Abstract
Malignant PEComas are an extremely rare subtype of soft tissue sarcomas. Here, we report the case of a man presenting with a perirectal PEComa and liver metastasis. Since the tumor harbored a tumor mutational burden of 23/Mb and a programmed death-ligand 1 tumor positivity score of 50%, the patient was treated with pembrolizumab as a second line of systemic therapy, in combination with everolimus. This combined therapy led to a near-complete response of the primary tumor and a partial response of the metastasis. Radioembolization of the liver metastasis was performed due to isolated liver progression, and the pelvic tumor was treated by radiotherapy because of pelvic symptoms. The disease is still stable after 13 months of pembrolizumab plus everolimus and multimodal treatment. This case shows that malignant PEComas can display molecular features associated with sensitivity to checkpoint inhibitors. The use of checkpoint inhibitors may be a relevant therapeutic strategy in these patients. It is also the first report on selective internal radiation therapy in PEComas.
Plain language summary
This article reports the case of a patient presenting with a very rare tumor type called “PEComa”. The tumor originated from the rectum and had disseminated to the liver. Since this tumor is very rare, there is a lack of knowledge on which treatments to use, and every case reporting the use of new treatments in PEComas is helpful. Here, the tumor displayed molecular alterations that suggested that it would respond to immunotherapy, such as a high number of mutations. Therefore, the patient was treated with an immunotherapy called pembrolizumab, in combination with another medication (everolimus). The rectal tumor nearly disappeared under treatment, and the liver metastasis decreased in size. The patient had radiotherapy of the rectum because of rectal bleeding. For the liver metastasis we used another technique called radioembolization, that consists in delivering radioactive compounds directly in the metastasis through the bloodstream. Now, he has received immunotherapy for 13 months and the disease is still under control. This case shows that immunotherapy can be a good treatment option in PEComas. It is also the first time that a medical team reports the using radioembolization to treat a PEComa.
Keywords
Introduction
Perivascular epithelioid cell tumors (PEComas) are an ultra-rare tumor type. Most PEComas are benign, but some have malignant potential and are then considered a rare subtype of soft tissue sarcomas. While the uterus is the most frequently affected site, several cases of malignant PEComas located along the gastrointestinal tract have been reported, such as in the rectum1,2 or liver. 3 The molecular signature of malignant PEComas involves recurrent loss of function mutations in TSC1 or 2.
Evidence on the use of systemic therapies for metastatic PEComa is low and relies mostly on retrospective cohorts and case reports. Advanced PEComas display a poor response to cytotoxic chemotherapy (around 20%). 4 Other treatment options include anti-angiogenic agents or mTOR (mammalian target of rapamycin) inhibitors. A single-arm phase II trial reports a response rate of 39% with prolonged responses in 31 patients treated with nab-Sirolimus for a malignant PEComa. 5 The presence of a TSC2 mutation was shown to be predictive of response to nab-Sirolimus. 5 There is no robust data on the use of other therapies, such as immunotherapy, in metastatic PEComa.
Here, we report a case of a patient with metastatic PEComa of the rectum treated with pembrolizumab. This article was written according to the CARE guidelines for case reports. 6
Case
We report the case of a 56-year-old man with a personal history of acute myeloid leukemia treated by allograft (11 years ago), squamous cell carcinoma of the tongue, and sebaceous carcinoma, both treated by surgery.
By the end of 2022, the patient presented with lower abdominal pain and recurrent fever. Abdominal computed tomography scan and magnetic resonance imaging (MRI) showed a pararectal mass of 12 × 9 × 10 cm invading the prostate and right seminal vesicle (Figure 1(a)). The positron emission tomography scanner showed hypermetabolic lymph nodes in the aorto-caval and lower mesenteric areas. The initial morphological analysis of a pararectal biopsy sample was suggestive of an epithelioid gastrointestinal stromal tumor (GIST). However, no mutation was found in the KIT or PDGFRA genes, no loss of expression of SHDB, and the disease progressed after 1 month of imatinib with the development of liver metastasis. This metastasis was biopsied, and the histological examination showed the same morphological features as in the primary tumor.

