Abstract
Background:
The efficacy of immune checkpoint inhibitors (ICIs) in cancer patients taking anti-hypertensive drugs is still not well established.
Objective:
To elucidate the effect of anti-hypertensive drugs on the clinical outcome of cancer patients receiving immunotherapy.
Design:
A retrospective cohort study and meta-analysis.
Method:
We conducted a real-world retrospective study of cancer patients treated with immunotherapy at two tertiary centers between January 2019 and June 2023, with primary outcomes being overall survival (OS) and progression-free survival (PFS). In addition, we performed a meta-analysis to synthesize currently relevant clinical studies.
Results:
A retrospective clinical study of 336 patients from 2 centers suggested that the use of anti-hypertensive drugs was related to a preferable OS (hazard ratio (HR) = 0.55, 95% confidence interval (CI): 0.33–0.90) compared to non-users. For PFS, no significant correlation was detected (HR = 0.71, 95% CI: 0.49–1.03). Further analysis revealed that renin–angiotensin system inhibitor (RASi) and calcium channel blocker (CCB) have a synergistic effect with ICIs. In addition, subgroup analysis found that the benefits of RASi or CCB in combination with ICIs are greater in women or patients ⩾65 years of age. There was better disease control in lung cancer patients using RASi, and a significantly longer OS was observed in patients with gastrointestinal tumors taking CCB. Meta-analysis suggested that anti-hypertensive drugs were associated with improved OS, but only the combination of RASi and immunotherapy showed a synergistic effect. No significant correlation with OS was found for other anti-hypertensive drugs, and there was no overall positive effect on PFS.
Conclusion:
Our study found that use of anti-hypertensive drugs, particularly RASi or CCB, was associated with improved OS in patients undergoing immunotherapy. The synergistic effects of RASi or CCB with ICIs were more pronounced in females or elderly. RASi or CCB exhibited different benefits in various types of tumors. These findings provide valuable insights for treating cancer patients with hypertension.
Keywords
Introduction
Significant advancements have been achieved in the field of cancer therapeutics through the application of immunotherapy. In contrast to conventional therapies, immune checkpoint inhibitors (ICIs) function by activating the host’s immune system to effectively eliminate tumor cells. 1 ICIs have been utilized in the treatment of various solid cancers, including but not limited to non-small-cell lung cancer (NSCLC), 2 melanoma, 3 breast cancer, 4 and renal cell carcinoma. 5 However, the efficacy of ICIs is restricted to a limited proportion of patients, with only approximately 20%–40% of cancer patients responding to ICIs. 6 As a result, it is essential to investigate the factors influencing the effectiveness of ICIs and to devise strategies to enhance the response to immunotherapy.
Currently, concomitant medication is recognized as a factor that can impact the effectiveness of immunotherapy. Studies have shown that certain medications, such as proton pump inhibitors (PPIs) and antibiotics, can have a detrimental effect on the outcomes of immunotherapy. 7 Anti-hypertensive drugs are a common concomitant medication among cancer patients receiving ICIs. 8 Therefore, there is a growing interest in the impact of anti-hypertensive drugs on immunotherapy. Preliminary preclinical studies have suggested that anti-hypertensive drugs, such as renin–angiotensin system inhibitor (RASi) 9 and beta-blocker, 10 may have beneficial effects on immunotherapy. However, previous clinical studies have investigated the association between anti-hypertensive medications and immunotherapy, which remains contentious. Some retrospective clinical studies have suggested that the use of RASi 11 or beta-blocker 12 may improve clinical outcomes in cancer patients receiving ICIs, while other studies have not found similar findings. 13 The influence of calcium channel blockers (CCBs) and diuretics on patient prognosis lacks robust support in the literature, with a lack of evidence-based medical evidence from clinical studies. To further elucidate the relationship between anti-hypertensive drugs and immunotherapy, we conducted a real-world retrospective study across two tertiary hospitals and performed a meta-analysis to synthesize the existing clinical evidence.
