Abstract
Background:
Real-world data on C-MET protein overexpression in non-small cell lung cancer (NSCLC) patients, particularly among the Asian Chinese population, are limited.
Objectives:
This study aimed to evaluate the clinicomolecular characteristics and prognosis of C-MET overexpression in Chinese NSCLC patients, focusing on those with positive C-MET overexpression (immunohistochemistry (IHC) 3+).
Design:
A retrospective and observational study.
Methods:
Data were collected from NSCLC patients diagnosed at the First Affiliated Hospital of Guangzhou Medical University between November 2006 and April 2021. We identified C-MET overexpression using IHC and C-MET overexpression positivity was defined as IHC 3+ with ⩾50% tumor cells. Additionally, patient genotypes were collected for subgroup analysis.
Results:
Data from 9785 NSCLC patients were collected. C-MET (−) accounted for 5% (503/9785), C-MET (+) for 27% (2654/9785), C-MET (++) for 36% (3464/9785), and C-MET (+++) for 32% (3164/9785). Genetic testing was available for 4326 patients. Wild-type was observed in 37% (1591 cases), with epidermal growth factor receptor (EGFR) abnormalities being the most common at 49% (2127 cases). Positive C-MET overexpression correlated significantly with women (p < 0.001), early-stage (p = 0.003), adenocarcinoma (p < 0.001), and driver mutations (p < 0.001). Patients with anaplastic lymphoma kinase (ALK) alterations had a higher occurrence of C-MET overexpression positivity (57.1%). Positive C-MET overexpression was significantly associated with EGFR (p < 0.001), ALK (p < 0.001), and KRAS alterations (p = 0.024). Compared to C-MET overexpression (IHC 0), C-MET overexpression (IHC 2+) (hazard ratio (HR) = 0.455, p < 0.001) and C-MET overexpression (IHC 3+) (HR = 0.569, p < 0.001) were correlated with better overall survival in overall NSCLC patients, especially for C-MET overexpression (IHC 2+).
Conclusion:
Our study elucidates the clinicomolecular characteristics and prognosis of C-MET overexpression in NSCLC patients, particularly those with positive C-MET overexpression (IHC 3+). This provides insight into the prevalence of C-MET overexpression in Chinese NSCLC patients and offers a basis for considering C-MET overexpression as a prognostic and predictive marker in NSCLC.
Keywords
Introduction
Lung cancer remains a leading cause of cancer-related mortality worldwide. In recent years, targeted therapies have significantly improved treatment outcomes in non-small cell lung cancer (NSCLC). In addition to epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK), the mesenchymal–epithelial transition (MET) has emerged as a crucial therapeutic target in NSCLC. 1 The MET proto-oncogene, a receptor tyrosine kinase located on chromosome 7, activates downstream pathways upon binding with its ligand, hepatocyte growth factor. Aberrant activation of this pathway can promote lung cancer growth and metastasis. 2 Abnormal MET pathway activations include MET exon 14 skipping mutations, MET amplification, C-MET protein overexpression, and MET gene fusion. 1
Clinical studies have demonstrated the efficacy of MET inhibitors, such as tepotinib, capmatinib, and savolitinib, in treating advanced NSCLC patients with MET exon 14 skipping mutations, yielding notable treatment outcomes.3–5 However, a standardized treatment approach for NSCLC patients with C-MET protein overexpression is still lacking internationally. C-MET protein overexpression can be classified as primary and secondary. Previous studies have indicated that the incidence of primary C-MET protein overexpression in NSCLC ranges from 13.7% to 63.7%,6,7 while the incidence of secondary C-MET protein overexpression ranges from 30.4% to 37.0%.8,9 Clinical trials investigating MET inhibitor treatment for C-MET overexpression in NSCLC have shown promising responses in immunohistochemistry (IHC) 3+ populations, as evidenced by the TATTON study, the NCT01610336 study, and the INSIGHT study.10–12 Hence, the clinical use of IHC to identify MET overexpression, particularly IHC 3+, holds significant therapeutic guidance value.
