Abstract
Background:
Plasma Epstein–Barr virus (EBV) DNA has been identified as a significant prognostic marker for nasopharyngeal carcinoma (NPC), yet there is limited research on the prognosis of NPC patients with negative EBV DNA.
Objectives:
We explore the prognostic value of comprehensive immune-inflammatory and nutritional indicators to offer personalized treatment recommendations and prognosis predictions for non-metastatic NPC patients with negative EBV DNA.
Design:
This was a retrospective study.
Methods:
This study retrospectively analyzed 257 non-metastatic NPC patients with negative EBV DNA between January 2015 and December 2019. The Kaplan–Meier survival curves evaluated survival endpoints, and group discrepancies were assessed with log-rank tests. Principal component analysis (PCA) reduced data dimensionality. Univariate and multivariate Cox regression analyses identified significant prognostic variables. Risk stratification was performed based on recursive partitioning analysis (RPA). A robust prognostic model was constructed by nomogram and evaluated by calibration curves, decision curves, and the time-dependent area under the curve analysis.
Results:
PCA was employed to compute the immune-inflammation index (III) and nutrition index (NI). Multivariate Cox regression analysis revealed lactate dehydrogenase, III, and NI as significant prognostic variables for overall survival (OS). Utilizing RPA, we stratified the risk into three categories: low-risk group (low III + high NI), middle-risk group (low III + low NI), and high-risk group (high III). Both the middle- (p = 0.025) and high-risk groups (p < 0.001) exhibited poorer OS compared with the low-risk group. The nomogram model exhibited superior predictive accuracy compared to tumor lymph node metastasis stage alone (C-index: 0.774 vs 0.679).
Conclusion:
Our study validated the prognostic significance of III and NI in non-metastatic NPC patients with negative EBV DNA. Additionally, a clinical risk stratification was constructed to offer valuable insights into the individualized treatment of these patients.
Plain language summary
Keywords
Introduction
In the head and neck region, a common malignant tumor called nasopharyngeal carcinoma (NPC) is frequently seen. This type of cancer exhibits distinctive geographical distribution patterns, primarily occurring in southern China, North Africa, and Southeast Asia. Based on the 2020 GLOB0CAN data, the global number of new NPC cases exceeded 130,000, and China contributed to about 46.8% of these cases. Therefore, the incidence of NPC in China was relatively high.1–3 The use of radiotherapy is crucial in the management of NPC. Clinical practice widely employs the tumor lymph node metastasis (TNM) staging system to assess the prognosis of NPC patients. Heterogeneity can cause survival rates to differ among patients receiving similar treatment regimens, despite having identical clinical staging. Nonetheless, the system based on anatomical staging fails to accurately forecast patient prognoses or treatment outcomes. So, precisely forecasting NPC patient outcomes and directing tailored treatment remains a challenge.
Tumor markers are crucial in the early detection of cancer and in monitoring treatment efficacy. There is a strong correlation between NPC and the infection by Epstein–Barr virus (EBV). So far, plasma EBV DNA has been proved as a dependable indicator of NPC, playing an important role in its detection, prompt diagnosis, and prognosis prediction, as well as serving as a valuable supplement to TNM staging.4–6 However, a proportion of NPC patients have negative EBV DNA, and there is a limited body of research on prognostic markers for these patients. Consequently, it is critically important to pinpoint predictive indicators for NPC patients who have negative EBV DNA.
Not only does inflammation contribute to cancer progression, but it also propels every stage of tumor formation. 7 A variety of markers related to immune inflammation have been identified as reliable indicators for predicting the outcome of NPC patients.8–10 Cancer patients often exhibit signs of malnutrition. During the period of intensity-modulated radiotherapy (IMRT), malnutrition is often correlated with unfavorable survival outcomes in NPC patients.11–14 Therefore, timely nutritional intervention can improve not only the survival outcome and quality of life but also reduce treatment-related toxicity.15–17 An interesting study revealed that inflammation is a key factor in malnutrition, and nutrition affects inflammation as well, and the relationship between the two is intricate. 18 Recent researches suggest that a mix of immune-inflammatory and nutritional markers is important for predicting survival outcomes in patients with NPC.19–22 Nevertheless, the predictive significance of comprehensive immune-inflammatory and nutritional markers in non-metastasis NPC patients with negative EBV DNA is still ambiguous.
Therefore, the study sought to examine the predictive significance of comprehensive immune-inflammatory and nutritional marks in non-metastatic NPC patients with undetectable EBV DNA, providing recommendations for personalized therapy.
