Abstract
Background:
Although dual blockade HER2-based neoadjuvant chemotherapy is associated with excellent outcomes for human epidermal growth factor receptor 2 (HER2)-positive breast cancer, pertuzumab is not available to all patients due to cost. The optimal neoadjuvant chemotherapy for HER2-positive breast cancer in the presence of a single HER2 blockade is unknown. This study aimed to compare the efficacy and safety of epirubicin/cyclophosphamide followed by docetaxel/trastuzumab (EC-TH) with docetaxel/carboplatin/trastuzumab (TCH) neoadjuvant setting for HER2-positive breast cancer under the single HER2 blockade.
Methods:
Patients with stage II-IIIC HER2-positive breast cancer were randomly assigned to either eight cycles of EC-TH every 3 weeks during all chemotherapy cycles, or six cycles of TCH every 3 weeks. The primary endpoint was pathological complete response (pCR) (defined as the absence of invasive tumor cells in breast and axilla, ypT0/is ypN0).
Results:
From May 2017 to November 2019, 140 patients were randomly assigned, and 135 patients were ultimately found evaluable for the primary endpoint. The pCR was recorded in 25 of 67 patients [37.3%; 95% confidence interval (CI), 25.8–50.0] in the EC-TH group and in 38 of 68 patients (55.9%, 95% CI, 43.3–67.9) in the TCH group (p = 0.032). The most common adverse events (AEs) were neutropenia in 24 of 67 (35.8%) patients in the EC-TH group versus 27 of 68 (39.7%) in the TCH group (p = 0.642), anemia in 33 of 67 (49.3%) patients in the EC-TH group versus 34 of 68 (50.0%) in the TCH group (p = 0.931), and thrombocytopenia in five of 67 (7.5%) patients in the EC-TH group versus 17 of 68 (25.0%) in the TCH group (p = 0.006).
Conclusion:
For patients receiving the single HER2 blockade trastuzumab for HER2-positive breast cancer, TCH regimen might be a preferred neoadjuvant therapy.
Trial registration:
This trial was registered with ClinicalTrials.gov identifier: NCT03140553) on 2 May 2017.
Background
Neoadjuvant therapy for breast cancer has been used to downstage disease, improve resectability, and reduce the extent of surgery. 1 Neoadjuvant therapy also enables a quick assessment of the effectiveness of systemic treatment.2,3 Pathological complete response (pCR) in the breast and lymph nodes after neoadjuvant therapy is associated with favorable outcomes in certain subtypes of breast cancer.1,3–5 The neoadjuvant setting also provides an opportunity to tailor adjuvant therapy options according to pathologic response.6,7 The US Food and Drug Administration also supports the use of pCR as an endpoint in clinical testing of neoadjuvant therapy. 8
The human epidermal growth factor receptor 2 (HER2) is overexpressed or amplified in 20–25% of all breast cancer cases, and is associated with poor prognoses.9–12 Neoadjuvant therapy has become the standard of care for most patients with HER2-positive early breast cancer.3–5 The anti-HER2 monoclonal antibody trastuzumab has significantly improved the prognosis in patients with HER2-positive breast cancer,13–15 and HER2-targeted therapies have become the backbone of standard treatment regimens for patients with HER2-positive breast cancer. The National Comprehensive Cancer Network (NCCN) guidelines recommend the use of several regimens, such as a sequential combination of an anthracycline-containing regimen and taxane or concurrent use of docetaxel and carboplatin, in combination with at least trastuzumab for the (neo-) adjuvant chemotherapy of stage II/III HER2-positive primary breast cancer with the possible addition of pertuzumab. 16 Although dual blockade HER2-based neoadjuvant chemotherapy is associated with excellent outcomes for HER2-positive breast cancer, the high cost of pertuzumab makes it unavailable to many patients globally.