Real-world data on C-MET protein overexpression in the NSCLC population remain limited, particularly among the Asian Chinese population. This study aims to elucidate the clinicomolecular characteristics and survival prognosis of C-MET overexpression detected through IHC in NSCLC, with a particular focus on cases with positive C-MET overexpression (⩾50% of tumor cells with IHC 3+). This research provides comprehensive insights into the prevalence of C-MET overexpression detected by IHC in Chinese NSCLC patients, thereby assisting in the exploration of therapeutic strategies.
Methods
Study populations
We retrospectively reviewed and compiled data from NSCLC patients who underwent surgical resection or biopsy at the First Affiliated Hospital of Guangzhou Medical University from November 2006 to April 2021. All NSCLC cases were confirmed through cytology or pathology. Patients with recorded C-MET protein overexpression in IHC at the time of initial diagnosis, performed as a part of routine practice, were included in the screening criteria. Clinical data on age, gender, histology, tumor stage, dates of surgical or biopsy and death, IHC, and gene testing results were extracted from the medical record information repository of the First Affiliated Hospital of Guangzhou Medical University. The institutional review board approved the study protocol, and informed consent was waived for this observational study.
C-MET overexpression IHC assay
IHC staining for C-MET protein expression in tumor cells was performed using the SP44 monoclonal antibody. Tumor cells were categorized into high and low C-MET expression groups, with IHC 2+ or 3+ defined as high expression. In this study, C-MET overexpression positivity was defined as ⩾50% of tumor cells showing IHC 3+. Specifically, IHC 0 indicated no staining or staining in <50% of tumor cells, IHC 1+ denoted weak positive staining in ⩾50% of tumor cells but <50% of them being positive, IHC 2+ indicated positive staining in ⩾50% of tumor cells with <50% displaying strong positivity, and IHC 3+ represented strong positive staining in ⩾50% of tumor cells. 13
Mutational analyses
Genetic alternations were assessed by targeted next-generation sequencing covering critical mutations in nine major drivers (EGFR, ALK, KRAS, ROS1, RET, BRAF, PIK3CA, MET, and HER2) in NSCLC. In brief, DNA was extracted and quantified using the QIAamp DNA FFPE Tissue Kit (Qiagen, Hilden, Germany) and the Qubit fluorometer (Thermo Fisher Scientific, Waltham, MA, USA) following the manufacturer’s instructions. Subsequently, the products were captured and sequenced on an Ion Proton Sequencer (Thermo Fisher Scientific), using a panel encompassing 22 cancer-related genes. Data analysis was performed using a customized bioinformatics pipeline (Otype). Somatic alterations encompassed single nucleotide variations, short insertions or deletions, copy number variations, and gene rearrangements. 14
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics 26.0 (SPSS, Chicago, IL, USA) and GraphPad Prism 8.0 (GraphPad Software, Inc., Boston, MA, USA). Data were presented as mean ± standard deviation (SD) or frequencies and proportions. Chi-square tests were utilized to compare C-MET protein overexpression among NSCLC patients stratified by gender, histological type, stages, and driver mutations. The association of C-MET overexpression with each driver mutation was assessed. Overall survival (OS) was defined as the time from surgery resection or biopsy to death from any cause. Kaplan–Meier survival curves were constructed, and survival outcomes were compared using the log-rank test, with C-MET (−) or C-MET overexpression (IHC 0) serving as the reference cohort. A multivariate Cox Proportional Hazards (COX) regression model, adjusting for variables including age, gender, histologic type, tumor stages, driver mutations, and C-MET overexpression, was employed to analyze survival prognosis trends. Statistical significance was set at a p-value < 0.05.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology Statement (Supplemental Table 1). 15
Results
Clinical and molecular characteristics of C-MET overexpression in NSCLC
A total of 9785 NSCLC patients with sufficient tissue for C-MET protein overexpression assessment through IHC were included in this analysis. Among them, 503/9785 (5%) were C-MET (−), 2654/9785 (27%) were C-MET (+), 3464/9785 (36%) were C-MET (++), and 3164/9785 (32%) were C-MET (+++) (Figure 1(a)). Among the patients, 5145 (52.6%) were men, 2500 (25.5%) were nonsmokers, with a mean age of 59.1 years, and 48.6% were aged older than 60 years. The majority of patients were diagnosed at early stages (7629/9785, 78.0%) and exhibited adenocarcinoma (8497/9785, 86.8%). The distribution of various levels of C-MET overexpression across gender, age, smoking status, stage, and histologic type is summarized in Table 1. Additionally, differences in C-MET overexpression between surgical and biopsy specimens were further explored (Supplemental Figure 1). Additional detailed information can be found in the Supplemental Materials.

