Abstract
Objective
Our study aimed to evaluate the main factors affecting the efficacy of anlotinib to determine the therapeutically dominant populations.
Methods
The medical records of patients with lung cancer who were treated with anlotinib from July 2018 to February 2020 at Renji Hospital, School of Medicine, Shanghai Jiaotong University were retrospectively reviewed. The optimal cutoff prognostic nutritional index (PNI) value for predicting efficacy was determined according to receiver operating characteristic curves. Progression-free survival (PFS) and overall survival (OS) were calculated and compared using the Kaplan–Meier method and log‐rank test. The prognostic values of each variable were evaluated with univariate and multivariate Cox proportional hazard regression analyses.
Results
The overall disease control rate of 44 patients with lung cancer was 93.2% (41/44). The median PFS was 5.0 months (95% [confidence interval] CI: 2.2–7.8), and the median OS was 6.5 months (95% CI: 3.6–9.3). The multivariate analysis results indicated that hand–foot syndrome and high PNI values were independent protective factors of PFS and OS.
Conclusions
Anlotinib was effective in treating locally advanced or advanced lung cancer. High pretreatment PNI scores and the presence of hand–foot syndrome after treatment were independent prognostic markers for favorable OS and PFS.
Keywords
Introduction
Lung cancer is the most common malignant tumor worldwide (11.6% of total cases) and the leading cause of cancer-related death (18.4% of total cancer-related deaths). 1 Because most patients with early-stage lung cancer are asymptomatic, the diagnosis is usually made at locally advanced or advanced stages. 2 Drug therapy, including chemotherapy, targeted therapy, and immunotherapy, is still the most important treatment for unresectable lung cancer. Recently developed biomarker-driven agents, such as targeted therapies and immunotherapies, have provided great benefits for patients with different stages of lung cancer in first-line and second-line settings. However, there are limited clinical research results for third-line treatments, and there is currently no standard treatment plan. In addition, first-line and second-line treatments are considerably limited for patients with negative biomarker status and chemotherapy insensitivity or intolerance. In recent years, immunotherapy has revolutionized cancer treatment. Nivolumab and pembrolizumab have shown survival benefits in patients with both adenocarcinoma and squamous cell carcinoma as second-line or subsequent therapies,3–5 but their widespread application remains difficult in China because of economic reasons.
Angiogenesis is essential for several aspects of tumor development, including tumor growth, invasion, and metastasis. 6 Anlotinib is a new small molecule multi-target tyrosine kinase inhibitor that effectively inhibits the activities of several growth factor receptors, leading to impaired tumor angiogenesis and growth.7,8 Studies have shown that anlotinib is a potent inhibitor of small-cell lung cancer (SCLC) and non-small-cell lung cancer (NSCLC) in the third-line or later-line setting.9–11 However, some patients are non-responsive. Therefore, identifying a biomarker that can predict the clinical response of patients with advanced lung cancer to anlotinib is an urgent issue that needs to be addressed to improve clinical outcomes. The purpose of this study was to evaluate the main factors affecting the efficacy of anlotinib and identify prognostic indicators to determine the therapeutically dominant populations.
Materials and methods
Study population
The medical records of patients with lung cancer treated with anlotinib from July 2018 to February 2020 at Renji Hospital, School of Medicine, Shanghai Jiaotong University were retrospectively reviewed. Patients with incomplete clinical data or those lost to follow up were excluded. All patients diagnosed with lung cancer were confirmed by histopathology. This study was approved by the medical ethics committee of Renji Hospital. The need for informed patient consent was waived because of the retrospective nature of the study, and the patient’s personal data have been secured. The reporting of this study conforms to the STROBE statement. 12
Study parameters
The general clinical data of patients were collected. In addition, data from hematologic tests carried out before treatment with anlotinib were obtained. The prognostic nutritional index (PNI) was calculated as serum albumin (g/L) + 5 × peripheral blood lymphocyte count (×109/L).
