Abstract
Background:
The limited efficacy of chemotherapy in improving survival in pancreatic ductal adenocarcinoma (PDAC) necessitates the exploration of novel strategies to overcome treatment resistance.
Objectives:
This study aimed to investigate the impact of combining renin–angiotensin system (RAS) blockers with chemotherapy on survival outcomes in patients with PDAC.
Design:
Patients with PDAC were enrolled in the retrospective study.
Methods:
We analyzed patients with PDAC (n = 384) at our institution between 2014 and 2021. Survival outcomes, including event-free survival (EFS) and overall survival (OS), were analyzed according to the concomitant use of RAS blockers.
Results:
Among the 384 patients in the study, 70 (18.2%) concomitantly received angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs). Patients in the ACEI/ARB group, characterized by older age and more comorbidities, displayed a significantly superior 12-month EFS rate (22.86% versus 13.69%, p = 0.008) compared to the non-ACEI/ARB group, while OS remained similar between the groups. In the multivariate analysis, the use of ACEI/ARB was associated with better 12-month EFS (hazards ratio = 0.71, 95% confidence interval: 0.52–0.96; p = 0.024). Poor performance, advanced disease status, and higher CA19-9 levels were associated with poor survival outcomes.
Conclusion:
Concomitant use of ACEIs/ARBs in patients with pancreatic cancer resulted in significantly better 12-month EFS. Age, performance status, disease status, and higher CA19-9 levels were independent predictors of survival. The combination strategy might provide better treatment outcomes in patients with PDAC.
Keywords
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is a major contributor to cancer-related mortality, with a significant increase in global incidence over the past decades. 1 Several risk factors, including cigarette smoking, alcohol consumption, obesity, diabetes, family history, chronic pancreatitis, and genetic susceptibility, have been linked to the development of pancreatic cancer.2,3 However, owing to the lack of effective screening programs, more than half of the patients are diagnosed with locally advanced or metastatic disease and have a discouraging 5-year survival rate of approximately 10% worldwide. 4
Although surgical resection remains the primary approach for improving survival outcomes in early-stage PDAC, the 5-year survival rate after the procedure remains relatively low, ranging from 10% to 25%. 5 Moreover, systemic therapies, particularly chemotherapy, have revealed limited efficacy in significantly improving survival rates over the past few decades. 6 Therefore, there is an urgent need to explore new strategies to overcome treatment resistance and extend survival outcomes in patients with pancreatic cancer.
The renin–angiotensin system (RAS) plays a crucial role in regulating blood pressure, 7 and medications such as angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) have been approved and are effective in treating hypertension. Preclinical studies have suggested that inhibition of the RAS pathway may suppress tumor growth and metastasis.8,9 They have also exhibited the survival benefits associated with RAS blockade in patients with pancreatic adenocarcinoma treated with chemotherapy.10,11 However, retrospective studies have reported conflicting results regarding survival outcomes and the use of RAS blockers.12,13 Therefore, we aimed to retrospectively evaluate patients diagnosed with PDAC at a tertiary medical center to investigate the effects of RAS blockade combined with chemotherapy on survival outcomes.
Materials and methods
Patient selection
Patients were retrospectively obtained from the cancer registry database and the medical records of patients treated at Taichung Veterans General Hospital between 2014 and 2021, with the follow-up period extending until 30 April 2022. Patients with histologically confirmed pancreatic adenocarcinoma were included in our study. Furthermore, patients were required to follow up for a duration of more than 30 days. Patients without histologically confirmed pancreatic adenocarcinoma, pathology reports indicating a different type of pancreatic cancer, lost to follow-up, inadequate clinical records, human immunodeficiency virus (HIV) infection, without systemic treatment for PDAC and early mortality within 30 days from the time of diagnosis were excluded. Patients were classified as operable at the time their disease was diagnosed. This study was conducted with the approval of the Ethics Committee of Taichung Veterans General Hospital, and the institutional review board waived the requirement for informed consent. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology Statement. 14
Statistical analysis
Patients’ characteristics are summarized using medians with the first and third quartiles for continuous variables and numbers with percentages for categorical variables. The primary endpoint for this study is the overall survival (OS), representing the period from diagnosis to death from any cause. The event-free survival (EFS) is defined as the duration from diagnosis to the occurrence of relapse in operated patients, progression in non-operated patients, or death. OS and EFS were estimated using the Kaplan–Meier method, and the log-rank test was used to compare the survival curves. Categorical variables were compared using Fisher’s exact test, while continuous variables were assessed using the appropriate statistical test: independent t-test, Mann–Whitney U test, or χ2 test.