HER2-targeted treatment with trastuzumab has been associated with cardiac dysfunction, 17 particularly when combined with anthracycline-containing chemotherapy at higher doses.15,17 A pivotal trial in metastatic disease revealed cardio toxicity in 16% of patients when trastuzumab was administered with doxorubicin. 13 Because of the overlap in cardiotoxicity caused by anthracyclines and trastuzumab, anthracycline-free regimens have been assessed.15,18–21 Moreover, the GeparQuattro trail showed that about 24.9% of patients with HER2-positive breast cancer experienced no change in clinical progressive disease after four cycles of epirubicin/cyclophosphamide (EC). In patients with no change in clinical progressive disease after four cycles of EC, the pCR rate was 16.7%, possibly indicating that this group benefited the most from docetaxel and trastuzumab treatment. 22 The BCIRG-006 trial directly compared doxorubicin and cyclophosphamide followed by docetaxel and trastuzumab (AC-TH) regimens and docetaxel/carboplatin/trastuzumab (TCH) regimens in an adjuvant setting. After 10.3 years of follow-up, the BCIRG006 trial reported similar disease-free and overall survival rates in patients treated with a TCH regimen but a reduced acute and long-term toxicity compared with those in patients treated with AC-TH. 23 However, the optimal neoadjuvant chemotherapy in terms of safety and efficacy for HER2-positive breast cancer with single HER2 blockade is still unknown.
Therefore, the neoCARH study was designed to directly compare the efficacy and safety of epirubicin/cyclophosphamide followed by docetaxel/trastuzumab (EC-TH) with TCH in the single HER2 blockade neoadjuvant setting for HER2-positive breast cancer.
Methods
Study design and patients
The neoCARH study was a randomized, open-label, multicenter, phase II, neoadjuvant trial (ClinicalTrials.gov identifier: NCT03140553). Eligible patients were female, aged >18, and had HER2-positive invasive breast carcinoma, clinical stage II–III C, and Eastern Cooperative Oncology Group performance status 0 or 1, with normal organ and heart function. Tumors had to be HER2 immunohistochemistry 3+ or 2+ and positive for fluorescence in-situ hybridization (FISH). Tumors with estrogen receptor or progesterone receptor expression of 10% or more were defined as hormone receptor positive. Key exclusion criteria were stage IV breast cancer, bilateral breast cancer, other malignancies, inadequate organ function, impaired cardiac function, uncontrolled hypertension, previous systemic therapy for the treatment or prevention of breast cancer, or previous excisional biopsy of a primary tumor or axillary lymph node. All included patients provided written informed consent. The study was sponsored by Guangdong Provincial People’s Hospital and was approved by Ethics Committee of Guangdong Provincial People’s Hospital [no. GDREC 2016423H(R1)], together with the ethics committees at each participating institution.
Randomization
Patients were randomly assigned (1:1) to either the EC-TH group or the TCH group. Patients and investigators were not masked to the study treatment. Pathologists assessing the amount of residual tumor after neoadjuvant treatment were blinded to patient information and were not informed about the treatment allocation.
Procedures
Patients in the EC-TH group were administered four cycles of epirubicin (90 mg/m²), and cyclophosphamide (600 mg/m²) intravenously, followed by four cycles of docetaxel (100 mg/m²) and trastuzumab every 3 weeks. Patients in the TCH group were treated with docetaxel (75 mg/m²) plus carboplatin (area under the curve, 6 mg/ml per min) administered every 3 weeks for six cycles concurrently with trastuzumab. In the two regimens, trastuzumab was initially administered at a loading dose of 8 mg/kg, followed by 6 mg/kg every 3 weeks to complete 1 year of trastuzumab treatment. Treatment was discontinued early in the event of progressive disease or unacceptable toxicity.
Adverse events (AEs) were assessed with each cycle of treatment according to Common Terminology Criteria for Adverse Events (CTCAE) version 4.03. Hematopoietic growth factors were recommended for primary prophylaxis of treatment-emergent neutropenia according to NCCN guidelines. In patients with combined thrombocytopenia and neutropenia, dose reduction was warranted. Chemotherapy was postponed with a subsequent 25% dose reduction. Trastuzumab was temporarily or permanently discontinued if the left ventricular ejection fraction (LVEF) was decreased by >15% from baseline or if it was decreased by ⩾10% with LVEF below the lower limit of normal.
Local and adjuvant therapy
Patients underwent surgery within 6 weeks after their final dose of chemotherapy. Surgery consisted of either breast-conserving surgery or mastectomy, depending on tumor characteristics and patient preferences. Patients were treated with adjuvant radiotherapy, endocrine therapy, and adjuvant anti-HER2 treatment according to NCCN guidelines. All enrolled and randomized patients remained in the study and follow-up unless consent was withdrawn.
