing DESTINY-Breast04, a phase III trial exploring trastuzumab deruxtecan (T-DXd) in HER2-low patients, showed a remarkable 50% reduction in the risk of progression compared to chemotherapy (hazard ratio (HR) for disease progression or death, 0.50; 95% confidence interval (CI), 0.40–0.63, p < 0.001). 8 Other promising ADCs are currently being studied and have demonstrated clinical activity in HER2-low BC, including sacituzumab govitecan (SG), MRG002, and RC48-ADC.15–17 Therefore, we performed a systematic review and meta-analysis to explore the efficacy and safety of ADCs in patients with HER2-low advanced/metastatic (a/m) BC.
Methodology
This systematic review and meta-analysis was performed according to the guidelines from the Cochrane Collaboration and the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA), 18 and it was registered in the International Prospective Register of Systematic Reviews (PROSPERO—CRD42024452962) on January 1st, 2024. The PRISMA checklist for the abstract and the manuscript are available for reference in Supplemental Table S1(A) and (B), respectively.
Data source and search strategy
The following databases were systematically searched on January 27, 2024, and updated on June 19, 2024: PubMed, Embase, and Cochrane and the American Society of Clinical Oncology, European Society for Medical Oncology (ESMO), and San Antonio Breast Cancer Symposium conference proceedings. The full search strategy used in each database is found in Supplemental Table S2. Relevant reviews and references of included studies were also manually checked.
Eligibility criteria
For inclusion in this systematic review and meta-analysis, we considered phase II and III clinical trials (CTs) and retrospective cohorts assessing the outcomes of interest in patients with a/m HER2-low BC treated with ADCs (e.g., SG, T-DXd, RC48-ADC, MRG002). We checked posters or conference presentations for all included abstracts. Studies that included only a subgroup within the HER2-low population (e.g., brain metastases) were considered for inclusion. Main exclusion criteria were as follows: (1) lack of outcomes stratified by HER2 expression; (2) early stage BC; (3) phase I or studies reporting exclusively safety data; (4) ongoing studies for which data were not available at the time the search was performed, (5) ADC sequencing studies; (6) non-original studies (case reports, case series, reviews, letters to the editor, and commentaries); and (7) studies written in languages other than English.
Data collection and outcomes
Two investigators (I.M. and M.I.D.) independently screened studies by title and abstract using Rayyan software, selected reports for full-read, extracted data, and conducted the risk of bias analyses. Inconsistencies were resolved by consensus or by consulting a third author (M.V.). For each eligible study, we extracted data on baseline characteristics and collected data on (1) objective response rate (ORR), (2) disease control rate (DCR), (3) clinical benefit rate (CBR), (4) progression-free survival (PFS), (5) overall survival (OS), (6) median time to response (TTR), (7) median duration of response (DOR), and (8) adverse events (AEs).
We performed comparative analyses (ADCs vs treatment of physician’s choice (TPC)) for ORR, DCR, CBR, PFS, and OS. In addition, proportional analyses for the same efficacy outcomes were conducted across individual ADCs. The following subgroup analyses were explored: (1) PFS in IHC1+ versus IHC2+; (2) ORR, DCR, and CBR according to the antibody component of ADCs (anti-HER2 vs anti-Trop2); (3) ORR and PFS in hormone receptor (HoR)-negative vs HoR-positive; and (4) intracranial (IC)-ORR and IC-CBR in patients with brain metastases. We preferably use updated results with a longer follow-up for all analyses, whenever available.19–21 For the only study including HER2-low and HER2-ultralow patients, we used results from the intention to treat (ITT) population including both groups. 22
Exploring heterogeneity
To identify the impact of each study on the overall effect, we performed a leave-one-out sensitivity analysis for the outcome including the higher number of studies and ADCs (i.e., ORR). We explored the contribution of each study to the overall heterogeneity through the Baujat plot. 23 Moreover, we did a meta-regression analysis considering the influence of the median number of prior lines of therapies on the ORR. In this analysis, one study originally included in the ORR plot could not be included due to the lack of information regarding the median number of prior therapies. 17 In addition, in some studies, the median number of previous therapies was unavailable for the HER2-low subgroup. Thus, we considered the values given for the total population of the study.15,24,25 We also carried out analyses stratified by the study design (CTs vs real-world) to further explore the heterogeneity observed in main analyses (overall ORR, DCR, CBR, PFS, and OS).
Quality assessment
Quality assessment in retrospective cohorts and non-randomized CTs was performed using the ROBINS-I tool. 26 For randomized studies, we used the ROB-2 tool. 27 Publication bias was assessed through the funnel plot of individual study weights against point estimates and the linear regression for asymmetry (Egger test).
