Abstract
The use of programmed cell-death protein 1 (PD-1)/programmed cell-death ligand 1 (PD-L1) inhibitors is the standard therapy for the first-line or second-line treatment of patients with non-small-cell lung cancer (NSCLC). In contrast to current traditional treatments such as chemotherapy or radiotherapy, anti-PD-1 and anti-PD-L1 treatments can directly attenuate tumour-mediated exhaustion and effectively modulate the host anti-tumour immune response
Keywords
Introduction
Lung cancer is the leading cause of cancer-related deaths and is associated with a poor prognosis.1,2 Non-small-cell lung cancer (NSCLC) accounts for 85% of all lung cancers, 15–30% of which are squamous-cell carcinomas (SQCs) of the lung.3,4 The pathogenic genes of NSCLC have been discovered and widely investigated, and genotyping-based targeted therapy has been developed as one of the most successful therapeutic methods.5–8 At present, with a greater understanding of tumour immunity, targeted molecular therapy and immunotherapy have become important strategies for the conventional treatment of NSCLC.9–12 In recent years, the successful application of immune-checkpoint inhibitors (ICIs) of cytotoxic T-lymphocyte antigen-4 (CTLA-4), and anti-programmed cell-death protein 1 (PD-1) and programmed cell-death protein ligand 1 (PD-L1) in various advanced cancers has attracted widespread attention in the field of immuno-oncology.13–18 Two antibodies against PD-1, nivolumab and pembrolizumab, and two antibodies against PD-L1, atezolizumab and durvalumab, have been approved by the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA) for treating advanced NSCLC.19–24 The PD-1/PD-L1 inhibitors licensed for NSCLC treatment are listed in Table 1.
The PD-1/PD-L1 inhibitors licensed for NSCLC treatment.

EMA, European Medicines Agency; FDA, US Food and Drug Administration; IgG, immunoglobulin G; mAb, monoclonal antibody; NSCLC, non-small-cell lung cancer; PD-1, programmed cell-death protein 1; PD-L1, programmed cell-death ligand 1.
In this review, we aimed to outline the recent development of PD-1/PD-L1 inhibitors as first-, second-, and third-line treatments, and to discuss driver-gene mutation in NSCLC patients. Moreover, we summarised current and ongoing clinical trials of PD-1 and PD-L1 inhibitors in China. In the absence of optimal prognostic biomarkers, we elaborate on the presence of immune-related adverse events (irAEs), primary and acquired drug resistance, and the challenge of administering immunotherapy for the treatment of advanced NSCLC.
Regulatory mechanisms of PD-1/PD-L1 in NSCLC
Modulation of immune checkpoints is an important mechanism for evading immune surveillance by inhibiting activated T cells. 25 For example, PD-1+ immune cells can interact with PD-L1+ malignant tumour cells or other host cells. Mechanistically, immune escape might occur by interfering with related anti-tumour functions. 26 In addition, PD-1 is expressed by CD4+ T cells, CD8+ T cells and some other immune cell types. PD-1 interaction with its ligand, PD-L1, can weaken the effector functions of T lymphocytes such as activation, proliferation and survival, promote antigen-specific T-cell apoptosis, and affect CD4+ and CD8+ T cells, natural killer (NK) cells, and CD11c+ M1 macrophage functions to promote tumour development.27–29 Antibodies that suppress the interaction between PD-1 and PD-L1 can largely improve and restore the functionality of exhausted T cells and enhance their anti-tumour immune response. 30 PD-1 is highly expressed by regulatory T cells (Tregs) and has a significant inhibitory effect on tumour immunity. 31 The immune-related regulatory mechanism of action of PD-1/PD-L1 inhibitors is shown in Figure 1. Modulation of PD-1/PD-L interactions effectively inhibits the anti-tumour response by weakening the inhibitory activity of Tregs. 32 In addition to T-cell immunity, tumour-associated macrophages,33,34 as well as NK cells and dendritic cells in the tumour microenvironment (TME) can enhance anti-tumour effects.35–37

Regulatory mechanism of programmed cell-death protein 1 (PD-1)/programmed cell-death ligand 1 (PD-L1) inhibitors.
PD-1/PD-L1 inhibitor therapy in advanced NSCLC
Although there are many ways to treat NSCLC, such as radiotherapy, platinum-based chemotherapy, surgical resection and targeted molecular therapy, the long-term efficacy of these treatments is still not ideal, and the 5-year survival rate of patients with NSCLC is less than 18%. 38 Thus, there is an urgent demand for novel clinical therapies that are associated with less toxicity and higher efficacy to further prolong the survival time of patients. At present, some anti-PD-1/PD-L1 antibodies have been approved by the FDA for clinical treatment of advanced NSCLC; 39 these clinical studies will have a very important role in guiding long-term clinical practice.
PD-1/PD-L1 inhibitors as first-line treatment of advanced NSCLC
Multiple KEYNOTE trials (either alone or as part of a combined study) are being investigated using PD-1/PD-L1 inhibitors in various tumour types, with a focus on NSCLC. In the open-label phase Ib KEYNOTE-001 trial, patients with advanced NSCLC expressing PD-L1 in tumour tissues who were randomly assigned to the pembrolizumab group exhibited an objective response rate (ORR) of 27%, a median progression-free survival (mPFS) of 6.2 months [95% confidence interval (CI) 4.1–8.6], and a median overall survival (mOS) of 22.1 months (95% CI 17.1–27.2). The ORR, 1-year mPFS, and 1-year mOS were significantly higher in patients with a PD-L1 tumour proportion score (TPS) ⩾50% than in the overall population. The results showed that pembrolizumab was well tolerated; only 12 cases (11.9%) experienced grade 3 treatment-related adverse events (AEs), and there were no treatment-induced deaths.