Materials and methods
Clinical cohort
Patients
We conducted a retrospective clinical study in two centers: The 960th Hospital of the People’s Liberation Army (PLA) and the Affiliated Cancer Hospital of Shandong First Medical University. Cancer patients included in the study were those who received ICIs either as monotherapy or in combination with chemotherapy at the two medical centers between January 2019 and June 2023. Exclusion criteria were defined as follows: (1) receiving ⩽2 cycles of treatment; (2) primary multiple tumors; (3) receiving radiotherapy or surgery; (4) participation in clinical trials; and (5) incomplete baseline or follow-up information. Basic patient characteristics including gender, age, tumor type, Eastern Cooperative Oncology Group Performance Status (ECOG-PS) score, smoking history, use of anti-hypertensive drugs, underlying disease, concomitant medications, and ICIs were recorded. The study was approved by the hospital’s ethical committees (Approval No. 2023-061). A waiver of informed consent was granted due to the retrospective nature of the study.
Outcomes
Patients underwent tumor marker detection at each treatment cycle and an imaging examination every 2–3 cycles to evaluate the response of the cancer therapies. The primary outcomes were overall survival (OS) and progression-free survival (PFS). OS was defined as the time from the start of ICI treatment to death from any cause, while PFS was defined as the time from the initiation of ICI treatment to the first occurrence of disease progression or death. Subsequently, patients were followed up either during outpatient visits or by telephone.
Statistical analysis
To describe patient characteristics, categorical variables were expressed as the number of cases and percentage and analyzed using the Chi-squared test or Fisher’s exact test (where appropriate); continuous variables were expressed as means and compared using t-tests. To ensure similarity in baseline characteristics between groups, the propensity score matching (PSM) method was used to match patients taking anti-hypertensive drugs and non-users. Matching with a ratio of 1:1 was conducted (caliper value is 0.02). Propensity scores were calculated based on age, smoking history, coronary heart disease (CHD), and diabetes mellitus (DM). We compared OS and PFS between anti-hypertensive drug users and non-users using Kaplan–Meier analysis and Cox proportional hazard model analysis. The analysis was performed using Free Statistics software, version 1.8, and SPSS 26.0 software (IBM Corporation, Armonk, NY, USA). Statistical significance was defined at p < 0.05.
Meta-analysis
Literature search
The analysis is based on Preferred Reporting Items for Systematic Reviews and Meta-Analyses (Supplemental Table S1). We systematically searched the following online databases for relevant literature: Web of Science, PubMed, Cochrane Library, and Embase from inception until July 2024. We chose “anti-hypertensive drug,” “immune checkpoint inhibitor,” “cancer,” and “tumor” as MeSH terms and keywords (Supplemental Table S2). To ensure the inclusion of all eligible studies, we also carefully reviewed the references and proceedings.
Literature selection and data collection
Meta-analysis incorporated studies that met the specified criteria: (1) adult individuals diagnosed with cancer who have received at least one ICI treatment; (2) using any anti-hypertensive drugs for non-oncological reasons at the initiation of ICI treatment; and (3) randomized controlled trials or observational studies. The following were the exclusion criteria: (1) patients receiving radiotherapy, surgery, or other invasive treatment; (2) reviews, case reports, or meta-analysis; (3) non-clinical studies; (4) no relevant clinical outcome data were reported; and (5) duplicate publication of the same study. The primary outcome was the hazard ratio (HR) for OS or PFS with 95% confidence intervals (CI), gained either directly from the article or estimated from the Kaplan–Meier survival curve. Two researchers independently extracted basic information from the included studies: author, country, publication year, age, tumor, sample size, ICIs, type of anti-hypertensive medications, and outcomes, and compiled this information into a comprehensive table. Two researchers conducted an in-depth examination of the studies and obtained relevant information, and any disagreements were resolved by discussion.
Statistical and bias analysis
The Newcastle–Ottawa Scale (NOS) was employed to assess the quality of included studies. Meta-analysis was conducted utilizing R 4.3.2 software (R Foundation for Statistical Computing, Vienna, Austria), with statistical significance set at p < 0.05. Cochran Q and I2 tests were used to detect the heterogeneity between different studies. When heterogeneity was moderate or high (>50%), the random effect model was applied. In other cases, the fixed-effects model was utilized. The stability of outcomes was evaluated through the sensitivity analysis, and the bias of publication was tested using Egger’s and Begg’s test and funnel plots.