Outcomes
The primary endpoint was the percentage of pCR (ypT0/is, ypN0), which was defined as the absence of any residual invasive cancer in both the breast and axillary lymph nodes. The secondary endpoints in this study were: clinical response rates, according to the Response Evaluation Criteria in Solid Tumors, version 1.1; the safety of each treatment regimen, according to CTCAE version 4.03; the percentage of patients who underwent breast-conserving surgery; event-free survival (defined as the time from the date of randomization to either the first documentation of progressive disease or death); disease-free survival (DFS; time from the first date of no disease to the first documentation of progressive disease or death); and overall survival (OS; time from randomization to death from any cause). Primary and secondary objectives and endpoints were prespecified in the protocol.
Statistical analyses
We hypothesized that the TCH regimen would improve the proportion of patients with pCR from 38% to 64% compared with the EC-TH regimen. The expected proportion of pCR in the TCH group was derived from our experience with a dose-dense paclitaxel, carboplatin, and trastuzumab regimen. 24 A sample size of 124 patients was sufficient for 80% power with a two-sided significance level of 0.05. To account for the lost to follow-up rate of 10%, we planned to enroll 140 patients. All patients who were administered at least one treatment were included in the efficacy and safety analyses. The proportions of patients with pCR were estimated and reported with 95% confidence intervals (CIs) obtained by the Clopper–Pearson method. Differences in pCR rate and other categorical variables between the groups were tested using the chi-square test or Fisher’s exact test. Patients without a reported assessment of pCR (e.g. those with disease progression or who did not undergo surgery) were categorized as not having achieved a pCR. We performed multivariate logistic regression analysis to adjust for the effects of covariates that might have been related to pCR. All statistical analyses were completed using SPSS for Windows, Version 16.0, (SPSS Inc., Chicago, USA) and R3.6.3, and statistical significance was defined as a p value < 0.05.
Results
From May 2017 to November 2019, 140 patients were randomly assigned from nine centers in China. Five patients refused treatment and were excluded from the analyses: three patients (two in the EC-TH group and one in the TCH group) refused treatment and were administered the dual blockade HER2-based neoadjuvant therapy; two patients in the TCH group refused treatment and underwent surgery in advance). The remaining 135 patients (66 in the EC-TH group, 67 in the TCH group) were included in the intention-to-treat populations. Four patients withdrew from the study (two in the EC-TH group because of grade 4 neutropenia, one in the TCH group because of grade 3 thrombocytopenia, one in the TCH group withdrew because of patient’s decision). One patient in the EC-TH group developed grade 4 neutropenia after one course of chemotherapy and underwent surgery in advance. One patient in the TCH group underwent surgery after two courses of chemotherapy because of patient’s decision, and achieved a pCR. The remaining two patients withdrew from the study after one course of chemotherapy and did not undergo surgery. Figure 1 shows the patient profile for the trial. Baseline patient characteristics were well balanced between the treatment groups (Table 1).

Patient selection for the trial.
Patient demographics at baseline.

ECOG, Eastern Cooperative Oncology Group; EC-TH, epirubicin/cyclophosphamide followed by docetaxel/trastuzumab; ER, estrogen receptor; PR, progesterone receptor; TCH, docetaxel/carboplatin/trastuzumab.
Efficacy
In the EC-TH treatment group, 25 of 67 patients (37.3%, 95%, 25.8–50.0) achieved a pCR, compared with 38 of 68 patients (55.9%, 95% CI, 43.3–67.9) in the TCH group [p = 0.032; Figure 2(a)]. Consistent with findings shown in previous studies, the pCR rate was higher in patients with hormone-receptor (HR)-negative tumors than in those with HR-positive tumors in both groups [Figure 2(b)]. Subgroup analyses of pCR by treatment in subgroups defined by clinically relevant baseline characteristics are shown in Figure 3. In multivariate logistic regression analysis to adjust for the effects of clinicopathological covariates, treatment with TCH, T1-2, and ER-negative and PR-negative were associated with higher odds of achieving a pCR [odds ratio (OR), 2.26; 95% CI, 1.09–4.8; OR, 4.99; 95% CI, 1.63–17.06; and 3.19; 95% CI, 1.46–7.31, respectively] with EC-TH, T3-4, and ER-positive and/or PR-positive status used as a reference (Table 2).