Statistical analysis
R software (version 4.2.2; R Foundation, Vienna, Austria) was used to run all statistical analyses. The following packages were used: “metafor,” “meta,” and “weight”. DerSimonian and Laird random-effects models were used in all analyses. Comparative meta-analyses were done using HR or odds ratio (OR) with 95% CIs. p-Values lower than 0.05 were considered statistically significant. Heterogeneity was explored using the I2 test and values ⩾25% were considered significant for heterogeneity. Proportional meta-analyses were used for dichotomous outcomes and reported in percentages, with 95% CIs. We used logit-transformation of data when the individual study proportion was <0.2 or >0.8. In the case of a study with zero events, we used the doubled-arcsine transformation. Pooled analysis of individual studies’ PFS and OS was carried out using the median values and 95% CIs. Studies in which the upper or lower CI was not reached were excluded from OS and PFS analyses.
Results
Systematic review
The initial search yielded 1990 results, of which 116 studies were comprehensively assessed. Most studies lacked HER2-low patients or were ongoing studies with no published results. A list of excluded studies after a comprehensive review can be found in Supplemental Table S3. In all, 14 studies with 19 related reports were included.8,15–17,19–22,24,25,28–36 Of these, five were observational studies and nine were CTs (four phase III studies and five phase II) (Figure 1).

PRISMA flow diagram of study screening and selection.
Baseline characteristics
A total of 2883 patients with a/m HER2-low BC were included, 2075 (72%) received ADCs and 808 (28%) TPC. The median age of patients on ADCs ranged from 48.1 to 59 years. HoR status was available in 11 studies (2021 patients), and 72% (n = 1452/2021) had HoR-positive tumors. Most patients had Eastern Cooperative Oncology Group Performance Status (ECOG PS) 0–1 (99%; n = 1219/1228). The ADCs used were T-DXd (81.5%, n = 1691), SG (15%, n = 310), MRG002 (2.7%, n = 56), and RC48-ADC (0.8%, n = 18). Follow-up time ranged from 9.5 to 38.4 months. HER2-low definitions varied slightly across studies. Among 11 studies with this information available, 8 studies defined it as a score of IHC1+ or ICH2+ with a negative ISH, whereas 2 studies considered ISH negative or untested. One of the included studies, DESTINY-Breast06, also included HER2-ultralow, defined as faint or incomplete membrane staining in up to 10% tumor (IHC-zero) (76 were patients on T-DXd and 76 on TPC). The number of previous therapies and other baseline characteristics are presented in Table 1.
Baseline characteristics of studies included in this systematic review and meta-analysis.
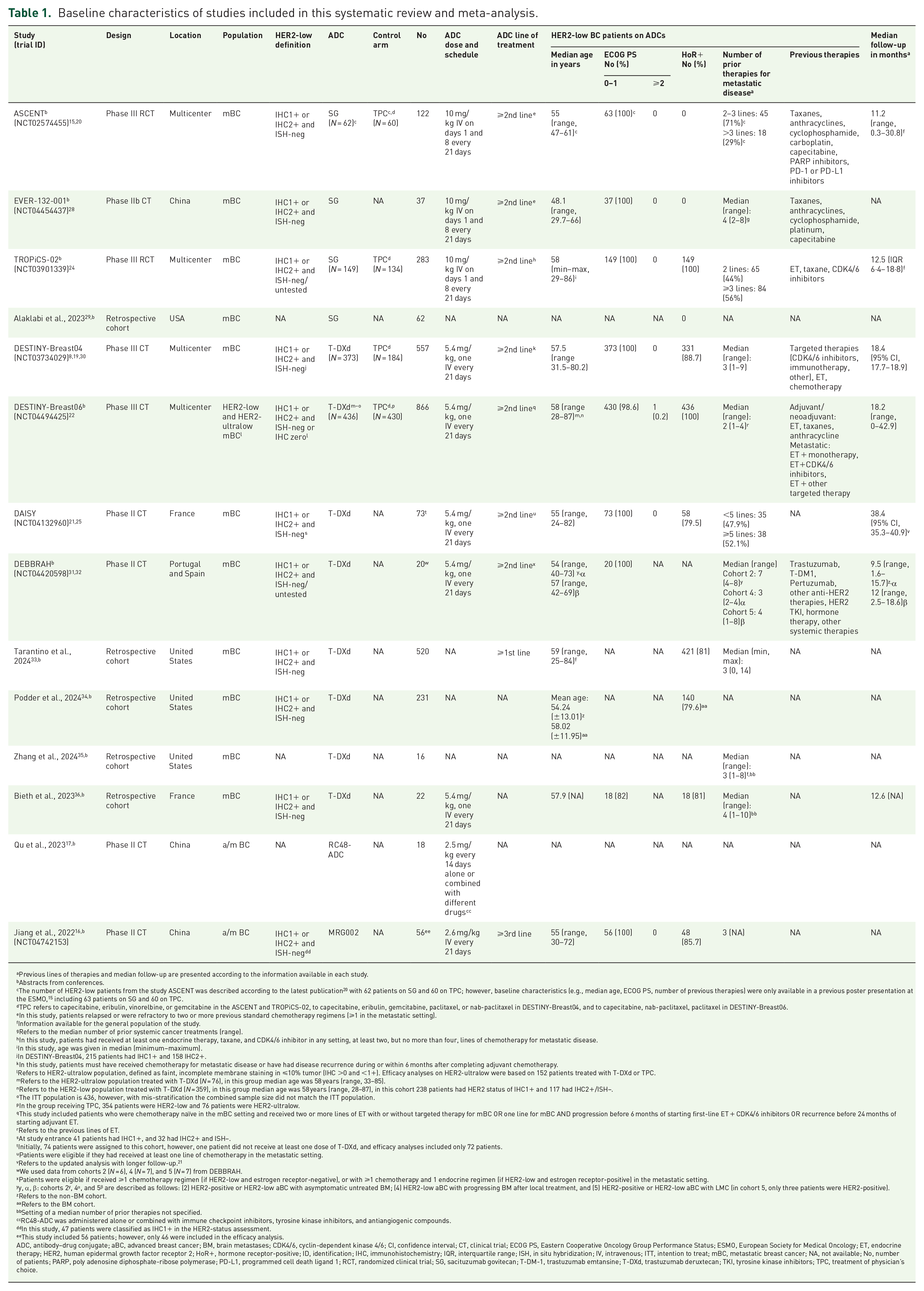
Previous lines of therapies and median follow-up are presented according to the information available in each study.
