Abstract
Background:
Human epidermal growth factor receptor 2 (HER2)-low has emerged as a potential new entity in breast cancer (BC). Data on this subset are limited, and prognostic results are controversial, evidencing the need of further data in a BC real-world cohort.
Methods:
Patients with HER2-negative stage I–III BC diagnosed between 2006 and 2016 were retrospectively reviewed in a single cohort from the Catalan Institute of Oncology Badalona. Demographics and clinicopathological characteristics were examined via medical charts/electronic health records. We aim to describe and compare HER2-0/HER2-low populations through Chi-square or Fisher test, and explore its prognostic impact using Kaplan–Meier curves and Cox regression models.
Results:
From a cohort of 1755 BC patients, 1401 invasive HER2-negative, stage I–III cases were evaluated. 87% were hormone receptor (HR)-positive versus 13% triple negative (TNBC). Overall, 43% were HER2-0 and 57% HER2-low (61% immunohistochemistry (IHC) 1+ and 39% IHC 2+). Comparing HER2-low versus HER2-0, HER2-low showed higher proportion of estrogen receptor (ER)-positive (91.6% vs 79.9%, p ⩽ 0.001) and progesterone receptor (PR)-positive (79.8% vs 68.9%, p ⩽ 0.001) cases. HER2-0 exhibited higher proportion of TNBC (20.1% vs 8.4%, p = 0.001), grade III tumors (28.8% vs 23.5%, p = 0.039), and higher Ki67 median value (26.47% vs 23.88%, p = 0.041). HER2-low was associated with longer time to distant recurrence (TTDR) compared to HER2-0 (67.8 vs 54.1 months; p = 0.015) and better BC-related survival (19.2 vs 16.3 years; p = 0.033). In the multivariable analysis, HER2-low was not an independent prognostic factor for TTDR and BC-related survival. ER expression showed a strong association with longer TTDR (Hazard Ratio: 0.425, p ⩽ 0.001) and improved BC-related survival (Hazard Ratio: 0.380, p ⩽ 0.001). PR expression was also associated with longer TTDR (Hazard Ratio: 0.496, p ⩽ 0.001), and improved BC-related survival (Hazard Ratio: 0.488, p ⩽ 0.001). Histological grade III was significantly associated with shorter TTDR (Hazard Ratio: 1.737, p = 0.002). Positive nodal status was the strongest factor correlated with worse BC-related survival (Hazard Ratio: 2.747, p ⩽ 0.001).
Conclusion:
HER2-low was significantly associated with HR-positive disease, whereas HER2-0 group had higher incidence of TNBC, histological grade III and higher Ki67%. Although HER2-low group was associated with longer TTDR and improved BC-related survival, these findings could be explained by the greater proportion of favorable prognostic features in this subgroup compared to HER2-0.
Introduction
Human epidermal growth factor receptor 2 (HER2) is an important prognostic and predictive biomarker in breast cancer (BC). 1 HER2-low tumors have been defined as an HER2 immunohistochemistry (IHC) expression of 1+ or 2+ without signals of gene amplification by in situ hybridization (ISH).2–9
HER2-low tumors represent a heterogeneous population including both hormone receptor (HR)-positive and HR-negative BC that may vary in prognosis and response to treatments. Up to approximately 50% of HER2-negative BC are considered as HER2-low.2,7,8
Traditionally, HER2-low tumors have not benefited from the use of anti-HER2 targeted therapies, such as trastuzumab, pertuzumab, or first-generation antibody-drug conjugate (ADC) trastuzumab emtansine (T-DM1).10–14 However, the novel ADC trastuzumab deruxtecan (T-DXd) has shown efficacy in pretreated patients with advanced or metastatic HER2-low BC, as observed in the phase III Destiny-Breast04 trial, evidencing a significant improvement in progression-free and overall survival over conventional chemotherapy. 15 These encouraging results led to the hypothesis that HER2-low tumors may represent a distinct BC biological subtype and a potential new target for novel directed therapies.