Materials and methods
Patients and data collection
We retrospectively included 257 patients diagnosed with non-metastatic NPC and tested negative for EBV DNA at the Fujian Cancer Hospital from January 2015 to December 2019. Patients were categorized according to their EBV DNA levels, and the threshold for distinguishing negative and positive categories was set at 0 copy/mL in accordance with the criteria established by Fujian Cancer Hospital’s Laboratory Medicine Center for this study. 23 Criteria for inclusion included: (1) patients with histopathologically confirmed NPC; (2) patients with complete medical records; (3) patients who underwent complete radical radiotherapy; (4) before treatment, patients should have completed a peripheral blood count; (5) patients testing negative for EBV DNA in each of the following three phases: pre-treatment, post-induction chemotherapy (IC), or pre-radiotherapy and post-radiotherapy. Criteria for exclusion included: (1) patients diagnosed with metastasis initially; (2) patients testing positive for EBV DNA; (3) patients having concurrent tumors other than NPC; (4) patients with prior anti-cancer treatments; (5) patients with pathological types not covered by World Health Organization (WHO); and (6) patients who were untraceable during follow-up. Fujian Cancer Hospital’s Ethics Committee examined and sanctioned the study (ID: K2024-289-01). Due to the retrospective design, further informed consent from patients relevant to this research was unnecessary. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology Statement 24 (Supplemental File).
Peripheral blood cells from all patients were collected within 1 week prior to therapy and tested for lactate dehydrogenase (LDH), neutrophil counts, lymphocyte counts, monocyte counts, platelet counts, and albumin (ALB). Plasma EBV DNA measurements were taken within 1 week before treatment, after IC or before radiotherapy and after radiotherapy. Information about the patient, including age, gender, tumor stage, lymph nodes stage, total stage, treatment strategies, as well as LDH, neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), systemic immune-inflammation index (SII), systemic inflammation response index (SIRI), pan-immune-inflammation value (PIV), ALB, and prognostic nutritional index (PNI) were collected. The formulas for the immune-inflammatory and nutritional indicators were detailed in Supplemental Material 1.
Evaluation and treatment
Baseline assessment of all patients before treatment encompassed gathering their medical history, conducting physical exams, hematological assessments, nasopharyngoscopy, head and neck magnetic resonance imaging (MRI), chest computed tomography (CT) scans, abdominal ultrasound, whole-body bone emission computed tomography, or positron emission tomography/X-ray CT. The number of EBV-DNA copies per milliliter of plasma was ascertained using reverse transcription-quantitative polymerase chain reaction technique. The imaging-based staging system was restaged by two experienced radiation oncologists according to the 8th edition of the American Joint Committee on Cancer (AJCC)/Union for International Cancer Control (UICC) system.
Treatment was conducted in accordance with the latest guidelines from the National Comprehensive Cancer Network, and every participant in the study was administered radical IMRT. Patients in stage I were treated solely with IMRT, while individuals in stages II–IVa primarily received concurrent chemoradiotherapy (CCRT) optionally combined with IC and/or adjuvant chemotherapy (AC).
Every patient underwent radical IMRT using six megavoltage photons over a period of 6–7 weeks. The total radiation doses given to the primary tumor varied between 67 and 78 Gray, while the involved neck area lymph nodes received doses ranging from 62 to 78 Gray. The guidelines in the RTOG 0225 protocol were used to establish dosage limits for organs at risk. 25 IC cycles usually depended on the clinical stage and chemotherapy tolerance. Platinum-based IC strategies were frequently utilized in conjunction with gemcitabine, paclitaxel, docetaxel plus 5-fluorouracil, or alternative strategies given on a tri-weekly basis. The administration of AC depended on tumor regression.
Follow-up and study endpoints
Patients were followed up every 3 months during the initial 2 years, followed by semi-annual check-ups from the third to the fifth year, and subsequently on an annual basis. Throughout the follow-up period, patients received a physical examination, lab tests, electronic nasopharyngoscope, MRI of head and neck, chest CT, abdominal ultrasound, and whole-body bone scans, among others. Survival and tumor status were recorded through imaging studies or phone consultations, leading to the last follow-up conducted on September 2, 2023. Whenever feasible, pathology was used to verify recurrence or distant metastasis, and in its absence, diagnosis relied on at least two imaging findings. Treatment was tailored for patients experiencing recurrence and/or distant metastasis, while the selection of salvage therapy determined by the clinicians’ decisions and patients’ preferences.
This research primarily focused on overall survival (OS), referring to the duration from the completion of radiation treatment to the final follow-up or occurrence of death without specific cause. The secondary endpoint included local–regional recurrence-free survival (LRRFS), denoting the duration from the end of radiation treatment to the occurrence of local–regional recurrence or the latest follow-up, and distant metastasis-free survival (DMFS), indicating the duration from the end of radiation treatment to the occurrence of distant metastasis or the latest follow-up. Progression-free survival (PFS) was described as the period from the completion of radiation treatment to the occurrence of progression, which included death, recurrence, distant metastasis, or the latest follow-up.