Abstracts from conferences.
The number of HER2-low patients from the study ASCENT was described according to the latest publication 20 with 62 patients on SG and 60 on TPC; however, baseline characteristics (e.g., median age, ECOG PS, number of previous therapies) were only available in a previous poster presentation at the ESMO, 15 including 63 patients on SG and 60 on TPC.
TPC refers to capecitabine, eribulin, vinorelbine, or gemcitabine in the ASCENT and TROPiCS-02, to capecitabine, eribulin, gemcitabine, paclitaxel, or nab-paclitaxel in DESTINY-Breast04, and to capecitabine, nab-paclitaxel, paclitaxel in DESTINY-Breast06.
In this study, patients relapsed or were refractory to two or more previous standard chemotherapy regimens (⩾1 in the metastatic setting).
Information available for the general population of the study.
Refers to the median number of prior systemic cancer treatments (range).
In this study, patients had received at least one endocrine therapy, taxane, and CDK4/6 inhibitor in any setting, at least two, but no more than four, lines of chemotherapy for metastatic disease.
In this study, age was given in median (minimum−maximum).
In DESTINY-Breast04, 215 patients had IHC1+ and 158 IHC2+.
In this study, patients must have received chemotherapy for metastatic disease or have had disease recurrence during or within 6 months after completing adjuvant chemotherapy.
Refers to HER2-ultralow population, defined as faint, incomplete membrane staining in ⩽10% tumor (IHC >0 and <1+). Efficacy analyses on HER2-ultralow were based on 152 patients treated with T-DXd or TPC.
Refers to the HER2-ultralow population treated with T-DXd (N = 76), in this group median age was 58 years (range, 33–85).
Refers to the HER2-low population treated with T-DXd (N = 359), in this group median age was 58 years (range, 28–87), in this cohort 238 patients had HER2 status of IHC1+ and 117 had IHC2+/ISH−.
The ITT population is 436, however, with mis-stratification the combined sample size did not match the ITT population.
In the group receiving TPC, 354 patients were HER2-low and 76 patients were HER2-ultralow.
This study included patients who were chemotherapy naïve in the mBC setting and received two or more lines of ET with or without targeted therapy for mBC OR one line for mBC AND progression before 6 months of starting first-line ET + CDK4/6 inhibitors OR recurrence before 24 months of starting adjuvant ET.
Refers to the previous lines of ET.
At study entrance 41 patients had IHC1+, and 32 had IHC2+ and ISH−.
Initially, 74 patients were assigned to this cohort, however, one patient did not receive at least one dose of T-DXd, and efficacy analyses included only 72 patients.
Patients were eligible if they had received at least one line of chemotherapy in the metastatic setting.
Refers to the updated analysis with longer follow-up. 21
We used data from cohorts 2 (N = 6), 4 (N = 7), and 5 (N = 7) from DEBBRAH.
Patients were eligible if received ⩾1 chemotherapy regimen (if HER2-low and estrogen receptor-negative), or with ⩾1 chemotherapy and 1 endocrine regimen (if HER2-low and estrogen receptor-positive) in the metastatic setting.
y, α, β: cohorts 2y, 4α, and 5 β are described as follows: (2) HER2-positive or HER2-low aBC with asymptomatic untreated BM; (4) HER2-low aBC with progressing BM after local treatment, and (5) HER2-positive or HER2-low aBC with LMC (in cohort 5, only three patients were HER2-positive).
Refers to the non-BM cohort.