Several studies have characterized the clinicopathological and molecular features of HER2-low tumors, with inconsistent results. It has been reported that HER2-low tumors are more frequently associated with HR-positive disease than HR-negative (TNBC) 8 also presenting larger tumors, 8 lower Ki67 scores2,8,16 and more likely to be clinical node-positive. 3 Retrospective studies have assessed the distribution of PAM50 intrinsic subtypes in HER2-low disease. In an analysis of 1576 HER2-negative BC tumors, 41.4% were luminal A, 24.6% luminal B, 26.3% basal-like, 4.6% HER2-enriched, and 3.1% normal like. 8 Within HR-positive disease, luminal A subtype was more frequent in HER2-low compared to HER2-0 (58.9% vs 2.8%), while luminal B and basal-like subtypes were less frequent in HER2-low compared to HER2-0. 8 No significant differences were observed in subtype distribution in TNBC according to HER2 status. 8 Agostinetto et al. 5 also presented results of PAM50 distribution in 789 tumor samples. Luminal A was the most represented subtype (50.8%) followed by luminal B (19.5%), and basal-like tumors (17.6%). Within HR-negative disease, intrinsic PAM50 subtypes varied significantly between HER2-0 and HER2-low tumors, 8 showing higher proportion of basal-like among HER2-0 and higher proportion of HER2-enriched in HER2-low subgroup. No statistically significant differences were observed in PAM50 distribution when comparing HR+/HER2-0 versus HR+/HER2-low tumors, 5 being luminal A followed by B the most frequent subtypes.
In both studies, among HR-positive tumors, ERBB2 gene and luminal-related genes determined by PAM50 platform were more commonly linked to HER2-low compared to HER2-0 disease.5,8
Concerning prognostic significance of HER2-low tumors, also discordant results have been reported. Diverse studies have found no significant differences in survival outcomes between patients with HER2-low versus HER2-0 tumors,3,5,8,17 whereas others have observed that HER2-low have a prognostic role presenting improved2,4 or worse outcomes.18–22
Differences in trial sampling size, patients’ characteristics, length of follow-up, and discordances in HER2-low definition and detection may explain some of these inconclusive results. However, these studies suggest that clinicopathological factors, such as HR status and nodal stage, could explain these differences. HR expression is a major biological driver of prognosis, 2 which can confound prognostic outcomes. 16
The aim of our study is to describe clinicopathological characteristics and survival outcomes in a retrospective cohort from the Catalan Institute of Oncology (ICO) Badalona, classically classified as HER2-negative, comparing HER2-low versus HER2-0 tumors, to identify clinical associations and investigate its role in BC prognosis. Our findings will allow one to elucidate if HER2-low could be considered as a new entity that shows different clinical behavior compared to HER2-0 subpopulation.
Materials and methods
Study design and participants
We retrospectively collected data from all patients diagnosed with stage I–III BC, between 2006 and 2016 in ICO Badalona; stages 0 (carcinoma in situ) and IV (metastatic disease) were excluded. The study was conducted in accordance with the ethics principles of the Declaration of Helsinki and approved by the Research Ethics Committee of Hospital Germans Trias i Pujol (ref. PI-23-153). All patients provided written informed consent, in accordance with Law 14/2007 on Biomedical Research, as well as Law 3/2018 on the Protection of Personal Data/European Data Protection Regulation 2016/679. This article was designed following STROBE guidelines.
We included patients diagnosed with an invasive BC in local or locally advanced stages. HER2-low subgroup was defined by HER2 IHC of 1+ or 2+ with lack of HER2 amplification by ISH, being HER2-0 defined by IHC of 0. Patients considered HER2-positive invasive carcinomas by ISH were excluded. The patient flow diagram for the study is shown in Figure 1.

Flow diagram of the study.
The following baseline clinicopathological characteristics were collected from medical records: age at diagnosis, gender, menopausal status, estrogen receptor (ER) status, progesterone receptor (PR) status, HER2 status (IHC and ISH), histologic subtype and grade, Ki67 percentage, and TNM staging. We also collected data regarding treatments offered (chemotherapy, hormonotherapy, surgery, and radiotherapy), date and site of disease recurrence, and survival status at last follow-up. Tumor stage was classified according to the TNM classification of the Union International Cancer Control. 23
Methods for determining HER2 and other molecular biomarkers
HER2, Ki67, and HR status were measured with standardized methods approved for routine clinical testing. HER2 immunohistochemistry staining was scored according to the criteria specified by DAKO (Agilent) for the interpretation of the HercepTest™ (https://www.agilent.com/en/product/pharmdx/herceptest-kits). Immunoreaction was determined to be strongly positive (3+) if a strong complete membrane staining was observed in more than 10% of neoplastic cells, or to be weakly positive (2+) if more than 10% of the tumor cells showed weak to moderate complete membrane staining. All other staining patterns were interpreted as negative (0/1+). Fluorescence situ hybridization (FISH) was performed by PathVysion LSI HER-2/neu SO/CEP 17 SGn probe on HER2 immunopositive cases (2+ or 3+), and the HER2 signal was analyzed by optical microscopy (Leica DMLS2). ER and PR levels were detected by IHC and were considered positive only if nuclear positivity was seen in more than 1% of neoplastic cells. Ki67 score was also measured by IHC.
