Abstract
Background:
Many patients with lung cancer have underlying chronic lung diseases. We assume that baseline lung functions might also affect the prognosis of non-small cell lung cancer (NSCLC) patients receiving immunotherapy.
Objectives:
We aimed to assess the impact of pretreatment clinical parameters, including lung function measures such as forced vital capacity (FVC), on the prognosis of patients with NSCLC following immune checkpoint inhibitors (ICIs) therapy.
Design:
Retrospective multicenter study.
Methods:
Study subjects were consecutively selected from a multicenter cohort of patients with NSCLC who were undergoing immunotherapy. Patients were selected regardless of their initial cancer stage and prior treatment. The primary outcome was immunotherapy-related overall survival (iOS), defined as the duration from the initiation of immunotherapy to the time patients were censored. Spirometry values were acquired before bronchodilator application and were performed within the year before the first ICI treatment
Results:
We selected 289 patients for evaluation. The median iOS was 10.9 months (95% confidence interval (CI), 7.5–14.3). Programmed death-ligand 1 (PD-L1) expression, tested by SP263, was <1% in 20.9%, 1%–49% in 44.3%, and ⩾50% in 32.6% of the patients. ICI was used most often as second-line treatment (70.2%), followed by first line (13.1%), and third line (11.4%). In the Kaplan–Meier analysis, the median iOS of the low FVC group was significantly shorter than that in the preserved FVC group (6.10 (95% CI, 4.45–7.76) months vs 14.40 (95% CI, 10.61–18.34) months,
Conclusion:
Pre-immunotherapy FVC (%) predicted immunotherapy-related outcomes in NSCLC patients, regardless of initial stage at diagnosis and prior treatment modalities.
Introduction
Lung cancer, particularly non-small cell lung cancer (NSCLC) adenocarcinoma and squamous cell carcinoma, is the leading cause of cancer-related deaths worldwide. 1 Most NSCLC patients are diagnosed at an advanced stage. 2 Recently, immune checkpoint inhibitors (ICIs) have become the main treatment modality for advanced NSCLC, alongside traditional platinum-based chemotherapy.3,4
Previous studies investigated several prognostic biomarkers and clinicopathological factors related to the prognosis of patients receiving immunotherapy. 5 High programmed death-ligand 1 (PD-L1) expression is a biomarker widely used to predict immunotherapy responses, and other potential biomarkers, including the tumor mutational burden, are being studied.6,7 Baseline patient factors such as sex, performance status, and body mass index (BMI) have been analyzed for their association with progression free survival (PFS) and overall survival (OS). Neutrophil-to-lymphocyte ratio, lactate dehydrogenase, and C-reactive protein levels before ICI, have been associated with unfavorable outcomes in several studies.8–10 However, other than PD-L1 expression, no biomarkers are currently in wide use to predict ICI efficacy.
In patients with NSCLC, comorbid lung diseases are common, and NSCLC patients often exhibit reduced pulmonary function compared with individuals without lung cancer. 11 Previous studies have shown an association between impaired forced expiratory volume in 1 s (FEV1) and poor outcomes in lung cancer patients receiving cytotoxic chemotherapy. 12 Furthermore, a study has suggested that decreased baseline forced vital capacity (FVC) is a risk factor for the occurrence of chemotherapy-associated acute exacerbation of interstitial lung disease (ILD), which can increase the mortality rate in patients with NSCLC. However, analyses of the association between baseline lung function and prognosis in NSCLC patients undergoing immunotherapy are insufficient. One retrospective study found that a low FEV1 is associated with an inferior survival outcome in advanced NSCLC patients receiving ICIs. 13