(a) The overall distribution of C-MET overexpression among 9785 NSCLC patients. (b) The overall distribution of driver mutations and wild-type status among 4326 NSCLC patients with C-MET overexpression.
Distribution of C-MET overexpression (IHC 0, 1+, 2+, 3+) across clinicopathological factors in 9785 NSCLC patients.

IHC, immunohistochemistry; NSCLC, non-small cell lung cancer; SD, standard deviation.
Among the 9785 NSCLC patients reviewed, 4326 had sufficient tumor tissue for genetic testing, with genetic alterations identified in 2735 patients. Specifically, 2127 cases (49%) exhibited EGFR alterations, 248 cases (6%) had KRAS alterations, and 72 cases (2%) showed ALK alterations. Other less common driver gene abnormalities were observed in 288 cases. Ninety patients presented with two or more coexisting driver mutations, with EGFR + PIK3CA co-mutations being the most prevalent (32 cases) (Figure 1(b) and Supplemental Figure 2). The distribution of different intensities of C-MET overexpression across wild-type genes and the nine oncogenic driver genes is detailed in Table 2.
Distribution of C-MET overexpression (IHC 0, 1+, 2+, 3+) in 4326 NSCLC patients undergoing genetic testing.
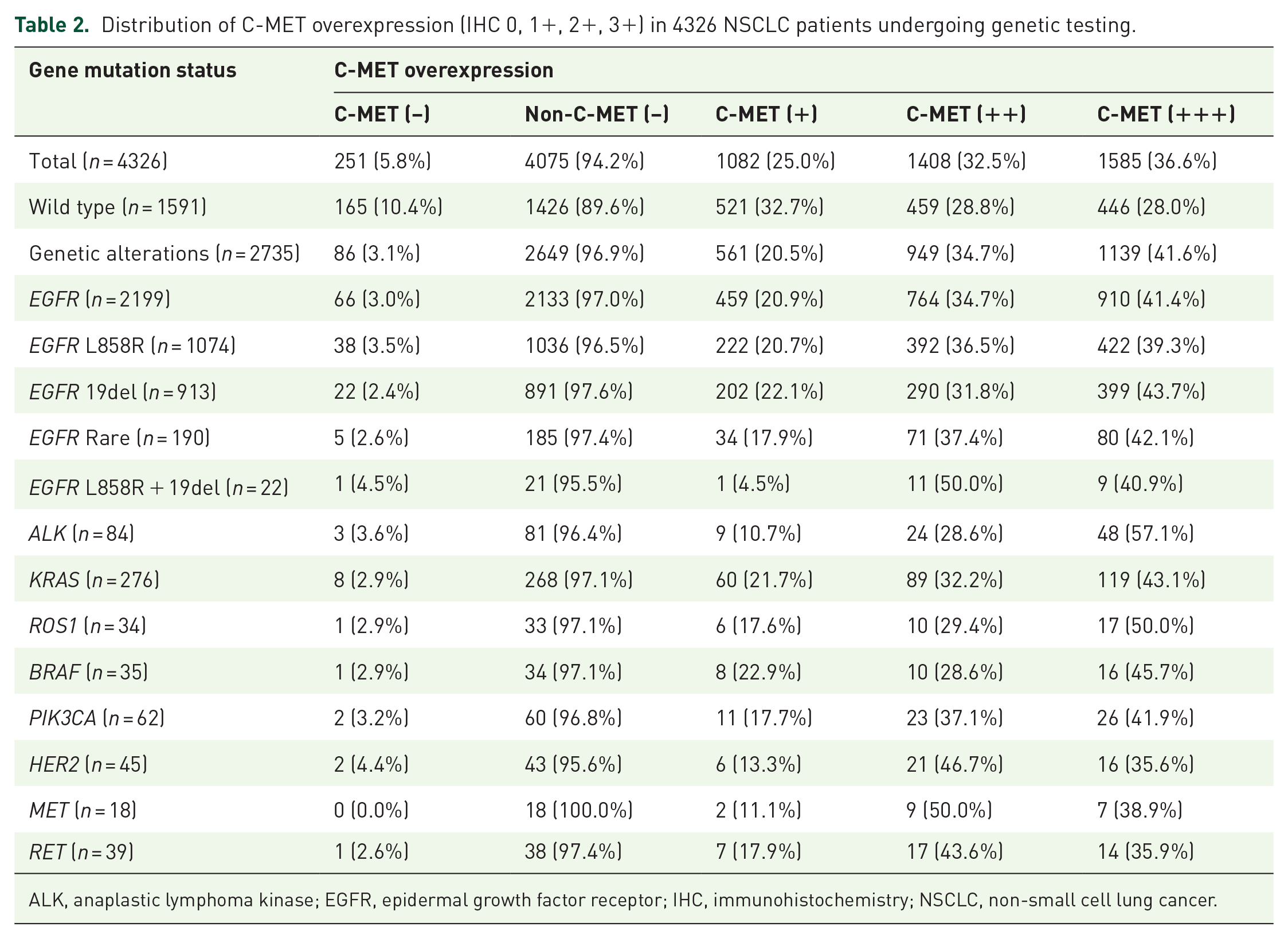
ALK, anaplastic lymphoma kinase; EGFR, epidermal growth factor receptor; IHC, immunohistochemistry; NSCLC, non-small cell lung cancer.
Clinical and molecular characteristics of positive C-MET overexpression (IHC 3+) in NSCLC
Subsequently, we focused on patients exhibiting positive C-MET overexpression (IHC 3+) to explore factors potentially influencing this phenomenon. Statistical analysis revealed that C-MET overexpression positivity (IHC 3+) was more prevalent among female patients (34.6% vs 30.3%, p < 0.001), early-stage NSCLC patients (33.1% vs 29.7%, p = 0.003), adenocarcinoma patients (36.0% vs 8.5%, p < 0.001), and those harboring driver mutations (41.6% vs 28.0%, p < 0.001) (Figure 2(a)–(d)).

C-MET overexpression positivity (IHC 3+) in NSCLC patients with respect to gender (a), tumor stage (b), histological type (c), and driver mutations (d).