Statistical analysis
A comprehensive cohort description analysis based on demographic, clinical, and biological data was performed using software SPSS 23.0 (SPSS, Chicago, IL, USA).
Categorical variables were summarized through frequencies and percentages, and quantitative variables using means and standard errors, or medians and interquartile ranges. Chi-square or Fisher’s test was used by comparing categorical variables, as well unpaired T-student test for comparing these with quantitative ones. p-values were calculated using a two-tailed test, considering a value under 0.05 as statistical significant, t-student test. Plots were drawn using GraphPad Prism.
Regarding survival analyses, different endpoints were analyzed: (i) time to recurrence (TTR) was defined as the time from the breast surgery to date of recurrence or the last follow-up, in cases of no relapse, and subclassified into time to local recurrence (TTLR) or time to distant recurrence (TTDR); and (ii) overall survival (OS) was defined as the time from date of BC diagnosis to date of death resulting from any cause. Breast cancer-related survival (CRS) was also evaluated, only considering BC-associated death. Survival analysis according to HER2-low status was performed by using Kaplan–Meier curves and log-rank function. Univariable and multivariable analyses considering different clinical covariables were performed using Cox regression models, represented by the Hazard Ratio with a 95% of confidence interval (95% CI). p-values under 0.05 were considered as statistically significant.
Results
Patient cohorts and clinicopathologic features
From a whole cohort of 1755 BC patients, a total of 1401 invasive HER2-negative BC patients stage I–III were included in the analysis. Total follow-up time was 250 months. The distribution of HER2-0 and HER2-low in this cohort was 606 (43%) and 795 (57%), respectively (Figure 1 and Table 1).
Clinicopathological features of the overall population and according to HER2 status.

HER2, Human epidermal growth factor receptor 2.
p-values in bold are considered statistically significant (p-value ≤ 0.05).
Overall, the median age at diagnosis was 58.69 years old. Menopausal status was confirmed in 941 patients (67.3%), while 458 (32.7%) were premenopausal. Most of the population were stages I and II (45.3% and 45.9%, respectively), followed by stage III (8.9%). Clinical nodal status was positive in 38.3% of patients (Table 1).
Regarding pathological characteristics, 81.6% were ductal carcinomas, 9.8% lobular, and 8.6% other special histologies (Supplemental Table 1). Histological grade was most commonly grade I and II (74.3%), whereas 25.7% were grade III. Ki67 median value was 25% (Table 1). Tumorectomy (68.1%) and sentinel lymph node biopsy (74.9%) were the preferred surgery approaches (Table 1).
Considering HER2 status, no significant differences were observed between HER2-0 and HER2-low in median age at diagnosis, menopausal status, clinical stage, clinical nodal status, histological subtype, neoadjuvant hormonotherapy, complementary chemotherapy and radiotherapy, and incidence of second neoplasms (Table 1 and Supplemental Table 1). There was a higher proportion of neoadjuvant chemotherapy in the HER2-0 group compared to HER2-low (18.1% vs 14.1%, respectively, p = 0.039; Supplemental Table 1).
A trend toward less aggressive surgical management among HER2-0 patients was also observed, including breast surgery (71% of tumorectomies in HER2-0 compared to 65.9% in HER2-low cohort, p = 0.045), and axillary surgery (sentinel lymph node biopsy was performed in 77.9% of HER2-0 patients compared to 72.6% in HER2-low, p = 0.027; Table 1).
HR expression, grade, and Ki67 according to HER2 status
HR was positive in 87% of cases, while 13% were TNBC (Figure 2(a)). Considering HR positive tumors, 487 (39.9%) presented HER2 IHC 0, 444 (36.5%) HER2 IHC 1+, and 288 (23.6%) IHC 2+. In the TNBC subgroup, 119 (65.4%) showed HER2 IHC 0, 38 (20.9%) HER2 IHC 1+, and 25 (13.4%) HER2 IHC 2+. These data show that HER2-low BCs account for 60.1% of HR-positive and 34.6% of TNBC (Figure 2(a)).

