Abstract
Objective:
To describe long-term outcomes of patients treated with nivolumab for advanced non-small cell lung cancer (aNSCLC) in everyday clinical practice in France, with a focus on patients aged ⩾80 years, patients with renal impairment and patients with brain metastases.
Methods:
The study included all patients with aNSCLC recorded in the French national hospital database, starting nivolumab in 2015–2016 and followed until December 2018. Patients were stratified by age, the presence of renal impairment and brain metastasis, as documented in the hospital discharge summaries. Information was retrieved on demographics, comorbidities and treatment history at baseline. Time to discontinuation of nivolumab treatment and overall survival were estimated using Kaplan–Meier survival analysis.
Results:
Overall, 10,452 patients were included, of whom 514 were octogenarians, 479 had renal impairment and 1800 had brain metastases at baseline. Median duration of nivolumab treatment was 2.8 months in the overall population and in both the octogenarian and renally impaired subgroups, and 2.3 months in patients with brain metastases. Median overall survival in these patient groups was 11.7 months (95% confidence interval: 11.3–12.2), 11.7 months (11.3–12.1), 11.7 months (11.3–12.2) and 9.9 months (9.0–10.9) respectively. Three-year overall survival rates were 19.1% (18.1–20.2) in the overall population, 16.5% (11.6–23.4) in octogenarians, 15.9% (11.8–21.4) in patients with renal impairment and 21.7% (19.4–24.2) in those with brain metastases.
Conclusion:
This large nationwide retrospective real-life cohort provided narrow estimates of long-term overall survival, which reached 19% at 3 years, consistent with data from phase III trials of nivolumab. Survival rates were comparable in the three special populations of interest and the overall population.
Introduction
Over the past 5 years, immune checkpoint inhibitors (ICI), such as nivolumab, atezolizumab and pembrolizumab, have become the standard of care for second-line treatment of advanced non-small cell lung cancer (NSCLC).1,2 These treatments offer substantially improved survival in pretreated patients compared with standard chemotherapy.3,4 The anti-PD-1 monoclonal antibody nivolumab is approved in Europe for the treatment of patients with locally advanced or metastatic NSCLC after prior chemotherapy. This ICI has demonstrated its efficacy at extending overall survival (OS) in patients with locally advanced or metastatic NSCLC after chemotherapy in large randomised studies in patients with advanced non-squamous (CheckMate057) and squamous (CheckMate017) NSCLC.5,6 In the long-term extensions of these studies, the 4-year OS rate with nivolumab was 14%. 7 The treatment benefit observed in randomised clinical trials has been replicated in real-world observational studies in everyday clinical practice (recently reviewed by Barlesi et al.). 8 These include the UNIVOC study in France, 9 a large cohort of >10,000 patients, corresponding to all patients with advanced NSCLC (aNSCLC) starting nivolumab treatment in the 2 years following the date when it was made available in France (2015).
The patients in the UNIVOC cohort were identified in the French National Health Data System (SNDS; Système National des Données de Santé), 10 which provides extensive data on healthcare resource use by all beneficiaries of the obligatory public health insurance system. Since patients remain in the database throughout their lives, the UNIVOC cohort provides an opportunity to determine long-term treatment outcomes in a large number of patients. As stated previously, the availability of long-term survival data is important to address some of the unanswered questions about the optimal use of ICIs in the management of aNSCLC. 11
The availability of this large cohort also enables outcomes to be evaluated in subgroups of patients who have not been widely evaluated in clinical trials or smaller observational studies, either because they were excluded or because they represent a small minority of treated patients. For example, the median age of patients enrolled in clinical trials of nivolumab is 10 years lower than that of patients with aNSCLC seen in clinical practice, 12 and very few patients aged >75 years were enrolled. However, since around 10% of patients newly diagnosed with NSCLC in France are octogenarians, 13 it is important to evaluate the effectiveness and tolerability in this population. In addition, patients with organ impairment were excluded from the pivotal clinical trials. 12 Patients with comorbid renal impairment represent 4% of the patients with aNSCLC and pose a particular challenge for management, 14 firstly since it appears to be associated with increased mortality,15,16 and secondly because certain standard chemotherapeutic agents for aNSCLC, such as cisplatin and pemetrexed, are not recommended in these patients.17,18 Finally, patients with brain metastases are another important group in which to evaluate the effectiveness of nivolumab. Such metastases are frequent in aNSCLC, occurring in around 30–40% of patients and are associated with poor prognosis and high morbidity.19,20 Immune checkpoint inhibitors may be beneficial in patients with brain metastases as they do not need to reach the tumour to exert their effect, since they act by priming cytotoxic T cells in the periphery.21–23
The objective of this study was to describe long-term outcomes of patients treated with nivolumab for aNSCLC in everyday clinical practice in France, using the SNDS database, for the overall population and for three specific populations, namely those aged ⩾80 years, those with renal impairment and those with brain metastases.