Furthermore, we performed additional analyses to explore the correlation between C-MET overexpression and TNM stage, including tumor size, lymph node metastasis, and distant metastasis. Initially, in the T stage, we focused on 7526 early-stage patients after excluding those with incomplete tumor size data. We analyzed the association between tumor size and C-MET overexpression (IHC 3+), revealing that C-MET overexpression (IHC 3+) was more common in the T1 stage compared to other T stages (34.0% vs 29.7%, p = 0.002). Conversely, the incidence of C-MET overexpression (IHC 3+) was lower in the T3 and T4 stages, particularly in the T4 stage (T3: 27.2% vs 33.4%, p = 0.046; T4: 13.3% vs 33.4%, p < 0.001). However, no significant correlation was observed between C-MET overexpression (IHC 3+) and tumor size in the T2 stage (Supplemental Figure 3(A)–(D)). Secondly, excluding patients with unevaluable lymph node metastasis in the N stage, we analyzed 8626 patients. We categorized them into N (−) (no lymph node metastasis) and N (+) (lymph node metastasis, regardless of ipsilateral or contralateral). The analysis demonstrated a higher prevalence of C-MET overexpression (IHC 3+) in patients with lymph node metastasis compared to those without (36.3% vs 32.9%, p = 0.010) (Supplemental Figure 3(E)). Lastly, for the M-stage analysis involving all 9785 patients, we observed a higher frequency of C-MET overexpression (IHC 3+) in NSCLC patients without distant metastasis compared to those with distant metastasis (33.3% vs 25.5%, p < 0.001) (Supplemental Figure 3(F)).
C-MET overexpression with respect to driver mutations in NSCLC
Overall, the incidence of C-MET overexpression across different driver genes exceeded the overall expression level (>94.2%). Analyzing the heterogeneity in various stains of C-MET overexpression among the nine oncogenic driver genes revealed that NSCLC patients with alterations in EGFR, ALK, KRAS, ROS1, BRAF, and PIK3CA exhibited a progressive increase in the frequency of various stains of C-MET overexpression (IHC 0, 1+, 2+, 3+). Notably, patients with ALK alterations exhibited a significantly higher prevalence of positive C-MET overexpression (IHC 3+) compared to other staining intensities (IHC 0, 1+, 2+), at 48/84 (57.1%) (Table 2 and Figure 3(a)).