Hormone receptor status, HER2-low status, and IHC scores distributions. (a) Percentage IHC scores distribution within the hormone receptor-positive (HR-positive) and negative (TNBC) population. Percentage of HER2-low tumors is shown in each population. (b) Percentage hormone receptor-positive (HR-positive) and negative (TNBC) population distributions within each IHC score (in percentage). Percentage of HER2-low tumors is shown in each population.
HR-positive tumors were more prevalent than TNBC tumors in both the HER2-0 and HER2-low groups (Figure 2(b)). Among HER2-0 tumors (IHC 0), 484 patients (79.9%) presented ER expression and 122 (20.1%) were considered ER-negative. Specifically, 77.9% presented ER expression higher than 10%, 2% from 1% to 10%, and 20.1% were <1%. In the HER2-low group, 728 (91.6%) had ER-positive and 67 (8.4%) ER-negative. Specifically, 89.3% expressed ER higher than 10%, 2.3% from 1% to 10%, and 8.4% <1% (Table 2). Regarding PR expression, 417 patients (68.9%) presented a positive expression and 188 (31.1%) were considered PR-negative within the HER2-0 group. Specifically, 59.8% presented PR expression higher than 10%, 9.1% from 1 to 10%, and 31.1% were <1%. In the HER2-low group, 634 (79.8%) had PR-positive and 161 (20.3%) PR-negative. Specifically, 67.7% expressed PR higher than 10%, 12.1% from 1 to 10%, and 20.3% <1% (Table 2).
Hormone receptor expression and response to hormone therapy of the overall population and according to HER2 status.

HR, Hormone receptor.
HER2-0 exhibited higher proportion of ER-negative (20.1% vs 8.4%, p < 0.001) and PR-negative tumors (31.1% vs 20.3%, p ⩽ 0.001), whereas HER2-low subgroup showed significant higher proportion of ER-positive (79.9% vs 91.6%, p ⩽ 0.001) and PR-positive (68.9% vs 79.8%, p ⩽ 0.001) cases. No differences were observed between HER2-low 1+ versus 2+ groups, highlighting that both entities can be classified under the category of HER2-low (Table 2).