Methods
This retrospective observational study was performed using data from the hospital discharge database (PMSI; Programme de Médicalisation des Systèmes d’Information) of the SNDS. 10 This database contains comprehensive data on healthcare resource consumption since 1 January 2011. The study included all patients hospitalised with lung cancer in France who initiated treatment with nivolumab between 1 January 2015 and 31 December 2016. The rules used for extraction of patient data have been described previously and are summarised briefly below. 9
Identification of patients and data extraction
Patients with a diagnosis of lung cancer were identified through an ICD-10 (International Classification of Diseases 10th Version) code for lung cancer (C34*) on the hospitalisation discharge summary. Prescription of nivolumab was identified from a list of anticancer drugs dispensed in hospitals. The date of the first treatment with nivolumab was taken as the index date. Data on hospitalisations between the opening of the database (1 January 2011) and 31 December 2018 were extracted. Information in hospital discharge summaries between the start of the database and the index date was used to document cancer history and comorbidities. Information collected throughout the period between the index date and 31 December 2018 was used to document the duration of nivolumab treatment and survival. This prospective follow-up period could thus range from 24 to 48 months, depending on the index date. The data collection periods are summarised in Figure 1.

Study periods.
Derived variables
The time since diagnosis of NSCLC was defined by the proxy variable of the interval between the first hospitalisation for NSCLC and the index date, as the precise date of diagnosis is not documented in the SNDS. A proxy variable of previous treatment with bevacizumab or pemetrexed was used to identify the histological type of NSCLC (non-squamous or squamous), since these are prescribed only for treatment of non-squamous disease. The presence of brain metastases at or prior to the index date was documented from the ICD-10 code C793 on the hospital discharge summary. Similarly, renal impairment was identified from relevant ICD-10 codes (N17 or N18) on hospital discharge summaries prior to the index date. The duration of treatment with nivolumab was defined as the interval between the index date and discontinuation, defined as previously described (no new treatment for at least 6 weeks after the previous treatment or death). 9 Deaths during hospital stays were identified; these account for over 80% of deaths from NSCLC in France. 24 OS was defined as the time from the start of treatment until in-hospital death from any cause or the last observation.
Statistical analysis
Data presentation is principally descriptive. Patient characteristics were compared between subgroups of interest using the χ² test for categorical variables or the Wilcoxon test for continuous variables. Time to treatment discontinuation (TTD) and OS rates were determined from Kaplan–Meier actuarial survival curves. Survival was compared between subgroups of interest using the log rank statistic. Statistical analyses were performed using R-3.6.1 software (Free Software Foundation, Boston, MA, USA).
Ethics
The study was conducted in accordance with International Society for Pharmacoepidemiology (ISPE) Guidelines for Good Pharmacoepidemiology Practices (GPP) and applicable regulatory requirements. Since this was a retrospective study of an anonymised database and had no influence on patient care, ethics committee approval was not required. The study was performed according to the MR006 guideline of the French data protection agency (Commission Nationale de l’Informatique et des Libertés; CNIL) with respect to the confidentiality of individual patient data.