CT scans showing the primary tumor and the liver metastasis. (a) Contrast-enhanced portal phase CT scan showing from left to right: baseline imaging, week 2 post-embolization with extensive tumoral necrosis, and 1-year follow-up after radiotherapy. (b) Contrast-enhanced portal phase CT scan showing from left to right: baseline imaging, 3-month partial response with decreased tumor enhancement.
The diagnosis of malignant PEComa was based on the coexpression of Human Melanoma Black-45 and smooth muscle markers in epithelioid and clear cells, together with atypia, mitoses, and a very high Ki67 index (Figure 2(a) and (b)). Tumor molecular profiling using next-generation sequencing identified a missense mutation in the exon 3 of TSC2 (p.Thr70Profs*36), as well as deleterious mutations in P53, RB1, and a high tumor mutational burden (TMB: 23/Mb). Moreover, a CHM::ATRX fusion transcript was identified by RNAseq analysis. Programmed death-ligand 1 (PDL1) was expressed on 50% of tumor cells, corresponding to a tumor positivity score (TPS) of 50%.
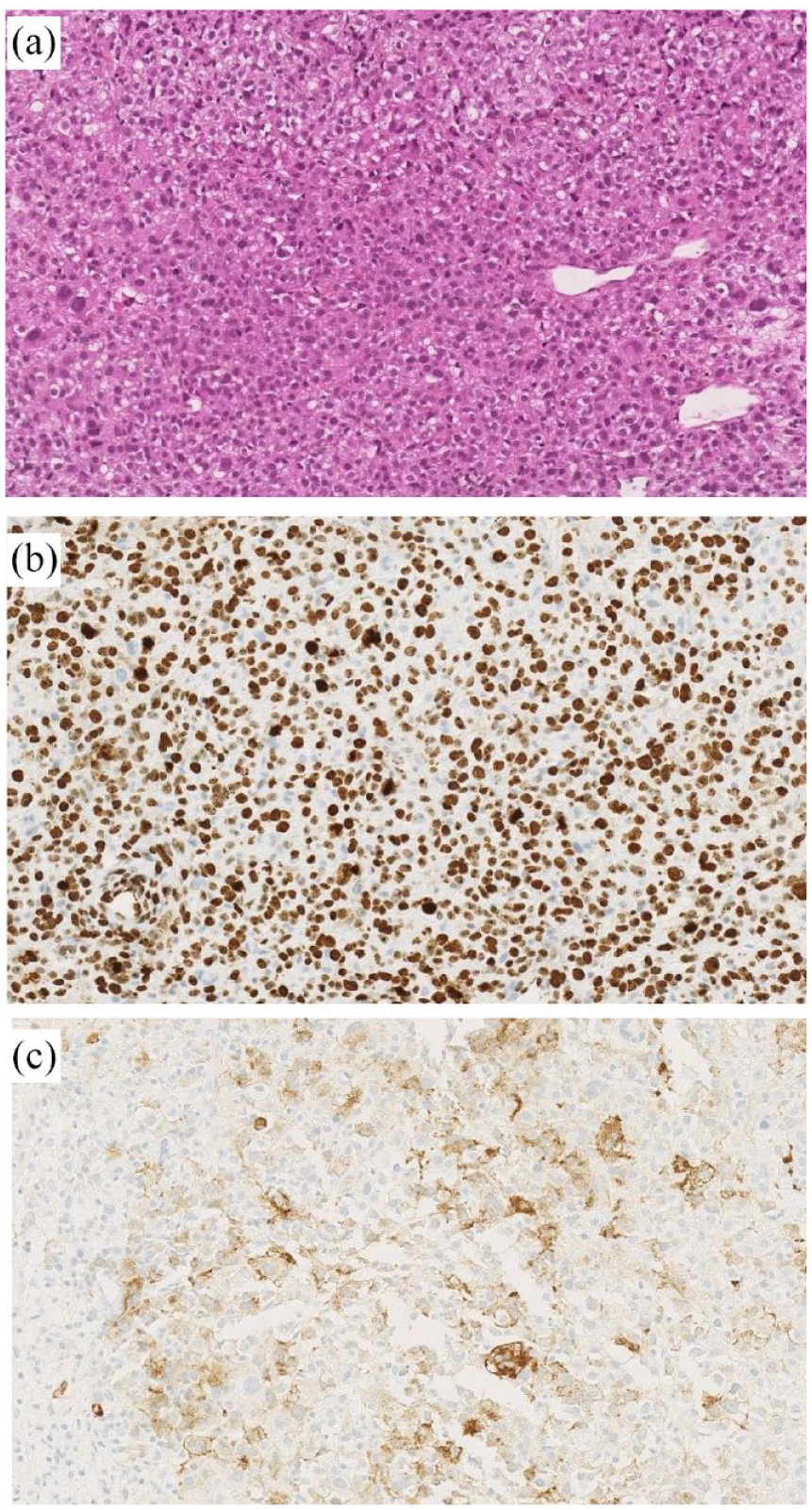
Pathological examination of the liver metastasis. (a) Hematoxylin and eosin-stained section showing a high density of epithelioid and clear cells displaying atypia and mitoses. (b) Ki67 staining. (c) PDL1 staining used for the computation of the PDL1 TPS score (50%).
The patient was hospitalized for lower gastrointestinal bleeding requiring repeated transfusion. The colonoscopy showed luminal invasion by the tumor. Radiological embolization of the superior rectal arteries was performed and stopped the bleeding. The patient also underwent a colostomy due to mechanical obstruction by the tumor.