HER2-0 group exhibited a statistically significant higher proportion of grade III (G3) tumors (28.8% vs 23.5%, p = 0.039; Figure 3(a)) and Ki67 median value (26.47% vs 23.88%, p = 0.041) compared to HER2-low group (Table 1). As previously described, 24 G3 tumors presented statistically significant higher Ki67 median values compared to G1/2 tumors in both HER2-0 and HER2-low, but no significant differences were observed in Ki67 median value between G1/2 and G3 within HER2-0 or HER2-low groups (Figure 3(b)).
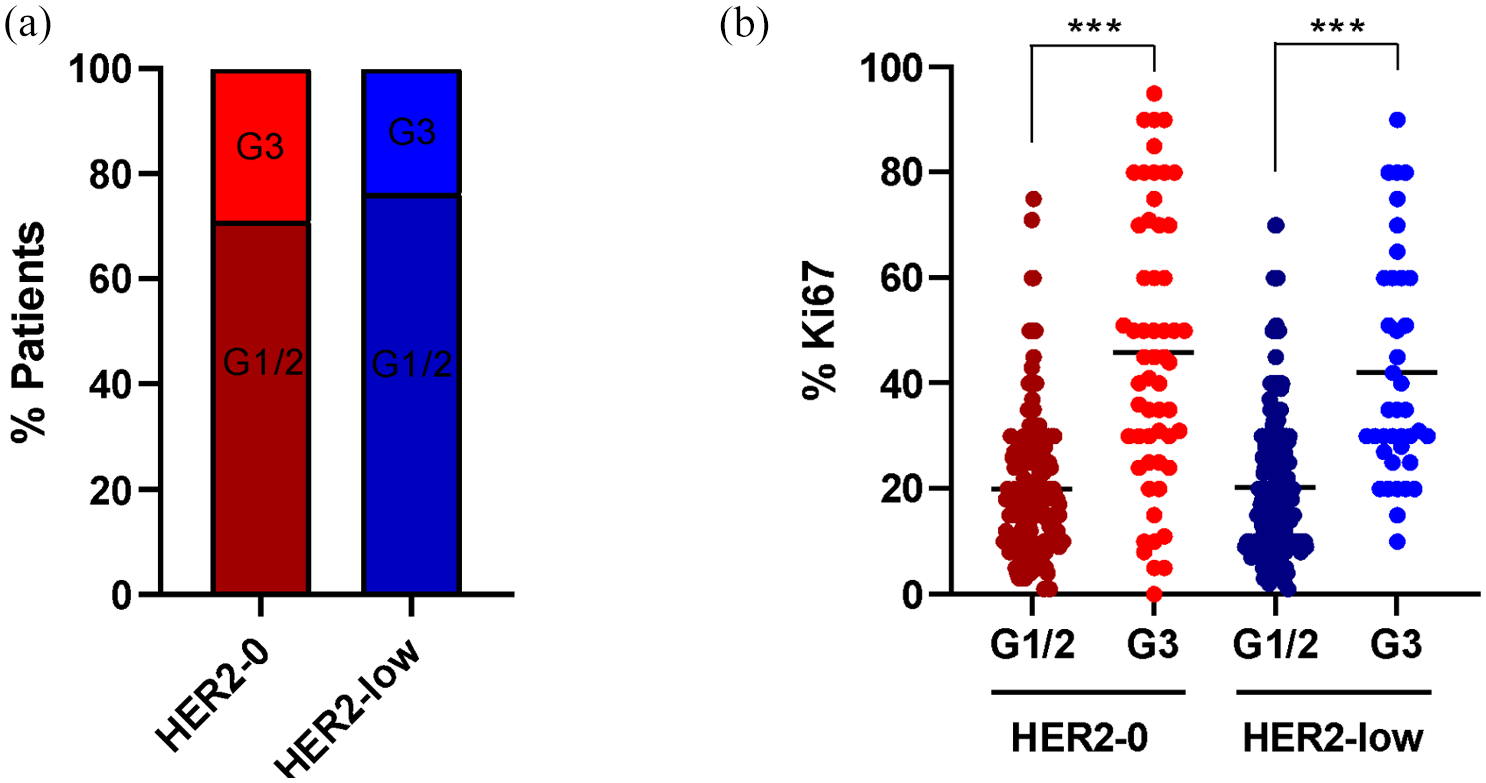
Histological grade and (a) Ki67 score (b) distribution in HER2-0 versus HER2-low tumors.
Survival outcomes according to HER2 status
There were no differences in terms of TTR, TTLR, and OS between HER2-low and HER2-0 groups. However, HER2-low subgroup was associated with a longer TTDR (Median: 54 vs 44 months; p-value: 0.011) and better BC-related survival (Median: not reached 50% survival; p-value: 0.020) compared to HER2-0 (Figure 4). Covariables that showed a significant impact in TTDR or BC-related survival together with HER2-low status, according to univariable analysis (Table 3), was included in multivariable analysis.

Time to recurrence, time to distant recurrence, and time to local recurrence, BC-related survival and OS in the global population.
Univariable analysis for TTDR and BC-related survival.
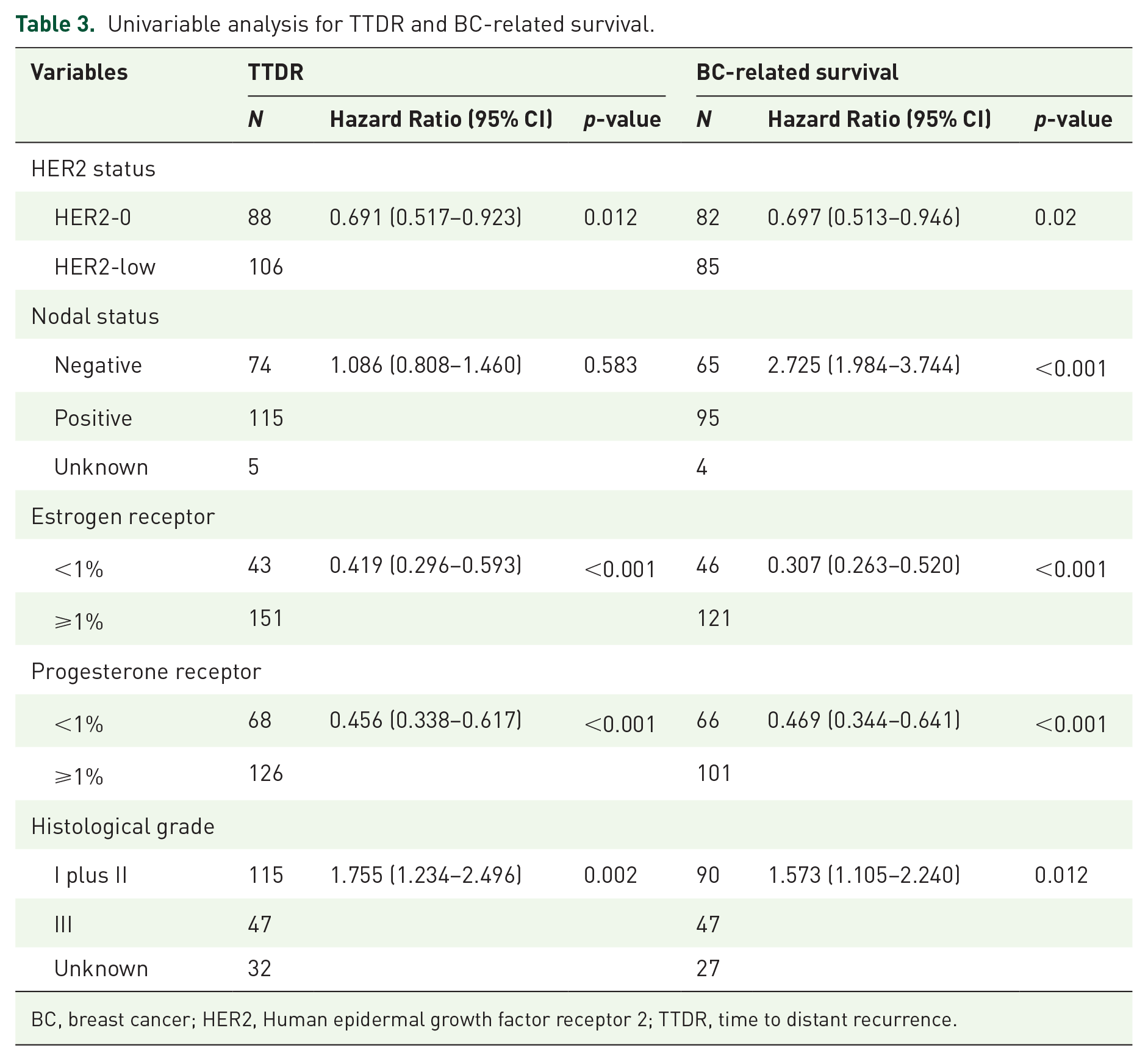
BC, breast cancer; HER2, Human epidermal growth factor receptor 2; TTDR, time to distant recurrence.