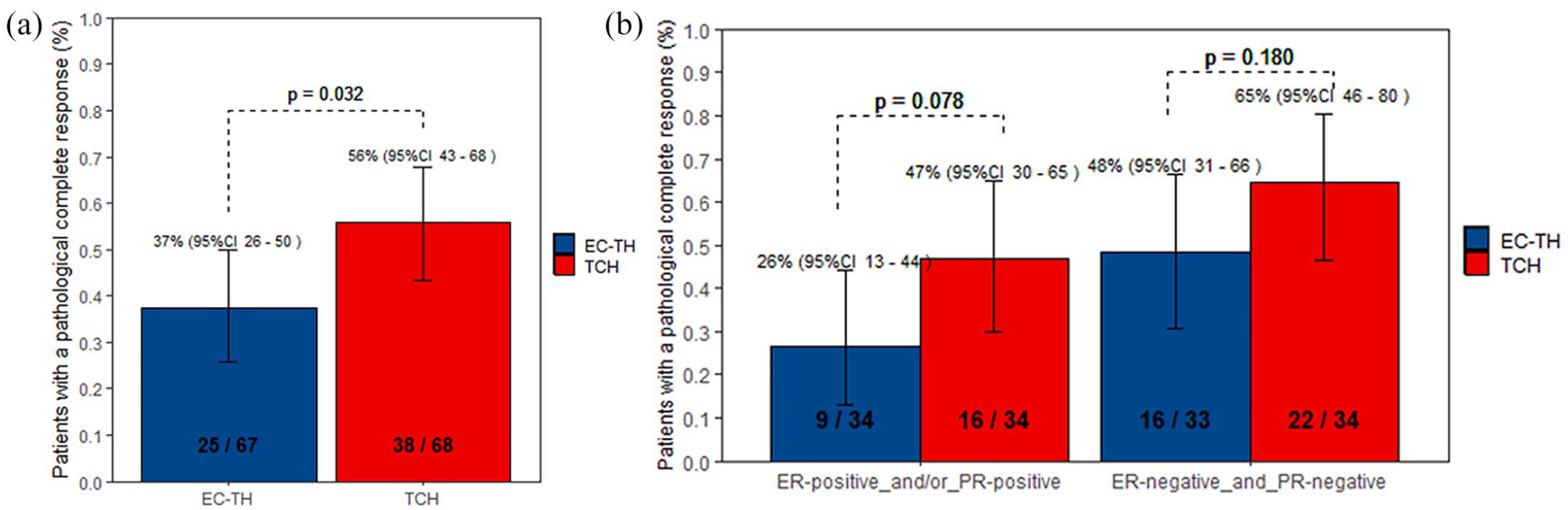
Pathological complete response in different groups. (a) Pathological complete responses according to treatment group. (b) Pathological complete responses according to treatment group and by hormone receptor status.

Pathological complete responses by subgroup.
Multivariate logistic regression analyses.

CI, confidence interval; EC-TH, epirubicin/cyclophosphamide followed by docetaxel/trastuzumab; ER, estrogen receptor; HR, hormone receptor; OR, odds ratio; PR, progesterone receptor; TCH, docetaxel/carboplatin/trastuzumab.
Clinical efficacy was determined by breast ultrasound and revealed that the patients in the TCH group had a higher response rate after four cycles of neoadjuvant chemotherapy than those the EC-TH group (Table 3). At surgery, objective responses were achieved in 59 of 66 patients in the EC-TH group (89.4%, 95% CI, 79.4–95.6) and 64 of 67 patients in the TCH group (95.5%, 95% CI, 87.5–99.1). In patients with SD or PD after four cycles of chemotherapy, the pCR rate was similar between the two groups (37.9% versus 33.3%, p = 1.0; Table 3).
Clinical and pathological response.

CR, complete response; EC-TH, epirubicin/cyclophosphamide followed by docetaxel/trastuzumab; PD, progressive disease; pCR, pathological complete response; PR, partial response; SD, stable disease; TCH, docetaxel/carboplatin/trastuzumab.