Results
Study populations
Overall, 10,452 patients hospitalised for lung cancer who initiated treatment with nivolumab between 1 January 2015 and 31 December 2016 were included. Of these, 514 were aged ⩾80 years, 479 had renal impairment and 1800 had brain metastases (Figure 2). The characteristics of these patients at the time of nivolumab initiation are presented in Table 1. Compared with patients aged <80 years, older patients were more frequently men, had a longer median time since first hospitalisation for lung cancer and presented more frequently with squamous aNSCLC, but less frequently with brain metastases. Patients with renal impairment were older and comorbid hypertension and more frequently men compared with patients without renal impairment. Patients with renal impairment also were more frequently undernourished, more frequently had hypertension or diabetes, but less frequently comorbid pulmonary disease. In contrast, patients with brain metastases were younger and more frequently women compared with patients without brain metastases. Patients with brain metastases more frequently presented with non-squamous aNSCLC and were more frequently undernourished.

Study populations of interest.
Patient characteristics at nivolumab initiation.

Time since first hospitalisation with cancer diagnosis.
Comorbidities are those identified within 1 year prior to patient inclusion.
Bold type indicates a significant difference (p < 0.005) between the pairs of subgroups (e.g. renal impairment versus no renal impairment).
COPD, chronic obstructive pulmonary disease; IQR, interquartile range.
Treatment outcomes
Treatment outcomes are presented in Table 2. The median time to nivolumab treatment discontinuation was 2.8 months. This was identical in patients aged ⩾80 years, in those aged <80 years and in patients with or without renal impairment. However, in patients with brain metastases, the median time was significantly shorter (2.3 months) than in those without brain metastases (p < 0.001).
Outcomes following initiation of nivolumab treatment.
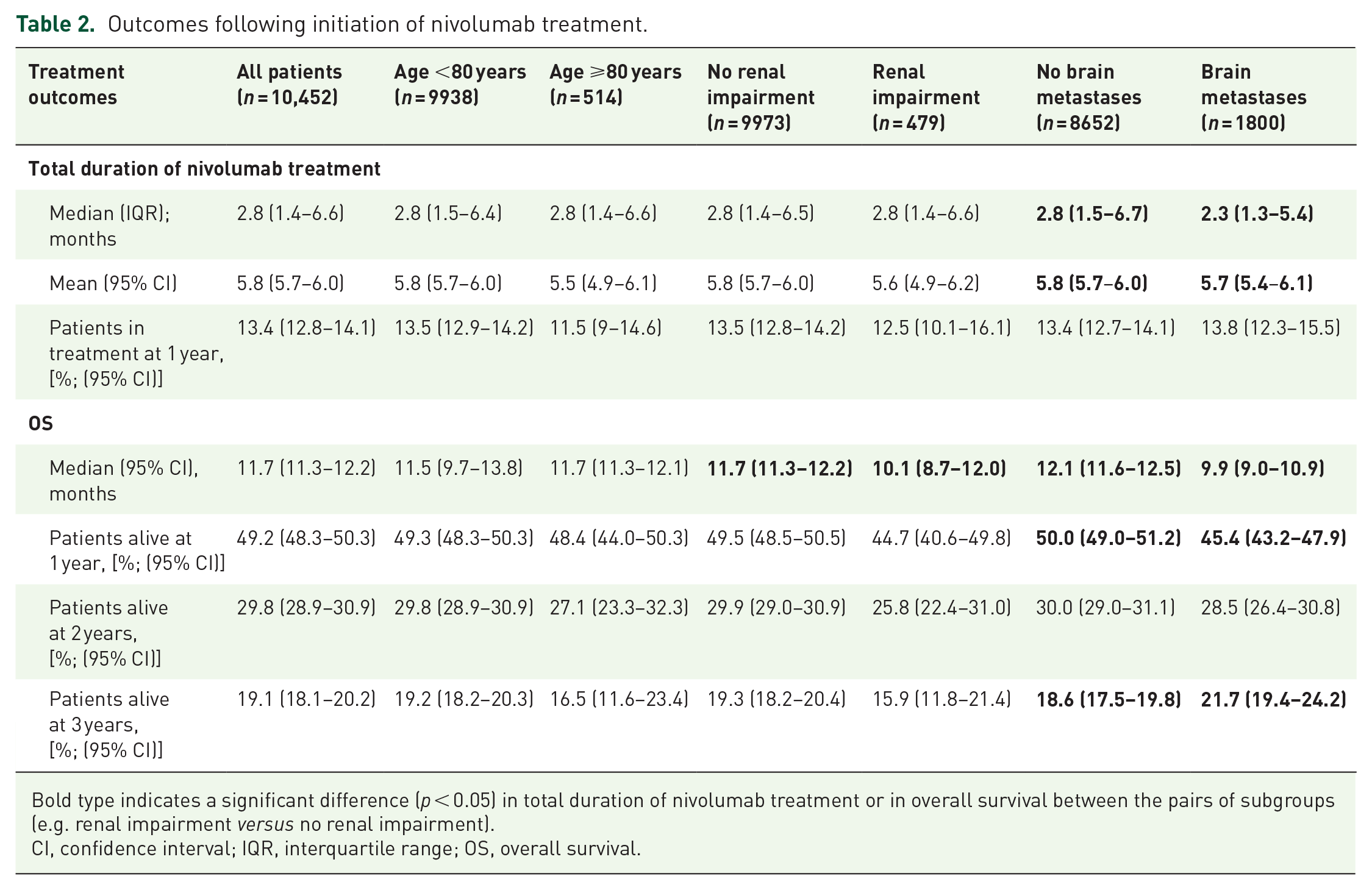
Bold type indicates a significant difference (p < 0.05) in total duration of nivolumab treatment or in overall survival between the pairs of subgroups (e.g. renal impairment versus no renal impairment).
CI, confidence interval; IQR, interquartile range; OS, overall survival.
In the total study population, median OS was 11.7 months; 30% of patients were still alive at 24 months and 19% at 36 months (Figure 3). OS was essentially similar in patients aged ⩾80 years and in younger patients (Figure 4A). In contrast, median OS was somewhat lower in patients with renal impairment (10.1 months; p = 0.03; Figure 4B) and in patients with brain metastases (9.9 months; p < 0.0001; Figure 4C).

OS in the study population. Data are presented as Kaplan–Meier survival curves with the 95% CIs of the survival estimates. The percentages below the curve represent survival rates at 12, 24 and 36 months.
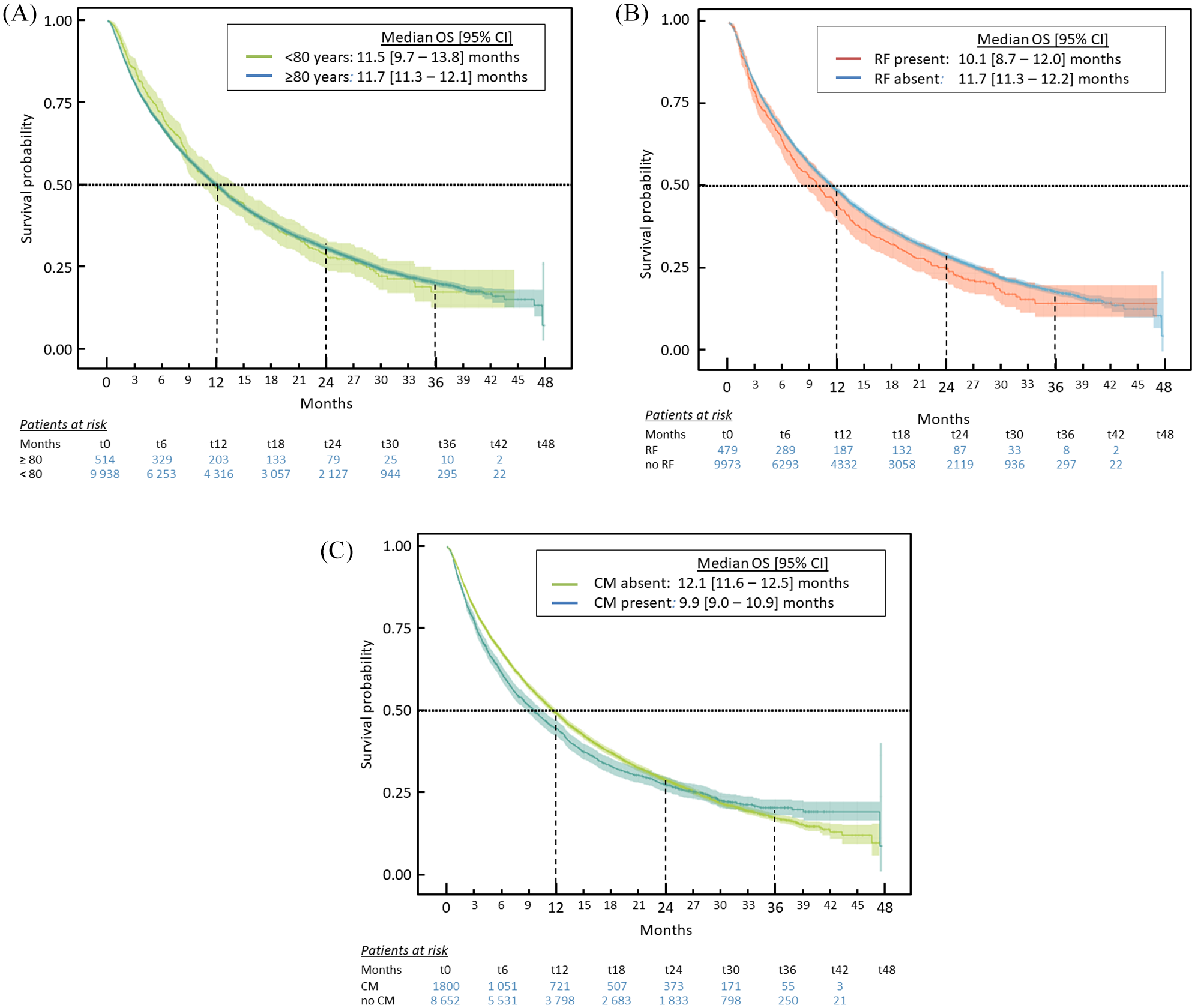
OS in subgroups of interest. (A) Older age. (B) RI. (C) BM. Data are presented as Kaplan–Meier survival curves with the 95% CIs of the survival estimates.