Statistical analysis
The median and interquartile range (IQR) of continuous variables were used, while proportions and whole numbers were used for classification and grade variables. Due to the high number of variables, we wanted to reduce the data dimensionality. If the Kaiser–Meyer–Olkin (KMO) value in the KMO test exceeds 0.5 and the p-value from the Bartlett’s test of sphericity is below 0.05, then principal component analysis (PCA) is deemed appropriate. Retention of principal components (PCs) depended on eigenvalues greater than 1, the scree plot, and cumulative variance contributions. 26 We included six immune-inflammatory indicators as well as two nutritional indicators in the PCA, and by extracting the PCs with eigenvalues greater than one, we obtained two PCs, which were named immune-inflammatory index and nutritional index, respectively.
Survival analysis using Kaplan–Meier was conducted to assess OS among different risk groups, and distinction between groups were analyzed through the log-rank test. Variables showing statistical significance with a p value below 0.05 in the univariate analysis were incorporated into the following multivariate COX regression analysis. Furthermore, variables exhibiting a p value less than 0.05 in the multivariate analysis were pinpointed as independent prognostic indicators for OS. Additionally, the outcomes of the univariate and multivariate COX regressions were displayed through forest plots. Variables that showed statistically significant association in the multivariate analysis were incorporated into an OS risk stratification model employing recursive partitioning analysis (RPA). The RPA was utilized to determine the optimal node in every division, allowing for the discovery of the most advantageous patient groups at each step. 27 The effect of therapeutic regimens on OS across various risk groups based on RPA was examined to provide individualized treatment decisions for these patients. Ultimately, the nomogram model was constructed using independent prognostic variables recognized through univariate COX regression analysis with a p value below 0.05, along with RPA risk groups. Evaluation of the constructed nomogram model involved the utilization of calibration curve, decision curve analysis, and time-dependent area under the curve (time AUC). Statistical significance was established when the p value was less than 0.05 in two sides. The data analysis was conducted utilizing IBM SPSS Statistics software version 25.0 (Armonk, NY: IBM Corp. https://www.spss.com/) and RStudio software version 2022.02.0 (https://www.r-project.org).
Result
Patient characteristics and follow-up
Among the 4173 patients with NPC who visited our center from January 2015 to December 2019, we excluded 3814 patients with positive EBV DNA, 77 patients with distant metastasis, 4 patients lacking complete medical records, 10 patients with prior anti-tumor therapy, 4 patients with non-WHO pathological types, 4 patients with other tumors, and 1 patient did not complete radical radiotherapy. Additionally, two patients were lost to follow-up. A total of 257 non-metastatic NPC patients with negative EBV DNA patients were enrolled in our study. The patient selection flow was shown in Supplemental Figure 1. The baseline characteristics of the patients are presented in Table 1. The median age of the cohort was 48 years old (IQR, 39–55 years). In total, 179 patients (69.65%) were men, while 78 (30.35%) were women. Out of 257 patients, 254 (98.83%) had non-keratinizing carcinoma, while only 3 (1.17%) had keratinizing squamous cell carcinoma. According to the 8th AJCC/UICC staging system, there were 89 (34.65%) in T1, 39 (15.18%) in T2, 86 (33.46%) in T3, and 43 (16.73%) in T4. There were 44 (17.12%) in N0, 121 (47.08%) in N1, 76 (29.57%) in N2, and 16 (6.23%) in N3. Twenty-two (8.56%), 67 (26.07%), 112 (43.58%), and 56 (21.79%) of them showed stage I, II, III, and IVA, respectively. The study’s treatment protocols included: 24 (9.34%) patients received only RT, 47 (18.29%) patients underwent IC followed by RT, 54 (21.01%) patients underwent CCRT, and 132 (51.36%) patients underwent IC combined with CCRT. The median LDH was 144 (IOR, 127–168). The median value of NLR, PLR, MLR, SII, SIRI, PIV, ALB, and PNI were 1.90 (IQR, 1.45–2.54), 117.14 (IQR, 91.97–155.75), 0.20 (IQR, 0.15–0.26), 472.55 (IQR, 337.79–674.63), 0.78 (IQR, 0.52–1.17), 186.21 (IQR, 120.96–296.11), 42.3 (IQR, 39.75–44.70), and 52.7 (IQR, 49.50–55.95).
Characteristics at baseline of non-metastatic NPC patients with negative EBV DNA (n = 257).

ALB, albumin; CCRT, concurrent chemoradiotherapy; EBV, Epstein–Barr virus; IC, induction chemotherapy; IQR, interquartile range; LDH, lactate dehydrogenase; MLR, monocyte-to-lymphocyte ratio; NLR, neutrophil-to-lymphocyte ratio; NPC, nasopharyngeal carcinoma; PIV, pan-immune-inflammation value; PLR, platelet-to lymphocyte ratio; PNI, prognostic nutritional index; RT, radiotherapy; SII, systemic immune-inflammation index; SIRI, systemic inflammation response index.