Local treatment
Of 135 patients, surgery was performed in 133 patients (66 in the EC-TH group and 67 in the TCH group) as two patients refused surgery. In the EC-TH group, 15 of 66 patients (22.7%) underwent breast-conserving surgery. In the TCH group, 23 of 67 patients (34.3%) underwent breast-conserving surgery. There was no difference in the proportions of patients undergoing breast-conserving surgery between the two treatment groups (p = 0.139). In the EC-TH group, 35 of 66 (53.0%) patients underwent axillary lymph node dissection. In the TCH group, 28 of 67 (41.8%) patients underwent axillary lymph node dissection. There was no difference in the proportions of patients undergoing axillary lymph node dissection between the two treatment groups (p = 0.194).
Adverse events
Most of the relevant toxicities reported during neoadjuvant treatment are summarized in Table 4. The most common adverse events (AEs) were neutropenia [24 of 67 (35.8%) patients in the EC-TH group versus 27 of 68 (39.7%) in the TCH group, p = 0.642], anemia [33 of 67 patients (49.3%) in the EC-TH group versus 34 of 68 (50.0%) in the TCH group, p = 0.931]. Most AEs were of grades 1–2. Nearly all of the most frequent AEs were deemed possibly related to study treatment. Incidences in the most common AEs were similar between the groups. There was a higher incidence of thrombocytopenia in the TCH group than in the EC-TH group [17 of 68 (25.0%) versus five of 67 (7.5%), p = 0.006]; most were grade 1 or 2, while four of 68 patients (5.9%) in the TCH group had grade 3–4 AEs. During neoadjuvant treatment, no symptomatic left ventricular systolic dysfunction was observed. LVEF decreased according to the commonly reported (non-CTCAE) definition of LVEF decline of 10% or more and LVEF below 50% was observed in three of 67 (4.5%) patients in the EC-TH group versus three of 68 (4.4%) patients in the TCH group (p = 1.0). One patient switched from TCH to TH after three cycles because of grade 3 thrombocytopenia.
Adverse events.

EC-TH, epirubicin/cyclophosphamide followed by docetaxel/trastuzumab; TCH, docetaxel/carboplatin/trastuzumab.
Discussion
This is the first, prospective, randomized, multicenter phase II trial comparing EC-TH and TCH regimens as neoadjuvant therapy for HER2-positive breast cancer. There was a higher pCR rate in the TCH arm than in EC-TH arm. Several studies directly compared anthracycline-containing regimens with anthracycline-free regimens. In the presence of dual HER2 blockade, both the TRYPHAENA study and TRAIN2 study did not record a significant increase in the proportion of patients achieving a pCR when treated with anthracyclines.20,25,26 The TRYPHAENA25,26 study assessed the tolerability and activity associated with trastuzumab and pertuzumab in combination with anthracycline- or carboplatin-based neoadjuvant systemic chemotherapy in patients with HER2-positive primary breast cancer. The pCR (ypT0/is ypN0) rates did not differ according to anthracycline use (notably, 63.6% for docetaxel/carboplatin/trastuzumab/pertuzumab versus 56.2% for fluorouracil/epirubicin/cyclophosphamide/trastuzumab/pertuzumab followed by docetaxel/trastuzumab/pertuzumab, versus 54.7% for fluorouracil/epirubicin/cyclophosphamide followed by docetaxel/trastuzumab/pertuzumab). Moreover, the TRYPHAENA trial did not detect any survival differences between anthracycline- or carboplatin-based neoadjuvant chemotherapy together with dual HER2 antibody blockade. In the TRAIN2, similarly high proportions of pCR were achieved with anthracyclines (67%) and without anthracyclines (68%), while a higher incidence of febrile neutropenia was recorded in the anthracycline group. 20 In the presence of a single HER2 blockade, TCH showed similar DFS and OS outcomes but resulted in reduced acute and long-term toxicity compared with AC-TH in the adjuvant BCIRG-006 trial. 23 In the neoadjuvant GeparQuattro Study, 22 patients with operable or locally advanced HER2-positive tumors were treated with four cycles of epirubicin/cyclophosphamide followed by four cycles of docetaxel with or without capecitabine [EC-T(X)] and trastuzumab every 3 weeks during all chemotherapy cycles. About 22.9% of patients showed no response after four cycles of EC, and might be considered as chemoresistant. The high pCR rate of 16.6% in this chemoresistant group might signal that this group benefited the most from docetaxel and trastuzumab treatment. Patients in the TCH group were administered docetaxel and trastuzumab as early and at the same dosage as those in the EC-TH group, yet the proportion of patients who achieved pCR was higher in the TCH arm than in the EC-TH arm. The subgroup analyses of pCR by treatment found that the pCR rate was higher in the TCH group than in the EC-TH group among the patients in the lymph node-positive tumors subgroup. This result suggested that patients with lymph node-positive tumors need upfront and high dose of docetaxel and trastuzumab like those used in the TCH regimen. However, the result is inconsistent with the apparent lesser benefit in stage III disease and may be a chance finding due to small sample size. In summary, with single HER2-blockade, the TCH regimen might be the preferred neoadjuvant therapy in patients with stage II HER2-positive breast cancer.