Refers to the BM cohort.
Setting of a median number of prior therapies not specified.
RC48-ADC was administered alone or combined with immune checkpoint inhibitors, tyrosine kinase inhibitors, and antiangiogenic compounds.
In this study, 47 patients were classified as IHC1+ in the HER2-status assessment.
This study included 56 patients; however, only 46 were included in the efficacy analysis.
ADC, antibody–drug conjugate; aBC, advanced breast cancer; BM, brain metastases; CDK4/6, cyclin-dependent kinase 4/6; CI, confidence interval; CT, clinical trial; ECOG PS, Eastern Cooperative Oncology Group Performance Status; ESMO, European Society for Medical Oncology; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; HoR+, hormone receptor-positive; ID, identification; IHC, immunohistochemistry; IQR, interquartile range; ISH, in situ hybridization; IV, intravenous; ITT, intention to treat; mBC, metastatic breast cancer; NA, not available; No, number of patients; PARP, poly adenosine diphosphate-ribose polymerase; PD-L1, programmed cell death ligand 1; RCT, randomized clinical trial; SG, sacituzumab govitecan; T-DM-1, trastuzumab emtansine; T-DXd, trastuzumab deruxtecan; TKI, tyrosine kinase inhibitors; TPC, treatment of physician’s choice.
Efficacy outcomes
In the pooled analysis of four randomized CTs (RCTs), a consistent benefit was observed in favor of ADCs (n = 1020, either T-DXd or SG) compared to TPC (n = 808) for ORR (OR, 3.7 (95% CI 2.5–5.6, I2 = 59%, p < 0.001), DCR (OR, 2.7 (95% CI 2.1–3.5, I2 = 0%, p < 0.001), and CBR (OR, 3.6 (95% CI, 2.7–5.2, I2 = 56%, p < 0.01) (Figure 2(a)–(c)). Moreover, the analysis of PFS and OS showed a 50% (n = 1828, HR 0.50, 95% CI, 0.36–0.68, I2 = 82%, p < 0.001) and 30% (n = 1546, HR 0.70, 95% CI, 0.57–0.86, I2 = 43%, p < 0.001) reduction in the risk of progression and death, respectively, for the ADC group (Figure 2(d) and (e)).

Efficacy outcomes in HER2-low BC patients on ADC versus TPC: (a) ORR; (b) DCR; (c) CBR; (d) median PFS; (e) median OS. Proportions for each trial are represented by a square and the horizontal line crossing the squares indicates the 95% CI. The diamonds represent the estimated overall effect of the meta-analysis based on random effects. The ADC used in each study is described in parentheses following the study name. For ASCENT, data from the final results publication 20 (Bardia, Rugo and Tolaney, 2024) were used for all analyses except for DCR in which data from the ESMO poster presentation 15 were used. We used updated survival results 19 for the PFS and OS analyses of DESTINY-Breast04. For DESTINY-Breast06, we used data for the ITT population including both HER2-low and HER2-ultralow.
Patients treated with any ADC had an ORR, DCR, and CBR of 39% (95% CI, 29%–48%), 79% (95% CI, 70%–86%), and 54% (95% CI, 42%–66%), respectively (Figure 3(a)–(c)). The subgroup of patients on T-DXd had higher responses in all three endpoints compared to those on SG, RC48-ADC, and MRG002 (Figure 3(a)–(c)). Yet, only one study evaluated RC48-ADC and MRG002. Overall, patients treated with T-DXd (four studies), SG (one study), or RC48-ADC (one study) achieved a median PFS of 7.1 months (95% CI, 5.5–9.0 months; test for subgroup difference, p = 0.02) (Figure 3(d)). Median OS in patients receiving either SG (one study) or T-DXd (four studies) was 12.3 months (95% CI, 8.8–17.1 months; Figure 3(e)). Median TTR and DOR ranged from 1.4 to 2.7 and 3.6 to 14.3 months, respectively (Supplemental Table S4).

Efficacy outcomes in HER2-low BC patients treated with an ADC: (a) ORR; (b) DCR; (c) CBR; (d) median PFS; (e) median OS. Proportions for each trial are represented by a square and the horizontal line crossing the squares indicates the 95% CI. The diamonds represent the estimated overall effect of the meta-analysis based on random effects. For ASCENT, we used data from the final results publication 20 for ORR and CBR, whereas for the DCR we used data from the ESMO poster presentation. 15 For DAISY, we used data from the updated analysis with a longer follow-up. 21 We used updated survival results 19 for the OS and PFS analyses of DESTINY-Breast04. For DESTINY-Breast06, we used data for the intention to treat the population including both HER2-low and HER2-ultralow. (a) The subgroup of patients with HoR-positive/HER2-low BC. (b) The subgroup of patients with HoR-negative/HER2-low BC. (c) The subgroup of patients without brain metastases. (d) The subgroup of patients with brain metastases.