The staging of patients with lung cancer was defined according to the eighth edition of the TNM classification of lung cancer (National Comprehensive Cancer Network). Performance status (PS) was assessed using the Eastern Cooperative Oncology Group criteria. The response to therapy was assessed according to the RECIST 1.1 criteria. Overall survival (OS) was calculated from the date of anlotinib initiation to the time of death (due to any cause) or until February 2020 for patients who remained alive. Progression-free survival (PFS) was defined as the duration between anlotinib initiation and objective tumor progression or death or until February 2020 for patients who remained progression-free. Adverse events were assessed according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events version 4.0. The disease control rate (DCR) was defined as the percentage of patients who were evaluated and achieved complete response (CR), partial response (PR), and stable disease (SD) for at least 4 weeks.
Safety assessment
Based on the most common adverse events associated with anlotinib, we selected those with an incidence of more than 10% in this study for drug safety analysis.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA). Descriptive analysis was used for all variables. Counting variables were presented as percentages. The optimal cutoff PNI value for predicting efficacy was determined according to receiver operating characteristic (ROC) curves and the areas under the ROC curve (AUC). Patients were divided into high PNI and low PNI groups based on cutoff values. PFS and OS were calculated and compared using the Kaplan–Meier method and log‐rank test. The prognostic value of each variable was evaluated with univariate and multivariate Cox proportional hazard regression analyses. P < 0.05 was considered statistically significant.
Results
Baseline data and efficacy
Forty-four patients with lung cancer were enrolled in our retrospective study. The median age was 66.5 years (range: 43–80 years). Baseline characteristics of the patients are listed in Table 1.
Demographic and clinical characteristics of patients (n = 44).

PS, performance status; EGFR, epidermal growth factor receptor; PR, partial response; SD, stable disease; PD, progressive disease.
The median PFS was 5.0 months (95% [confidence interval] CI: 2.2–7.8), and the median OS was 6.5 months (95% CI: 3.6–9.3). The median PFS of patients with SCLC (9 patients) was 5.0 months (95% CI: 2.6–7.4), and the median OS was 7.8 months (95% CI: 2.6–12.9). The median PFS of patients with NSCLC (33 patients) was 4.0 months (95% CI: 0.0–8.1), and the median OS was 6.5 months (95% CI: 0.0–13.7).
No patients achieved CR, 9.1% (4/44) achieved PR, 84.1% (37/44) had SD, and 6.8% (3/44) had progressive disease. The overall DCR was 93.2% (41/44), the DCR of SCLC was 88.9% (8/9), and the DCR of NSCLC was 97.0% (32/33).
Safety
The safety analysis of anlotinib is shown in Table 2. The most common adverse events were hand–foot syndrome (40.9%), hypertension (29.5%), diarrhea (22.7%), albuminuria (20.5%), liver dysfunction (13.6%), and oral mucositis (9.1%).
Safety analysis.

Univariate and multivariate analyses
Univariate and multivariate analyses of PFS and OS were performed using Cox regression models, and factors considered included age, gender, smoking status, pathological type, disease stage, PS, metastatic sites, treatment history, adverse events, and pretreatment PNI levels. The cutoff value for the stratification of age was 65 years. The pathological type was stratified into SCLC and NSCLC. The disease stage was stratified into stage III and stage IV, and the PS was stratified into 0 to 1 and ≥2. Because the PNI had no reference value, the optimal cutoff PNI value for predicting efficacy was 35 according to the ROC curve. The sensitivity, specificity, AUC, and P value were 0.88, 1.00, 0.911, and 0.019, respectively.
Only factors with statistically significant differences in the univariate analysis are listed in Table 3 and Table 4. In the univariate analysis, we found that PS 0 to 1 (P = 0.037), relatively earlier disease stages (P = 0.003), presence of hand–foot syndrome (P < 0.001), and high PNI scores (P < 0.001) were associated with longer PFS (Figure 1a–d), whereas liver metastasis (P = 0.038) and contralateral lung metastasis (P = 0.017) were associated with shorter PFS (Figure 1e–f). The presence of hand–foot syndrome (P < 0.001) and high PNI scores (P < 0.001) were associated with longer OS (Figure 1g–h). The multivariate analysis results indicated that hand–foot syndrome (P < 0.001) and high PNI scores (P < 0.001) were independent protective factors for PFS and OS.