40
The 3 years of data from KEYNOTE-001 showed that 79.7% patients with a PD-L1 TPS ⩾50% reached the mOS of 34.9 months (95% CI 20.3–not reached).
41
The KEYNOTE-189 study is a phase III clinical trial of non-squamous NSCLC patients without first-line sensitised epidermal growth-factor receptor (EGFR) or anaplastic lymphoma kinase (ALK) mutations. These patients received platinum-based drugs plus either pembrolizumab or placebo. The mPFS of the monoclonal antibody (mAb) group was significantly longer than the control group (8.8
For patients with advanced squamous NSCLC, the KEYNOTE-407 trial indicated that concomitant administration of carboplatin plus paclitaxel and pembrolizumab provided longer mOS and mPFS than traditional chemotherapy alone.
44
The efficacy outcome from the protocol-specified final analysis of KEYNOTE-407 showed that, after a median follow up of 14.3 months, pembrolizumab plus chemotherapy exhibited a clinically meaningful improvement over placebo plus chemotherapy for mOS (17.1
Another phase I/II trial, KEYNOTE-021, was designed to assess the efficacy and frequency of grade 3–4 AEs associated with combination treatment with platinum-based doublet chemotherapy with pembrolizumab. The results demonstrated that combination chemotherapy with pembrolizumab was associated with fewer grade 3–4 AEs compared with platinum-based chemotherapy or pembrolizumab monotherapy.
46
A randomised, open-label, phase II, multi-cohort study, KEYNOTE-021, was performed with 123 patients with primary stage IIIb, or IV NSCLC without EGFR or ALK mutations. The mPFS of the pembrolizumab plus chemotherapy group was 13.0 months (95% CI 8.3–not reached), whereas the mPFS of the group receiving chemotherapy alone was only 8.9 months (95% CI 4.4–10.3).
47
After a median time of 49.4 months from KEYNOTE-021, ORR (58%
In the KEYNOTE-024 trial, compared with platinum-based chemotherapy alone, pembrolizumab significantly improved OS (30.0
In the CheckMate 012 trial, 52 patients received nivolumab until disease progression or toxicity was experienced. The ORR was 28% in patients regardless of the tumour expression level of PD-L1, and the mPFS and mOS were 3.6 months (95% CI 0.1–28.0) and 19.4 months (95% CI 0.2–35.8), respectively. 53 In another CheckMate 012, open-label, phase I trial, 78 patients who received either nivolumab every 2 weeks plus ipilimumab every 12 weeks, or nivolumab every 2 weeks plus ipilimumab every 6 weeks, were assessed. The use of nivolumab plus ipilimumab as first-line treatment resulted in good tolerability and safety, and encouraging clinical efficacy, with a better ORR in each group. 54 Based on CheckMate 012, in the CheckMate 227 trial, nivolumab plus ipilimumab as first-line therapy significantly prolonged the mPFS compared with chemotherapy alone in patients with advanced NSCLC.55,56 CheckMate 568, a phase II trial of nivolumab plus ipilimumab in NSCLC patients, defined a cut-off of the tumour mutational burden (TMB) of at least 10 mutations per base pair for selecting patients most likely to respond to treatment, regardless of the PD-L1 expression level. 57 A TMB ⩾10 was associated with a high ORR and mPFS. However, in the CheckMate 026 study, compared with first-line chemotherapy alone in advanced NSCLC patients, patients that were treated with antibody therapy did not obtain significant survival benefits. 58
Atezolizumab and durvalumab are engineered and humanised mAbs against PD-L1 immunoglobulin G1 (IgG1), which can combine with PD-L1 to block the PD-1/PD-L1-mediated signalling pathway. BIRCH was designed to measure the efficacy of atezolizumab in advanced NSCLC as first-, second- and third-line therapies. Of 139 patients with advanced NSCLC who underwent first-line treatment with atezolizumab, the ORR, mPFS and mOS were 22%, 5.4 months (95% CI 3.0–6.9) and 20.1 months (95% CI 20.1–not reached), respectively.
59
IMpower150 was an open-label phase III study that aimed to assess the therapeutic efficacy of atezolizumab combined with bevacizumab and chemotherapy as first-line treatment in patients with advanced NSCLC. Patients received a combination therapy of atezolizumab + carboplatin + paclitaxel (ACP), bevacizumab + carboplatin + paclitaxel (BCP), or atezolizumab + BCP (ABCP) every 3 weeks for a total of four to six cycles. Following this, they then received atezolizumab, bevacizumab, or both, for maintenance treatment. Regardless of the expression levels and genetic status of PD-L1, the mPFS and mOS of patients with metastatic non-squamous NSCLC in the ABCP group were significantly improved when compared with the ACP or BCP group.
60
In the IMpower132 trial, atezolizumab combined with carboplatin and the chemotherapeutic agent pemetrexed significantly decreased mortality and improved the mPFS and mOS compared with chemotherapy alone in patients with advanced non-squamous NSCLC.