Results
Clinical cohort
Patient characteristics
We collected a cohort of 336 cancer patients receiving ICIs from the 960th Hospital of PLA and the Affiliated Cancer Hospital of Shandong First Medical University. The study population was divided into two groups based on their utilization of anti-hypertensive medications. The cohort included 151 patients on anti-hypertensive therapy and 185 patients without such treatment. Among the patients taking anti-hypertensive drugs, 101 were administered RASi, 75 were on CCB, 19 were given beta-blockers, and 16 were taking diuretics. The baseline clinical characteristics are detailed in Table 1. The mean age of the total cohort was 63.3 years. Notably, the subgroup receiving anti-hypertensive drugs had a higher average age (65.0 years) compared to the non-users (61.9 years), which was a statistically significant difference. The majority of the cohort was male (74.1%), with the most prevalent tumor types being gastrointestinal (GI) tumors (56.5%) and lung cancer (36.9%). Patients concurrently on anti-hypertensive drugs exhibited a higher prevalence of comorbidities, including CHD and DM. A total of 87.8% of patients were treated with a combination of immunotherapy and chemotherapy, with platinum-based chemotherapy being the most common. First-line immunotherapy was administered to 73.5% of the patients, and the most frequently used immunotherapy drugs were sintilimab and camrelizumab. Other than age, smoking history, comorbidities, and anti-hypertensive medications, no significant differences were observed in other variables between the two groups.
Clinical characteristics of the patients in the cohort.

ECOG-PS, Eastern Cooperative Oncology Group Performance Status; ICIs, immune checkpoint inhibitors; PD-1, programmed cell death protein 1; PD-L1, programmed cell death-ligand 1.
After PSM, a total of 119 cases and their corresponding 119 controls were included in the survival analysis, totaling 238 participants. Baseline demographic and clinical characteristics before and after matching are presented in Table 2. After matching, there was no significant difference between groups for each matching variable (p > 0.05). Furthermore, 93 pairs of patients who received RASi were successfully matched with non-users of RASi (Supplemental Table S4). Similarly, 71 pairs of patients treated with CCB were successfully matched with those not undergoing CCB treatment (Supplemental Table S5).
Baseline characteristics of patients before and after propensity score-matched analysis.

CHD, coronary heart disease; DM, diabetes mellitus.
Effect of anti-hypertensive drugs on immunotherapy
The follow-up observation of the survival of patients in the clinical retrospective cohort revealed that those taking anti-hypertensive drugs had a longer median OS compared to non-users (18.9 vs 35.4 months, p = 0.009, Figure 1(a)). The survival curve showed that patients taking anti-hypertensive drugs also had a tendency toward extending the median PFS (11.8 vs 20.5 months, p = 0.128, Figure 1(b)), although not statistically significant. Analysis of different types of anti-hypertensive drugs showed that RASi patients had a significant benefit on OS (15.8 vs not reached, p < 0.001, Figure 1(c)) and PFS (11.2 vs 20.5 months, p = 0.006, Figure 1(d)). An extended median OS was also observed in those taking CCB (15.8 vs 35.4 months, p < 0.001, Figure 1(e)). But no similar benefit was observed in the median PFS (11.8 vs 21.3 months, p = 0.055, Figure 1(f)). To further determine the factors influencing the efficacy of immunotherapy, we conducted univariate and multivariate analyses. In the univariate analysis, anti-hypertensive drugs decreased mortality risk by 45%, suggesting a significant association between anti-hypertensive drug use and OS (HR = 0.55, 95% CI: 0.35–0.89). The use of RASi and CCB was also associated with an extended OS (RASi: HR = 0.58, 95% CI: 0.34–0.99; CCB: HR: 0.43, 95% CI: 0.22–0.84). After adjusting for gender, age, tumor type, ECOG-PS, metastatic site, treatment line, and chemotherapy, a multivariate analysis revealed that the use of anti-hypertensive drugs conferred a stable benefit on OS of patients receiving immunotherapy (Table 3). For controlling disease progression, it was found that anti-hypertensive drugs reduced disease progression by 30% (HR = 0.70, 95% CI: 0.49–0.99). No significant effect on PFS was observed among patients treated with RASi (HR = 0.67, 95% CI: 0.45–1.00) and CCB (HR = 0.71, 95% CI: 0.46–1.11). In multivariate analysis, it was found that only the use of RASi could delay disease progression, and CCB had no significant effect on PFS (Table 4). We did not observe an effect on survival with either beta-blockers (OS: HR = 0.56, 95% CI: 0.18–1.79; PFS: HR = 0.98, 95% CI: 0.46–2.10) or diuretics (OS: HR = 1.94, 95% CI: 0.78–4.83; PFS: HR = 1.10, 95% CI: 0.48–2.49). In addition, it was found that ECOG-PS, treatment line, and combination chemotherapy were factors affecting the prognosis of tumor patients.

