Abstract
Objective
Chaperonin containing TCP1 subunit 5 (CCT5) encodes the CCT5 protein subunit of chaperonin-containing TCP-1 (CCT/TRiC) complex, and is shown to be upregulated in tumour pathogenesis. The study aim was to investigate the differential expression of CCT5 between nasopharyngeal carcinoma (NPC) and noncancerous nasopharyngeal tissues, and the correlation between CCT5 expression and clinicopathological parameters/prognosis in patients with NPC.
Methods
Microarray assay data were evaluated for differential expression between NPC and noncancerous nasopharyngeal tissues. CCT5 expression in NPC and noncancerous nasopharyngeal tissues was determined at mRNA and protein levels by quantitative reverse transcription–polymerase chain reaction (qRT–PCR) and immunohistochemistry. Relationships between CCT5 expression in NPC, clinical parameters, and prognosis were statistically analysed. CCT5-mediated cell proliferation was assessed using EdU and cell counting kit-8. Western blot and co-immunoprecipitation were utilized to explore E3 ubiquitin-protein ligase parkin (PARK2)-induced degradation of CCT5.
Results
Microarray data showed CCT5 levels to be significantly increased in NPC versus noncancerous nasopharyngeal tissues, which was confirmed by qRT-PCR and immunohistochemical assays. Increased CCT5 protein levels positively correlated with tumour size, tumour recurrence, and clinical stage, and inversely correlated with patient’s overall survival. Multivariate Cox regression analysis showed that enhanced CCT5 protein expression is an independent prognostic factor for patients with NPC. Overexpression of CCT5 markedly induced NPC cell proliferation. Finally, PARK2, as a suppressive E3 ubiquitin-ligase enzyme, was shown to bind CCT5 and induce degradation in NPC.
Conclusions
Increased CCT5 may be an unfavourable factor promoting NPC growth. Binding of PARK2 to CCT5 was associated with CCT5 degradation, suggesting that PARK2 is an upstream negative modulator in NPC.
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumour with distinctive geographic distribution, and is most prevalent in Southern China and Southeast Asia. Epstein–Barr virus (EBV) infection is the leading cause of NPC, with detection of the EBV genome in almost all NPC cases in endemic regions. The consumption of salt-preserved fish, genetic family history, and certain human leukocyte antigen class I genotypes are also significant risk factors for NPC.1–4 These factors alone or together cause the abnormal expression of various genes eventually leading to NPC pathogenesis.5–9
Chaperonin-containing TCP-1 (CCT/TRiC) is a multi-subunit complex consisting of eight paralogous subunits (CCT1–8) that mediates the folding of newly synthesized proteins. 10 Investigations have shown that CCT/TRiC interacts with many proteins involved in the proliferation, migration, and apoptosis of tumour cells, such as cyclin B, cyclin E, actins, tubulins, p21, the von Hippel-Lindau tumour suppressor, Kirsten rat sarcoma viral proto-oncogene (KRAS), and signal transducer and activator of transcription 3 (STAT3).11–13 CCT5 is a critical subunit of the CCT/TRiC complex, and studies suggest that CCT5 might be an oncogene in some cancers.14,15 However, its specific function in NPC remains unexplored.
E3 ubiquitin-protein ligase parkin (also known as PARK2), an E3 ubiquitin ligase, was first elucidated as a key gene involved in autosomal recessive juvenile Parkinson’s disease. However, increasing evidence indicates PARK2 as a tumour suppressor participating in a variety of cancers via ubiquitination and degradation of various tumour promoters, such as yes-associated protein (YAP) in oesophageal squamous cell carcinoma, 16 and apoptosis regulator BCL2 in breast cancer. 17 However, whether PARK2 binds to CCT5 and induces degradation has not been documented in NPC.
In a previous study, the present author group used a microarray to screen differentially expressed genes between NPC and noncancerous nasopharyngeal tissues.18,19 Several genes, including programmed cell death 4 (PDCD4) and cellular communication network factor 2 (CTGF) were found to be significantly differentially expressed, and the molecular function of the above mentioned genes was fully explored.20–22 Re-analysis of the microarray data revealed that CCT5 mRNA levels were markedly elevated in NPC. Thus, the aim of the present study was to validate the differential expression of CCT5 between NPC and noncancerous nasopharyngeal tissues, and to analyse the correlation of CCT5 expression with clinicopathological parameters and prognosis of patients with NPC.