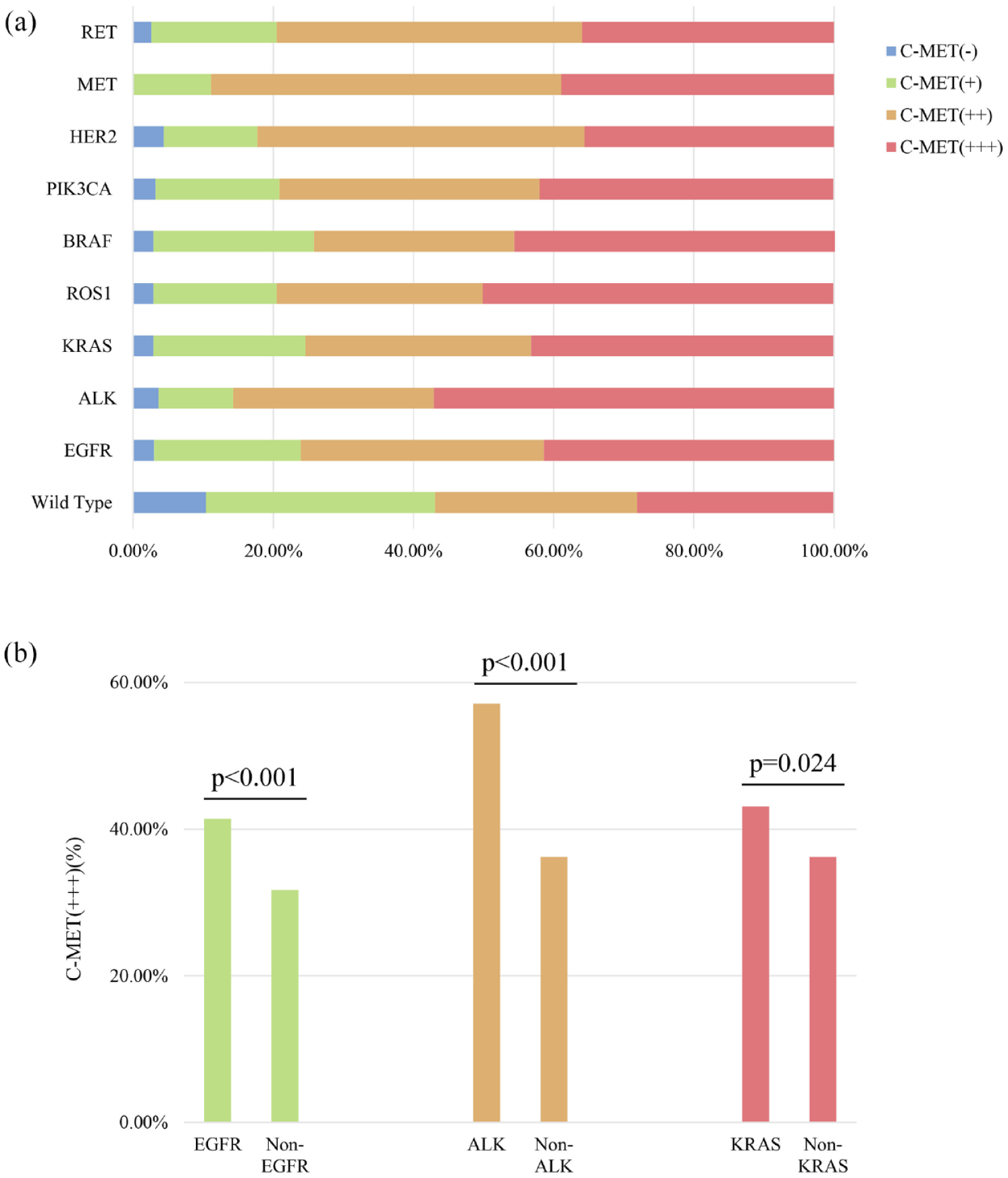
(a) The overall distribution of various stains of C-MET overexpression (IHC 0, 1+, 2+, 3+) among NSCLC patients with various driver mutations and wild-type status. (b) C-MET overexpression positivity (IHC 3+) in NSCLC patients with respect to different driver mutations.