The relationship between anti-hypertensive drugs and immunotherapy. (a and b) Kaplan–Meier curves for the association of anti-hypertensive drug use with OS and PFS. (c and d) OS and PFS in RASi versus non-RASi patients. (e and f) OS and PFS in CCB versus non-CCB patients (after matched).
Univariate and multivariate analyses to determine the factors affecting OS.

Significant p values (p<0.05) are indicated in
CCB, calcium channel blocker; 95% CI, 95% confidence interval; ECOG-PS, Eastern Cooperative Oncology Group Performance Status; HR, hazard ratio; OS, overall survival; RASi, renin–angiotensin system inhibitor.
Univariate and multivariate analyses to determine the factors affecting PFS.

Significant p values (p<0.05) are indicated in
CCB, calcium channel blocker; CI, confidence interval; ECOG-PS, Eastern Cooperative Oncology Group Performance Status; HR, hazard ratio; PFS, progression-free survival; RASi, renin–angiotensin system inhibitor.
Subgroup analysis of the influence of RASi or CCB on immunotherapy
To further explore the potential benefits of RASi or CCB use in different patients, subgroup analysis was conducted and stratified by gender, age, and tumor type. Regarding RASi (Figure 2), the female patients using RASi exhibited a significant prolongation of OS (HR = 0.22, 95% CI: 0.07–0.72) and PFS (HR = 0.37, 95% CI: 0.16–0.84) compared to non-users. However, for male patients, the use of RASi was not associated with improved OS and PFS (OS: HR = 0.81, 95% CI: 0.42–1.57; PFS: HR = 0.70, 95% CI: 0.43–1.15). When stratified by age, RASi use did not show an association with patient prognosis in patients <65 years of age (OS: HR = 0.66, 95% CI: 0.31–1.39; PFS: HR = 0.73, 95% CI: 0.43–1.25), but patients ⩾65 years of age, the use of RASi was linked to a positive OS (HR = 0.23, 95% CI: 0.08–0.62) and PFS (HR = 0.47, 95% CI: 0.24–0.91) compared to non-users. In terms of tumor type, no association between RASi use and OS was found in lung cancer and GI tumors (lung cancer: HR = 0.39, 95% CI: 0.11–1.40; GI cancer: HR = 0.55, 95% CI: 0.25–1.22). However, the use of RASi demonstrated better disease control in lung cancer patients compared to non-users (HR = 0.37, 95% CI: 0.17–0.79), as evidenced by longer PFS, although similar results were not found in GI cancer (HR = 0.79, 95% CI: 0.45–1.38).

Subgroup analysis of the effect of RASi use on OS (a) and PFS (b) in patients receiving ICIs.
An association between CCB use and OS was observed in women and elders compared to men or patients <65 years of age. In lung cancer patients, no benefit of CCB use in OS and PFS was found. However, we observed a longer OS in GI cancer patients using CCB compared to non-users (HR = 0.24, 95% CI: 0.07–0.87), the similar results were not found in PFS (Figure 3).