Materials and methods
Microarray analysis
Differential gene expression between NPC and noncancerous nasopharyngeal tissues was assessed in a previous study using 8K Human cDNA microarrays from Shenzhen Chipscreen Biosciences Limited (Shenzhen, China). 19 The microarray data were re-evaluated to search for new potential NPC biomarkers.
Study population and sample collection
A total of 16 fresh NPC samples and 11 fresh noncancerous nasopharyngeal samples were collected from patients who underwent biopsy at the People’s Hospital of Zhongshan City, at the time of diagnosis, before treatment. Inclusion criteria were: patients suspected of having NPC who had provided informed consent for study inclusion and tissue sample collection, and patients with adequate tissue for analysis. Patients who had received prior cancer therapy were excluded. The NPC group comprised those with confirmed pathological diagnosis of NPC, and the noncancerous nasopharyngeal group comprised those with confirmed noncancerous nasopharyngeal tissue. All samples were immediately preserved in liquid nitrogen. Further, 158 paraffin-embedded NPC specimens and 45 noncancerous nasopharyngeal specimens were obtained from the People's Hospital of Zhongshan City. Written informed consent was obtained from each patient who provided tissue samples for the study, and the study was approved by the ethics committee of the People’s Hospital of Zhongshan City. All NPC specimens were staged according to the 1997 World Health Organization NPC staging system.
RNA extraction and quantitative real-time reverse transcription–polymerase chain reaction (qRT–PCR)
Total RNA was extracted from 16 fresh NPC tissues and 11 noncancerous nasopharyngeal tissues using a Total RNA Isolation Kit (TaKaRa Bio, Shiga, Japan) following the manufacturer’s protocol, then 1 µg RNA per sample was reverse transcribed into cDNA using a reverse transcription reagent kit (Vazyme, Nanjing, China) with the following parameters: 37°C for 15 min, 85°C for 40 s, then 4°C. The cDNA was then amplified by real-time qRT–PCR to determine the mRNA level of CCT5 using an SYBR Premix Ex Taq kit (TaKaRa Bio) with an Mx3000P real-time PCR system (Stratagene, CA, USA). The reaction mix included 100 ng cDNA, SYBR green master mix, 0.2 µM reverse and forward primers, and RNase-free water to adjust to final volume. GAPDH was used as an internal control. The primer sequences were as follows: CCT5 (forward, 5′-CCTTGGGACCCACTATCGAG-3′; reverse, 5′-TTCTAACAAGGCACCAGCCA-3′); GAPDH (forward, 5′-GCACCGTCAAGGCTGAGAAC-3′; reverse, 5′-TGGTGAAGACGCCAGTGGA-3′). The reaction cycling parameters were: 95°C for 30 s, and 40 cycles of 95°C for 5 s and 60°C for 30 s. Relative gene expression levels were calculated using the 2−ΔΔCt method.
Immunohistochemical analysis
Immunohistochemical analysis was performed as described previously.23,24 Briefly, paraffin-embedded sections (3 µm) of NPC and noncancerous nasopharyngeal tissue were deparaffinized and hydrated in xylene and descending ethanol series, followed by heat-induced antigen retrieval in 10 mM citrate buffer for 2 min at 100°C. Endogenous peroxidase activity and nonspecific antigen were then blocked with 3% hydrogen peroxide and serum. The sections were incubated with rabbit anti-human CCT5 antibody (1:100; 11603-1-AP, Proteintech, Rosemont, IL, USA) for 1 h at 37°C, followed by horseradish peroxidase (HRP)–conjugated secondary antibody (Maixin, Fuzhou, China) for 30 min at room temperature. Finally, the sections were visualized with DAB and counterstained with haematoxylin. The images were captured using a microscope under 200 × magnification.
The immunostained tissue sections were analysed by two pathologists (SYW and LRP). The staining level was evaluated based on the staining intensity and the proportion of positively stained areas in the whole section. The staining intensity was classified as 0 (negative), 1 (weak), 2 (moderate), and 3 (strong). The areas of staining were defined as 0 = < 10%, 1 = 10%–25%, 2 = 26%–50%, 3 = 51%–75%, and 4 =>75%. The staining intensity and percentage scores were added up to obtain the total score for CCT5 staining. Total scores between 0 and 5 were defined as low expression and ≥6 as high expression.
Cell culture
5-8F cells and Hone1 cells from stock stored at the Cancer Research Institute of the Southern Medical University (Guangzhou, China) were cultured in RPMI1640 cell culture media (VivaCell, Denzlingen, Germany) supplemented with 10% fetal bovine serum premium (Cegrogen, Stadtallendorf, Germany) and 1% penicillin-streptomycin (Solarbio, Beijing, China). Cells were cultured in a 37°C, 5% CO2 incubator. Culture dishes were purchased from Jet Biofil (Guangzhou, China).