We used the Cox proportional hazards model to adjust for covariates. Hazards ratios (HRs) were calculated to estimate the effect of ACEIs/ARBs on survival outcomes by adjusting for important prognostic factors including age, sex, performance status based on the Eastern Cooperative Oncology Group (ECOG) performance scale, disease status (operable, advanced, or metastatic), body mass index (categorized as <18.5, ⩾18.5 and <25, and ⩾25 according to the national health insurance standards), pretreatment carbohydrate antigen 19-9 levels, comorbidities, blood type, and ACEI/ARB intervention. Prognostic factors (p < 0.05) in the univariate analysis were included in the multivariate analysis to assess their independent impact on survival outcomes. Variables with statistical significance in the univariate analysis were subsequently included in a multivariate analysis to determine their correlation and identify those with statistically significant differences. All statistical tests were two-sided, and p < 0.05 was considered statistically significant.
Results
Patient characteristics and comparison
Initially, 800 patients were included in our study. However, 354 patients were excluded from the analysis for various reasons, including the lack of tissue biopsies to confirm the diagnosis of pancreatic adenocarcinoma, pathology reports indicating a different type of pancreatic cancer, loss of follow-up, inadequate clinical records, HIV infection, and early mortality within 30 days from the time of diagnosis. In addition, 142 patients were excluded because they did not receive systemic treatment for PDAC. Furthermore, patients were required to undergo follow-up in our institution for a duration exceeding 30 days. In the end, our study included 384 patients with pancreatic cancer who received systemic chemotherapy (Figure 1). The median follow-up duration was 10.6 months, ranging from 1 to 73.8 months. Among these patients, 70 (18.2%) were treated with RAS blockers, which included ACEIs or ARBs. The remaining 314 (81.8%) patients received other antihypertensive drugs, such as calcium channel blockers and β-blockers, or no antihypertensive treatment (n = 155, 40.4%).

Flow diagram of the selected patients with pancreatic ductal adenocarcinoma receiving systemic chemotherapy.
Table 1 presents the characteristics of the patients included in this study. The median ages at diagnosis for patients who did and did not receive ACEI/ARB was 70 years [interquartile range (IQR): 63.8–78.0 years] and 68 years (IQR: 60.0–73.3 years), respectively (p = 0.023). This indicates that patients in the ACEI/ARB group were significantly older at the time of diagnosis. In addition, a higher proportion of patients in the ACEI/ARB group had comorbidities, such as hypertension, chronic kidney disease, and diabetes. However, other baseline characteristics were comparable between the two groups.
Characteristics of patients with pancreatic cancer in ACEI/ARB (+) and ACEI/ARB (−) groups.

Continuous data are expressed as the median and IQR. Categorical data are expressed as numbers and percentages.
ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin receptor blockers; BMI, body mass index; CA19-9, Carbohydrate antigen 19-9; CEA, Carcinoembryonic antigen; ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range.
Prognostic impact of ACEI/ARB use in patients with pancreatic cancer
To evaluate the impact of ACEIs/ARBs on survival outcomes, we analyzed the EFS and OS. In our cohort, the median OS for all patients was 13.3 months, and the median EFS was 4.5 months. Our findings revealed a significant improvement in EFS among patients receiving ACEI/ARB therapy compared to those not receiving ACEI/ARB treatment. The 12-month EFS rates were 22.86% and 13.69% in the ACEI/ARB group and non-ACEI/ARB group, respectively (p = 0.008). Moreover, the ACEI/ARB group presented superior EFS rates at 6 months (55.7% versus 39.5%, p = 0.019) and 24 months (11.4% versus 2.9%, p = 0.005) compared with the non-ACEI/ARB group [Figure 2(a)].