Since the diagnosis of PEComa was initially not available, a platinum-based doublet was chosen as first-line chemotherapy, as recommended for cancers of unknown primary. 6 Chemotherapy using the FOLFOX regimen (5-fluorouracil (5FU) and oxaliplatin) led to an initial partial response on the rectal tumor, but the disease progressed, and the bleeding recurred after 2 months of chemotherapy.
Due to the high TMB and the TPS of 50%, the checkpoint inhibitor pembrolizumab was chosen as second-line therapy, in combination with hemostatic pelvic radiotherapy (30 Gy in 10 fractions). After 3 months, the pelvic mass and liver metastasis remained stable according to iRECIST. Since the mutation in TSC2 is predictive of sensibility to mTOR inhibitors, everolimus was added in combination with pembrolizumab. After 2 months of treatment with everolimus and 5 months of pembrolizumab, the patient had a nearly complete response on the pelvic tumor (not measurable vs 137 × 95 mm before pembrolizumab) and a 30% decrease in the liver metastasis (Figure 1(a) and (b)).
However, an isolated liver progression occurred after 4 months of combined therapy. The liver metastasis was treated by selective internal radiation therapy (SIRT) while systemic therapy was continued. The post-procedure liver MRI, performed 1 month after SIRT, showed stability of the liver metastasis, and the most recent evaluation, 3 months after SIRT (13 months since the beginning of immunotherapy), shows signs of tumor necrosis and a non-significant increase in the size of the lesion (82 vs 77 mm).
Concerning the primary pelvic tumor, after 7 months of combined therapy, the patient experienced a local progression causing pelvic pain, and a second pelvic stereotactic radiotherapy was performed (20 Gy in five fractions). To date, the patient has received pembrolizumab for 13 months and everolimus for 10 months.