There was a median follow-up time of 77.0 months (IQR, 51.1–86.0 months) in the study. At the date of the last follow-up day, there were 26 (10.12%) cases of locoregional recurrence, 24 (9.34%) of distant metastasis, and 35 (13.62%) died from any disease. There were 92.22% and 88.72% of OS in the general population at 3 and 5 years, respectively. There were 93.4% and 90.66% of LRRFS at 3 and 5 years, respectively. The DMFS percentage at 3 and 5 years were 92.22% and 91.44%, respectively. The PFS percentage at 3 and 5 years were 87.16% and 84.05%, respectively (Figure 1).
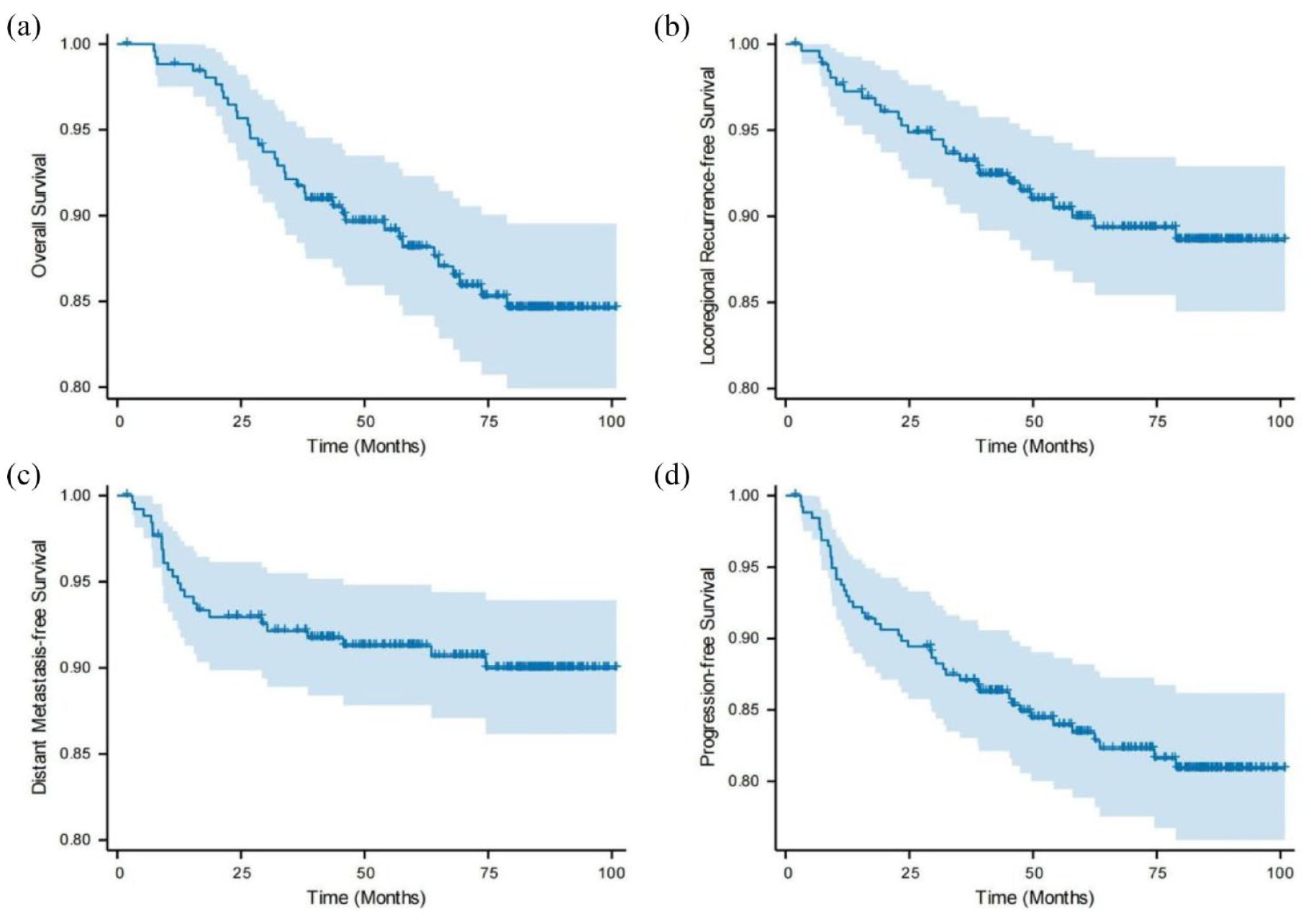
The Kaplan–Meier survival curve of overall survival (a), local–regional recurrence-free survival (b), distant metastasis-free survival (c), progression-free survival (d).