The 55.9% of pCR rate in patients treated with TCH in our study was higher than those shown in previous studies employing trastuzumab in combination with docetaxel and carboplatin.21,27,28 In the GETN(A)-1 trial, 27 all 70 patients had T2/T3 (100%) tumors or clinical N1/2 nodes (53%), and their HER2 was tested only by immunohistochemistry. In the GETN(A)-1 trial, the pCR rate was 39% in intention-to-treat patients and 43% in retrospective centralized HER2 3+/HER2 FISH-positive tumor patients. Meanwhile in the TRAIN and Huang et al. studies,21,28 about 41% of patients were T3–4, and 80% were clinical lymph node-positive. The high pCR rate in our study without dual blockade can be explained by the fact that we included patients with less advanced clinical tumor–node–metastasis stage. The 55.9% pCR rate in our study in patients treated with TCH was lower than those shown in other studies employing dual HER2-blockade in combination with docetaxel and carboplatin.19,20,26 In the KRISTINE trial, only 38% of patients were ER-negative and PR-negative, and 17% of patients were IIIB–IIIC. pCR was achieved in 123 of 221 (55.7%) patients in the docetaxel, carboplatin, and trastuzumab plus pertuzumab group. 19 In the TRAIN-2 study, the pCR rate was 68% of patients in the non-anthracycline group who were treated with nine cycles of paclitaxel, carboplatin, and trastuzumab plus pertuzumab. 20 The APT study assessed trastuzumab plus weekly paclitaxel only in node-negative patients with mainly stage I disease (91%) and reported a three-year recurrence-free survival rate of 99%. 29 Thus, the challenge remains to define the omission of anthracyclines and the best combinations of anti-HER2 drugs. In patients with stage I disease, TH is an excellent treatment regimen; in patients with stage II disease without lymph node metastases, the addition of carboplatin seems warranted; meanwhile, in most stage II patients and those with stage III disease, the dual HER2-blockade in combination with docetaxel and carboplatin is recommended. Among patients with HER2-positive early breast cancer and residual invasive disease after completion of neoadjuvant chemotherapy plus HER2-targeted therapy, adjuvant treatment with Trastuzumab emtansine (T-DM1) resulted in a 50% lower risk of recurrence of invasive disease or death than adjuvant continuation of trastuzumab. 6 However, dual HER2-blockade is not universally accessible, and challenge remains to define the best combinations of anti-HER2 drugs and the clinical outcomes of omitting anthracyclines to avoid the long-term cardiotoxicity. Both our study and the BCIRG006 trial assessed the omission of anthracyclines and BCIRG006 trial showed survival benefit of the non-anthracycline TCH regimen. 23 Future trials should also be designed to address de-escalation in pCR patients.
Despite the high pCR rate, the TCH neoadjuvant chemotherapy did not result in a significantly higher rate of breast-conserving surgery (EC-TH versus TCH, 22.7% versus 34.3%, respectively). Breast conservation depends on several parameters, such as breast size, tumor location, presence of ductal carcinoma in situ, contraindication to radiation therapy, and patient willingness. Therefore, these differences in breast-conserving surgery between the two groups cannot be attributed only to differences in treatment activity.