Transfection of CCT5-Flag fusion expression plasmid and PARK2
To assess the effects of CCT5 overexpression, 5-8F and Hone1 cells were transfected with a fusion expression vector containing human CCT5 and Flag (Weizhen Company, Shandong, China). After amplification in Escherichia coli DH5α, the plasmid was extracted using a plasmid extraction kit (Beyotime Biotechnology, Shanghai, China), according to the manufacturer’s instructions. 5-8F and Hone1 cells at 40% confluence were then transfected with 1 µg CCT5-Flag or control-Flag using Lipofectamine 2000 Transfection Reagent (Invitrogen, Carlsbad, CA, USA), according to the manufacturer’s instructions. The expression efficiency of the fusion protein was detected using Flag antibodies via western blot, as described later.
Additionally, at 12 h prior to transfection, the 5-8F and Hone1 cells were plated into 6-well plates (Nest Biotech, China) and cultured to 30–50% confluence. PARK2 overexpression plasmid and respective control plasmids (Genechem, Shanghai, China) were then transfected using Lipofectamine 2000 Transfection Reagent (Invitrogen) according to the manufacturer’s protocol.
Cell proliferation assays
Cell proliferation was assessed by cell counting kit-8 (CCK-8) and 5-ethynyl-2′-deoxyuridine (EdU) assays in 5-8F and Hone1 cells.
For CCK-8, the 5-8F and Hone1 cells were harvested and seeded into 96-well culture plates at a density of 4000 cells/well. Cells were allowed to adhere for 12 h at 37°C/5% CO2, then viability was assessed by CCK-8 assay at 0 h, 24 h, 48 h, 72 h, and 96 h after adhesion. CCK-8 reagent (Vazyme, Nanjing, China) was added to the medium (10 µl/well), incubated at 37°C for 1 h, and absorbance was measured at 450 nm using a Multiskan SkyHigh microplate spectrophotometer (ThermoFisher, Waltham, MA, USA). The experiment was repeated 3 times.
The EdU incorporation assay was performed using the EdU Apollo567 in vitro imaging kit (RiboBio, Guangzhou, China), according to the manufacturer's protocol. The 5-8F and Hone1 cells were seeded at 8000 cells/well into 96-well plates and allowed to adhere for 12 h at 37°C/5% CO2 before incubating with 10 µM EdU for 2 h followed by fixation with 4% paraformaldehyde. After permeabilization with 0.3% Triton X-100, the cells were stained with Apollo fluorescent dyes and cell nuclei were stained with DAPI. Positive staining was visualised and quantified by counting three random fields under fluorescence microscopy. The experiments were repeated 3 times.
Western blot
To extract protein for western blotting, cells were harvested and lysed in RIPA cocktail buffer (RIPA with protease inhibitor and phosphatase inhibitor; Beyotime, Shanghai, China). Protein concentrations were determined by BCA protein assay before adding sodium dodecyl sulphate (SDS)-loading buffer to each protein sample followed by denaturation at 95°C for 5 min. Denatured protein samples were separated by 10% SDS-polyacrylamide gel electrophoresis (PAGE) and transferred to polyvinylidene difluoride membranes (MilliporeSigma, Burlington, MA, USA). The membranes were blocked in tris-buffered saline (containing 0.05% tween-20; TBST) with 5% defatted milk (for normal protein test) or bovine serum albumin (for phosphorylated protein test) for 1 h at room temperature. The relevant primary antibody was added and the membranes were incubated at 4°C overnight, then washed 3 times with TBST (5 min per wash). The secondary antibody was then added and the membranes incubated at room temperature for 1 h before again washing 3 times in TBST (5 min per wash). Immunosignals were stimulated by Chemiluminescent HRP substrate (MilliporeSigma), according to the manufacturer’s instructions, and detected with a ChemiDoc™ CRS Molecular Imager (Bio-Rad, Hercules, CA, USA). The following antibodies were used for western blots: polyclonal rabbit anti-CCT5 (11603-1-AP), polyclonal rabbit anti-PARK2 (14060-1-AP), polyclonal rabbit anti-GAPDH (10494-1-AP), all purchased from Proteintech (Wuhan, China), and monoclonal mouse anti-flag (F1804; Sigma-Aldrich, St. Louis, MO, USA). HRP-conjugated goat anti-rabbit IgG(H+L) (SA00001-2; Proteintech) and HRP-conjugated goat anti-mouse IgG(H+L) (SA00001-1; Proteintech) were used as the secondary antibody. CCT5 protein polyubiquitination was examined by immunoblot analysis with polyclonal rabbit anti-ubiquitin antibody (10201-2-AP; Proteintech, Wuhan, China). Experiments were repeated 3 times.
