Abstract
Background:
Alveolar soft-part sarcoma (ASPS) is a rare tumor driven by the ASPSCR1-TFE3 fusion protein, with a propensity for metastasis. Prognostic factors remain poorly understood, and traditional chemotherapies are largely ineffective. Recent interest lies in immune checkpoint inhibitors (ICIs), yet predictive biomarkers for treatment response are lacking. Previous studies have shown promising results with ICIs in ASPS, indicating a need for further investigation into biomarkers associated with immune response.
Objectives:
To identify prognostic biomarkers in ASPS and to explore the role of immune-related markers, particularly L1CAM, in predicting patient outcomes.
Design:
A retrospective cohort study of 19 ASPS patients registered in the GEIS database. The study involved the collection of clinical and histopathological data, followed by an analysis of immune markers and gene expression profiles to identify potential prognostic indicators.
Methods:
Clinical and histopathological data were retrospectively collected from the GEIS-26 study cohort of 19 ASPS patients. Immunohistochemistry was performed to evaluate immune markers programmed death-1 ligand (PD-L1), programmed death-1, FAS, FASL, CD8, CD3, and CD4. An HTG ImmunOncology panel was conducted on formalin-fixed paraffin-embedded samples to explore gene expression. Effects of differentially expressed genes on survival were explored by Kaplan–Meier.
Results:
PD-L1 positivity was widely observed (63%) in tumors, and CD8+ lymphocytic infiltration was common. High CD8 density correlated with greater overall survival (OS) while not statistically significant. No associations were found for other immune markers.
Conclusion:
High
Plain language summary
Why was the study done? Alveolar soft-part sarcoma (ASPS) is a rare cancer with limited treatment options. Our study aimed to understand how the immune system responds to ASPS and explore potential treatments, as current therapies are often ineffective. What did the researchers do? We analyzed data from 19 ASPS patients to investigate immune response and potential treatment targets. We examined the expression of immune markers and genes related to immune response to identify factors influencing patient outcomes. What did the researchers find? We found that most tumors showed signs of an active immune response, with a protein called PD-L1 being present. We also noticed that many tumors had a type of immune cell called CD8+ lymphocytes. Although having more of these CD8+ cells seemed to be linked to better survival, this connection wasn’t strong enough to be certain. We didn’t find any clear links with other immune markers we looked at. However, we did find that a protein called L1CAM was more common in patients who had fewer CD8+ cells in their tumors, and this was linked to poorer overall survival. What do the findings mean? Our study sheds light on the immune response in ASPS and identifies potential targets for therapy. By understanding these mechanisms, we hope to pave the way for more effective treatments and improve outcomes for ASPS patients in the future.
Keywords
Introduction
Alveolar soft-part sarcoma (ASPS) is an exceedingly rare entity, molecularly characterized by the genetic rearrangement t(X;17)(p11;q25), resulting in the ASPSCR1-TFE3 fusion protein. 1 This fusion protein acts as an aberrant transcription factor, which results in an upregulation of angiogenesis and cell proliferation-related genes.2,3 This entity affects predominantly young patients and, although it shows an indolent growth pattern, it has a high metastatic potential, with more than half of the patients developing distant metastasis in the lungs, bones, and the central nervous system, even many years after the initial diagnosis.4,5 There is scarce data regarding ASPS prognostic factors: stage, size, and age at diagnosis have been described as prognostic in retrospective series and registries.4,6,7 There are no molecular prognostic factors described for ASPS to date, apart from the cell proliferation Ki-67 marker. 8 ASPS is not a tumor that is sensitive to classic cytotoxic drugs, but it has a notable sensitivity to antiangiogenic molecules, such as sunitinib, 9 cediranib,10,11 or pazopanib. 12 However, the responses to these drugs are limited in time and all patients with advanced disease will eventually succumb to it, 13 so new therapeutic options like immunotherapy are desperately needed for these patients.