In this scenario, ER and PR status showed the strongest association with longer TTDR (Hazard Ratio: 0.425, 95% CI 0.300–0.620, p ⩽ 0.001) and (Hazard Ratio: 0.496, 95% CI (0.345–0.635), p ⩽ 0.001), respectively. Histological grade III was associated with shorter TTDR (Hazard Ratio: 1.744, 95% CI 1.220–2.473, p = 0.002). HER2-low status did not show to be an independent prognostic factor in TTDR (Figure 5).

Multivariable analysis including major clinicopathological factors for TTDR. ER and PR positive expression were significantly associated with longer TTDR (Hazard Ratio 0.425, p ⩽ 0.001) and (Hazard Ratio 0.469, p ⩽ 0.001), respectively, while histological grade III (Hazard Ratio 1.744, p = 0.002) was associated with worse prognosis.
Regarding BC-related survival, the multivariable analysis showed that ER (Hazard Ratio: 0.389, 95% CI 0.275–0.551, p ⩽ 0.001) and PR (Hazard Ratio: 0.488, 95% CI 0.357–0.669, p ⩽ 0.001) were significantly associated with longer BC-related survival. In contrast, positive nodal status was the strongest factor related to worse BC-related survival (Hazard Ratio: 2.747, 95% CI 2.000–3.744, p ⩽ 0.001) followed by histological grade III (Hazard Ratio: 1.543, 95% CI 1.082–2.199, p = 0.017). Again, HER2-low status was not an independent prognostic factor in BC-related survival (Figure 6).

Multivariable analysis including major clinicopathological factors for BC-related survival. ER and PR positive expression were significantly associated with improved BC-related survival (Hazard Ratio 0.389, p ⩽ 0.001) and (Hazard Ratio 0.0.488, p ⩽ 0.001), respectively. Positive nodal status (Hazard Ratio 2.747, p ⩽ 0.001) and histological grade III (Hazard Ratio 1.943, p = 0.017) were associated with worse BC-survival.