Analyses stratified by the antibody component of ADCs revealed a better ORR and CBR for patients on anti-HER2 ADCs compared to the anti-Trop2 ADC (test for subgroup difference, p < 0.01; Supplemental Figure S1).
Three studies were included for a PFS analysis stratified by IHC status, with 524 patients classified as IHC1+ and 592 as IHC2+ (Figure 4). The median PFS for the IHC1+ group was 10.6 months (95% CI, 8.2–13.7 months) and 9.7 months (95% CI, 6.1–15.6 months) for the latter. No significant difference between groups was observed (p = 0.74).
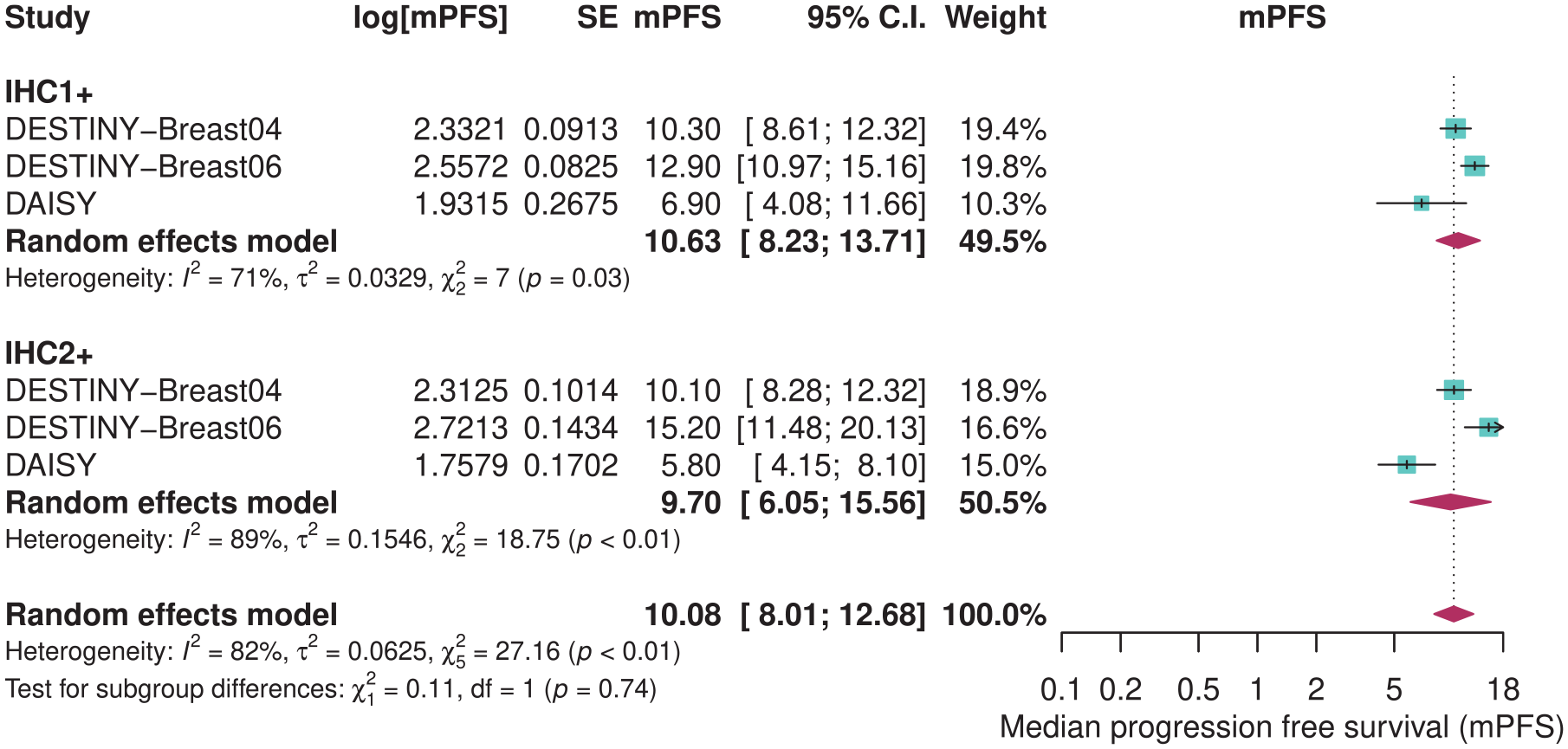
mPFS in HER2-low patients receiving T-DXd according to IHC status.
The subgroup analysis for ORR according to HoR expression included seven studies with 151 HoR-negative and 934 HoR-positive patients (Supplemental Figure S2(A)). Objective responses were numerically higher in the HoR-positive compared to the HoR-negative group (48% vs 38%), yet it did not reach statistical significance (p = 0.24). The median PFS considering both groups also revealed a tendency to a better, but nonsignificant benefit for the HoR-positive cohort (6.0 vs 10.1 months, p = 0.13) (Supplemental Figure S2(B)).