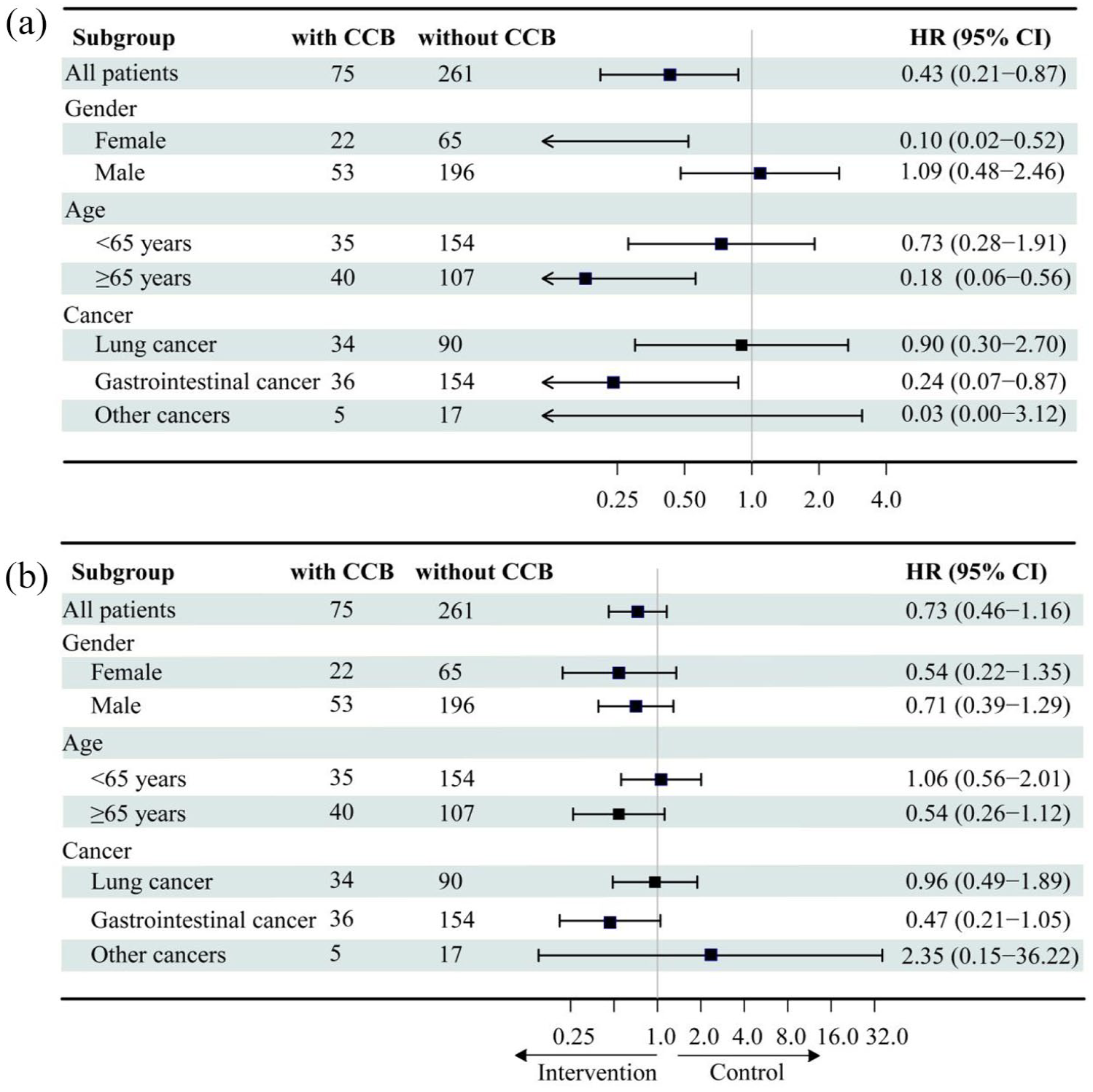
Subgroup analysis of the effect of CCB use on OS (a) and PFS (b) in patients receiving ICIs.
Meta-analysis
Study characteristics
The meta-analysis included a total of 17 studies, enrolling 13,707 patients. A flowchart illustrating the screening process for literature is presented in Figure 4. Anti-hypertensive drugs, commonly used in hypertensive patients, include RASi, CCB, beta-blockers, and diuretics. However, the effect of diuretics in patients receiving immunotherapy has only been shown in a single study. The most prevalent cancers among the included studies were lung cancer and melanoma. Patients were from various regions, primarily the United States, Europe, and Asia. The studies were published from 2016 to 2024. The median patient age ranged from 57.6 to 73.7 years. The analysis included only retrospectively conducted studies. The general characteristics of the included studies are shown in Table 5. The NOS was used to assess the quality of the 17 studies, all of which were of moderate or high quality (Supplemental Table S3).

PRISMA flow chart.
Baseline characteristics of studies included (n = 17).

Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CCB, calcium channel blocker; CI, confidence interval; CTLA-4, cytotoxic T lymphocyte antigen 4; HCC, hepatocellular carcinoma; HR, hazard ratio; ICIs, immune checkpoint inhibitors; MRCC, metastatic renal cell cancer; mUC, metastatic urothelial carcinoma; NA, not available; NOS, Newcastle–Ottawa Quality Assessment Scale; NSCLC, non-small-cell lung cancer; OS, overall survival; PD-1, programmed cell death 1; PD-(L)1, programmed cell death (ligand) 1; PFS, progression-free survival; RASi, renin–angiotensin system inhibitor.
Effect of anti-hypertensive drugs
Seventeen studies examined the impact of anti-hypertensive medications on OS among patients treated with ICIs. In comparison to patients not receiving anti-hypertensive medication, those who were administered anti-hypertensive drugs had an extended OS (HR = 0.94, 95% CI: 0.89–0.99, p = 0.0134, Figure 5). Heterogeneity in studies was low (I2 = 45%, p = 0.01); thus, a fixed-effects model was used to combine effect sizes. However, in a pooled meta-analysis of 12 studies on PFS, no significant association was observed between the use of anti-hypertensive medication and PFS (HR = 0.97, 95% CI: 0.85–1.09, p = 0.567, Figure 6) and a random-effects model was used to analyze PFS due to the high heterogeneity observed among the studies (I2 = 59%, p < 0.01).

Forest plots of the hazard ratio of OS in patients receiving immunotherapy combined with anti-hypertensive drugs.

Forest plots of the hazard ratio of PFS in patients receiving immunotherapy combined with anti-hypertensive drugs.
To investigate the impact of different types of anti-hypertensive drugs on immunotherapy and potential factors affecting heterogeneity, we conducted subgroup analyses stratified by type of anti-hypertensive drugs and tumor type. For the effect of different anti-hypertensive drugs on OS (Figure 7) or PFS (Figure 8), the use of RASi was found to be associated with longer OS (HR = 0.89, 95% CI: 0.84–0.94, p < 0.01). However, no improvement was observed for PFS (HR = 0.91, 95% CI: 0.70–1.20, p = 0.421). When taking CCB to treat hypertension, the use of CCB was not associated with OS (HR = 1.04, 95% CI: 0.92–1.18, p = 0.492) or PFS (HR = 1.03, 95% CI: 0.92–1.16, p = 0.670). The same results were observed in patients receiving beta-blockers (OS: HR = 1.02, 95% CI: 0.93–1.11, p = 0.706; PFS: HR = 0.96, 95% CI: 0.83–1.12, p = 0.642). For tumor type (Figures 9 and 10), no significant correlation was observed between the use of anti-hypertensive drugs and PFS (HR = 0.98, 95% CI: 0.75–1.29) or OS (HR = 1.04, 95% CI: 0.92–1.27) in lung cancer treated with ICIs. Similarly, anti-hypertensive drugs did not show a significant association with the prognosis of melanoma patients undergoing immunotherapy (OS: HR = 0.90, 95% CI: 0.65–1.23, p = 0.503; PFS: HR = 0.88, 95% CI: 0.67–1.15, p = 0.580). However, in the case of urothelial cancer, anti-hypertensives were associated with a favorable OS (HR = 0.44, 95% CI: 0.28–0.70, p = 0.001).

Forest plots of HR of OS in patients receiving ICIs combined with different anti-hypertensive drugs.

Forest plots of the HR of PFS in patients receiving ICIs combined with different anti-hypertensive drugs.

Forest plots of the HR of OS with regard to cancer.

Forest plots of the HR of PFS with regard to cancer.
Stability of result
The results indicated the absence of publication bias in HR analysis of PFS (Begg’s test: p = 0.2629, Egger’s test: p = 0.4669, Supplemental Figure 1) and OS (Begg’s test: p = 0.3587, Egger’s test: p = 0.7729, Supplemental Figure 2). The sensitivity analysis results indicated that the HR for OS and PFS was not significantly impacted by any individual study (Supplemental Figures 3 and 4). Collectively, these findings demonstrate the stability of the study outcomes, which appear to be unaffected by potential publication bias.