Patients exhibiting a restrictive spirometry pattern with reduced FVC often have sequelae of previous lung infection or injury.14,15 Additionally, there is a prior research indicating that restrictive spirometry is associated with systemic inflammation. 16 Furthermore, a decrease in FVC is an indicator of lung function distinct from FEV1, as it can be influenced by factors like subclinical ILD, pleural abnormalities, or impaired lung growth. 17 FVC reduction not only reflects the state of lung parenchyma but also reflects poor performance status and is associated with various systemic comorbidities.18,19 Considering the previous evidences, FVC can be a valuable and more comprehensive clinical parameter, which had been understudied in terms of its impact on immunotherapy in NSCLC patients. In real-world management of lung cancer patients, treatment modalities, especially immunotherapy, are administered according to pre-specified guidelines, status of concurrent genetic alterations, or PD-L1 expression.20,21 However, clinicians should not only focus on the genetic and molecular pathological characteristics of the cancer but also consider a range of patient baseline factors and underlying conditions when predicting immunotherapy outcomes. Considering that the majority of lung cancer patients are more likely to have impaired lung functions compared to other solid tumors, a more personalized approach considering patients’ lung conditions is necessary. 22 Chronic lung diseases, such as emphysema, entail decreased lung function and accompany chronic inflammatory conditions of the lung parenchyma, which can affect the tumor microenvironment. In the airways and alveolar lumen of chronic obstructive pulmonary disease (COPD) patients, the number of CD4+ T cells increases significantly with airflow limitation and emphysema staging. 23 Studies have suggested that lung cancer patients with COPD may benefit more than non-COPD lung cancer patients in terms of clinical outcome, with more favorable PFS and response rates.24,25 While COPD has received much clinical attention for its prognostic value in lung cancer patients undergoing immunotherapy, other pulmonary function parameters are yet to be explored. In this context, assessing patients who have undergone immunotherapy with the consideration of high or low baseline FVC will be significant, as it can more comprehensively cover both comorbidities and lung functions compared to other pulmonary function parameters.
This study aims to assess the impact of pretreatment clinical parameters, including lung function measures such as FVC, on patient prognosis following ICI therapy, by analyzing a consecutively enrolled, multicenter cohort of patients with NSCLC.
Materials and methods
Patient selection
Study participants were patients with NSCLC who received ICI treatment, regardless of the number of prior non-immunotherapy treatment lines or their initial cancer stage (stage I–IV). These patients were consecutively selected from a multicenter cohort (Figure 1). Between January 2016 and December 2021, 1958 patients were diagnosed with lung cancer at seven university hospitals in South Korea: Seoul St. Mary’s Hospital, Yeouido St. Mary’s Hospital, Bucheon St. Mary’s Hospital, Eunpyeong St. Mary’s Hospital, St. Vincent Hospital, Incheon St. Mary’s Hospital, and Uijeongbu St. Mary’s Hospital. After excluding 217 patients diagnosed with small cell lung cancer (SCLC), 1741 patients remained. Out of that group, 289 patients who underwent ICI treatment were enrolled in this study cohort. Inclusion criteria for the study were: (1) diagnosed with NSCLC and (2) underwent immunotherapy. The exclusion criterion was: (1) patients with SCLC and (2) patients without consent for data use.

Study flowchart.
OS and PFS
Immunotherapy-related overall survival (iOS) was defined as the time between the date of ICI initiation and the date of death from any cause or last known date alive. PFS was defined as the duration from ICI initiation to the date of initial disease progression. Patients underwent computed tomography scans after every two treatment cycles to evaluate their response. This evaluation was conducted by the treating physicians and independent radiologists, and the response evaluation was performed using Response Evaluation Criteria in Solid Tumors version 1.1.