Co-immunoprecipitation (Co-Ip)
The interaction between endogenous CCT5 and PARK2 in 5-8F and Hone1 cells was assessed using a co-immunoprecipitation (Co-Ip) kit (Pierce Biotechnology, Rockford, IL, USA), according to the manufacturer's instructions. The cells were lysed in cold lysis buffer with a phosphatase inhibitor cocktail (Cwbio, Taizhou, China) and protease inhibitor cocktail (Cwbio), and the supernatant was collected to measure protein concentration after centrifuging at 10 000 g for 20 min. Samples were then incubated with polyclonal rabbit anti-CCT5 (11603-1-AP, Proteintech), polyclonal rabbit anti-pPARK2 (14060-1-AP, Proteintech), or normal rabbit IgG (#2729, Cell Signaling Technology, Danvers, MA, USA) antibodies overnight at 4°C. Protein A-G agarose beads from the Co-Ip kit were added and maintained at room temperature for 40 min. After being washed with lysis buffer 4 times, the beads were eluted and denatured in SDS-PAGE loading buffer. Finally, the precipitated proteins were analysed by western blot assay. Experiments were repeated 3 times.
Cycloheximide chase
Cycloheximide (CHX) pulse-chase experiments were conducted to determine CCT5 protein half-life. 5-8F cells were seeded into 6-well plates in 2 ml culture medium and cultured to 70% confluence. Cells were then treated with CHX (50 µg/ml) and harvested at 0, 2, 4, 6, 8, 10 h. Total cell lysates were separated by SDS-PAGE and protein levels were analysed by immunoblot. The experiment was repeated 3 times.
MG132 experiments
The proteosome inhibitor MG132 (MedchemExpress, Monmouth Junction, NJ, USA) was used to detect the stability of CCT5. 5-8F cells were seeded into 100-mm cell culture dishes. Once at 70% confluence, cells were treated with MG132 (20 µM) and then harvested after 8 h. Total cell lysates were separated by SDS-PAGE and protein levels were analysed by immunoblot. The experiments were repeated 3 times.
Statistical analyses
All statistical analyses were performed using SPSS software, version 20.0 (IBM, Armonk, NY, USA). Data are presented as n (%) prevalence or mean ± SD. Differences in CCT5 expression between NPC and noncancerous nasopharyngeal tissues was assessed with χ2-test. Differences in demographic and clinicopathological characteristics between patients with NPC and high or low CCT5 expression were also assessed with χ2-test. Overall survival time was analysed using the Kaplan–Meier method, and correlations between various parameters and overall survival were analysed by univariate and multivariate Cox regression. Between-group comparisons of proliferation or proportion of cells in S phase were performed using two independent samples t-test. A P value < 0.05 indicated statistical significance.
Results
Microarray analysis
Analysis of microarray data revealed that CCT5 mRNA levels were significantly elevated in eight pooled samples of NPC tissues (4 samples per pool) compared with 24 pooled noncancerous nasopharyngeal samples (Figure 1(a)). Sample processing has been described previously. 19

Levels of chaperonin containing TCP1 subunit 5 (CCT5) mRNA were upregulated in nasopharyngeal carcinoma (NPC) versus noncancerous nasopharyngeal tissues: (a) heat map of microarray results showing differently expressed genes between NPC and noncancerous nasopharyngeal samples (green indicates downregulated, red indicates upregulated); and (b) column scatter plot of quantitative real-time reverse transcription–polymerase chain reaction, showing significantly upregulated CCT5 mRNA levels in fresh NPC versus noncancerous nasopharyngeal tissues.
Increased CCT5 mRNA level in NPC
Real-time RT–PCR was used to determine CCT5 mRNA levels in 16 freshly collected NPC tissues and 11 noncancerous nasopharyngeal tissues, and showed that CCT5 mRNA levels were significantly higher in NPC versus noncancerous nasopharyngeal tissues (P = 0.0318; Figure 1(b)).