Further analysis of the impact of different driver mutations on positive C-MET overexpression (IHC 3+) revealed that patients with EGFR, ALK, and KRAS alterations were more likely to exhibit positive C-MET overexpression (EGFR: 41.4% vs 31.7%, p < 0.001; ALK: 57.1% vs 36.2%, p < 0.001; KRAS: 43.1% vs 36.2%, p = 0.024). However, the differences in positive C-MET overexpression among patients with other driver mutations in NSCLC did not reach statistical significance (Figure 3(b)).
Relationship between C-MET overexpression and survival prognosis in NSCLC
Survival analysis evaluating the impact of C-MET overexpression on the prognosis of 8762 NSCLC patients revealed that those with C-MET overexpression (IHC 2+) (hazard ratio (HR) = 0.455, p < 0.001) and C-MET overexpression (IHC 3+) (HR = 0.569, p < 0.001) exhibited a better OS (Figure 4(a)). The same survival trend was observed in a COX multivariate model analysis adjusting for factors such as gender, age, stage, and histologic type. Especially, compared to patients with C-MET overexpression (IHC 0), those with C-MET overexpression (IHC 2+) (HR = 0.794, p = 0.026) and C-MET overexpression (IHC 3+) (HR = 0.896, p = 0.287) appeared to have a reduced risk of mortality (Table 3).
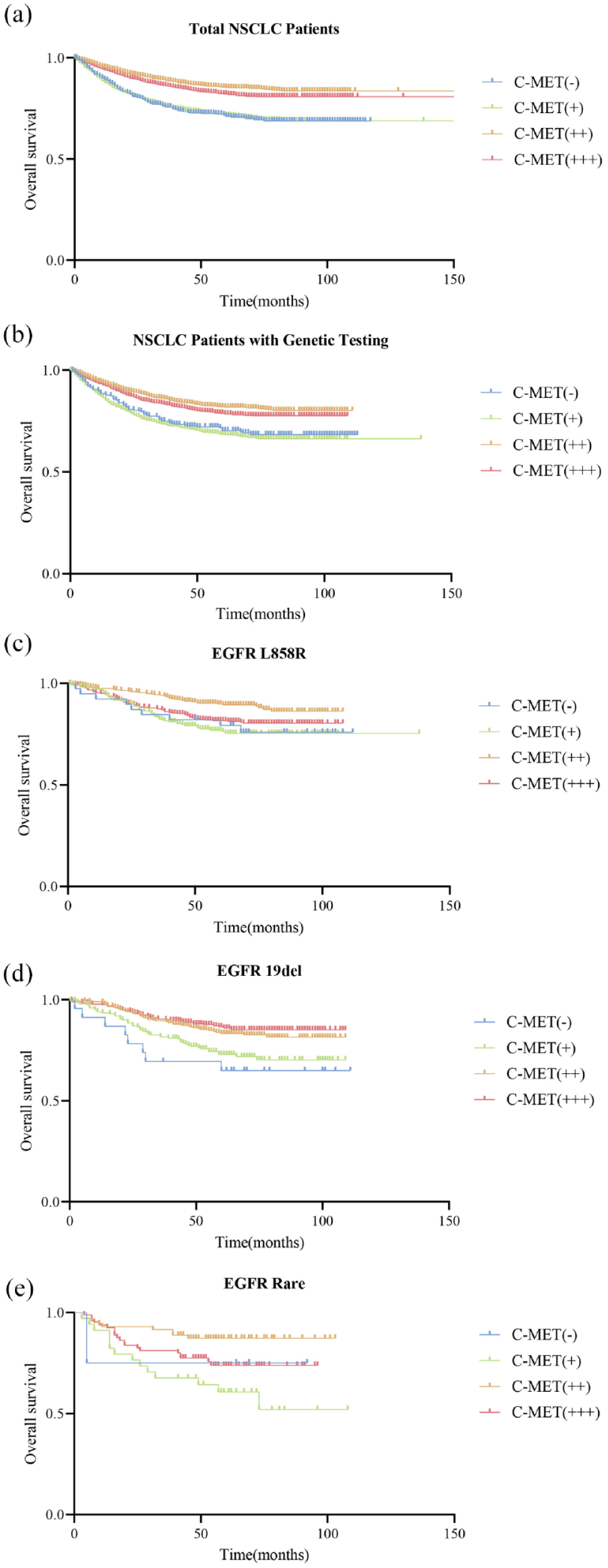
OS based on various stains of C-MET overexpression (IHC 0, 1+, 2+, 3+) in 8762 NSCLC patients (a), 4298 NSCLC patients with genetic testing (b), 1092 NSCLC patients with EGFR L858R mutations (c), 931 NSCLC patients with EGFR 19del mutations (d), and 189 NSCLC patients with EGFR rare mutations (e).