26
Among the selected patients, 141 patients were eligible for the PFS analysis. If patients’ best response was a partial response or complete response, they were classified as
Assessments and data collection
The following clinicopathological data were collected for all enrolled patients: age, sex, smoking history, Eastern Cooperative Oncology Group performance status (ECOG PS), tumor characteristics (histology, cancer stage, and mutation status), and treatment modalities. Spirometry values were acquired before bronchodilator application and were performed within the year before the first ICI treatment. The spirometry tests were performed using a VMAX22 ENCORE spirometer and V62J plethysmographic cabinet, VYAIRE-USA, VMAX Carefusion software, and were performed by qualified technicians. The reporting data were interpreted in accordance with the American Thoracic Society/European Respiratory Society guidelines. 27 The cut-off value used to distinguish between the low FVC group and the preserved FVC group was set at 70% of the predicted FVC. 28
For treatment data, regimens other than pembrolizumab + pemetrexed + platinum, and monotherapy regimen of pembrolizumab, nivolumab, durvalumab, or atezolizumab were classified as “other regimens.” This group included regimens such as mk7684a + pembrolizumab, pemetrexed + platinum + durvalumab, gemcitabine + platinum + durvalumab, atezolizumab or placebo + nab-paclitaxel + platinum, nivolumab + ipilimumab, durvalumab + vactosertib, and bevacizumab + atezolizumab + paclitaxel + platinum. The reporting of this study conforms to the STROBE Statement (Supplemental Material).
Subgroup analysis
In order to evaluate outcomes in a less heterogeneous subgroup, we excluded patients harboring epidermal growth factor receptor (EGFR) or anaplastic lymphoma kinase (ALK) mutations and those who underwent regimens including platinum chemotherapy. A subgroup of 195 patients underwent separate analyses for general clinical characteristics, baseline factors associated with FVC < 70%, and survival analysis.
Statistical analysis
The baseline demographics and clinical features of patients were compared between the low FVC and preserved FVC groups. Additionally, the entire patient cohort was divided into responders and non-responders based on their best responses to ICI treatment. Data for continuous variables are presented as means with standard deviations. The Chi-square test was performed to compare categorical parameters, and continuous variables were compared using two-sided
Results
Patient characteristics and treatment
We enrolled 289 patients in this study, and the median time from ICI treatment initiation to disease progression was 5.03 ± 0.93 months (95% confidence interval (95% confidence interval (CI)), 3.21–6.58 months). The median iOS was 10.90 ± 1.71 months (95% CI, 7.55–14.25 months). Within the study population, 253 patients underwent pulmonary function testing (PFT) prior to their first ICI, and those results placed 197 (77.9%) patients in the preserved FVC group and 56 patients (22.1%) in the low FVC group. The baseline characteristics of all participants and the two FVC groups are summarized in Table 1. The mean age, sex, BMI, and smoking history did not differ significantly between the groups. The proportion of patients with ECOG PS ⩾ 2 was higher in the low FVC group than the preserved FVC group (8.9% vs 2.5%,
Clinical characteristics of the overall study population.

Continuous variables are presented as mean ± standard deviation, and categorical variables as number (%),
Others include NSCLC not specified, adenosquamous carcinoma, large cell carcinoma, and sarcomatoid type.
ALK, anaplastic lymphoma kinase; BMI, body mass index; DLco, diffusing lung capacity for carbon monoxide; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; FEV1, forced expiratory volume in 1 s; FEV1/FVC, FEV1-to-FVC ratio; FVC, forced vital capacity; LN, lymph node; MPE, malignant pleural effusion; NSCLC, non-small cell lung cancer; PD-L1, programmed death-ligand 1; PFT, pulmonary function test; RV/TLC, residual volume/total lung capacity.
Table 2 compares lung cancer treatment data between the low and preserved FVC groups. The median number of ICI cycles administered to all patients was 4 (range, 1–42 cycles), and 203 (70.2%) patients received ICI as second-line treatment. The most frequently used regimen was atezolizumab monotherapy (32.2%). The proportion of patients who underwent surgery prior to ICI was significantly higher in the preserved FVC group (5.5% vs 25.0%,
Patient characteristics based on treatment modality.

Continuous variables are presented as mean ± standard deviation, and categorical variables as number (%).
FVC, forced vital capacity; pemed, pemetrexed; PFT, pulmonary function test.
Clinical factors associated with low FVC
To identify risk factors associated with low FVC, a logistic regression analysis was performed, and the results are presented in Table 3. In the univariate logistic regression analyses, poor PS (ECOG score ⩾2; odds ratio (OR), 3.765;
Risk factors associated with low FVC in study patients.

Categorical variables represented as number (%).