Discussion
OS in this large (>10,000) cohort of patients with aNSCLC treated with nivolumab following prior chemotherapy in clinical practice in France was consistent with long-term survival reported from a pooled analysis of data from four clinical studies of nivolumab in the same indication, including the two pivotal trials CheckMate 017 and 057. 7 In the present analysis of the UNIVOC cohort, median survival was 11.7 months and the survival rate at 36 months was 19%. These data are similar to those from long-term extensions of CheckMate 017 and 057, showing a median survival of 11.1 months and a 36-month survival rate of 17%. 7
With respect to older age, the numbers of patients aged over 75 years in the CheckMate 017 and 057 studies was very low (31 out of a total of 427 patients: 7%). Whether ICIs are beneficial in older patients with aNSCLC is an important question since effectiveness may be compromised by immunosenescence,25,26 which may develop from the age of 65 years. 27 A number of specific studies have investigated nivolumab in patients over 70 years of age. For example, Felip et al. 28 evaluated 811 patients with squamous aNSCLC, of whom 278 were aged ⩾70 years and 125 were aged ⩾75 years. Median OS was essentially similar irrespective of age: 10.0 months in all 811 treated patients, 10.0 months in patients aged ⩾70 years and 11.2 months in those aged ⩾75 years. An Italian cohort study enrolled 371 patients, of whom 175 were aged ⩾65 years and 70 aged ⩾75 years. 29 This study reported that objective response rates and disease control rates were similar among patients aged <65, 65, <75 and ⩾75 years and in the overall study population. However, median OS was reduced in patients aged ⩾75 years (5.8 months) compared with the overall population (7.9 months). 29 More recently, Galli et al. reported similar progression-free survival and OS following ICI treatment in 180 patients aged <70 years, 94 aged 70–79 years and 16 aged ⩾80 years. 30 Finally, an insurance claims database study from the United States (US) evaluated 1256 patients aged ⩾65 years starting nivolumab at a median age of 75 years and reported a similar median OS to that previously described in younger patients, although there was no internal control group in this study. 31 The present UNIVOC study describes one of the oldest cohorts of patients yet studied, aged ⩾80 years, and found no difference in treatment duration or in OS in these patients compared with younger ones. This is consistent with the findings of a Cox analysis of survival data from a prospective study (EVIDENS) of 1420 patients with aNSCLC followed for a median duration of 18 months, in which age ⩾80 years was not associated with poorer survival. 8 Taken together, the available data thus suggest that older patients may derive a benefit similar to younger patients. However, it should be noted that the patients aged ⩾80 years did not have more pulmonary comorbidity, had less cerebral metastases and were no less malnourished than younger patients, which may suggest some patient selection by the physicians, with nivolumab being offered to octogenarians who were relatively healthy. However, as for many other studies conducted to date in this indication, the patients did not have a documented geriatric evaluation to assess vulnerability. To assess the relationship between outcome and vulnerability would be a relevant goal for future research in this field. In this respect, structured evaluation with instruments such as the Comprehensive Geriatric Assessment may be useful.32,33