In the TRAIN-study, bone-marrow toxicity was frequently seen, neutropenia grade 3 occurred in 49% of patients, and grade 4 occurred in 19% of patients. Less than 5% (5/109) of patients experienced febrile neutropenia. Other grade 3–4 bone-marrow toxicities were thrombocytopenia (43%) and anemia (15%). 21 In our study, the most common AEs were neutropenia [24 of 67 (35.8%) patients in the EC-TH group versus 27 of 68 (39.7%) in the TCH group, p = 0.642] and anemia [33 of 67 (49.3%) patients in the EC-TH group versus 34 of 68 (50.0%) in the TCH group, p = 0.931]. Most AEs were of grades 1–2. Incidences of most of the common AEs were similar between the groups. While there was a higher incidence of thrombocytopenia in the TCH group than in the EC-TH group [17 of 68 (25.0%) versus 5 of 67 (7.5%), p = 0.006]; most events were grade 1 or 2, while four of 68 patients (5.9%) in the TCH group had grade 3–4 events. Granulocyte-colony stimulating factor support was also used in the study and improved tolerability. The cardiac safety profile was excellent in both groups with no reported systolic dysfunction or symptomatic LVEF decrease during the study. LVEF decrease according to the commonly reported (non-CTCAE) definition of LVEF decline of 10% or more and LVEF below 50% was observed in three of 67 (4.5%) patients in the EC-TH group versus three of 68 patients (4.4%) in the TCH group (p = 1.0). However, the long-term clinical consequence of low-grade cardiac toxicity is not well known and warrants further study.
The neoCARH study has several strengths and weaknesses. The neoCARH is thus far the first multicenter prospective randomized phase II trial to address the question of an optimal chemotherapy backbone for single targeted HER2 therapy in HER2-positive breast cancer. The study used two standard-type neoadjuvant chemotherapies. However, long-term efficacy and safety data are not yet available. Although dual blockade HER2-based neoadjuvant chemotherapy is associated with excellent outcomes for HER2-positive breast cancer, the high cost of pertuzumab makes it unavailable to many patients globally. Researchers nowadays aim at investigating in which patient cohorts treatment escalation might be the preferred option and in which settings de-escalation could be appropriate. Other limitations of our study include the absence of a central pathology review of HER2 status and hormone receptor status as well as the lack of quality-of-life endpoints. Due to the lack of sufficient follow-up for most patients, results on event-free survival are not shown.
Conclusions
In conclusion, we found a similar incidence of AEs but a higher pCR rate in the TCH arm compared with the EC-TH arm. In the single HER2-blockade, a TCH regimen might be the preferred treatment regimen in patients with stage II-IIIC HER2-positive breast cancer. However, this is valid only for counties where pertuzumab is not available. Long-term follow-up is required to confirm these results.
Footnotes
Acknowledgements
The authors thank all of the patients analysed in this study. The funding source had no role in this study. All of the authors have final responsibility for the decision to submit this manuscript for publication.
Author contributions
Availability of data and materials
Due to Informed Consent Form, data privacy, and Intellectual Property Rights-related restrictions, the clinical data cannot be made public, that is, accessible for anyone, for any purpose without a review process and without putting an agreement in place. Nevertheless, raw data are available upon request and any requests can be directed to the neoCARH team.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics statement
The study was sponsored by Guangdong Provincial People’s Hospital and was approved by Ethics Committee of Guangdong Provincial People’s Hospital [no. GDREC 2016423H(R1)], together with the ethics committees at each participating institution.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by grants from Science and Technology Planning Project of Guangzhou City (202002030236), Science and Technology Special Fund of Guangdong Provincial People’s Hospital (No. Y012018218), National Natural Science Foundation of China (grant number 81871513), CSCO-Hengrui Cancer Research Fund (Y-HR2016-067), Guangdong Provincial Department of Education Characteristic Innovation Project (2015KTSCX080), Guangdong Basic and Applied Basic Research Foundation (grant number 2020A1515010346) and Guangdong Medical Science and Technology Research Fund (grant number A2019494, A2019252). Funding sources had no involvement in the study design, data collection, analysis and interpretation, writing of the report, and decision regarding article submission for publication.
Informed consent
All patients provided written informed consent before study entry.