CCT5 protein levels are upregulated in NPC
Immunohistochemical staining was performed to analyse CCT5 expression in terms of protein levels and subcellular localization in 158 paraffin-embedded NPC samples and 45 paraffin-embedded noncancerous nasopharyngeal samples (Figure 2). CCT5 was mainly expressed in the cytoplasm of NPC and noncancerous nasopharyngeal epithelial cells. CCT5 protein levels were classified as high in 115 out of 158 NPC samples (72.8%) but in only 13 out of 45 noncancerous nasopharyngeal samples (28.9%), indicating that CCT5 expression was significantly upregulated at the protein level in NPC tissues compared with noncancerous nasopharyngeal tissues (P < 0.001; Table 1).

Representative photomicrographs showing immunostaining for chaperonin containing TCP1 subunit 5 (CCT5) protein in nasopharyngeal carcinoma (NPC) and noncancerous nasopharyngeal tissues: (a) noncancerous nasopharyngeal tissue section showing low CCT5 protein levels; (b) noncancerous nasopharyngeal tissue section showing high CCT5 protein levels; (c and d) NPC tissue sections showing low CCT5 protein levels; and (e and f) NPC tissue sections showing high CCT5 protein levels (original magnification in all photomicrographs, × 200).
Expression of chaperonin containing TCP1 subunit 5 (CCT5) in paraffin embedded nasopharyngeal carcinoma (NPC) and noncancerous nasopharyngeal tissue samples.

Data presented as n prevalence.
Between-group differences in CCT5 expression (χ2-test).
Relationship between CCT5 expression and clinicopathological features
The relationship between demographic and clinicopathological characteristics of patients with NPC and CCT5 expression in NPC tissues is summarized in Table 2. Statistically significant differences in CCT5 protein levels were shown according to tumour size (T1–2 versus T3–4, P = 0.017), clinical stage (I–II versus III–IV, P < 0.001), and tumour recurrence (P = 0.011) in patients with NPC. No significant differences in CCT5 protein levels were shown regarding patient's age, sex, lymph node metastasis (N) classification, or distant metastasis (M) classification (Table 2).
Correlation between demographic and clinicopathological characteristics and chaperonin containing TCP1 subunit 5 (CCT5) expression in tissue from patients with nasopharyngeal carcinoma.

Data presented as n prevalence.
NS, no statistically significant between-group difference (P > 0.05; one-way analysis of variance).
Correlation between CCT5 and overall survival
The correlation between CCT5 expression at the protein level and patients' overall survival was assessed using Kaplan–Meier survival curves, to explore the prognostic value of CCT5 in patients with NPC. CCT5 expression was observed to inversely correlate with overall survival in patients with NPC (Figure 3). Patients with high levels of CCT5 expression showed significantly shorter survival time compared with those with low expression (P = 0.001). Multivariate Cox regression analysis indicated that CCT5 expression at the protein level may be an independent and unfavourable prognostic factor in patients with NPC (P = 0.02; Table 3).

Kaplan-Meier curves of overall survival in 158 patients with nasopharyngeal carcinoma with high or low chaperonin containing TCP1 subunit 5 (CCT5) expression levels.
Univariate and multivariate Cox regression analysis of individual parameters correlated with overall survival in 158 patients with nasopharyngeal carcinoma.

HR, hazard ratio; CI, confidence interval; CCT5, chaperonin containing TCP1 subunit 5.
NS, no statistically significant correlation (P > 0.05).
CCT5 promotes NPC proliferation
To further elucidate the role of CCT5 in NPC, CCT5-Flag was transfected into 5-8F and Hone1 cells to promote CCT5 overexpression. The expression efficiency CCT5-Flag fusion protein was assessed using western blot with Flag antibody (Figure 4(a)). The effects of CCT5 on NPC cell viability and proliferation were subsequently assessed. The CCK-8 and EdU assays revealed that CCT5 significantly promotes proliferation and cell cycle G1/S phase transition, respectively, in cells overexpressing CCT5 compared with control vector cells (P < 0.01; Figure 4(b) and 4(c)).

Chaperonin containing TCP1 subunit 5 (CCT5) promotes nasopharyngeal carcinoma (NPC) cell proliferation: (a) representative western blot showing successful co-transfection and expression of CCT5-FLAG in 5-8F and Hone1 cell lines; (b) cell cycle kit-8 proliferation assay results showing increased absorbance (450 nm) readings over 4 days in cells transfected with CCT5 (CCT5 overexpression) versus control vectors and (c) representative photomicrographs showing EdU incorporation (scale bar: 100 µm) in 5-8F and Hone1 cells transfected with CCT5 (CCT5 overexpression) versus control vectors. ***P < 0.001 and **P < 0.01 versus controls (NC [CCT5]).