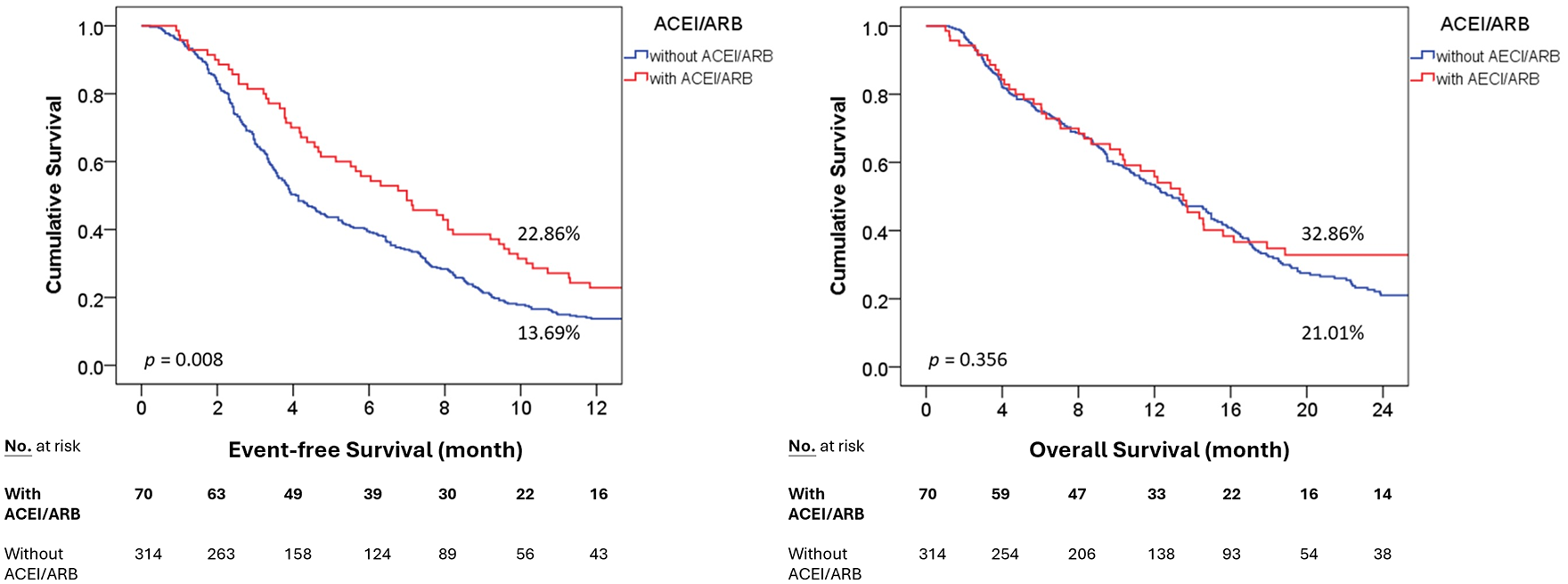
The utilization of ACEI/ARB in patients with pancreatic cancer prolonged the event-free survival. The Kaplan–Meier survival curves for (a) 12-month EFS and (b) OS in patients with pancreatic cancer with or without ACEI/ARB use. The p values have been analyzed by the log-rank test.
However, the OS rates did not differ significantly between the two groups, regardless of the follow-up duration [Figure 2(b)]. The 12-month OS rate was 32.86% and 21.01% in the ACEI/ARB and non-ACEI/ARB groups, respectively (p = 0.356). These results suggest that ACEI/ARB treatment is associated with improved EFS but does not significantly impact OS.
Prognostic factors of 12-month EFS in patients with pancreatic cancer
To investigate the prognostic factors influencing survival outcomes, we analyzed the clinical variables associated with 12-month EFS rates. Our findings revealed that the use of ACEIs/ARBs was an independent favorable prognostic factor for 12-month EFS [HR: 0.71; 95% confidence interval (CI): 0.52–0.96; p = 0.024]. Conversely, poor ECOG performance status, advanced disease status, and higher levels of the tumor marker CA19-9 were identified as significant adverse prognostic factors for 12-month EFS.
In addition, age, sex, BMI, comorbidities, and blood type did not significantly affect the 12-month EFS (Table 2). Thus, our results indicated that the use of ACEI/ARB, ECOG performance status, disease status at diagnosis, and CA19-9 levels were crucial factors influencing 12-month EFS rates.
Univariate and multivariate analyses of 12-month event-free survival.

Data were analyzed using a Cox proportional hazards regression model.
p < 0.05. **p < 0.01.
ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin receptor blockers; BMI, body mass index; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazards ratio.