Treatment with nivolumab has not previously been evaluated in patients with renal impairment and these were explicitly excluded from studies in the clinical development programme. Unlike certain treatment options for patients with aNSCLC, nivolumab is not contraindicated in patients with renal impairment. Although acute immune-related nephritis and renal dysfunction have been reported in patients treated with ICIs used in monotherapy or in association with chemotherapy,34,35 these reactions are uncommon. In the UNIVOC cohort, the time to nivolumab discontinuation did not differ between patients with or without renal impairment, suggesting that there is no major tolerability issue in this population. This would be consistent with data from previous case series of patients with renal disease treated with various ICIs, which reported that these treatments had no obvious negative impact on renal function.36–38 Median OS was somewhat shorter in patients with renal impairment, although this may reflect increased cancer-related mortality in these patients rather than a suboptimal benefit of nivolumab. 15
Concerning patients with brain metastases, 1800 were included in the UNIVOC cohort, which is a large number compared with previous studies. The median treatment duration and median OS were both significantly shorter in patients with these metastases than in patients without. The reason for the reduced time to nivolumab discontinuation cannot be identified from the SNDS database, but could represent more rapid disease progression in patients with brain metastases. Interestingly, the hazard function seemed to vary over time, with the excess mortality in patients with brain metastases accruing principally during the first 18 months of treatment, after which long-term OS appeared similar to the group with no brain metastases. Indeed, at 2 years, the presence of brain metastases did not seem to influence the OS rate significantly [28.5% (26.4–30.8) in patients with brain metastases compared with 30.0% (29.0–31.1) in those without]. Data on nivolumab in patients with brain metastases are limited and somewhat contradictory. 22 A recent Cox analysis of survival data from a study of 67 patients with aNSCLC treated with nivolumab has reported that symptomatic brain metastases were associated with reduced OS in patients treated with nivolumab. 39 Subsequently, a similar association was described in the much larger multicentre, prospective EVIDENS study. 8 On the other hand, a Japanese study reported that the presence of brain metastases, unlike lung or liver metastases, was not associated with reduced OS. 40 Such discrepancies between studies may be explained by a potential impact of corticosteroid treatment in patients with symptomatic lesions, which has been suspected to impair the effectiveness of immunotherapy. 41 The differences between studies may also be attributable to differences in the types of patients included or in the specific treatments that they were receiving for the brain metastases. With regard to the evolution of brain metastases themselves, there is some evidence suggesting that this can be improved by nivolumab treatment.42–44