PARK2 ubiquitinates CCT5 leading to its protein degradation
The Co-Ip results showed that CCT5 interacts with PARK2 in the 5-8F NPC cell line (Figure 5(a)). To determine whether PARK2 ubiquitinates and causes degradation of CCT5 protein, 5-8F cells were incubated with the proteosome inhibitor MG132. The presence of MG132 rescued PARK2-overexpression-induced CCT5 protein loss (Figure 5(b)). Further, when 5-8F cells overexpressing PARK2 were treated with 50 µg/ml CHX to block protein synthesis, the half-life of CCT5 was significantly shorter than in control cells not overexpressing PARK2 (Figure 5(c)). A ubiquitination experiment was conducted to further investigate whether PARK2 ubiquitinates CCT5 and affects its stability, and showed that the ubiquitination level of CCT5 was increased by PARK2 overexpression in 5-8F cells (Figure 5(d)). Finally, MG132 suppressed PARK2-mediated protein degradation of CCT5. Together, these results demonstrated that PARK2 ubiquitinated CCT5 protein leading to its degradation.
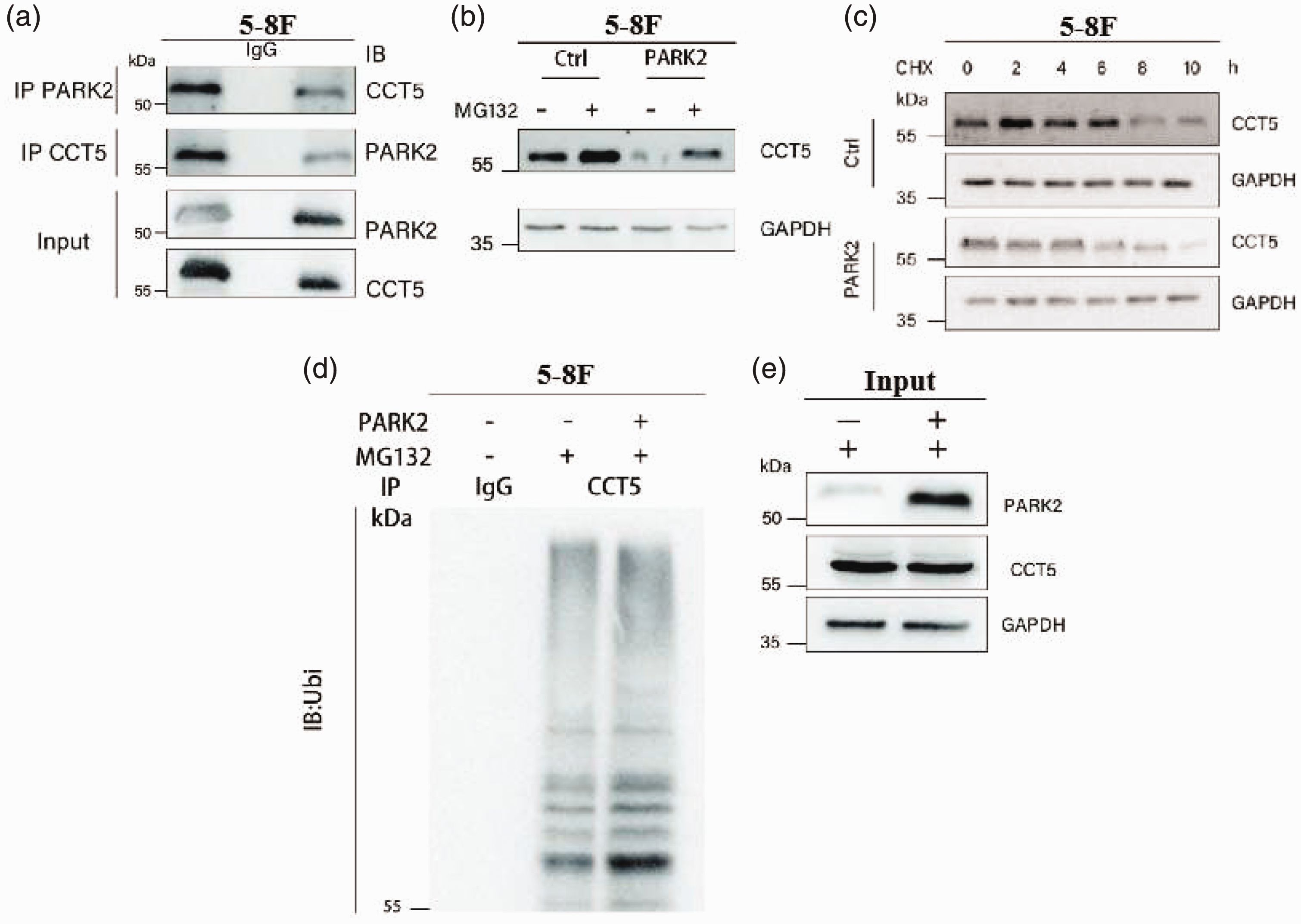
E3 ubiquitin-protein ligase parkin (PARK2) ubiquitinates and induces degradation of chaperonin containing TCP1 subunit 5 (CCT5) protein: (a) representative immunoblots showing co-immunoprecipitation of CCT5 and PARK2 components in 5-8F cells; (b) representative immunoblots of CCT5 in 5-8F cells transfected to overexpress PARK2, or controls (Ctrl) without PARK2 overexpression, treated with MG132 (20 µM) for 8 h, showing higher CCT5 levels in the presence of MG132; (c) representative immunoblots of time-course analysis of CCT5 protein levels in 5-8F cells with or without PARK2-overexpression incubated with cycloheximide (CHX), showing increased degradation of CCT5 in cells overexpressing PARK2; (d) polyubiquitination of CCT5 was analysed in 5-8F cells overexpressing PARK2. 5-8F cells with or without PARK2 overexpression were treated with MG132 (20 µM) for 8 h. Ubiquitin-conjugated CCT5 proteins from 5-8F lysates were immunoprecipitated by anti-CCT5 antibody and subjected to immunoblot with ubiquitin (IB:Ubi) antibody; and (e) representative immunoblots showing that MG132 suppresses PARK2-induced CCT5 protein degradation in 5-8F cells. IB, immunoblot (western blot); IP, immunoprecipitation; Input, positive controls.