ALK, anaplastic lymphoma kinase; BMI, body mass index; COPD, chronic obstructive pulmonary disease; DLco, diffusing lung capacity for carbon monoxide; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; FEV1, forced expiratory volume in 1 s; FEV1/FVC, FEV1-to-FVC ratio; FVC, forced vital capacity; PD-L1, programmed death-ligand 1.
Treatment response and PFS in patients treated with immunotherapy
Table 4 compares patients based on their best response to ICI, categorized as responders and non-responders. A total of 223 patients were eligible for the immunotherapy treatment response evaluation. Among them were 161 (72.2%) non-responders and 62 (27.8%) responders. No significant differences between groups were observed in the clinicopathological factors. Although responders demonstrated higher mean FVC, FEV1, and DLco values than non-responders, those differences were not statistically significant.
Patient characteristics based on best treatment response.

Continuous variables are presented as mean ± standard deviation, and categorical variables as number (%).
Others include NSCLC not specified, adenosquamous carcinoma, large cell carcinoma, and sarcomatoid type.
ALK, anaplastic lymphoma kinase; COPD, chronic obstructive pulmonary disease; CR, complete response; DLco, diffusing lung capacity for carbon monoxide; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; FEV1, forced expiratory volume in 1 s; FEV1/FVC, FEV1-to-FVC ratio; FVC, forced vital capacity; NSCLC, non-small cell lung cancer; PD, progressive disease; PD-L1, programmed death-ligand 1; PFT, pulmonary function test; PR, partial response; SD, stable disease.
In the Kaplan–Meier analysis, the low FVC group had a significantly shorter median PFS than the preserved FVC group (2.99 (95% CI, 1.25–4.74) months vs 5.26 (95% CI, 2.85–7.68) months,
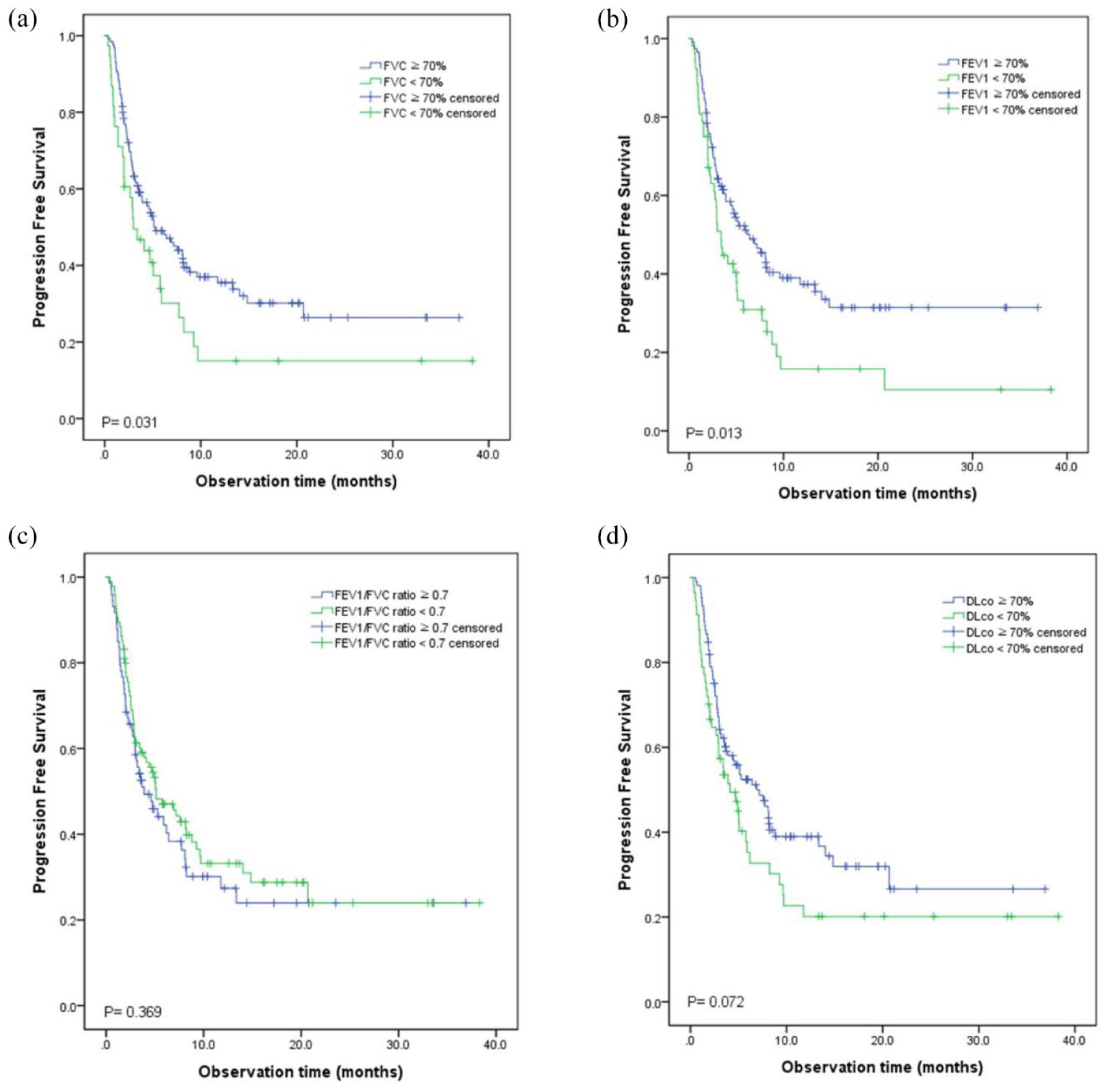
Kaplan–Meier curves for PFS by (a) FVC < 70% predicted. (b) FEV1 < 70% predicted. (c) FEV1/FVC ratio < 0.7. (d) DLco < 70% predicted as cut-off values.