The tumor expression of the immune checkpoints programmed death-1/programmed death-1 ligand (PD-1/PD-L1) has been described as a prognostic factor for some epithelial neoplasms.14,15 In sarcomas, its prognostic role is still controversial, with some studies suggesting a worse prognosis for patients with PD-L1-positive sarcomas,16–19 and other studies not being able to show this prognostic role. 20 However, the sarcoma family encompasses more than 80 different subtypes, so there are clear limitations to the generalization of these results for all histologic subtypes. In recent years, several immunomodulatory drugs have been widely developed in oncology, showing improvements in patient survival, especially in melanoma, lung, and genitourinary carcinomas. Noteworthy, the tumor expression of PD-L1 has been described as a predictive factor of response to these drugs. 21 In sarcomas, we already have some data on check-point inhibitors coming from clinical trials and clinical experience, suggesting that these drugs may be active in some specific sarcoma subtypes,22–24 such as ASPS.
Indeed, we have shown in a retrospective analysis that immune checkpoint inhibitors (ICIs) are active in the treatment of advanced ASPS, with patients achieving an overall response rate (ORR) of 54.4% and an extended overall survival (OS) (median not reached vs 34.7 months) after treatment. 25 In line with these results, a recent clinical trial showed considerable activity of the ICI atezolizumab in patients with advanced ASPS, with 19 out of 52 (37%) objective responses, 1 of them a complete response, and a median progression-free survival (PFS) of 20.8 months, 26 granting the FDA approval of atezolizumab for the treatment of patients with advanced ASPS. Curiously, responses were noted independently of the baseline expression of PD-1 or its ligand PD-L1. Likewise, combinations of ICIs have proven successful, as evidenced by a phase II trial regarding the combination of durvalumab (an anti-PD-L1 inhibitor) and tremelimumab (an anti-Cytotoxic T-lymphocyte associated protein 4 inhibitor). Among 10 ASPS patients evaluated using immune-related RECIST and immune-related response criteria, ORRs stood at 40% and 50%, respectively. Notably, two patients achieved complete responses. 27 Similar combinations, like nivolumab/ipilimumab in ASPS, have reported a prolonged response of 27 months. 28 Additional studies explore other combinations like tyrosine kinase inhibitors (axitinib) with anti-PD-1 (pembrolizumab), which have demonstrated an ORR of 50.4% in the 11 evaluable patients with ASPS. 29 Similar combinations like sunitinib/nivolumab reported partial responses in four out of seven patients (57%). 24 However, there remains a need for biomarkers of response to immune treatment to identify patients who would benefit from this therapy.
This study aims to analyze different protein and genomic markers, as well as the association between them, related to immune response in a retrospective series of ASPS patients.
Methods
Patients
Patients diagnosed with ASPS between December 1994 and July 2016, and registered in the online database of the Spanish Group for Research on Sarcoma (GEIS), were retrospectively analyzed. Clinical and histopathological data, treatment modalities, and survival outcomes were systematically collected. Ethical clearance was secured from the Ethics Committee of each participating institution under the principles outlined in the Helsinki Declaration. Tumor samples and clinical data were collected after the patients signed the informed consent form. OS was assessed from the time of diagnosis to the date of last follow-up or death, with only tumor-associated deaths considered as events. Disease-free survival (DFS) was calculated from the date of initial treatment to the time of recurrence. Relapse-free survival (RFS) was calculated for patients with localized disease, from the date of surgery to the time of relapse. One patient was lost to follow-up but was censored at their last known contact and included in the analysis for death, relapse, or recurrence. Median DFS and OS were calculated using the Kaplan–Meier method, with intergroup differences assessed via the log-rank test. The cutoff point for high (
Immunohistochemistry
The diagnosis was confirmed in all cases by an expert pathologist. Protein expression was tested on complete 4-µm sections of archived formalin-fixed paraffin-embedded (FFPE) blocks using PD-L1 (ab205921; Abcam, Cambridge, UK), PD-1 (ab52587; Abcam), FAS (C18C12; Cell Signaling, Danvers, MA, USA), FASL (NOK-1; Novus Biological, Centennial, CO, USA), CD8 (ab4055; Abcam), CD3 (ab16669; Abcam), and CD4 (EP204; Master Diagnostica, Granada, Spain) antibodies. Staining of PD-1, FAS, and FASL was categorized as positive or negative, based on the presence of staining. PD-L1 positivity cutoff was defined as at least 5% of cells showing membrane staining of any intensity. 30 For lymphocyte infiltration, we defined the following score: Negative (0%), low infiltration (1%–4% of total cells), and high infiltration (⩾5% of total cells). Results were revised by two expert pathologists in sarcomas.