Univariate and multivariate analysis of PFS.
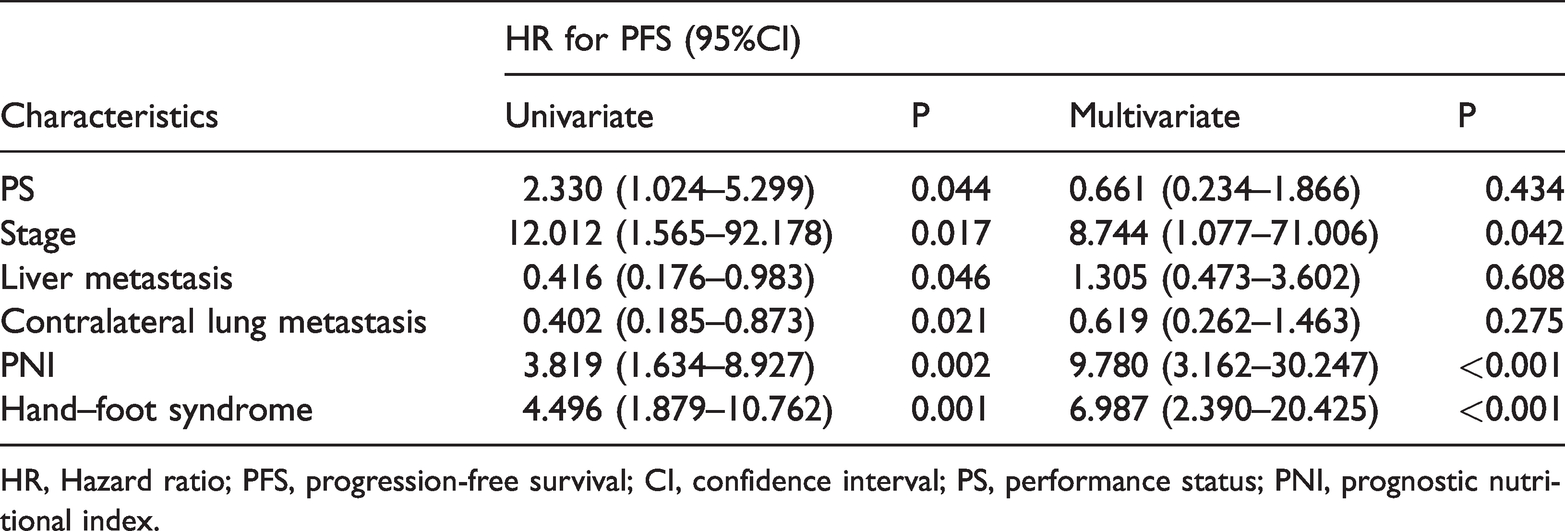
HR, Hazard ratio; PFS, progression-free survival; CI, confidence interval; PS, performance status; PNI, prognostic nutritional index.
Univariate and multivariate analysis of OS.