61
The IMpower130 trial reported an obvious improvement in mOS and mPFS, and no change in AEs were found in the group receiving atezolizumab plus chemotherapy compared with the group receiving chemotherapy alone as the first-line treatment of patients with metastasised non-squamous NSCLC without driver gene mutations.
62
The IMpower131 study showed that an atezolizumab plus platinum-based chemotherapy significantly improved mPFS in patients with first-line squamous NSCLC; however, the mOS was similar between groups.
63
Impower110 was performed to analyse the efficacy and safety of atezolizumab, as compared with platinum-based chemotherapy, as a first-line treatment for patients with metastatic NSCLC. The primary measurement was the OS among patients in the intention-to-treat population. This study found that the mOS was longer in the atezolizumab group than in the chemotherapy group (20.2
In a phase Ib trial, durvalumab and the anti-CTLA-4 antibody tremelimumab were administered to patients with advanced NSCLC. Regardless of the level of PD-L1 expression, the ORR was 23% in the group that received durvalumab combined with tremelimumab.
66
The MYSTIC trial was an open-label, phase III trial of first-line treatment with durvalumab with or without tremelimumab
Efficacy of anti-PD-1/PD-L1 agents for advanced NSCLC in first line.

ABCP, atezolizumab plus BCP; AE, adverse event; ACP, atezolizumab plus carboplatin plus paclitaxel; APP, atezolizumab plus pemetrexed plus carboplatin or cisplatin; BCP, bevacizumab plus carboplatin plus paclitaxel; CI, confidence interval; CnP, carboplatin + nab-paclitaxel; HR, hazard ratio; mOS, median overall survival; mPFS, median progression-free survival; NA, not applicable; NSCLC, non-small-cell lung cancer; NR, not reached; ORR, objective response rate; PD-1, programmed cell-death protein 1; PD-L1, programmed cell-death ligand 1; PP, pemetrexed plus carboplatin or cisplatin; TMB, tumour mutational burden; TPS, tumour proportion score.
PD-1/PD-L1 inhibitors as second-line treatment in advanced NSCLC
The human anti-PD-1 mAb nivolumab was the first to be approved as second-line therapy for stage IV NSCLC. 69 CheckMate 017, a randomised, open-label, phase III study assessed the efficacy and safety of nivolumab for advanced squamous NSCLC; greater efficacy was obtained during or after first-line chemotherapy. Regardless of PD-L1 expression levels, the mOS, ORR, and mPFS were markedly better in the nivolumab group than in the docetaxel group.69,70 The CheckMate 057 trial evaluated the efficacy of nivolumab in patients with advanced non-squamous NSCLC who experienced obvious disease progression after first-line treatment. Compared with docetaxel, nivolumab treatment resulted in a remarkably longer mOS, with a more favourable safety profile.71,72 In both the CheckMate 017 and CheckMate 057 trials, the 2-year OS rates of patients with squamous NSCLC treated with nivolumab compared with docetaxel were about 23% and 8%, respectively. In those with non-squamous NSCLC, the 2-year OS rates of those treated with nivolumab were 29% and 16% in the CheckMate 017 and CheckMate 057 trials, respectively. 73 The CheckMate 078 trial compared the safety and efficacy of nivolumab with docetaxel in patients with squamous or non-squamous NSCLC. 74 The results were consistent with those of the CheckMate 017 and CheckMate 057 trials. Another Argentinian study was conducted to explore the efficacy and safety of nivolumab in previously treated patients with advanced NSCLC; the results showed that nivolumab was well tolerated, with promising prognostic outcomes in patients with NSCLC who had undergone previous treatment. 75 A multicentre retrospective observational study in Japan was performed to evaluate the efficacy of nivolumab in second-line treatment in NSCLC patients. The mOS was 14.6 months (95% CI 12.3–15.9); 1-year survival rate was 54.3%; mPFS was 2.1 months (95% CI 1.9–2.4); and ORR was 20.5%. Subgroup analysis showed that PS, EGFR mutation status, smoking status, and PD-L1 were associated with the effectiveness of nivolumab. 76
KEYNOTE-010, an open-label, randomised phase II/III trial, included pretreated NSCLC patients with an expression level of PD-L1 ⩾1% in tumour cells who had received treatment with pembrolizumab or docetaxel. This study showed that treatment with pembrolizumab significantly prolonged mOS in patients with previously treated, advanced NSCLC, especially in those with high PD-L1 expression levels.
77
Patients in the KEYNOTE-010 trial were followed up to assess long-term outcomes. At a median follow-up time point of 42.6 months (range 35.2–53.2 months), those treated with pembrolizumab experienced a significantly improved mOS compared with those treated with docetaxel. The 3-year OS rates were 34.5%
A randomised phase III clinical trial, OAK, also showed that regardless of the expression level of PD-L1, the survival rate following atezolizumab treatment was significantly improved compared with docetaxel. Based on the OAK findings, atezolizumab has been approved by the FDA and the EMA for further treatment of those previously treated for advanced NSCLC. For long-term survivors in the atezolizumab group, the ORR was 14% and the mOS was 13.8 months (95% CI 11.8–15.7).81,82 In the POPLAR study, an open-label, phase II trial, atezolizumab treatment significantly improved OS compared with docetaxel in patients with NSCLC who were previously treated.
83
The BIRCH study also proved the efficacy of atezolizumab in patients with advanced NSCLC as second-line therapy. The ORR, mPFS, and mOS were 19%, 2.8 months (95% CI 1.5–3.9), and 15.5 months (95% CI 12.3–not reached), respectively.