Discussion
In previous studies, many genes have been reported to be involved in the carcinogenesis of NPC, however, the abnormal expression of these genes does not fully explain the pathogenesis of NPC. To find new biomarkers of NPC, the present group’s previously published microarray data of differentially expressed genes between NPC and noncancerous nasopharyngeal tissues was re-assessed and showed that CCT5 was upregulated in NPC tissues. 19 CCT5 is a molecular chaperone and a subunit of TRiC complex, which exerts critical functions in efficiently folding nascent and stress-denatured proteins. Dysfunction of the TRiC complex may lead to the cellular accumulation of misfolded proteins and induce diseases including cancers. 25 Being an important part of the CCT/TRiC complex, the role of CCT5 in various types of cancer is far from fully characterized to date. Xu et al. 26 reported that CCT5 mRNA levels were significantly elevated in breast cancer tissues compared with normal breast tissues, and high CCT5 expression was significantly associated with poor prognosis in patients with breast cancer. Furthermore, CCT5 expression was remarkably upregulated in p53-mutated breast cancer and associated with chemoresistance to docetaxel. Silencing CCT expression enhanced docetaxel-induced apoptosis. 27 In addition, increased CCT5 expression was also found in hepatocellular carcinoma and considered as an unfavourable prognostic factor. Gene set enrichment analysis, based on TCGA data, revealed that overexpressed CCT5 might be involved in the dysregulation of Myc target genes and cell cycle in hepatocellular carcinoma.28,29 In nonsmall cell lung cancer (NSCLC), not only was CCT protein upregulated in cancer tissues, but also the anti-CCT5 autoantibody level increased in sera, suggesting that CCT5 may be used as a biomarker in NSCLC diagnosis. 30 The aforementioned findings indicate that CCT5 is an oncogene in various human cancers; however, its specific role in human NPC remains unknown.