Few studies reported the IC benefit of ADCs (Supplemental Figure S3). The subgroup analysis of 3 studies with 52 patients with brain metastases revealed an IC-ORR of 26% (95% CI, 9%–55%). The IC-CBR considering two studies with 28 patients was 58% (95% CI, 40%–76%).
Supplemental Tables S5–S7 describe the PFS and OS from all studies, including those that could not be pooled for main and subgroup analyses.
Adverse events
Treatment-emergent adverse events (TEAEs) of SG and T-DXd are shown in Supplemental Figure S4. TEAEs grade 3 or higher were more commonly reported in the SG group, whereas the T-DXd group had a higher rate of TEAEs leading to treatment discontinuation (p < 0.01).
The most common all-grade AEs in patients receiving T-DXd were nausea (67%) and fatigue (48%), followed by alopecia (42%), neutropenia (36%), and interstitial lung disease (ILD, 10%) (Supplemental Figure S5). Severe (i.e., grade ⩾ 3) nausea, fatigue, and neutropenia were observed in 1%, 3%, and 17% of patients, respectively. In DESTINY-Breast06, 6 patients among 434 on T-DXd experienced grade ⩾ 3 ILD, whereas DESTINY-Breast04 registered 8 cases of grade ⩾ 3 ILD. These were consistent with the known toxicity profile of T-DXd (Supplemental Figure S5).
In most reports, AEs were not fully described for the SG group since HER2-low patients were a post hoc or subgroup analysis. Limited data were available on the safety profile for MRG002 and RC48-ADC. Nevertheless, they were often associated with hematotoxicity and gastrointestinal reactions.
Heterogeneity
We found no significant association between the median number of prior therapies and the ORR in the meta-regression analysis (p = 0.22) (Supplemental Figure S6). The small number of studies could have impacted this finding. In addition, a high heterogeneity persisted even when adjusting for this variable (I2 = 86.1%, p < 0.0001).
The leave-one-out analysis yielded similar results for the ORR pooled analysis (Supplemental Figure S7). Three studies were considered outliers based on the Baujat plot analysis (Supplemental Figure S8).8,22,24 DESTINY-Breast06 and TROPiCS-02 greatly contributed to both effect size and heterogeneity, whereas DESTINY-Breast04 only to the effect size.8,22,24 Few real-world studies were included in this meta-analysis. However, considerable heterogeneity remained in the sensitivity analysis stratified by study design (Supplemental Figure S9). The inclusion of clinical studies at different phases and differences in eligibility criteria likely influenced this finding.
Quality assessment
All 10 nonrandomized studies included in this meta-analysis were considered to have a moderate risk of bias (Supplemental Table S8).16,17,21,25,28,29,31–36 They predominantly lacked adjustment for confounding factors, failing to meet the specified criteria for the first domain. The four RCTs met most criteria for all domains and were determined to be at low risk of bias (Supplemental Table S8).8,15,22,24 The funnel plot analysis for the ORR revealed a wide distribution of studies (Supplemental Figure S10). Nonetheless, Egger’s test did not indicate the presence of publication bias (t = −1.99, p = 0.0812).
Discussion
Out of 2883 patients with a/m HER2-low BC evaluated in both CTs and real-world settings, the ORR, DCR, and CBR of patients treated with any ADC were 39%, 79%, and 54%, respectively. Patients treated with T-DXd achieved significantly higher responses than those who received SG, RC48-ADC, and MRG002, although the group on SG consisted of a heavier pretreated population compared to other ADCs. In the pooled analysis of four RCTs, T-DXd and SG demonstrated a greater antitumor response than TPC. Also, ADCs were associated with a 50% and 30% reduction in the risk of progression and death, respectively, compared to chemotherapy. The subgroup analyses according to IHC and HoR status were statistically non-significant. However, all groups seem to derive benefits from ADCs.