PCA to construct comprehensive immunoinflammatory and nutritional index
To evaluate the immune-inflammatory and nutritional status of patients with NPC in the cohort, we standardized the eight indicators of NLR, PLR, MLR, SII, SIRI, PIV, ALB, and PNI and then applied them to the PCA. The eigenvalues of the first two PCs exceeded 1, with the cumulative variance contribution rate for these PCs at 78.54%, which indicated that these PCs accounted for a significant proportion of the variance in the multi-dimensional immune-inflammatory and nutritional indicators (Supplemental Table 1). The correlation coefficients presented in Table 2 revealed that PC1, had named immune inflammation index (III), predominantly comprised NLR, PLR, MLR, SII, SIRI, and PIV, while PC2, had named nutrition index (NI), consisted primarily of ALB and PNI. (Table 2, Figure 2). The computation formulas for III and NI were as follows:

Rotated component matrix displayed the values of the loadings of each variable on different components.

ALB, albumin; MLR, monocyte-to-lymphocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PIV, pan-immune-inflammation value; PLR, platelet-to lymphocyte ratio; PNI, prognostic nutritional index; SII, systemic immune-inflammation index; SIRI, systemic inflammation response index.

PCA load plot that visualized the relationship between variables and principal components and the contribution value of each principal component.
Identifying independent prognostic factors for OS
The univariate Cox regression analysis revealed significant associations of these variables with OS in the total cohort: age (p = 0.017), tumor stage (p = 0.012), node stage (p = 0.004), total stage (p = 0.007), IC (p = 0.007), LDH (p = 0.035), III (p = 0.002), and NI (p = 0.027) (Figure 3). After univariate analysis, every statistically significant variable was included in the multivariate Cox regression analysis. The multivariate analyses revealed that LDH (hazard ratio (HR): 1.014, 95% confidence interval (CI): 1.003–1.024, p = 0.012), III (HR: 1.174, 95% CI: 1.012–1.362, p = 0.034), and NI (HR: 0.079, 95% CI: 0.525–0.957, p = 0.024) were served as independent predictors worse OS (Figure 4).

Univariate COX regression analysis of OS for non-metastatic NPC patients with negative EBV DNA.

Multivariate COX regression analysis of OS for non-metastatic NPC patients with negative EBV DNA.
RPA model building and comparison OS of different risk groups
To further predict the prognosis of this population, we subjected the independent prognostic factors, LDH, III, and NI to RPA. According to the RPA results, III < 1.3968 was low III, and III ⩾ 1.3968 was high III. Low NI was defined as NI < 0.2567, and high NI was defined as NI ⩾ 0.2567. Finally, non-metastatic NPC with negative EBV DNA was divided into three risk stratification based on RPA analysis, including low-risk group (low III + high NI, n = 79), middle-risk group (low III + low NI, n = 127), and high-risk group (high III, n = 51) (Figure 5(a)).
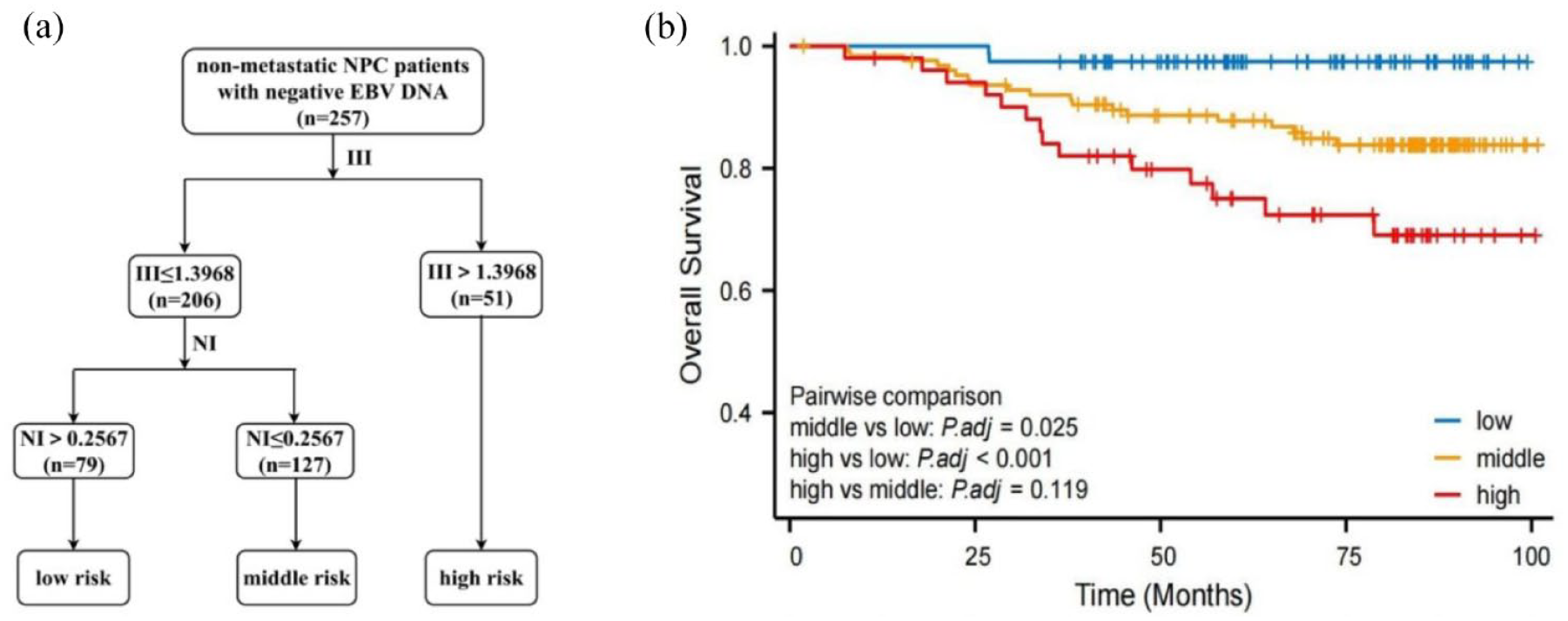
Construction of RPA prognostic risk stratification in non-metastatic NPC patients with negative EBV DNA for overall survival (a). The Kaplan–Meier survival curve of overall survival for the comparison of different risk groups (b).