Discussion
Based on a retrospective cohort study and meta-analysis, we performed a comprehensive and systematic investigation of the impact of anti-hypertensive drugs on immunotherapy and had two key findings. First, we found that anti-hypertensive treatment can work synergistically to improve the antitumor efficacy of ICIs, significantly extending patients’ OS and PFS. Notably, RASi or CCB demonstrated a stronger synergistic effect. Second, we identified for the first time that the synergistic benefits of RASi or CCB on ICIs were concentrated in females or patients ⩾65 years of age. It is worth noting that RASi or CCB exhibited different benefits in various types of tumors. In lung cancer patients, RASi showed a tendency to be associated with better clinical outcomes, while the benefit of CCB was more pronounced in patients with GI tumors.
For RASi, Drobni et al. 16 enrolled 5910 cancer patients who were taking anti-hypertensive drugs and receiving immunotherapy. A total of 57.9% of the patients were on RASi. They found that RASi was related to better prognosis (HR = 0.92, 95% CI: 0.85–0.99), which aligns with our findings. The results remained stable even after adjusting for baseline characteristics, tumor type, ECOG PS, tumor metastasis, treatment line, and combination chemotherapy. Although some previous studies have reported negative results, our meta-analysis of pooled studies still supports the synergistic effect of RASi on immunotherapy.
For CCB, we found that using CCB significantly improved OS in patients undergoing immunotherapy, but previous studies have not found similar results. The proportion of CCB use ranged from 8% to 23% in the four available studies, with two focusing on NSCLC 11 and melanoma, 17 and the others examining multiple cancer types.13,19 In our study, the percentage of CCB users was 22.3%, with a majority having GI tumors (48%). The discrepancy with previous studies might be attributed to sample size limitations and tumor heterogeneity, yet our findings offer valuable insights. Future studies with larger sample sizes are necessary to address the research gap in this field. However, the number of beta-blocker or diuretic users was minimal in our study. Only one study mentioned that the use of diuretics did not affect patient survival. 19 Therefore, further large-scale studies are required to determine the effect of beta-blockers or diuretics on the efficacy of immunotherapy.
Based on real-world data, the present study is the first attempt to explore the patient populations potentially benefiting from the synergistic effects of RASi or CCB with ICIs. A subgroup analysis was performed, taking into account variables such as gender, age, and tumor type. Surprisingly, our findings revealed notable disparities in the efficacy of RASi or CCB among patients with different tumor types. In lung cancer patients, RASi showed potential in controlling disease progression. In preclinical studies, RAS may facilitate the development of a tumor immunosuppressive microenvironment by upregulating the expression of PD-L1 28 or influencing myeloid cells and fibroblasts. 29 Consequently, RASi has the potential to enhance the efficacy of immunotherapy by mitigating immunosuppression. This may explain why RASi can enhance the efficacy of ICIs and lead to a significant extension of PFS in lung cancer patients. 11 However, our meta-analysis did not reveal a significant association between the use of anti-hypertensive drugs and prognosis in lung cancer patients. This discrepancy may be attributed to the limited sample size of our clinical cohort. Therefore, we believe that larger-scale studies are needed to draw more comprehensive and precise conclusions. CCB has been found to improve the prognosis for patients with GI tumors, as demonstrated by significantly longer OS. In colorectal cancer mice, CCB effectively reduced the expression of programmed cell death ligand 1 (PD-L1) in tumor cells and programmed cell death 1 (PD-1) in T lymphocytes. 30 This indicates a potential to boost the antitumor effects when combining CCB with ICIs. However, more research is needed to validate these findings. Currently, there is a lack of studies specifically investigating the prognostic effects of CCB among patients receiving immunotherapy for GI tumors, and there is insufficient data for meta-analysis. The present study aptly fills the gap. In the future, we will continue to investigate the potential effects of anti-hypertensive drugs on the prognosis of GI cancer patients. In addition, we discovered that RASi or CCB exhibit stronger synergistic effects in women or patients ⩾65 years of age. However, there is limited research on how gender or age influences the relationship between anti-hypertensive drugs and immunotherapy. Future prospective studies are necessary to strengthen the validity of our results. These findings highlight the potential for personalized treatment strategies that combine RASi or CCB with ICIs and emphasize the need for further research to optimize treatment approaches for various patient subgroups.