Immunomodulation factors
Gene expression was assessed using an ImmunOncology (IO) panel, targeting 549 different mRNAs, using the HTG Edge Seq System (HTG Molecular, Tucson, AZ, USA) on a 5-μm FFPE section sample, according to the manufacturer’s protocol.31,32 We explored differential gene expression in different prognostic groups: localized/metastatic, PD-L1+/−, and lymphocytic infiltration >10%.
Guidelines
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement: guidelines for reporting observational studies 33 (Supplemental File 1).
Results
Clinical characteristics and survival
In all, 19 patients with ASPS were included in our study cohort. The median age at diagnosis was 22.8 years (6–62), with a female predominance: 13 females (68%) and 6 males (32%). The majority of tumors arose in the extremities (15, 79%). More than half of the patients were diagnosed with localized disease (14, 73%), while 5 (26%) already had distant metastasis at diagnosis. In total, 13 of 19 patients (68%) experienced metastatic spread either at baseline or during follow-up. Further details are shown in Table 1.
Clinical characteristics of the series and outcome.

GI, Gastro-Intestinal.
With a median follow-up from diagnosis of 83 months (12–147), 9 out of 14 (64%) patients with localized disease at diagnosis suffered a disease relapse, with a median RFS of 55 months (95% confidence interval (CI) 1–108) (Supplemental Figure 1). In this group of patients, survival rates were 100% at 24 months, 93% at 48 months, and 79% at the end of the follow-up.
Among those patients diagnosed with advanced disease, three patients died, survival rates were 80% at 24 months, 60% at 48 months, and 40% at the end of the follow-up.
Immunohistochemistry
Twelve out of 19 patients (63%) had PD-L1-positive tumors, and in all cases, some grade of lymphocytic infiltration was found. Nine patients presented with low CD8-positive density (up to 4% of cells), while 10 patients showed high density. The high CD8 group exhibited greater OS [Not reached (NR) vs 138 months] and DFS (54.9 vs 10.2 months) compared to the low group. However, no statistically significant differences were observed for either. Similarly, using the median DFS as a cutoff, patients with low DFS exhibited a mean CD8 density of 6.5% compared to 9.1% in the high DFS group, albeit with no statistically significant differences. Results from all immunohistochemistry are summarized in Table 2 and some examples are shown in Figure 1.
Immunohistochemistry results.

Low infiltration: up to 4% of total number of cells. High infiltration ⩾5% of total cells.
Results in 15 cases.
Results in 18 cases.
PD-1, programmed death-1; PD-L1, programmed death-1 ligand.