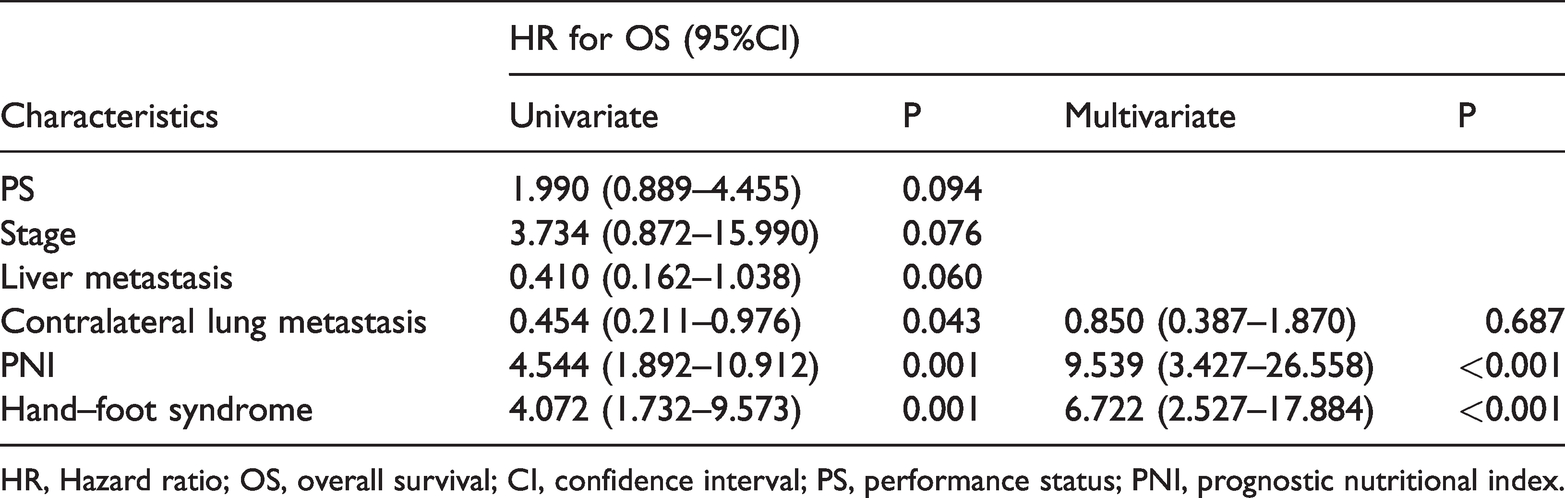
HR, Hazard ratio; OS, overall survival; CI, confidence interval; PS, performance status; PNI, prognostic nutritional index.

Kaplan–Meier plots of prognostic factors according to overall survival (OS) and progression-free survival (PFS).
Discussion
Anlotinib is a new anti-tumor drug developed independently in China. Studies have confirmed its efficacy and safety in both SCLC and NSCLC.10,11 However, the inclusion criteria for clinical trials are strict, and there are limited studies related to the treatment of anlotinib in a real-world setting. In this study, we retrospectively evaluated the real-world data of patients with lung cancer who were treated with anlotinib in China to assess the efficacy and toxicity of anlotinib. The median PFS and OS of patients with lung cancer in our study were different from those reported in previous studies.9–11 One possible explanation is that the inclusion criteria of this study were relatively broad. In addition, the sample size of this study was small, and some patients had a short follow-up period and did not reach disease progression or death.
In the era of immunotherapy, an increasing number of studies have focused on therapy combined with immune checkpoint inhibitors. Liu et al. 13 reported that anlotinib ameliorated the immuno-microenvironment by downregulating programmed death-ligand 1 expression on vascular endothelial cells to inhibit tumor growth, providing theoretical and experimental evidence for the combination of anlotinib with immunotherapy. At the 2019 World Conference of Lung Cancer, Professor Bao-hui Han presented a report on sintilimab combined with anlotinib as first-line therapy for advanced NSCLC. 14 The results revealed an objective response rate (ORR) as high as 72.7%, similar to the results of targeted therapy, and the DCR was 100%. In a retrospective study of 101 patients with NSCLC who were treated with anlotinib combined with immunotherapy as third-line therapy, 15 the ORR was 18.8%, and the DCR was 79.2%. The median PFS was 6.7 months, which was longer than that reported in ALERT 0303 10 (the median PFS was 5.4 months), and the addition of immunotherapy did not increase the incidence of adverse events. A prospective, large-sample study is needed to confirm the efficacy of immunotherapy combined with anlotinib.