Due to the widely demonstrated impact of HR status in survival, and the observed enrichment of both ER and PR in HER2-low group in our cohort, we also analyzed the effect of HER2-low status in survival corrected by HR expression. Again, no statistically significant differences in TTDR or BC-related survival were observed when analyzing HER2-low versus HER2-0 and stratifying by HR status (Supplemental Figures 1 and 2).
Discussion
The rise of novel ADCs in BC has changed the classical dichotomous classification of HER2 disease. Efficacy results obtained with T-DXd in HER2-low metastatic BC patients 15 have opened a promising treatment landscape in this subset of patients and have raised questions about whether HER2-low disease should be considered a new prognostic entity.
In this work, we presented a retrospective analysis including classically considered HER2-negative tumors, comparing clinicopathological features and survival outcomes between HER2-0 and HER2-low subgroups.
We observed a significant association between HER2-low tumors and HR expression, in line with previously published studies.2,3,8 In contrast, HER2-0 tumors were significantly associated with HR-negative disease, higher histological grades, and higher Ki67 scores, which is also consistent with previous literature.2,8,16
Results showed significantly more neoadjuvant chemotherapy treatments among HER2-0 tumors, which could be partially explained by the enrichment of triple negative tumors in this population. A trend toward a less aggressive surgical management was also observed in the HER2-0 group. These results could be directly related to the higher use of neoadjuvant chemotherapy in this subgroup of patients. In the line of our findings, it has been reported in the bibliography that HER2-low cases exhibit larger tumor sizes and more nodal involvement compared to HER2-0.8,19 However, other studies exhibited opposite findings, evidencing smaller tumor sizes in HER2-low tumors.25,26
Here, we have not observed differences between HER2-low 1+ and 2+ groups in terms of HR expression, which leads to the conclusion that both entities can be classified under the category of HER2-low. However, we cannot be sure that a more accurate classification of HER2 expression will change this paradigm because conventional IHC appears to have become limited in this context. 27 A more sensitive technique is crucial to reclassify HER2-negative BC accurately, establishing various levels of low HER2 expression and determining the minimum HER2 levels for benefiting from HER2-targeted ADCs. In the phase II Daisy trial, efficacy of T-DXd was evaluated in HER2-overexpressing, HER2-low, and HER2-0 tumors. 28 Notably, antitumor activity was observed in patients with HER2-0 tumors, indicating that these patients could benefit from novel HER2-directed ADC therapies. These data suggest that IHC-0 cases may also have low levels of HER2 protein expression, known as HER2 ultralow, considered as expression > 0 and < 1+ by IHC. Based on these results, IHC may not be the most accurate test to determine HER2 expression to predict efficacy in patients with HER2-low BC, and current detection and classification techniques should be optimized.
The ongoing DB-06 trial (NCT04494425) evaluates T-DXd efficacy in metastatic HER2-low/HR-positive BC, including patients with HER2-0 or ultra-low. This trial could provide evidence for defining the threshold of HER2 levels needed to benefit from T-DXd therapy and determine the clinical significance of distinguishing HER2-low BC from HER2-0 tumors with current testing methods. Alternatively, while more sophisticated and cutting-edge techniques are being developed, amplifying IHC signals via amplification systems, like avidin-biotin complex (ABC), labeled streptavidin-biotin (LSAB), and polymer-based methods,29–31 and implementing comprehensive evaluation criteria could be good cost-effective strategies for rapid clinical integration.
Regarding survival outcomes, we evidenced a longer TTDR and BC-related survival in HER2-low compared to HER2-0 tumors. Hence, better survival outcomes for HER2-low disease have also been reported in previous studies.2,4,25,32–38 These results may be explained by the enrichment of HR-positive tumors in HER2-low group, which is known to be a major biological driver of prognosis in BC and may confound prognostic outcomes.2,16 This fact is consistent with our findings from the multivariable analysis, in which ER and PR expression are postulated as the strongest independent prognostic factors of longer TTDR and BC-related survival. Prognostic significance of HER2-low status remains unclear, with lack of solid conclusions, but our results and previous literature suggest that the classification as HER2-low does not seem to have an intrinsic prognostic value, but rather associated with other factors such as the status of the HR 39 and lower histological grades. At molecular level, it has been described that only in HR-positive BC (and not in HR-negative), HER2-low tumors upregulate luminal-related genes (luminal A and B) and downregulate proliferation-related genes, basal-like genes, tyrosine kinase receptors, and PAM50 signatures (i.e., HER2-enriched, basal-like, normal-like).5,8 These observations suggest that the intricate interplay between HER2 (even at low levels) and HR pathways via PI3K/AKT/mTOR and MAPK downstream signaling pathways could define the specific molecular biology of HER2-low BC, and in consequence, its prognosis, compared to HER2-0 tumors.