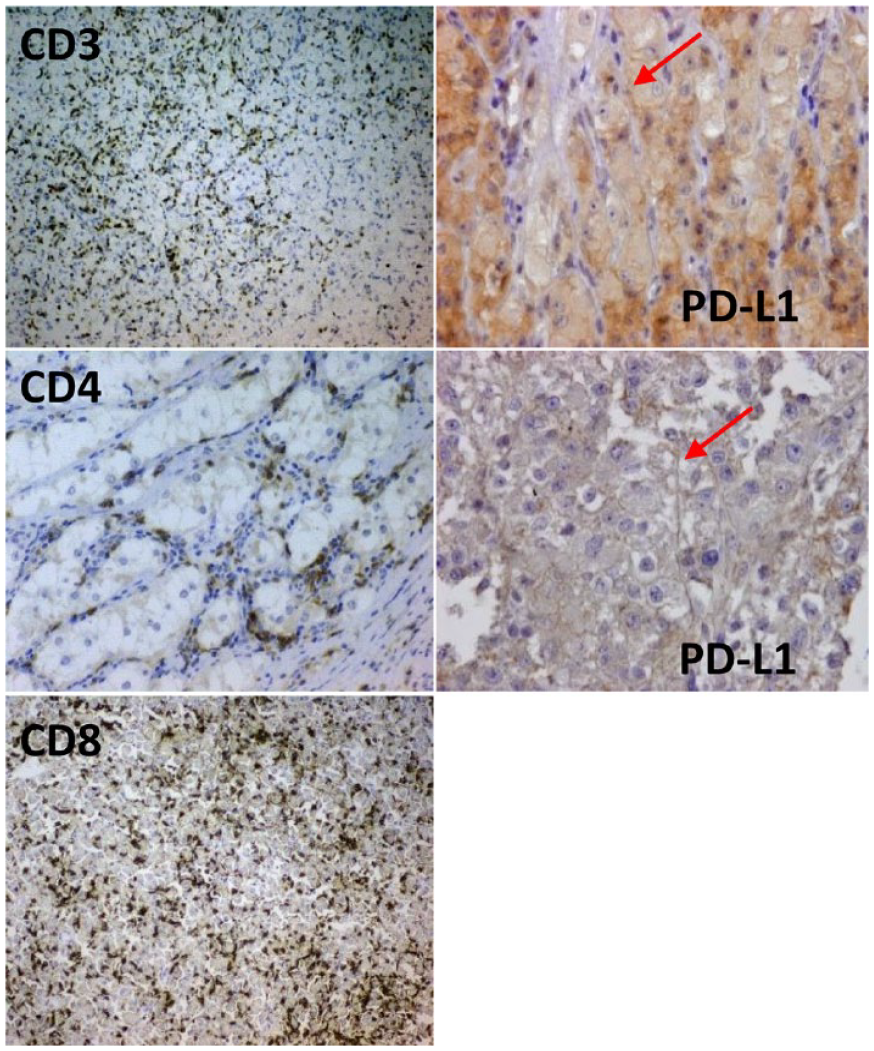
Left: lymphocytic infiltration. Right: two examples with membrane-positive staining for PD-L1.
Immune-related genes
A total of 17 available tumor samples were used for transcriptomic analysis using the IO array. Due to a lack of biological material, 2 of the 19 patients were not included in this analysis. No differentially expressed genes (DEGs) were identified for metastasis at diagnosis, relapse, or death. In addition, no DEGs were obtained for patients with positive versus negative CD3 or FAS protein expression by Immunohistochemistry. Two DEGs,
DEGs identified when comparing CD8-high versus CD8-low groups.

Genes marked in bold correspond to genes that were used for survival analysis.
Prognostic role
Protein expression of PD-L1, FAS, FASL, and CD8 showed no prognostic information in terms of DFS or OS (Table 4). Although not statistically significant, there were notable differences in median DFS between FAS-positive (55 months) and FAS-negative cases (10 months), as well as between CD8-positive (55 months) and CD8-negative cases (10 months). Expanding the analysis to DEGs according to the expression of the aforementioned proteins, no significant differences were observed for OS or DFS. However,
Outcome (disease-free survival and overall survival) according to IHQ.

No statistics due to lack of events.
CI, confidence interval; DFS, disease-free survival; IHQ, immunohistochemistry; OS, overall survival; PD-L1, programmed death-1 ligand.

L1CAM upregulation correlates with worse OS in ASPS patients. The high L1CAM expression group presents a median OS of 27.0 months (95% CI 22.8–31.2) compared to 151 months (95% CI NR) of patients with low L1CAM expression (
Discussion
Here we present the results of a series of 19 cases of ASPS, a very rare sarcoma subtype.