The PNI calculated based on the serum albumin level and total lymphocyte count in peripheral blood is a widely used nutritional and immunological index in which the lymphocyte count reflects the immunological status, and the albumin concentration reflects the nutritional status. Several studies have assessed the PNI in patients with both SCLC and NSCLC. In a study of 220 patients with SCLC who received first-line platinum-based chemotherapy, Go et al. 16 reported an association between low pretreatment PNI scores and poor survival. In a meta-analysis published in 2018 by Hu et al., 17 the PNI was reported as a prognostic marker in patients with NSCLC, but most of the studies included in these analyses were performed in patients with early-stage, resected NSCLC treated with chemotherapy or radiotherapy, and the number of studies in patients with advanced disease was limited. Bozkaya et al. 18 reported that pretreatment PNI values were an independent prognostic factor for OS and PFS in patients with metastatic NSCLC treated with first-line chemotherapy. Current studies are investigating the use of the PNI as an indicator of clinical benefit in patients receiving chemotherapy. Few studies have been conducted on the prognostic value of the PNI in patients with lung cancer treated with other therapies. Shoji et al. 19 reported that pretreatment PNI values were significantly associated with responses to immune checkpoint inhibitor therapy in patients with NSCLC, and the PNI was an independent prognostic factor for PFS. Recently, a retrospective study showed that the pretreatment PNI value was an independent prognostic factor for OS in patients with extensive-stage SCLC who were treated with anlotinib. 20 Our study showed that the PNI remained a good predictor of prognosis for patients with lung cancer who were treated with anlotinib.
Chest radiotherapy is a common strategy to improve the stability of local chest tumors. Wang et al. 21 found that patients who received chest radiotherapy had a longer PFS. This might be because chest radiotherapy alters the immune microenvironment of cancer, thereby potentially increasing the efficiency of anlotinib. The univariate and multivariate analysis in our study found no correlation between chest radiotherapy and PFS or OS in patients with lung cancer who were treated with anlotinib, which may be related to the small sample size of this study. A prospective, large-sample study is needed to clarify the prognostic impact of chest radiotherapy in patients with lung cancer who are treated with anlotinib.
The targeting of multiple proteins and signaling pathways by anlotinib may help overcome the acquired resistance induced by previous treatments. However, compared with mono-target agents, anlotinib may cause more adverse reactions. This study showed that the incidence of adverse reactions after treatment with anlotinib was high, but most were mild and treatable, similar to previous studies.10,22 Only three cases had serious adverse reactions. One case experienced serious hand–foot syndrome and underwent dose reduction due to a grade 3 adverse reaction. One case experienced grade 3 diarrhea, but their condition improved after symptomatic treatment. One case experienced hepatic failure and died. However, the patient had multiple metastases, including to the liver, at the time of diagnosis. This study suggests that more attention should be paid to adverse effects during the future application of anlotinib.
Although anlotinib simultaneously functions as an anti-angiogenic agent and epidermal growth factor tyrosine kinase inhibitor (EGFR-TKI), its associated adverse events have been demonstrated to be tolerable and transient. Similar to EGFR-TKIs, 23 treatment with anlotinib also resulted in hand–foot syndrome, which was considered to be a predictor of good PFS. Similar to anti-angiogenic agents, 24 anlotinib caused hypertension, which was reported to be a good predictor of PFS. Nan et al. 25 reported that patients with advanced NSCLC who developed hand–foot syndrome had longer OS and PFS compared with patients who did not develop this syndrome during third-line or further anlotinib therapy. Our study further analyzed the relationship between adverse reactions and prognosis. Our results showed that hand–foot syndrome was a predictor of good PFS and OS after anlotinib treatment, whereas no significant correlation was found between hypertension and PFS or OS. Because our study is a retrospective study with a small sample size, a large-sample prospective study is needed to confirm the relationship between adverse reactions and the prognosis of patients treated with anlotinib.
Conclusion
Our study showed that anlotinib was effective in treating locally advanced or advanced lung cancer and was well tolerated. High pretreatment PNI values and the presence of hand–foot syndrome after treatment were independent prognostic markers for good OS and PFS. Thus, the PNI may be used as a cost-effective and simple prognostic tool in routine clinical practice. In addition, immune-nutritional support before or during anlotinib therapy may potentially improve the response and outcomes of patients with lung cancer to anlotinib. A prospective study is needed to verify the usefulness of immune-nutritional support in patients with lung cancer patients treated with anlotinib.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant for a clinical innovation topic from Renji Hospital Affiliated to the Medical College of Shanghai Jiao Tong University (No. 2019NYLYCP0103).