In the univariate Cox analyses, male sex, poor PS, the presence of a concurrent intrathoracic lesion at the time of diagnosis, and PD-L1 (SP263) expression <1% were significant predictive factors for poor PFS. Conversely, high FVC (%) and FEV1 (%) values were associated with improved PFS (Supplemental Table S1). Based on those findings, the multivariable analysis, which included a total of 158 patients, indicated that male sex, poor PS, and the presence of a concurrent intrathoracic metastasis at the time of diagnosis were significantly associated with poor PFS, and PD-L1 (SP263) expression ⩾50% was associated with improved PFS. FVC (%) did not demonstrate a statistically significant association with PFS in the multivariable analysis, and when FVC (%) was replaced with FEV1 (%), FEV1 (%) also did not show a statistically significant relationship with PFS.
iOS in patients treated with immunotherapy
During the observation period from January 2016 to February 2022, among the 253 patients who underwent PFT, 38 patients (67.9%) in the low FVC group and 94 patients (47.7%) in the preserved FVC group died. In the Kaplan–Meier analysis, the median iOS of the low FVC group was significantly shorter than that in the preserved FVC group (6.10 (95% CI, 4.45–7.76) months vs 14.40 (95% CI, 10.61–18.34) months,

Kaplan–Meier curves for iOS in NSCLC patients who received ICI, stratified by FVC with a cut-off value of 70% predicted. (a) All study patients. (b) Initially stage IV patients.

Kaplan–Meier curves for iOS when stratifying the cohort based on COPD status and PFT parameters. (a) COPD reported in the medical records. (b) COPD defined by spirometry. (c) FEV1 < 70% predicted. (d) DLco < 70% predicted as cut-off values.
The results of the univariate and multivariable Cox analyses for iOS are shown in Table 5. Among the pulmonary function parameters, low FVC (%; hazard ratio (HR), 0.982; 95% CI, 0.973–0.991;
Univariate and multivariable Cox regression analyses of clinicopathologic variables affecting iOS in NSCLC patients receiving ICI.