59
It is well known that approximately 10% of patients with NSCLC are complicated with comorbid interstitial pneumonia, with a poor prognosis. The Thoracic Oncology Research Group 1936/AMBITIOUS study is an ongoing, multicentre, single-arm, phase II trial to assess the safety and efficacy of atezolizumab for pretreated advanced/recurrent patients with NSCLC complicated with idiopathic chronic fibrotic interstitial pneumonia. We are looking forward to the results of this clinical study.
84
Another multicentre, phase II, open-label, single-arm VinMetAtezo trial was designed to assess the safety and efficacy of metronomic oral vinorelbine in combination with atezolizumab in advanced NSCLC progressing after first-line platinum-based chemotherapy. The primary outcome was PFS at 4 months.
85
In France, Marine
Efficacy of anti-PD-1/PD-L1 agents for advanced NSCLC in second line and third line.
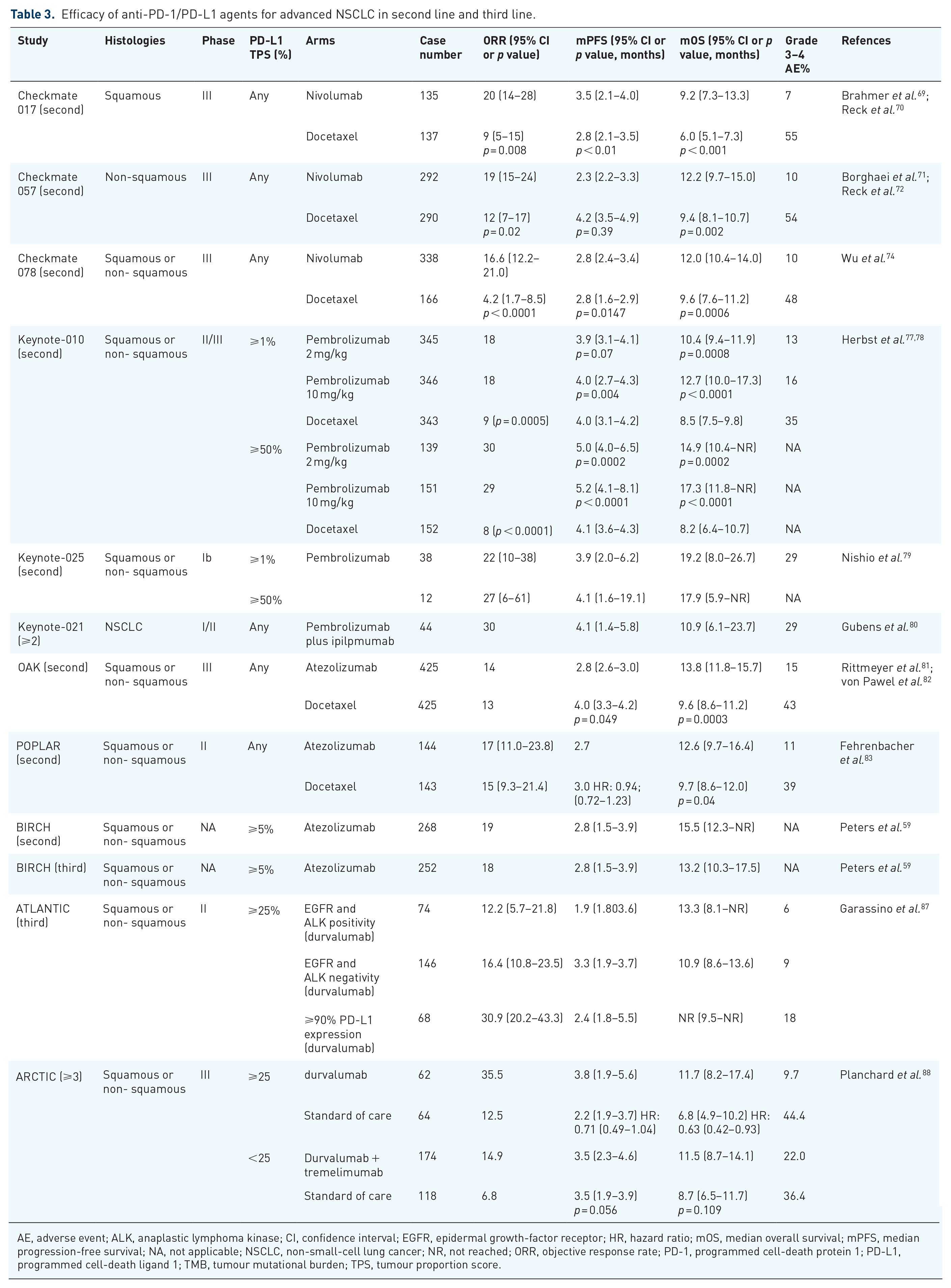
AE, adverse event; ALK, anaplastic lymphoma kinase; CI, confidence interval; EGFR, epidermal growth-factor receptor; HR, hazard ratio; mOS, median overall survival; mPFS, median progression-free survival; NA, not applicable; NSCLC, non-small-cell lung cancer; NR, not reached; ORR, objective response rate; PD-1, programmed cell-death protein 1; PD-L1, programmed cell-death ligand 1; TMB, tumour mutational burden; TPS, tumour proportion score.