Prognostic factors influencing the OS in patients with pancreatic cancer
Cox univariate and multivariate analyses were performed to determine prognostic factors influencing the OS. Our results revealed that age >65 years was a significant prognostic factor for OS, with an HR of 1.48 (95% CI: 1.13–1.95; p = 0.005). Moreover, poor performance status, advanced disease status at diagnosis, higher levels of CA19-9 (HR: 1.01; 95% CI: 1.001–1.01; p = 0.018), and the presence of heart failure (HR: 1.60; 95% CI: 1.07–2.39; p = 0.023) were identified as crucial prognostic factors affecting OS (Table 3).
Univariate and multivariate analyses of overall survival.

Data were analyzed using a Cox proportional hazards regression model.
p < 0.05. **p < 0.01.
ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin receptor blockers; BMI, body mass index; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazards ratio.
However, ACEIs/ARBs did not have a protective effect on OS. In summary, our analysis indicated that older age, poor performance status, advanced disease status, and the presence of heart failure were associated with poor OS outcomes.
Subgroup analysis of the use of ACEI/ARB in patients with pancreatic cancer
Regarding the potential benefits of concomitant ACEI/ARB use in patients with pancreatic cancer, our findings revealed that the administration of ACEI/ARB had a protective effect on 12-month EFS, particularly in specific subgroups (Supplemental Table 1). These subgroups included women, patients aged >65 years, those with operable disease, individuals with a BMI between 18.5 and 25, patients with an ECOG PS of 1 or higher, those with CA19-9 levels >90 ng/mL, individuals with diabetes, and patients with blood type O. These findings suggest that ACEI/ARB therapy might have a protective effect on EFS in patients with pancreatic cancer with abovementioned characteristics.
Discussion
In the present study, we observed significant protective effects associated with the concomitant use of ACEIs/ARBs in patients with pancreatic cancer receiving systemic chemotherapy, which were consistent in the 6-, 12-, and 24-month EFS rates. In our study, most patients with pancreatic adenocarcinoma (75.6%) received a gemcitabine-based regimen as first-line treatment, followed by S-1 monotherapy and the FOLFIRINOX regimen. These results align with prior studies exhibiting that ACEI/ARB use is linked to better EFS and OS in advanced pancreatic cancer patients treated with gemcitabine alone.15,16
The desmoplastic microenvironment plays a crucial role in the poor prognosis of pancreatic adenocarcinoma, with the transforming growth factor β (TGF-β) signaling pathway implicated in tumor progression and stromal fibrosis.17,18 The loss of SMAD4, a key mediator of TGF-β signaling, disrupts TGF-β-induced cell cycle arrest and is associated with chemoresistance and TGF-β-mediated tumorigenesis. 19 Using data from The Cancer Genome Atlas dataset, 20 our analysis revealed that high mRNA expression levels of SMAD4 were associated with significantly better relapse-free survival (HR: 0.32; 95% CI: 0.14–0.75; p = 0.0053). Conversely, high mRNA expression levels of TGF-β were associated with poor relapse-free survival (HR: 4.74; 95% CI: 1.10–20.42; p = 0.022, Supplemental Figure 1). Recently, ACEIs/ARBs have been studied for their inhibitory effects on the TGF-β signaling pathway to reduce tumor fibrosis. 21 Previous in vivo studies have presented that the use of ACEIs/ARBs enhances drug delivery by decompressing tumor vessels and reducing hypoxia.11,22 Taken together, targeting the RAS may have potential therapeutic effects in pancreatic adenocarcinoma.
Furthermore, several studies have reported encouraging clinical outcomes in patients with PDAC treated with ACEI/ARBs. A large cohort study (n = 8158) utilizing healthcare databases revealed a noteworthy 20% reduction in mortality risk (HR: 0.80; 95% CI: 0.72–0.89) among patients exposed to ARBs or ACEIs after pancreatic cancer diagnosis.13,23 This study found ARBs or ACEIs to be most beneficial within the first 3 years post-diagnosis. Typically, initial gemcitabine treatment results in a median survival of 4.9 months for advanced cancers. 24 When chemotherapy was combined with ACEIs/ARBs, the results presented better clinical outcomes for median EFS and OS (8.7 and 15.1 months, respectively) in a retrospective study. 15 In the study, the patients received gemcitabine alone as a frontline treatment without surgery or radiation before chemotherapy. In another study of patients with pancreatic cancer without metastasis, a significantly longer OS (median OS, 36.3 versus 19.3 months, p = 0.011) was observed in the ACEI/ARB subgroup. Our study presented that ACEIs or ARBs extended EFS in patients with pancreatic cancer, whether operable or metastatic, without significantly impacting OS.