ADCs bind to targeted antigens and internalize into tumor cells where they release a cytotoxic payload. 37 Several mechanisms may influence ADC efficacy, including payload selection, antigen density, and tumor microenvironment. 37 Interestingly, for HER2-low tumors, even modest expression of HER2 may be sufficient to allow ADC uptake. 38 Another rationale for their potent antitumor activity is the bystander effect, by which surrounding non-target cells also experience cytotoxic effects of the released payload.37–40 This phenomenon holds particular relevance in tumors with heterogeneous antigen expression such as HER2-low.37–39 However, it is unclear whether the efficacy of ADCs results from HER2 blockade by the antibody or if this component mainly transports the payload into HER2-expressing cells. 37
The groundbreaking DESTINY-Breast04 trial proved the benefit of next-generation ADCs in metastatic patients expressing low levels of HER2. 8 T-DXd, a humanized antibody linked to a topoisomerase I blocking agent (deruxtecan), was associated with responses superior to 50% in this study. 8 Moreover, the group of 373 HER2-low mBC patients on the ADC had a reduction of 49% and 36% in the risk of disease progression and death, respectively, compared to chemotherapy. 8 This was soon followed by studies demonstrating the consistent activity of T-DXd in heavily pretreated populations. DAISY, a phase II trial, studied T-DXd in a cohort of 73 HER2-low patients who received a median of five prior lines of therapy. 21 The authors reported a meaningful ORR of 37.5% and a median PFS of 6.8 months with longer follow-up (38.4 months). 21
Recently, primary results of phase III DESTINY-Breast06 have expanded the treatment landscape of T-DXd. 22 In this study, T-DXd was administered in earlier lines compared to DESTINY-Breast04. It included chemotherapy naïve patients in the metastatic setting, with HoR+ HER2-low and ultralow disease previously treated with endocrine therapy. 22 In the HER2-low cohort composed of 359 patients, T-DXd was associated with a median PFS of 13.2 versus 8.1 months for 354 patients on TPC (HR, 0.62; 95% CI, 0.51–0.74; p < 0.0001). 22 In the ITT population, including both HER2-low and HER2-ultralow patients (n = 436), median PFS was similar, with 13.2 months for those on T-DXd and 8.1 months for 430 patients treated with chemotherapy (HR, 0.63; 95% CI, 0.53–0.75; p < 0.0001). 22 These findings support the superior activity of T-DXd in earlier treatment lines and an extension of benefit to even lower HER2 receptor expression. 22
The meaningful efficacy of T-DXd has also shed light on other ADCs.8,41 In three studies initially planned in HER2-negative BC patients treated with SG, post hoc analyses were performed according to the IHC score.15,24,28 The phase III ASCENT trial analysis revealed a PFS and OS benefit of 56% and 57%, respectively, in 123 patients with HoR-negative HER2-low tumors receiving SG compared to those on TPC. 15 The analysis by the TROPiCs-02 trial revealed a 42% PFS benefit in 283 patients with HoR-positive HER2-low BC treated with SG. 24 The phase II EVER-132-001 trial reported a CBR of 40.5% and a median PFS of 5.5 months in 37 HER2-low patients. 28 In our pooled analysis of all three studies, SG elicited an ORR and DCR in 28% and 74% of patients, respectively.
Other ADCs being studied for HER2-low patients are MRG002 and disitamab vedotin (RC48-ADC).16,17 For the first, the antibody component is composed of modified trastuzumab, and for the latter, of hertuzumab.16,17 Both are coupled with a microtubule inhibitor monomethyl auristatin E payload.16,17 MRG002 was studied in a phase II trial on 56 a/m HER2-low BC patients, most were HoR-positive, and 34.7% achieved an ORR. 16 A single-arm phase II study tested RC48-ADC alone and in combination with immune checkpoint inhibitors in a cohort of 38 patients with HER2-low BC. 17 The authors found an ORR of 29% and a median PFS of 3.6 months. 17 Results from an ongoing phase III CT on RC48-ADC versus TPC in HER2-low mBC are awaited to understand its antitumoral activity better (NCT04400695).
Despite the proven efficacy of ADCs, there is still uncertainty about whether HER2-low constitutes a separate subtype.42–46 The meta-analysis by Molinelli et al. 46 including 1,797,175 patients reported a slightly higher OS in favor of HER2-low than the HER2-negative, irrespective of HoR status in the metastatic setting. The authors highlight that differences between groups were limited and possibly driven by HoR status. 46 In this meta-analysis, we included a large retrospective cohort by Tarantino et al. 33 which included 520 HER2-low patients. This study reported a median PFS of 7.8 and 4.5 months for HoR-positive and -negative cohorts, respectively. In the pooled analysis of three studies, we also found a higher PFS in favor of the HoR positive, although it was nonsignificant.
The prognostic value of the IHC score also remains unsettled. 47 Retrospective data support favorable outcomes for IHC1+ compared to IHC2+ tumors regardless of HoR status in early-stage BC. 47 Scores of IHC1+ were associated with increased survival compared to IHC0, although this was not extended for IHC2+ compared to IHC0. 48 On the other hand, in DESTINY-Breast04 and DAISY trials, both IHC1+ and IHC2+ subsets derived benefit from T-DXd, suggesting this test may not be accurate in predicting efficacy.8,25 In this meta-analysis, three studies were pooled for analysis according to IHC status (524 patients were classified as IHC1+ and 592 as IHC2+).8,22,33 A similar median PFS was found for both groups.