PD-1/PD-L1 inhibitors as third-line or greater treatment in advanced NSCLC
The BIRCH study examined the clinical efficacy of atezolizumab as third-line or greater therapy in advanced NSCLC. The mOS, mPFS, and ORR were 13.2 months (95% CI 10.3–17.5), 2.8 months (95% CI 1.5–3.9), and 18%, respectively as third-line treatment, suggesting that atezolizumab has good clinical efficacy as third-line therapy for advanced NSCLC. 59
The ATLANTIC trial was an open-label, phase II, multicentre clinical trial conducted to assess the efficacy of durvalumab as third-line or greater therapy for advanced NSCLC. The ORR was 12.2% in EGFR+ and ALK+ patients who had expression levels of PD-L1 ⩾25% and received durvalumab, which was lower than that of patients who were EGFR− and ALK− with the same expression level of PD-L1 ⩾25% (16.4%) or those with expression of PD-L1 ⩾90% (30.9%). The mPFS rates in the three groups were 1.9 months, 3.3 months, and 2.4 months, respectively. 87
ARCTIC was a phase III, randomised, open-label study that evaluated the efficacy of durvalumab with or without tremelimumab
PD-1/PD-L1 inhibitors in cases of advanced NSCLC with driver-gene mutations
Inhibition of the PD-1/PD-L1 axis has resulted in favourable responses in NSCLC patients with EGFR mutations; however, the efficacy was significantly lower in these patients compared with those without EGFR mutations.89,90 Although high expression levels of PD-L1 in patients with advanced NSCLC may be associated with EGFR mutations, 91 studies have shown that EGFR-tyrosine kinase inhibitors (TKIs) downregulate the messenger ribonucleic acid (mRNA) expression of PD-L1, suggesting that EGFR-TKIs may activate T-cell immunocompetence in the TME of NSCLC patients with EGFR-mutations.92–94 A phase II clinical trial showed that pembrolizumab was less effective in advanced NSCLC patients who were naïve to TKIs and were PD-L1+, with EGFR mutations, even those with a PD-L1 expression level ⩾50%; this indicated that anti-PD-1/PD-L1 treatment was not the ideal first choice for patients with EGFR mutations. 95
The phase I/II KEYNOTE-021 trial analysed the feasibility of administering erlotinib or gefitinib combined with pembrolizumab as first-line treatment in patients with stage IIIb/IV EGFR-mutant NSCLC. Pembrolizumab plus gefitinib combination therapy had to be continued because of severe AEs, and another study showed that pembrolizumab plus erlotinib administration did not improve the ORR when compared with pembrolizumab plus gefitinib. 96 The CheckMate 370 trial evaluated the safety of first-line treatment with nivolumab combined with crizotinib in patients with ALK+ NSCLC. Again, the combination treatment had to be discontinued, and enrolment was closed because of observed grade ⩾3 hepatic toxicities. 97
A subgroup analysis of advanced NSCLC patients with EGFR mutations and ALK rearrangements was performed to assess the use of anti-PD-1/PD-L1 therapy as second-line treatment in the CheckMate 057, KEYNOTE-010, POLAR and OAK studies. Their results revealed that EGFR mutations and ALK rearrangements in patients with advanced NSCLC were associated with beneficial outcomes and prolonged mOS from anti-PD-1/PD-L1 treatment compared with chemotherapy when administered as second-line treatment.71,72,77,78,81–83 In a study assessing the effects of nivolumab, the ORR was significantly lower in patients with EGFR-mutant non-squamous NSCLC than in patients without the mutation (8.8%
One of the studies intended to identify a biomarker for patients most likely to positively respond to immunotherapy. Tumour deoxyribonucleic acid (DNA) was derived from advanced NSCLC patients receiving nivolumab or pembrolizumab therapy as second- or third-line treatment. The most common mutated genes observed in these patients were tumour protein p53 (TP53; 49%), Kirsten rat sarcoma 2 viral oncogene homolog (KRAS; 43%), and v-erb-b2 erythroblastic leukaemia viral oncogene homolog 2 (ERBB2; 13%). This study confirmed that patients with KRAS mutations experienced a longer mPFS and mOS than patients without KRAS mutations. Furthermore, patients with ERBB-family mutations failed to respond to treatment with PD-1 antibodies. 104 The efficacy data of anti-PD-1/PD-L1 for advanced NSCLC with driver gene mutations is shown in Table 4.
Efficacy of anti-PD-1/PD-L1 agents for advanced NSCLC with driver gene mutations.

ABCP, atezolizumab plus bevacizumab plus carboplatin plus paclitaxel; AE, adverse event; ALK, anaplastic lymphoma kinase; CI, confidence interval; EGFR, epidermal growth-factor receptor; mOS, median overall survival; mPFS, median progression-free survival; NA, not applicable; NSCLC, non-small-cell lung cancer; NR, not reached; ORR, objective response rate; PD-1, programmed cell-death protein 1; PD-L1, programmed cell-death ligand 1; TPS, tumour proportion score; WT, wild type.
Application of PD-1/PD-L1 inhibitors in advanced NSCLC cases in China
Toripalimab (also known as JS001 or TAB001) is a recombinant human anti-PD-1 mAb developed by Shanghai Junshi Biotechnology Co., Ltd. that selectively inhibits PD-1 and PD-L1/PD-L2 interactions to facilitate T-cell activation.