Our analysis revealed that ACEIs/ARBs offer protective benefits in pancreatic cancer patients, with survival outcomes affected by factors like performance status, disease stage, and CA19-9 levels. Previous studies have reported an increased incidence of pancreatic cancer among patients with a history of diabetes, as well as among those with blood types A, B, or AB, compared to blood type O individuals. 25 In the context of adjuvant S-1 therapy, patients with blood type O have favorable survival outcomes. 26 Another study exhibited patients with blood type O had better survival rates in resected pancreatic cancer than those with other blood types. 27 On the other hand, elevated serum CA19-9 and CEA levels are indicative of an unfavorable prognosis. However, our study did not identify blood type O as a significant factor affecting OS outcomes. Nevertheless, we presented that high CA19-9 levels were associated with poor survival outcomes in the entire cohort. By contrast, the use of ACEIs/ARBs in subgroups including females, individuals aged >65 years, those with operable disease, ECOG PS ⩾1, higher CA19-9 levels, diabetes mellitus, and blood type O provided better event-free outcomes.
In recent decades, significant advances have been made in the development of effective combination chemotherapeutic regimens for pancreatic cancer.28,29 Furthermore, salvage chemotherapy has become increasingly prevalent, contributing to improved survival outcomes.30,31 Although there was no significant improvement in OS observed in our study, this could be due to the influence of salvage chemotherapy and molecular pathways beyond the TGF-β signaling pathway. Notably, ACEIs/ARBs, which are commonly employed for the management of hypertension and heart failure, have presented a favorable safety profile. Thus, concurrent administration of ACE/ARB has the potential to synergistically enhance pancreatic cancer treatment outcomes.
There were several limitations to our study. First, the retrospective nature of the study limited our ability to comprehensively assess the total duration of ACEIs/ARBs use following pancreatic cancer diagnosis, making it challenging to clearly determine the long-term impact of ACEIs/ARBs on OS. Second, many ACEIs/ARBs were used in clinical practice, and it was difficult to identify the suitable drug and ideal dosage. Multiple first-line chemotherapy regimens make it hard to identify which benefits most from ACEIs/ARBs use. Further clinical trials are necessary to understand ACEIs/ARBs’ benefits in pancreatic cancer treatment.
Conclusion
Our retrospective study highlighted the potential benefits of concurrent ACEI/ARB use in patients with pancreatic cancer, yielding improved EFS outcomes. Notably, this advantage is particularly pronounced in specific subgroups, including female patients, older individuals, those with poor performance status, elevated CA19-9 levels, diabetes, and blood type O. However, further prospective investigations are essential to comprehensively elucidate the therapeutic potential of RAS inhibitors in pancreatic cancer management.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241247019 – Supplemental material for The concomitant use of the renin–angiotensin system inhibitors and survival outcomes of patients with pancreatic adenocarcinoma: an analysis from a tertiary center
Supplemental material, sj-docx-1-tam-10.1177_17588359241247019 for The concomitant use of the renin–angiotensin system inhibitors and survival outcomes of patients with pancreatic adenocarcinoma: an analysis from a tertiary center by Kuan-Yu Tseng, Chiann-Yi Hsu, Yu-Hsuan Shih, Hsin-Chen Lin, You-Cheng Li, Chieh-Lin Jerry Teng and Cheng-Wei Chou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-1-tam-10.1177_17588359241247019 – Supplemental material for The concomitant use of the renin–angiotensin system inhibitors and survival outcomes of patients with pancreatic adenocarcinoma: an analysis from a tertiary center
Supplemental material, sj-jpg-1-tam-10.1177_17588359241247019 for The concomitant use of the renin–angiotensin system inhibitors and survival outcomes of patients with pancreatic adenocarcinoma: an analysis from a tertiary center by Kuan-Yu Tseng, Chiann-Yi Hsu, Yu-Hsuan Shih, Hsin-Chen Lin, You-Cheng Li, Chieh-Lin Jerry Teng and Cheng-Wei Chou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