The risk stratification based on RPA demonstrated a satisfactory prognosis for OS of non-metastatic NPC patients with negative EBV DNA as shown in Figure 5(b) (middle-risk group vs low-risk group: HR: 5.645, 95% CI: 2.331–13.671, p = 0.025; high-risk group vs low-risk group: HR: 11.434, 95% CI: 4.166–31.381, p < 0.001; high-risk group vs middle-risk group: HR: 2.032, 95% CI: 0.939–4.397, p = 0.119).
Relationship between different RPA risk groups and therapy strategies
We investigated the longevity advantages of IC across various risk stratification groups. The results indicated that in the whole cohort, patients treated with IC had a lower OS in contrast to those didn’t treat with IC (IC, YES, vs NO, p = 0.001) (Supplemental Figure 2(A)). The middle-risk group showed a similar result (IC, YES, vs NO, p = 0.044) (Supplemental Figure 2(C)), whereas there was no significant difference between those with IC-YES and those with IC-NO in the low-risk group (p = 0.241) (Supplemental Figure 2(B)), mirroring the findings in the high-risk group (p = 0.087) (Supplemental Figure 2(D)).
Development and evaluation of nomogram model for predicting OS
To better predict the OS of the non-metastatic NPC patients with negative EBV DNA, a nomogram model was developed in Figure 6(a). The nomogram for predicting OS incorporated several prognostic factors, such as age, tumor stage, lymph node stage, total stage, IC, LDH, and RPA group. Points were allocated to each variable to reflecting its prognostic significance. A higher cumulative point score, representing the sum of points for all factors, indicated a poorer prognosis.

Development and evaluation of nomogram model for OS. Nomogram score model (a); the 1-, 3-, and 5-year calibration curves (b); the decision curve (c); the time AUC curves (d).
The calibration curve demonstrated strong predictive performance for the nomogram model, evidenced by a C-index of 0.774 (95% CI: 0.734–0.814) (Figure 6(b)), surpassing the total stage, which had a C-index of 0.679 (95% CI: 0.638–0.720). The decision curve analysis revealed that at a threshold probability greater than 0.05, the nomogram model provided a higher net benefit, signifying its substantial clinical application and superiority over other indicators (Figure 6(c)). In addition, we compared the nomogram model with other prognostic factors for the ability to predict OS. The findings showed that the AUC value of the nomogram model surpassed that of the other indicators at any time point, demonstrating its enhanced capacity to discriminate between survival and death compared to the total stage. It supported the conclusion that the nomogram model more accurately predicted survival outcomes for non-metastatic NPC patients with negative EBV DNA (Figure 6(d)).
Discussion
In this study, we validated the independent prognostic significance of III and NI for OS in non-metastatic NPC patients with negative EBV DNA. Subsequently, based on these indicators, an RPA risk stratification was performed. The results demonstrated that compared with the low-risk group, the high- and middle-risk groups had significantly lower survival rates. Moreover, we observed that both the total cohort and middle-risk group did not benefit from IC, while no significant conclusion could be drawn for the low- and high-risk groups. Finally, a nomogram model was constructed incorporating RPA stratification, TNM stage, and other clinical parameters to further predict patients’ prognosis. This model exhibited superior prognostic performance compared to the TNM stage and other individual factors.