105
Yang
Sintilimab is a human IgG4 mAb coupled to PD-1; it was jointly developed by Innovent Biologics and Eli Lilly and Company. Sintilimab was approved for treating patients with classical Hodgkin’s lymphoma who had relapsed or were refractory after at least two rounds of systemic chemotherapy in China. 107 A phase Ib study was performed to assess the safety and efficacy of sintilimab as a neoadjuvant therapy for NSCLC; the results showed that sintilimab was well tolerated in NSCLC patients, with an encouraging pathologic response rate. 108
Camrelizumab, a PD-1 inhibitor, developed by Jiangsu Hengrui Pharmaceutical Co., Ltd., has recently been approved for the treatment of relapsed or refractory classical Hodgkin’s lymphoma in China. 109 A study evaluated the efficacy and safety of camrelizumab combined with microwave ablation (MWA) in patients with metastatic NSCLC. The results indicated that the ORR was 33.3%, with two patients achieving a complete response (CR) and five patients achieving a partial response (PR). The mPFS was 5.1 months; OS was not reached. The most common AE related to camrelizumab administration was reactive capillary hyperplasia of the skin 110 that could be alleviated by substitution with apatinib. 111
The anti-PD-1 antibody, tislelizumab, has demonstrated clinical tolerability and safety in treating solid tumours, including NSCLC in Chinese patients. 112 Toripalimab, sintilimab, camrelizumab and tislelizumab are all currently undergoing clinical trials for the treatment of various solid tumours, including advanced NSCLC; these are summarised in Table 5.
Application of PD-1/PD-L1 inhibitors in the advanced NSCLC in China.
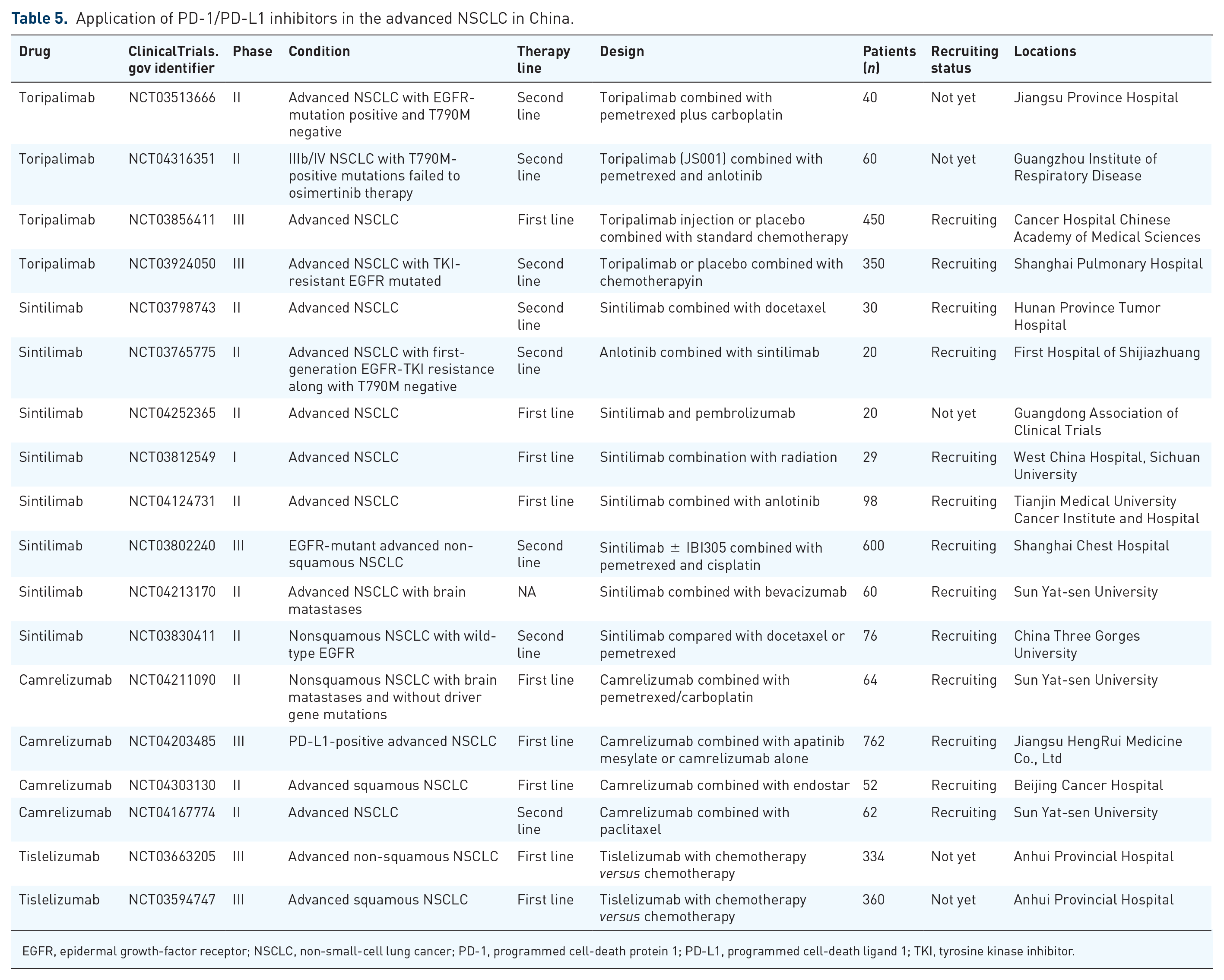
EGFR, epidermal growth-factor receptor; NSCLC, non-small-cell lung cancer; PD-1, programmed cell-death protein 1; PD-L1, programmed cell-death ligand 1; TKI, tyrosine kinase inhibitor.