In the present study, qRT-PCR was applied to analyse CCT5 mRNA levels in fresh NPC and noncancerous nasopharyngeal tissues. Consistent with the microarray data and results of aforementioned studies, a significantly increased CCT5 mRNA level was found in NPC tissues compared with noncancerous nasopharyngeal tissues. Moreover, immunohistochemical analysis concurred with mRNA expression results, showing upregulated CCT5 protein levels in NPC tissues, with a 72.8% rate of high expression level in NPC samples versus 28.9% in noncancerous nasopharyngeal samples. The relationship between CCT5 protein levels and tumour progression was further analysed. Higher levels of CCT5 protein were found in patients with NPC who had larger tumours, advanced clinical stage, and recurrent tumours. However, CCT5 expression had no correlation with age, sex, and lymph node metastasis. These data demonstrated that overexpressed CCT5 may accelerate the clinical progression of NPC, suggesting it may be a risk factor in patients with NPC. Previous studies indicated that CCT5 overexpression was an unfavourable prognostic factor in various tumours,14,15 and the present evidence showed that elevated CCT5 protein levels inversely correlated with overall survival in patients with NPC. Patients with higher levels of CCT5 protein had shorter overall survival time. Multivariate Cox regression analysis confirmed that abnormal CCT5 expression was an independent prognostic factor for patients with NPC, suggesting that CCT5 as an oncogene may be a potential therapy target for NPC.
Rapid proliferation is a major feature of malignant tumours, including NPC. Hence, identifying biomarkers that promote tumour growth may help provide therapeutic targets for NPC treatment. CCT/TRiC is involved in cell proliferation due to its essential role in folding cell cycle-related proteins, including oncoproteins, such as cyclin E, cyclin B, and tumour suppressor protein p21 and p27. 11 P53 is a tumour suppressor gene frequently mutated in cancers, whose mutation alters the expression of various genes, leading to the malfunctioning of cell cycle arrest and apoptosis to promote tumour growth. Significantly increased CCT5 expression was found in p53-mutated breast cancer, indicating that CCT5 might be regulated by mutated p53, contributing to tumour proliferation. 27 In addition, increased CCT5 induces the malignant phenotype of tumours and is also shown in other tumour types.14,15,26 In the present study, high CCT5 expression was associated with larger tumour size based on clinical sample assay, suggesting CCT5 as the potential tumour growth-promoting factor in NPC. CCT5 was also shown to be a cell-proliferative gene stimulating NPC growth, intimating that CCT5 may be a potential oncogene in NPC.
To the best of the authors’ knowledge, the degradation mechanism of CCT5 protein in tumours has not been reported in previous studies. PARK2 is a tumour suppressive E3 ubiquitin-ligase enzyme that participates in the proteasome pathway to degrade other proteins.31,32 Inactivation of PARK2 is associated with a variety of cancers, and PARK2 depletion promotes the growth of liver, lung, and breast cancer cells.33,34 In addition, PARK2-deficient mice are more likely to develop tumours. 35 Interestingly, through overexpression, PARK2 was found to interact with CCT5 in the present study. Furthermore, PARK2 was shown to ubiquitinate and induce degradation of CCT5 protein. This result demonstrated that PARK2 may be an upstream factor negatively regulating CCT5 expression in NPC.
Conclusion
In summary, the present data demonstrated that an elevated CCT5 expression level may be an unfavourable factor promoting NPC pathogenesis. CCT5 is degraded by a PARK2-mediated ubiquitination pathway leading to suppression of NPC cell proliferation. These data suggest that CCT5 may be a potential oncogene that can be degraded by PARK2 in NPC.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241271754 - Supplemental material for Increased CCT5 expression is a potential unfavourable factor promoting the growth of nasopharyngeal carcinoma
Supplemental material, sj-pdf-1-imr-10.1177_03000605241271754 for Increased CCT5 expression is a potential unfavourable factor promoting the growth of nasopharyngeal carcinoma by Shaoyu Wu and Lingrong Peng in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605241271754 - Supplemental material for Increased CCT5 expression is a potential unfavourable factor promoting the growth of nasopharyngeal carcinoma
Supplemental material, sj-pdf-2-imr-10.1177_03000605241271754 for Increased CCT5 expression is a potential unfavourable factor promoting the growth of nasopharyngeal carcinoma by Shaoyu Wu and Lingrong Peng in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605241271754 - Supplemental material for Increased CCT5 expression is a potential unfavourable factor promoting the growth of nasopharyngeal carcinoma
Supplemental material, sj-pdf-3-imr-10.1177_03000605241271754 for Increased CCT5 expression is a potential unfavourable factor promoting the growth of nasopharyngeal carcinoma by Shaoyu Wu and Lingrong Peng in Journal of International Medical Research
Footnotes
Acknowledgements
We thank Prof. Weiyi Fang for providing the microarray data and paraffin-embedded samples of patients with NPC.
Author contributions
Shaoyu Wu, study conception and design; Lingrong Peng, critical article revision for important intellectual content, and final approval of the version to be submitted.
Data availability statement
A request for raw data may be made directly to the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