Several recently published meta-analysis have also reported improved OS in HER2-low disease compared to HER2-0, which remained significant after correcting for HR expression.36–38 Similarly, improved disease-free survival in HER2-low subgroup was also observed in the global population and in HR-positive subgroup, whereas no significant results were observed within the HR-negative.
In any case, HER2-low is a clinically relevant biomarker for treatment with T-DXd, given the remarkable survival benefits observed with T-DXd compared to standard chemotherapy in this population.
Our study represents a large cohort of HER2-negative patients, with extensive collected data regarding clinical and pathological features. This volume of data allowed us to comprehensively analyze our cohort and obtain representative results of our population.
However, our study presents some limitations. In line with previously published literature, data are reported from retrospective databases. The lack of molecular data of included patients represents also a limitation in our analysis. On the other hand, stage III disease was scarcely represented in our cohort, which could have underpowered the results. However, the incidence of stage III in our cohort is similar to the observed during that period in our environment.40,41 Finally, variability among pathologists in the detection and classification of HER2-low may also be a limitation to consider when interpreting these results, and pathologists and clinicians, therefore, should be aware that an IHC 0 versus IHC 1+ result has a new treatment implication in the metastatic setting.
Conclusion
This study observes that there are clinical and survival differences between HER2-low and HER2-0 tumors. However, these findings could be explained by a higher incidence of favorable prognostic features in HER2-low subgroup, as suggested by multivariable analysis. New therapies for HER2-0 disease are an unmet medical need. Future research is needed to determine best practices for testing and scoring HER2 and to identify patients who will benefit from HER2-targeted therapies in the HER2-low setting.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241290720 – Supplemental material for Deciphering HER2-low breast cancer (BC): insights from real-world data in early stage breast cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359241290720 for Deciphering HER2-low breast cancer (BC): insights from real-world data in early stage breast cancer by Anna Pous, Adrià Bernat-Peguera, Assumpció López-Paradís, Beatriz Cirauqui, Vanesa Quiroga, Iris Teruel, Eudald Felip, Angelica Ferrando-Díez, Milana Bergamino, Laia Boronat, Margarita Romeo, Gemma Soler, Christian Mariño, Paula Rodríguez-Martínez, Laura Pons, Ester Ballana, Anna Martinez-Cardús and Mireia Margelí in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-2-tam-10.1177_17588359241290720 – Supplemental material for Deciphering HER2-low breast cancer (BC): insights from real-world data in early stage breast cancer
Supplemental material, sj-tif-2-tam-10.1177_17588359241290720 for Deciphering HER2-low breast cancer (BC): insights from real-world data in early stage breast cancer by Anna Pous, Adrià Bernat-Peguera, Assumpció López-Paradís, Beatriz Cirauqui, Vanesa Quiroga, Iris Teruel, Eudald Felip, Angelica Ferrando-Díez, Milana Bergamino, Laia Boronat, Margarita Romeo, Gemma Soler, Christian Mariño, Paula Rodríguez-Martínez, Laura Pons, Ester Ballana, Anna Martinez-Cardús and Mireia Margelí in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-3-tam-10.1177_17588359241290720 – Supplemental material for Deciphering HER2-low breast cancer (BC): insights from real-world data in early stage breast cancer
Supplemental material, sj-tif-3-tam-10.1177_17588359241290720 for Deciphering HER2-low breast cancer (BC): insights from real-world data in early stage breast cancer by Anna Pous, Adrià Bernat-Peguera, Assumpció López-Paradís, Beatriz Cirauqui, Vanesa Quiroga, Iris Teruel, Eudald Felip, Angelica Ferrando-Díez, Milana Bergamino, Laia Boronat, Margarita Romeo, Gemma Soler, Christian Mariño, Paula Rodríguez-Martínez, Laura Pons, Ester Ballana, Anna Martinez-Cardús and Mireia Margelí in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