Predictive biomarkers in anti-PD-1/PD-L1 immunotherapy
In order to predict the response to immunotherapy, the biomarkers initially used by the researchers were the expression of PD-1 receptor and PD-L1, and the prediction method was assessed only by immunohistochemistry. 113 More recently, with the development with newer technology, there are many available biomarkers that could be used to further predict the efficacy and prognosis of immunotherapy.113–117 These include PD-L1 expression, the number of tumour-infiltrating lymphocytes (TILs), the TMB, mutations in certain driver genes, microsatellite instability (MSI)/defective mismatch repair (dMMR), peripheral blood-based biomarkers and interferon gamma (IFN-γ) levels (Figure 2).

Predictive biomarkers and challenges facing anti-programmed cell-death protein 1 (PD-1)/programmed cell-death ligand 1 (PD-L1) treatment in advanced non-small-cell lung cancer (NSCLC).
Level of PD-L1 expression and the number of TILs
Currently, the level of PD-L1 is commonly used as a tumour biomarker to evaluate the efficacy of anti-PD-1/PD-L1 therapy in patients with NSCLC.34,118 The KEYNOTE-024 and KEYNOTE-042 trials49–51 suggested that pembrolizumab could significantly improve the survival rate in patients with high expression levels of PD-L1. The KEYNOTE-001 study also indicated that patients with a PD-L1 TPS ⩾50% would likely experience better outcomes, including improved ORR, mPFS, and mOS.
40
Therefore, pembrolizumab has been approved by the FDA for advanced NSCLC patients without driver gene mutations as a first-line treatment. In a single-arm study assessing early ICIs in selected patients with high expression levels of PD-L1, Ito
The TME plays a vital role in the growth and progression of cancer cells and distant metastasis, and affects the efficacy of immunotherapy in clinical oncology.
123
TILs mainly include NK cells and cytotoxic T cells, both of which are routinely assessed in histopathology reports as predictive biomarkers.124,125 In hyperprogressive disease patients, a high number of CD2− CD4 T lymphocytes during the first and second line of immunotherapy was associated with a lower mOS.
126
A retrospective study by Chen
TMB
The TMB is representative of the number of tumour mutations; the higher the frequency of mutations in somatic exonic regions, the greater the number of corresponding tumour-related antigens that are expressed.128,129 For patients with advanced NSCLC, a high TMB detected by next-generation sequencing seems to predict the lasting clinical effects of PD-1 or PD-L1 blockades.
130
This results in the subsequent increase in activation of lymphocytes to maintain an immunological anti-tumour response. A previous study evaluated the predictive role of the TMB in 76 NSCLC patients treated with anti-PD-1/PD-L1 therapy. Patients with a high TMB exhibited significantly longer mPFS and mOS.
131
In a Chinese study, the TMB was analysed in NSCLC patients receiving anti-PD-1/PD-L1 treatment; the mPFS was significantly longer at 10.6 months in the high TMB group compared with just 3.9 months in the low TMB group (
Driver-gene alterations
Next-generation sequencing technology has been able to identify thousands of mutations in large cohorts for various cancers.
138
Ono
MSI/dMMR
In addition to PD-1/PD-L1 expression analysis, the presence of MSI is also a key reaction marker. 144 Studies have shown that genetic mutations in the dMMR pathway can lead to MSI, and high MSI is closely related to the increased immunogenicity and immune response produced by tumours. MSI is an excellent predictive biomarker. Therefore, the FDA has approved pembrolizumab to treat unresectable solid tumours with high MSI or dMMR. 145 The DNA MMR pathway includes the mutL homolog 1 (MLH1), MSH (mutS homolog)-2, postmeiotic segregation increased 2 (PMS2) and MSH-6 proteins. When this pathway is defective (dMMR), it can be used as a means to select patients for immunotherapy. In addition, MSI frequency can also be used to predict NSCLC. 117 Similarly, NSCLC patients with DNA polymerase epsilon (POLE) and DNA polymerase delta 1 (POLD1) gene mutations also have an objective clinical response to pembrolizumab treatment and a corresponding degree of association.146,147 Therefore, clinicians can further determine whether a patient can use the epidemiology of the patient’s MSI/dMMR to predict NSCLC prognosis. 148
Peripheral blood-based biomarkers
Because biopsies of lung cancer tissue samples are difficult to obtain, some alternative methods include minimally invasive biopsy of peripheral blood for practical reasons.
149
Peripheral blood contains DNA, RNA, and proteins released from tumour tissues reflecting the dynamic changes in the TME. The level of ctDNA in the peripheral blood in the early stage of treatment provides valuable clues to the therapeutic effect. In various cancer types, including NSCLC, the detection of highly mutated ctDNA is positively correlated with a better clinical response to immune-checkpoint blockade.
150
Zhang
IFN-γ
IFN-γ is a vital immune-regulatory molecule in the TME released by tumour-reactive T cells.
154
Recent studies have suggested that IFN-γ is an important driver of PD-L1 expression in tumours, and it can significantly improve the response to anti-PD-1 therapy.