In separate multivariable analyses, each of FEV1 (%), DLco (%), and absolute DLco was entered in place of FVC (%). All of these parameters showed a significant association with iOS in multivariable analyses. FEV1 (%)
BMI, body mass index; CI, confidence interval; DLco, diffusing lung capacity for carbon monoxide; ECOG PS, Eastern Cooperative Oncology Group performance status; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; HR, hazard ratio; ICI, immune checkpoint inhibitor; iOS, immunotherapy-related overall survival; NSCLC, non-small cell lung cancer; PD-L1, programmed death-ligand 1; PFT, pulmonary function test.
Subgroup analyses (patients who do not harbor EGFR or ALK mutations or underwent platinum-containing regimens)
General characteristics
After excluding patients with EGFR or ALK mutations and those who underwent platinum-containing regimens, a total of 195 patients were analyzed. When stratified by an FVC cutoff of 70%, 150 patients had FVC ⩾ 70%, and 45 patients had FVC < 70%. There was no significant difference between the two groups regarding baseline patient factors, other than pulmonary function parameters (Supplemental Table S2).
Supplemental Table S3 shows a comparison of treatment-related parameters between the two groups. Significant differences were observed in the immunotherapy regimen and the best response to immunotherapy. The FVC < 70% group had a significantly higher proportion of patients treated with pembrolizumab (40.0% vs 27.3%,
Association with FVC < 70% in the subgroup
When various parameters including age, BMI, ECOG score, smoking history, and initial clinical stage were analyzed for their association with FVC < 70%, only DLco (%) < 0.7 showed a significant association (OR = 7.655,
Comparison between responders and non-responders
Among the subgroup, there were 157 patients evaluable for immunotherapy response assessment. There were 122 patients included in the non-responders group and 35 patients included in the good responders group. There were no significant differences between the baseline parameters, including pathology, stage at diagnosis, PD-L1 expression, and pulmonary functions parameters (Supplemental Table S5).
iOS analysis in the subgroup
In the univariate analysis, significant factors included sex, ECOG performance score, PD-L1 expression, stage at diagnosis, FVC (%), FEV1 (%), and DLco (%). In the multivariable analysis, where FVC (%) was included among pulmonary function parameters, the significant predictors of iOS were male sex (HR: 3.020, 95% CI: 1.542–5.914,
Discussion
This study evaluated the impact of baseline pulmonary function on the iOS of NSCLC patients undergoing immunotherapy. Older age, male sex, poor PS, and decreased baseline lung function were identified as factors associated with poor iOS in NSCLC patients treated with ICIs. Low FVC, FEV1, and DLco were identified as significant factors associated with poor iOS. However, the FEV1/FVC ratio was not associated with PFS or iOS. In separate analyses based on the stage at diagnosis, pathologic type, smoking status, and PD-L1 expression, we consistently observed that the low FVC group, defined as patients with FVC < 70%, exhibited significantly lower iOS in each subgroup. The low FVC group showed a higher prevalence of concurrent intrathoracic metastasis, malignant pleural effusion, and poor PS. In addition, the low FVC group exhibited significantly lower mean FEV1 and DLco values.