Discussion
Here, we chose the checkpoint inhibitor pembrolizumab as the second line of systemic therapy in a patient with metastatic PEComa. This choice relies on the fact that the tumor harbored a high TMB (23 mut/Mb) and elevated PDL1 TPS score (50%), two features predictive of sensitivity to checkpoint inhibitors. For example, a TPS > 1% was shown to predict a benefit from pembrolizumab monotherapy in patients with stage III or IV non-small-cell lung cancer, 7 and the same threshold of 1% is used to guide the prescription of combined chemo-immunotherapy in squamous cell oesophageal carcinoma. 8 In these trials, the magnitude of benefit drawn from pembrolizumab was even broader in the subgroups of patients with TPS > 10 or TPS > 20 than those with TPS > 1. The high tumor molecular burden is also predictive of the sensitivity to checkpoint inhibitors in solid tumors: for example, a TMB > 10 mut/Mb was shown to predict the response to pembrolizumab in a tumor-agnostic trial. 9
There are very few reports on the use of checkpoint inhibitors for malignant PEComa. 10 In a retrospective cohort of patients with locally advanced or metastatic malignant PEComa, the median TMB was 2.5 (range: 1–17). 2 Among these patients, three received immune checkpoint inhibitors: one patient had progressive disease on nivolumab + ipilimumab, one had a partial response to pembrolizumab + radiotherapy, and one had a complete response to pembrolizumab monotherapy. The patient exhibiting a complete response had a TMB of 17, while the TMB was <2 in the other two patients. 2 In another case report, a patient was treated with pembrolizumab for lymph node recurrence after surgical resection of a PEComa of the abdominal wall. 10 A checkpoint inhibitor was chosen for this patient because the tumor displayed a ‘high PD1 expression’. The patient had a complete response. Finally, a patient presenting with metastatic recurrence of a renal angiomyolipoma (a rare subtype of PEComa) was treated with nivolumab monotherapy, due to a high PDL1 expression (>50%). 11 This patient also experienced a complete response, and nivolumab was stopped after 2 years of therapy. There is no published case of combined use of pembrolizumab plus everolimus in malignant PEComa. In our patient, pembrolizumab plus everolimus in combination with radiotherapy induced a nearly complete response on the primary perirectal tumor and a partial response on the liver metastasis. This resulted in a complete disappearance of fever, bleeding, and pelvic pain, as well as an improvement in performance status.
Due to the delayed diagnosis of PEComa, this patient was treated with 5FU and oxaliplatin as first-line chemotherapy, a regimen that is rarely used in PEComas and not recommended in soft tissue sarcomas in general. Anthracycline-based chemotherapy could therefore be considered an alternative treatment option for this patient. However, in an international retrospective cohort on advanced PEComas, 4 in which 23 patients received anthracyclines, an anthracycline-based regimen showed poor efficacy, with a median PFS of 3.2 months.
Another innovative aspect of this patient is the use of SIRT as a local treatment of liver metastases. To our knowledge, there are no reports on the use of this technique in malignant PEComa. Here, the use of SIRT permitted local control of a liver metastasis that displayed isolated progression while the primary tumor was still responding to systemic therapy. The extensive tumor necrosis seen on the post-intervention MRI suggests that PEComas may be sensitive to this technique.
In our patient, molecular profiling of the tumor identified a CHM::ATRX fusion transcript. Because of a frameshift, this fusion is expected to result in a loss of ATRX expression. The CHM::ATRX fusion has not yet been reported in cancers, and it is absent from public repositories. However, loss of function mutations in ATRX are among the most prevalent mutations in PEComas 12 and other sarcoma subtypes such as leiomyosarcoma, 13 together with mutations in TP53 and RB1. ATRX (alpha thalassemia/mental retardation syndrome X-linked) acts as a tumor suppressor by regulating the elongation of telomeres, and the loss of this gene could also promote immune escape. 14
Conclusion
This case shows that malignant PEComas can express markers predictive of sensitivity to checkpoint inhibitors, such as elevated TPS/CPS scores or a high TMB. Therefore, we suggest that evaluating TMB and TPS scores should be considered in all patients presenting with malignant PEComas. The combination of checkpoint inhibitors with everolimus is safe and well tolerated; it can induce partial responses in at least some patients with malignant PEComas. In our patient, a multimodal approach combining local therapies (radiotherapy, SIRT) and systemic therapies allowed for a prolonged control of an otherwise aggressive disease.