155
The increased IFN-γ levels at baseline and at 3 months after anti-PD-1 therapy were significantly correlated with improved mOS.
156
Recent studies have shown that in 17 NSCLC patients treated with nivolumab, mPFS was obviously prolonged in those with high expression levels of IFN-γ, in contrast to those with low expression of IFN-γ (5.1
Other potential biomarkers
Other potential biomarkers being investigated in anti-PD-1/PD-L1 immunotherapy on NSCLC include the pretreatment haemoglobin level, 158 serum vascular endothelial growth factor (VEGF) concentration, 159 serum lactate dehydrogenase level, 160 gut microbiome composition 116 and neo-antigen load. 161
Challenges for anti-PD-1/PD-L1 treatment in advanced NSCLC
Currently, there are still many unresolved problems associated with the clinical use of anti-PD-1/PD-L1 therapies (Figure 2). First, PD-L1 expression is a predictive biomarker of outcomes of anti-PD-1/PD-L1 treatment, as it is correlated with the efficacy of PD-1/PD-L1 inhibitor therapy in NSCLC; however, this association can be quite variable. For example, the improvement in mOS by nivolumab and durvalumab treatment for NSCLC was independent of PD-L1 expression levels.71,162 In immunohistochemical analysis, the expression levels of PD-L1 are dynamic and heterogeneous, and this is further complicated by issues related to antibody specificity and detection techniques. 163 In the future, more valuable biomarkers may be identified by neoantigen prediction techniques, microbiota signatures, or high microsatellite instability (MSI), all of which should be further explored in clinical medicine.
The second challenge is the risk of irAEs associated with the use of anti-PD-1/PD-L1 monotherapy for advanced NSCLC. Although the safety profile of individual antibodies is favourable compared with standard chemotherapy; the frequency of irAEs in combination therapies was much higher (Tables 2–4). For example, 53.6% of patients who received atezolizumab plus chemotherapy developed irAEs, whereas only 29.1% of patients in the group receiving chemotherapy alone did. 61 Therefore, better strategies to reduce the risks of serious irAEs need to be explored in future studies.
The third challenge of immunotherapy is deciding how to effectively and accurately select the best combination therapy for patients with advanced NSCLC. Anti-PD-1/PD-L1 treatment combined with chemotherapy is proven to have great potential for improving ORRs.164,165 Apart from chemotherapy, treatments with VEGF, CTLA-4, chimeric antigen receptor T cell, CRISPR-Cas9 gene editing or lymphocyte-activation-protein-3 therapies could provide additional choices for combination strategies in future clinical studies.17,166–169 Therefore, it is important to establish a standard to guide and determine the most appropriate combination therapy for treating advanced NSCLC.
The fourth challenge is that some patients who are resistant to anti-PD-1/PD-L1 therapy experience a poor prognosis after treatment. The underlying resistance mechanism is a very complicated process, and little is known about the potential tumour-related antigens associated with TMB resistance to anti-PD-1/PD-L1 therapy or the genetic determinants of a good prognosis.
Conclusion
To date, four anti-PD-1/PD-L1 drugs, including pembrolizumab, nivolumab, atezolizumab and durvalumab, have been registered and approved for the neoadjuvant, first-line and second-line treatment of NSCLC. With the approval of ICIs, immune-checkpoint therapy based on modulating PD-1/PD-L1 signalling provides more choices for treating advanced NSCLC patients. However, the ORR after ICI treatment is only 14–20% in NSCLC patients who are not identified beforehand as being likely to experience a good prognosis. Therefore, identification of predictive biomarkers is crucial for selecting patients with NSCLC who would most benefit from anti-PD-1/PD-L1 therapy. Although the clinical application of anti-PD-1/PD-L1 therapy is still at an early stage, there are an increasing number of ongoing clinical trials to assess the efficacy of anti-PD-1/PD-L1 treatment. EGFR mutation rates and microbiome differences may affect the efficacy of anti-PD-1/PD-L1 treatment. ALK rearrangements in patients with advanced NSCLC are associated with beneficial outcomes and prolonged mOS from anti-PD-1/PD-L1 treatment compared with chemotherapy, when administered as second-line treatment. Patients with KRAS mutations experienced a longer mPFS and mOS than patients without a KRAS mutation using anti-PD-1/PD-L1 treatment, thus immunotherapy may be a promising strategy with KRAS mutation of NSCLC owing to regulating immune response. Therefore, with the continuous development of clinical trials, more retrospective and prospective clinical studies need to comprehensively evaluate more underlying biomarkers to improve treatment efficacy and enable more NSCLC patients to benefit from ICIs for anti-PD-1/PD-L1 therapy.
Footnotes
Acknowledgements
The authors thank Prof. Nong Yang from the Lung Cancer and Gastroenterology Department, Hunan Cancer Hospital, Affiliated Tumour Hospital of Xiangya Medical School of Central South University for technical help this manuscript. In addition, we would like to thank SAGE Author Services (![]() ) for English language editing.
) for English language editing.
Authors’ contributions
JZ and JQ wrote the manuscript; QM, LC, LL, TC and PW collected the references and modified the manuscript; LC and JZ designed the manuscript and approved the final manuscript for publication.
Availability of data and materials
Please contact the corresponding author for data requests.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the National Natural Science Foundation of China (81802278, 81900563, 81670017, 81960232, and 82001282) and the Natural Science Foundation of Hunan Province (2019JJ50361 and 2020JJ4418).