As per the WHO, NPC is classified into three pathological subtypes: keratinizing squamous, non-keratinizing, and basaloid squamous. The keratinizing subtype is a minority, representing less than 1% of cases in endemic regions such as southern China.28,29 Conversely, the non-keratinizing subtype is the most prevalent in endemic areas, accounting for over 95% of cases, and is primarily linked to EBV infection. 1 Hence, early detection of EBV infection can facilitate timely identification and management of NPC. However, the use of serological testing for anti-EBV IgA antibodies, such as early antigen (EA IgA), anti-EBV capsid antigen (VCA IgA), and anti-EBV nuclear antigen 1 (EBNA1 IgA) for the identification of NPC is hindered by low sensitivities and specificities, limiting their effectiveness in screening asymptomatic individuals. 30 The presence of EBV DNA is crucial for the early detection, diagnosis, and prognostication of NPC due to its high sensitivity (97.1%) and specificity (98.6%). 31 Despite the gold standard status of the Epstein-Barr virus encoded small RNA (EBER) in situ hybridization test for detecting EBV infection, this research center did not consistently identify this indicator in the initial stages, leading to a large number of data loss. Consequently, NPC with negative EBV DNA was chosen as the focus of this study. While the majority of research indicates that EBV infection is primarily linked to non-keratinizing NPC rather than keratinizing NPC, a minority of studies have proposed a potential association between EBV infection and keratinizing NPC.32,33 Therefore, we conducted a study on patients diagnosed with non-metastatic NPC who tested negative for EBV DNA, irrespective of their histological types.
The pathogenesis of NPC is closely linked with EBV infection in regions prone to epidemics. 3 Several reports have indicated that patients with negative EBV DNA before treatment exhibit superior survival rates and prognosis compared to those with elevated plasma EBV DNA levels.34–36 However, other studies has indicated that NPC patients with negative EBV DNA exhibited similar survival rates and prognoses to those with positive EBV DNA, 37 yet the predicators of these patients remain rarely explored. The results of our study revealed that both III and NI held predictive significance in NPC patients with negative EBV DNA. Notably, our analysis suggested that the TNM stage did not independently predict the prognostic for NPC patients with negative EBV DNA, which corresponded well with the results demonstrated by Yuan et al. 38 Nonetheless, the study by Yao et al. 39 suggested that N stage independently predicted outcomes for these patients. In this light, there remains controversy regarding the prognostic utility of TNM staging system in predicting OS for NPC patients with negative EBV DNA. Consequently, the TNM stage was included as an indicator in the construction of the nomogram model, despite its lack of statistical significance in the multivariate COX regression analysis.
A retrospective study of 1106 patients diagnosed with non-metastatic advanced NPC revealed that distant metastasis was the predominant cause of treatment failure in patients with positive EBV DNA. The study suggested that neoadjuvant chemotherapy followed by CCRT was the most effective treatment approach for these patients, whereas CCRT alone was deemed adequate for patients with negative EBV DNA. 34 For individuals in stage II NPC presenting undetectable levels of pretreatment EBV DNA, the survival rates achieved through the application of IMRT as the sole treatment modality seemed to be on par with those resulting from CCRT. 40 In conclusion, there is a possibility that patients who have low levels of EBV DNA before treatment might experience advantages from a decreased intensity of chemotherapy. This inference aligns with the results of our research, which indicated that patients across the entire cohort and within the middle-risk group didn’t benefit from IC. However, significant results were not obtained for the low- and high-risk groups. This may be attributed to the limited sample size, additional research on a larger scale in the future may be required. As indicated by the univariate COX regression analysis, CCRT didn’t emerge as a standalone predictive factor for OS in this population. Consequently, our analysis only focused on examining the potential benefits of IC in different risk subgroups of the population.
Numerous hematological biomarkers have demonstrated prognostic significance in different types of cancer. Specially, prognostic systems based on immune-inflammation and nutrition had been extensively researched. In the progression of cancer, inflammation plays a crucial role and is closely associated with cellular changes, growth, viability, formation of new blood vessels, as well as the spread of cancer cells.41,42 Neutrophils and platelets are critical regulators promoting cancer growth, angiogenesis, and distant metastasis. 43 Platelets also have a crucial function in protecting cancer cells against apoptosis induced by chemotherapy and in preserving the integrity of tumor blood vessels. 44 Conversely, lymphocytes can inhibit cancer proliferation and migration, thus playing a significant role in protective immunity. 43 Monocytes have the capability to secrete monocyte chemoattractant protein-1, which indirectly stimulates the development, vascularization, and distant spread of cancerous tumors. 45 Certain immune-inflammatory markers derived from these peripheral blood cell counts, including NLR, PLR, LMR, SII, and SIRI, have been proven to possess strong prognostic value in NPC patients. Multiple research findings have indicated a strong correlation between elevated NLR, increased PLR, and decreased LMR with an unfavorable outlook on the survival of individuals diagnosed with NPC.8,10,46 In 2017, the initial study by Jiang et al. utilized propensity score matching (PSM) techniques to investigate the relationship between SII and NPC prognosis. The findings indicated that an elevated SII, before and after PSM adjustment, independently predicts a poorer OS outcome among individuals with NPC. 47 Two subsequent meta-analyses have affirmed the significant correlation between an increased SII and inferior OS, PFS, and DMFS in individuals with NPC.48,49 Yuan et al. suggested that in NPC patients with negative EBV DNA, there was a negative correlation observed between SIRI and the prognosis of OS and PFS. Conversely, SII did not show any significant correlation with the prognosis in such patients. 38 The predictive significance of PIV has been widely acknowledged in various types of malignancies,50–54 but its potential to forecast survival results in NPC has not been investigated. In the research, we aforementioned immune-inflammatory markers using PCA to derive the III and discovered that an elevated III showed a notable correlation with decreased OS among non-metastatic NPC patients who tested negative for EBV DNA.