A decreased FVC is associated with intrinsic lung factors such as lung parenchymal damage and various lung diseases that cause problems in the pulmonary parenchyma, including different types of ILDs, post-infection sequelae, pneumoconiosis, drug-induced pneumonitis, and pulmonary vasculitis.29,30 In addition to lung parenchymal abnormalities, factors such as respiratory muscle weakness, rib cage deformities, and pleural effusion can also lead to a decrease in FVC, which is associated with an increased risk of all-cause mortality.31,32 In the general population, FVC < 80% in adults is a risk factor for increased risk of cardiovascular mortality. 33 Furthermore, low FVC is associated not only with functional impairment but also with various comorbid conditions, including metabolic syndrome, diabetes, hypertension, and stroke.34–36 Thus, FVC serves as an indicator of a different aspect of pulmonary status from FEV1, and it also holds distinct clinical significance in representing the overall systemic condition and underlying diseases of patients, differing from the implications of FEV1. Given that NSCLC patients undergoing immunotherapy are dealing with systemic disease, FVC’s ability to reflect the patient’s overall systemic condition beyond pulmonary status suggests its potential as an independent risk factor. In our study, the FVC < 70% group showed a significantly higher proportion of patients with poor ECOG performance scores, intrathoracic metastases at diagnosis, and larger tumor mass, as demonstrated in the comparative analysis. Beyond the impact of decreased lung function, these concurrent conditions could have also contributed to the worse survival observed in this group. Previous research on lung function in lung cancer patients showed that a low FVC is associated with worse OS in NSCLC patients undergoing curative resection. 37 Furthermore, several studies have reported associations between pretreatment FVC impairment and acute exacerbation of ILD 38 and ICI-pneumonitis 39 in NSCLC patients receiving cytotoxic chemotherapy. To the best of our knowledge, few studies have investigated the association between FVC as a marker of pre-immunotherapy lung function and clinical outcomes such as iOS in patients undergoing immunotherapy. To minimize the impact of concurrent driver mutations (EGFR and ALK mutations) and variability in treatment regimens, we conducted a subgroup analysis excluding patients with targetable mutations and those who received platinum-based treatments. The independent association between FVC and iOS remained significant in the subgroups analysis. While prospective studies are necessary to confirm the association between FVC and immunotherapy-related outcomes, clinicians identifying potential risk factors associated with decreased FVC (%) before immunotherapy could be beneficial to NSCLC patients. Additionally, regular surveillance for the aggravation of concurrent comorbidities, including chronic lung disease, should be conducted when managing NSCLC patients undergoing immunotherapy with decreased baseline FVC.
In addition to findings on FVC, our study revealed significant observations about the impact of COPD on NSCLC patients receiving immunotherapy. Previous reports indicate that NSCLC patients with coexisting COPD undergoing immunotherapy may experience longer PFS compared to those without COPD.40,41 Mark et al. demonstrated, based on resected human lung tissue and a murine cigarette smoke exposure model, that COPD-affected lung tissue displayed increased Th1 differentiation and that PD-1 expression was elevated in tumors of patients with COPD. 40 However, the influence of underlying chronic lung diseases on the efficacy of immunotherapy in NSCLC remains controversial. In our study, when patients were categorized by self-reported COPD, those with COPD had significantly worse iOS compared to non-COPD patients, suggesting that the presence of COPD does not positively impact immunotherapy-related outcomes. We believe that the presence of COPD, along with other comorbidities and decreased lung functions, may negatively impact patient prognosis. Thus, the adverse effects associated with COPD likely outweigh any potential positive impact on the tumor microenvironment.
In this study, the low FVC group showed significantly lower mean FEV1 and DLco values than patients with preserved FVC. In the multivariable Cox analysis, substituting FVC (%) with FEV1 (%), DLco (%), or absolute DLco showed that each pulmonary function factor significantly associated with iOS. This suggests that a decrease in lung function parameters, including FVC, prior to ICI can contribute significantly to poor prognosis in NSCLC patients receiving ICI. A previous study demonstrated that patients with advanced NSCLC receiving ICI therapy had worse PFS and OS when their FEV1 was less than 80%. 13 The present study supports those findings by showing that a low FEV1 is associated with unfavorable outcomes in NSCLC patients receiving ICI.
A low DLco is associated with CT-detected emphysema and an increased risk of lung cancer mortality in COPD patients.42,43 It has been reported that chemotherapy and radiotherapy can induce pulmonary toxicity, leading to a significant decline in DLco in NSCLC patients that is further exacerbated by their combination.44,45 The PFTs, including DLco, used in this study, were performed within 1 year before the initiation of ICI, with 86.9% of patients starting immunotherapy as second-line therapy or beyond and 25.7% of patients receiving concurrent chemoradiotherapy. Therefore, the decrease in DLco might have been influenced by pre-ICI treatments, indicating that our analysis alone is insufficient to consider DLco as a standalone predictive factor for ICI. Further investigation is needed to show the potential prognostic value of DLco in patients undergoing ICI therapy.