Several differences in patient characteristics were observed at inclusion. Certain of these are expected, such as the higher frequency of hypertension and diabetes in patients with renal impairment, the high level of use of radiotherapy in patents with brain metastases and the association of renal impairment or brain metastases with age. Others, however, are more noteworthy. For example, the proportion of patients with non-squamous cell disease (adenocarcinoma) was higher in younger patients than in those aged ⩾80 years and in those with brain metastases. The higher proportion of patients with adenocarcinoma in the younger patients may represent a cohort effect, associated with changes in smoking habits over time (reduction in smoking rates and a move towards low-tar products). 45 Population-based studies have shown an increase over time in the proportion of adenocarcinoma at the expense of squamous cell cancer, 46 which is more specifically related to smoking than adenocarcinoma. 47 The higher proportion of men in patients aged ⩾80 years with lung cancer probably also represents a cohort effect, since smoking became commonplace in women many decades later than in men. 45 The higher proportion of adenocarcinoma in patents with brain metastases is consistent with previous observations that adenocarcinoma is more likely to metastase to the brain than squamous cell cancer. 48 The higher proportion of undernourished patients in the renal impairment group probably reflects the muscle wasting and protein loss that typically occurs in chronic kidney disease. 49 The over-representation of men in the renal impairment group is somewhat surprising given that chronic kidney disease is more common in women than in men, but may reflect the fact that smoking, which is the principal lifestyle risk factor for lung cancer and more common in men, is also a risk factor for chronic kidney disease. 50 The higher proportion of undernourished patients in the brain metastases group is consistent with previous findings that low nutritional status in lung cancer is correlated with the burden of brain metastases. 51
The strengths of this study are the large number of patients enrolled, which makes it possible to evaluate minority subgroups with sufficient power, and the exhaustive inclusion of all patients with aNSCLC treated with nivolumab in France, which obviates any issues of representativeness of the study population. The limitations relate to the absence of information on clinical decision-making and outcome in the database, as well as its retrospective nature. Notably, no data are available in the database on PD-L1 expression, certain comorbidities or concomitant