In clinical practice, patients are often treated with multiple drugs. To eliminate the influence of other concomitant medications, we specifically analyzed the impact of drugs known to influence immunotherapy, such as antibiotics, 31 glucocorticoids, 32 and PPI. 33 However, we did not obtain any significant effect of antibiotics and glucocorticoids on patients’ prognosis, which may be attributed to the limited sample size and the brief duration of their use. Our results showed that using PPI was associated with improved clinical outcomes. Previous research has discovered a connection between PPI and ICI monotherapy. A recent study suggested that patients taking PPI in combination with chemotherapy and ICIs experienced more favorable outcomes compared to those undergoing immune monotherapy. 34 However, only 25% of the patients receiving ICIs and chemotherapy in the cohort of this study were on PPI. Our clinical cohort demonstrated that 87.8% of patients received chemotherapy in combination with immunotherapy, with over half of these patients concurrently taking PPI, which may have contributed to the positive results observed in our study. Upon conducting multivariate analysis while controlling for PPI use, we found that the association between anti-hypertensive medications and ICIs remained stable. However, the conflicting results need to be verified through further experiments.
Despite the medical importance of anti-hypertensive drugs in cancer remaining inconclusive, the outcomes from cellular and animal experiments are encouraging. The Ang II/Angiotensin 1 signaling axis plays a crucial role in regulating the cellular matrix, which, in turn, affects tumor perfusion and promotes an immunosuppressive microenvironment.9,35 So, the use of RASi may represent a viable strategy for adjunctive antitumor therapy. Recently, clinical trials are underway that investigate the combination of losartan with nivolumab for the treatment of pancreatic cancer. Furthermore, the combination of verapamil with a PD-1 inhibitor has been associated with improved survival rate in a mice model of cervical cancer. 36 This indicated that CCB may offer a promising therapeutic option for cervical cancer patients with hypertension. In addition, the inhibition of beta-adrenergic signaling has the potential to enhance antitumor immune responses. 37 Kokolus et al. 10 demonstrated that the combination of propranolol and PD-1 significantly slowed tumor growth in melanoma mice. Further clinical studies are needed to validate the above findings.
Our study is limited by several factors. First, the inclusion of a small clinical cohort restricts the ability to evaluate the relationship between beta-blockers or diuretics and the efficacy of immunotherapy, as well as the impact of anti-hypertensive drugs on prevalent tumors or various ICIs. Prospective validation in a larger group is necessary to enhance confidence in the conclusions. Second, a chemotherapy control group should be established to determine the relationship between immunotherapy and anti-hypertensive drugs, but the limited number of patients receiving ICIs as monotherapy, mainly elderly patients, and confounding baseline characteristics in the preliminary patient analysis hindered a controlled study. Future clinical studies should aim to rectify these issues and establish a control group. Third, our analysis did not include data regarding the dose–response relationships of the anti-hypertensive drugs, due to being underpowered to analyze the dose on the outcome (data not presented). Prospective studies are required to further explore this relationship. Lastly, the 17 studies included in the meta-analysis were retrospective and exhibited inadequate control of variables due to reporting and selection biases, resulting in a lack of comparability at baseline. Although we performed meta-regression to address heterogeneity among studies, no factors explaining the observed heterogeneity were identified. In summary, additional randomized clinical trials are necessary to validate the relationship between hypertensive medications and immunotherapy.
Conclusion
Overall, based on clinical trials and meta-analysis, we found that taking anti-hypertensive drugs was associated with a favorable OS in cancer patients receiving ICIs, with RASi or CCB demonstrating superior synergy effects. However, due to the inherent limitations of current research, a significant number of prospective and fundamental studies are required to elucidate the effects of RASi or CCB on immunotherapy. The objective of our study was to provide more precise clinical guidance aimed at enhancing cancer patient outcomes in clinical practice.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241292227 – Supplemental material for Anti-hypertensives associated with survival in cancer patients receiving immunotherapy: new evidence from a real-world cohort study and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241292227 for Anti-hypertensives associated with survival in cancer patients receiving immunotherapy: new evidence from a real-world cohort study and meta-analysis by Ping Ma, Zhihuan Zhang, Mengying Qian, Hao Jiang, Yu Zhao, Qing Shan, Xia Liu, Tianming Yao and Jinmin Guo in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