It was also found that historical parameters associated with iOS were consistent in our studies. Age, PD-L1 expression at diagnosis, and ECOG score showed significant association with iOS in multivariable analysis. Age, as with other chemotherapy treatments for NSCLC, was shown to be an independent predictor for iOS. PD-L1 expression was associated with iOS in patients undergoing immunotherapy, consistent with previous findings.46,47 Poor ECOG scores have been shown to be associated with PFS and OS in meta-analyses. 48
This study has several limitations. First, the study was retrospective, and sample heterogeneity may not have been adequately accounted for, so we performed a subgroup analysis in a group with less variability. The absence of rigorous inclusion and exclusion criteria may limit final results. More efforts should be conducted in a possible prospective trial. Second, although recruiting patients from all stages, 36 out of the enrolled participants lacked PFT values. In an effort to maximize the sample size and investigate the presence of other risk factors beyond PFT, these patients were also included in the analysis. A subsequent study with a larger cohort of patients is warranted in the future. Third, although previous studies have suggested an association between low FVC and immune-related adverse events (irAEs), this study was unable to confirm that association due to the small number of patients exhibiting irAE. Finally, FVC values could have been influenced by pre-immunotherapy treatment toxicities or recent infections. Although only a small percentage of patients received prior radiotherapy to lung lesions in the present study, these factors should be considered in future prospective studies. The results of this study should be interpreted with the understanding that patient heterogeneity could have influenced the outcomes. For example, in patients who underwent EGFR Tyrosine kinase inhibitor (TKI) treatment prior to immunotherapy, the type of EGFR mutation or TKI regimen could have impacted the FVC value. 49
Despite those limitations, our study is multi-institutional and has demonstrated that a low FVC, independent of treatment line and stage, is associated with worse outcomes following immunotherapy. This finding indicates FVC’s potential as a clinical factor that can be used with proven biomarkers such as PD-L1 to predict ICI treatment outcomes. Nonetheless, it requires a separate validation study. Additional large-scale prospective studies to examine patterns in FVC changes before and after ICI treatment, as well as the long-term prognosis associated with decreased FVC, could clarify the correlation between pulmonary function and prognosis in patients receiving ICIs. After validating the potential of FVC as a biomarker through such studies, the use of PFTs before ICI administration, irrespective of the presence of dyspnea, could provide clinicians with comprehensive prognostic information for NSCLC patients undergoing ICI.
Conclusion
In conclusion, pre-immunotherapy FVC (%) was associated with immunotherapy-related outcomes in NSCLC patients, regardless of the initial stage at diagnosis and prior treatment modalities. Conducting pretreatment spirometry in NSCLC patients receiving ICI could assist in predicting their prognosis. Further research is needed to investigate whether improving lung function through pulmonary rehabilitation before and during ICI treatment can improve the clinical outcomes of patients with NSCLC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241281480 – Supplemental material for Low pre-immunotherapy forced vital capacity is associated with poor outcomes in non-small cell lung cancer patients receiving immunotherapy regardless of prior treatment history
Supplemental material, sj-docx-1-tam-10.1177_17588359241281480 for Low pre-immunotherapy forced vital capacity is associated with poor outcomes in non-small cell lung cancer patients receiving immunotherapy regardless of prior treatment history by Jeong Uk Lim, Hye Seon Kang, Chang Dong Yeo, Ju Sang Kim, Sung Kyoung Kim, Jin Woo Kim, Seung Joon Kim and Sang Haak Lee in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241281480 – Supplemental material for Low pre-immunotherapy forced vital capacity is associated with poor outcomes in non-small cell lung cancer patients receiving immunotherapy regardless of prior treatment history
Supplemental material, sj-docx-2-tam-10.1177_17588359241281480 for Low pre-immunotherapy forced vital capacity is associated with poor outcomes in non-small cell lung cancer patients receiving immunotherapy regardless of prior treatment history by Jeong Uk Lim, Hye Seon Kang, Chang Dong Yeo, Ju Sang Kim, Sung Kyoung Kim, Jin Woo Kim, Seung Joon Kim and Sang Haak Lee in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