The analysis of the IO HTG panel provided us with a substantial quantity of data on the expression of immune-related genes in ASPS. We found one gene significantly related to a poorer prognosis: the overexpression of L1CAM, a DEG associated with lower infiltration of CD8. L1CAM, also known as L1 cell adhesion molecule, is a transmembrane protein that belongs to the L1 protein family and is encoded by the
In the context of sarcoma, L1CAM has been suggested as a potential surface target for Chimeric antigen receptor T-cells (CAR-T) therapy in rhabdomyosarcoma, given its detection in the majority of samples analyzed. Notably, patients with high
Overall, in sarcoma and various cancers,
Moreover, there is evidence supporting our findings that L1CAM may influence CD8 infiltration. In breast cancer, a four-gene signature, including L1CAM, has been identified to impact CD8 levels in high-risk patient groups. 43 In addition, L1CAM has been associated with tumor progression through direct recruitment of regulatory T cells, which subsequently inhibit the antitumor microenvironment, including CD8 cells. 44
We describe here the expression in this entity of apoptotic proteins FAS/FASL, and PD-1 and PD-L1 and we found out that the expression of PD-L1 in ASPS is relatively high, with 63% of cases showing positivity for this immune marker. This percentage is higher than the one reported in the literature for other series of sarcoma,20,45 suggesting that this histology may be more prone to immunomodulatory treatments. We also observed lymphocytic infiltration in all cases, with a clear predominance of CD8+ lymphocytes, which is in line with a report on a series of 10 ASPS. 46 All these data together suggest a role for immunomodulatory interventions in this entity, which is supported by recent studies.26,27 In our series, we did not find a statistically significant prognostic role for PD-L1, FAS, FASL, or CD8 expression. However, FAS-positive tumors showed notably longer DFS when compared with FAS-negative tumors. This observation is consistent with previously published results from a larger cohort including other STS subtypes. 47 High-density (>5% of total cells) lymphoid infiltration by cytotoxic CD8+ lymphocytes was also related to longer DFS and OS in our series, although not reaching statistical significance. The lack of significance in our study could be attributed to the small sample size, which may have reduced the statistical power.
The limited size of this series could explain in part these results, but ASPS is such a rare disease that a bigger series could only be obtained within an international collaboration. Further research with larger cohorts is warranted to validate these findings and explore additional therapeutic strategies.
In conclusion, our study emphasizes the importance of L1CAM expression in ASPS and its potential as a therapeutic target. Future preclinical studies could further investigate the potential protumoral role of L1CAM in ASPS cells. In addition, conducting in vivo studies and clinical trials could reveal the potential benefits of combining ICIs with L1CAM-targeting drugs or CAR-T cells. The findings related to the immune microenvironment suggest potential avenues for immunomodulatory interventions.
Conclusion
Our study highlights the potential of L1CAM expression in predicting survival in ASPS patients. Future preclinical studies could further investigate the potential protumoral role of L1CAM in ASPS cells. In addition, conducting in vivo studies and clinical trials could reveal the potential benefits of combining ICIs with L1CAM-targeting drugs or CAR-T cells. The findings related to the immune microenvironment suggest potential avenues for immunomodulatory interventions.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241293951 – Supplemental material for Exploratory analysis of immunomodulatory factors identifies L1CAM as a prognostic marker in alveolar soft-part sarcoma
Supplemental material, sj-docx-1-tam-10.1177_17588359241293951 for Exploratory analysis of immunomodulatory factors identifies L1CAM as a prognostic marker in alveolar soft-part sarcoma by José L. Mondaza-Hernandez, Nadia Hindi, Antonio Fernandez-Serra, Rafael Ramos, Ricardo Gonzalez-Cámpora, María Carmen Gómez-Mateo, Javier Martinez-Trufero, Javier Lavernia, Antonio Lopez-Pousa, Nuria Laínez, Jeronimo Martinez-Garcia, Claudia Valverde, María Ángeles Vaz-Salgado, Gabriel Garcia-Plaza, Isabel Marin-Borrero, Jaime Carrillo-Garcia, Marta Martin-Ruiz, Pablo Romero, Antonio Gutierrez, Jose A. López-Guerrero, David S. Moura and Javier Martin-Broto in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank the contribution of Andres Carranza-Carranza, Ramiro Alvarez-Alegret, and Patricio Ledesma for data management and shipping coordination.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