medications that may influence survival, the reasons for treatment discontinuation, disease progression, performance status, outcomes other than death in hospital or the occurrence of immune-related adverse events. In the particular case of the subgroup of patients with renal impairment, this is identified by the ICD-10 code on the hospital discharge summary, but no information is available on the severity through measures of creatinine clearance. There may also be an issue with under-reporting of brain metastases due to incomplete coding. In addition, the date of first cancer diagnosis is defined by a proxy variable and this may introduce some error into the estimates.
In conclusion, this study assessed the effectiveness of nivolumab in potentially vulnerable subgroups of pretreated patients with aNSCLC. In a real-world treatment setting, OS consistent with that observed in interventional trials could be achieved. OS was not compromised in patients aged ⩾80 years and only marginally in patients with renal impairment. In consequence, nivolumab may be offered to patients aged ⩾80 years and should remain an option in patients with renal impairment. Regarding patients with brain metastases, further studies using other types of design are merited to explore the benefits and safety of nivolumab in this patient subgroup.
Footnotes
Acknowledgements
Writing and editorial assistance was provided by SARL Foxymed.
Conflict of interest statement
JBA was supported by grants from Fondation pour la Recherche Médicale (FRM). CC reports consultancy fees from Astra Zeneca, Boehringer Ingelheim, MSD, Pierre Fabre Oncology, Lilly, Roche, Bristol- Myers Squibb, Novartis, Lilly, Pierre Fabre Oncology and Boehringer Ingelheim. RC reports consultancy fees from Astra-Zeneca, Bristol- Myers Squibb, Roche and Takeda. MGL reports consultancy fees/research funding from Bristol-Myers Squibb, Astra Zeneca, MSD, Roche and Novartis. FEC, CYC, and AFG are employed by Bristol Myers Squibb. BJ and RJ are employees of HEVA.
Data availability
The source database (PMSI) contains personal health data with potentially identifying and sensitive patient information. According to French law (Decree N° 2016-1871 dated 28th December 2016, concerning the processing of personal data in the SNDS), PMSI data are available exclusively from the database holder, the CNAMTS (Caisse nationale de l’assurance maladie des travailleurs salariés), to institutions who meet the criteria for access to confidential data, following procurement of consent from the National Health Data Institute (INDS) and the French data protection authority (CNIL). Publication of individual patient data is not permitted. The INDS, which is responsible for access to health data in France, is a one-stop-shop window for access to the SNDS database (including the PMSI database). The contact address for the INDS is Institut National des Données de Santé (INDS), 19 rue Arthur Croquette, 94220 Charenton-le-Pont, Telephone: +33 1 45 18 43 90; Email: ![]() .
.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Bristol Myers Squibb (Princeton, NJ) and ONO Pharmaceutical Company Ltd. (Osaka, Japan).