Malnutrition is reported to affect 35%–60% of NPC patients, which significantly impacts their responses to treatment. 55 In NPC patients, there was a notable correlation observed between a decreased serum ALB level and unfavorable outcomes in terms of OS and DMFS. 56 An analysis of 10 studies involving 4511 individuals diagnosed with NPC indicated that a decreased PNI functioned as a standalone prognostic element for both OS and DMFS. Consequently, this analysis suggested employing PNI as a prognostic marker for the survival results of NPC patients in both the immediate and extended time frames. 57 Our research involved the NI using serum ALB levels and PNI. The results we obtained indicated that NI emerged as a significant independent predictor of OS among non-metastatic NPC patients who tested negative for EBV DNA.
Several limitations were identified in our research. First, inherent to the intrinsic characteristics of retrospective studies, the occurrence of selection bias is a possible outcome, as the inclusion of patients was contingent upon their fulfillment of predetermined inclusion and exclusion criteria. Second, this was a study with a limited patient cohort from a single institution. Prospective analysis with a larger patient cohort is warranted. Third, EBER is considered the gold standard for detecting EBV infection. However, initially, EBER testing was not consistently performed at our center, resulting in missing data. Hence, we selected non-metastatic NPC with EBV DNA-negative as the focus of our study. In the future, we aim to investigate the prognosis of patients with EBER negative NPC through the implementation of routine EBER testing. Fourth, the levels of biomarkers exhibit dynamic alterations during the advancement of the disease and in response to treatment measures. In this research, only the correlation between inflammatory and nutritional indicators before treatment and the outlook was investigated. Therefore, additional exploration is warranted to assess the comparative predictive significance of these indicators prior to, during, or following treatment, which could potentially steer the trajectory of forthcoming research endeavors.
Nevertheless, our findings are significant as they represent the inaugural investigation into the clinical relevance of integrated immune-inflammatory and nutritional indicators for the prediction of OS in non-metastatic NPC with negative EBV DNA. Additionally, it holds referential value for informing treatment decision-making in this patient group.
Conclusion
In conclusion, our study had confirmed that III and NI can function as significant predictive indicators for non-metastatic NPC patients who test negative for EBV DNA. Furthermore, our investigation revealed that a nomogram model, which integrated III, NI, and TNM staging, could offer valuable complementary information to existing TNM staging protocol. This nomogram model had the potential to predict the individualized survival of this patient population more accurately, providing dependable prognostic insights and aiding the customization of treatment plans to optimize patient outcomes.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241286489 – Supplemental material for Prognostic value of immune-inflammatory and nutrition indicators in non-metastatic nasopharyngeal carcinoma with negative plasma Epstein–Barr virus DNA
Supplemental material, sj-docx-1-tam-10.1177_17588359241286489 for Prognostic value of immune-inflammatory and nutrition indicators in non-metastatic nasopharyngeal carcinoma with negative plasma Epstein–Barr virus DNA by Youliang Weng, Lishui Wu, Ying Li, Jing Wang, Zijie Wu, Xinyi Hong, Xiaoyong Liu, Jinghua Lai, Jun Lu and Sufang Qiu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241286489 – Supplemental material for Prognostic value of immune-inflammatory and nutrition indicators in non-metastatic nasopharyngeal carcinoma with negative plasma Epstein–Barr virus DNA
Supplemental material, sj-docx-2-tam-10.1177_17588359241286489 for Prognostic value of immune-inflammatory and nutrition indicators in non-metastatic nasopharyngeal carcinoma with negative plasma Epstein–Barr virus DNA by Youliang Weng, Lishui Wu, Ying Li, Jing Wang, Zijie Wu, Xinyi Hong, Xiaoyong Liu, Jinghua Lai, Jun Lu and Sufang Qiu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