The clinical activity of ADCs in HER2-low BC patients with brain metastases is still to be explored. In patients with HER2-positive tumors, T-DXd was shown to elicit remarkable IC responses.49–51 In the HER2-low population, the phase II DEBBRAH trial is currently assessing antitumor responses of patients with CNS involvement and variable HER2 expression treated with T-DXd. 31 In cohorts 2 and 4 with 12 patients with HER2-positive or HER2-low BC and asymptomatic or progressive BMs, 50% achieved an ORR. 32 In cohort 5, the IC-CBR was 71.4% in 7 patients with leptomeningeal disease. 31 In our pooled analysis of three studies, 26% out of 52 patients achieved an IC-ORR on T-DXd. The analysis, including 2 studies with 28 patients, revealed an IC-CBR of 58%. These findings suggest that ADCs may be effective in HER2-low brain metastases. However, further evidence on this subset is warranted.
Concerning safety, we found higher frequencies of TEAE grade ⩾ 3 and TEAE leading to drug discontinuation for T-DXd compared to SG. Interestingly, T-DXd and SG share a payload composed of topoisomerase inhibitors, and some AEs are common to both. 52 Nonetheless, the lack of individual patient data prevented us from exploring this association further. Understanding toxicity in the scenario of ADC sequencing is particularly relevant.52,53 Whether toxicity is cumulative and its impact on the decision of the next ADC remains unknown.52,53 Ongoing clinical studies are likely to explore some of these gaps and provide guidance on how to mitigate ADC toxic effects (NCT03742102, NCT06188559, and NCT05520723).
The introduction of next-generation ADCs has drastically changed the treatment landscape of metastatic HER2-low BC treatment. Despite initial clinical benefit, a subset of patients will eventually progress on ADCs.54,55 Mechanisms of acquired resistance are not fully elucidated but appear to be mainly mediated by the antibody or payload components or alterations in the tumor microenvironment.56,57 Previously, T-DM1 resistance was associated with loss of HER2 expression, resulting in poor antigen–antibody binding. 58 Similar results were found in DAISY, the first study to unravel some of the T-DXd resistance mechanisms. 25 Yet, in this study, four out of six patients still exhibited intratumoral T-DXd uptake, suggesting that this may not be the leading mechanism of resistance. 25 In triple-negative BC and other tumors, upregulation of efflux pumps and disruptions in the payload’s target were also shown to play a role in developing resistance.59,60 The identification of predictive biomarkers and development of strategies such as ADC-combined regimens may offer alternatives to overcoming or preventing ADC-acquired resistance.54,57
Understanding mechanisms behind acquired resistance is one among several unmet challenges surrounding ADC treatment in HER2-low BC. 61 HER2-status variability during disease evolution, considerable intratumoral heterogeneity, and methodological and analytical divergences in pathology assays may make standardization of HER2-low definitions difficult, rendering appropriate patient selection challenging.9,61 Moreover, in this meta-analysis, we included only phase II CTs, but several phase I studies are currently investigating other ADCs in HER2-low BC (NCT02277717, NCT03523572, NCT02980341, and NCT03451162). With the expanding range of ADCs available, the optimal sequencing for those progressing on prior ADCs or patients who may be eligible for multiple ADCs is yet to be clarified. 45 In the ADC era, advances in molecular imaging techniques may offer alternatives in measuring tumor target expression and help to personalize clinical decisions. 62
This study has certain limitations. First, many of the reports included in this meta-analysis were abstracts or conference presentations with preliminary or not fully matured results. For some ADCs, only one study was available or were small studies with limited patient numbers. A high heterogeneity was seen in some analyses, likely due to the inclusion of retrospective cohorts and studies with different eligibility criteria. Due to the lack of data from individual studies, we could not perform an analysis based on important factors such as previous treatments or metastatic sites. To address some of these limitations, we used random-effect models across all analyses, performed multiple subgroup analyses, and a meta-regression according to the median number of prior therapies. Lastly, we performed sensitivity analyses (according to study design and leave-one-out analysis) and explored heterogeneity using the Baujat plot.
Conclusion
Our systematic review and meta-analysis supports the efficacy of ADCs (T-DXd, SG, RC48-ADC, and MRG002) in patients with a/m BC whose tumors express low levels of HER2. Particularly, we found remarkable responses in patients treated with any ADC and a significant improvement in all efficacy outcomes—ORR, DCR, CBR, OS, and PFS—compared to standard therapy. Consistent antitumor activity was seen for HER2-low patients on ADCs regardless of HoR and IHC status. Future studies should focus on bringing ADCs into earlier lines of therapy, developing accurate HER2-testing tools, and unraveling mechanisms of resistance and ADC sequencing. Strategies focusing on toxicity mitigation also warrant development.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359241297079 – Supplemental material for Antibody–drug conjugates in patients with advanced/metastatic HER2-low-expressing breast cancer: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-tam-10.1177_17588359241297079 for Antibody–drug conjugates in patients with advanced/metastatic HER2-low-expressing breast cancer: a systematic review and meta-analysis by Isabella Michelon, Maria Inez Dacoregio, Maysa Vilbert, Jonathan Priantti, Caio Ernesto do Rego Castro, Lucas Vian, Paolo Tarantino, Evandro de Azambuja and Ludimila Cavalcante in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