COX’s multivariable analysis between C-MET overexpression and overall survival in 8762 NSCLC cases.

95% CI, 95% confidence interval; HR, hazard ratio; IHC, immunohistochemistry; NSCLC, non-small cell lung cancer.
A total of 4298 cases underwent genetic testing for further prognostic analysis. The results indicated that patients with C-MET overexpression (IHC 2+) (HR = 0.565, p < 0.001) and C-MET overexpression (IHC 3+) (HR = 0.673, p = 0.002) had better OS (Figure 4(b)). A COX multivariate model analysis was performed, adjusting for confounders, which indicated that the survival trend had not changed despite lacking statistical significance (Table 4).
COX’s multivariable analysis between C-MET overexpression and overall survival in NSCLC patients harboring gene mutations.

95% CI, 95% confidence interval; HR, hazard ratio; NSCLC, non-small cell lung cancer.
In addition, we performed a survival analysis across different genetic subgroups. Due to the limited sample sizes, we were unable to perform survival analysis on gene types other than the common EGFR mutations. Therefore, we compared the prognosis of different levels of C-MET overexpression within the EGFR L858R, EGFR 19del, and EGFR Rare subgroups. Our findings indicated that compared to cases with C-MET overexpression (IHC 0), those with C-MET overexpression (IHC 2+) showed better OS (EGFR L858R: HR = 0.468, p = 0.038; EGFR 19del: HR = 0.421, p = 0.023; EGFR Rare: HR = 0.446, p = 0.444), as did those with C-MET overexpression (IHC 3+) (EGFR L858R: HR = 0.825, p = 0.583; EGFR 19del: HR = 0.347, p = 0.005; EGFR Rare: HR = 0.936, p = 0.948) (Figure 4(c) –(e)). Similar results were observed in COX multivariate analysis for the EGFR L858R and EGFR 19del subgroups. However, the results of COX multivariate analysis in the EGFR Rare subgroup were unstable, likely due to the limited sample size (Supplemental Tables 2–4).
Moreover, we conducted a prognosis analysis for C-MET-positive patients (IHC 3+) carrying gene mutations. The results showed that compared to wild-type patients, C-MET-positive patients with gene mutations had better OS (HR = 0.530, p < 0.001) (Supplemental Figure 4(A)). Further analysis of nine gene mutations indicated differences in OS among C-MET-positive patients (IHC 3+) with different gene mutations (Log-rank, p = 0.014). However, due to the limited sample sizes of gene mutations other than EGFR, these specific prognosis results might require further exploration (Supplemental Figure 4(B)). The results of the COX multivariate analysis are presented in Supplemental Tables 5 and 6.
Discussion
Currently, targeted molecular therapy is an effective treatment for lung cancer, with increasing attention on the MET target. However, there is still a lack of well-established therapeutic strategies for C-MET protein overexpression in NSCLC, and no consensus on the clinical association and survival prognosis of C-MET overexpression in NSCLC. In this study, we present a cohort of NSCLC patients with C-MET overexpression prior to treatment from a single institution. To our knowledge, this study represents the largest scale investigation to date into the clinicomolecular characteristics and prognosis analysis of C-MET overexpression by IHC in NSCLC, particularly within the Asian Chinese population.16–18
Our study explored the clinical and pathological features of 9785 NSCLC patients with varying degrees of C-MET overexpression, focusing on factors such as gender, age, tumor stage, and histological type. The overall incidence of primary C-MET overexpression in NSCLC in our study was 95%, significantly higher than previously reported rates (13.7%–63.7%).6,7 We speculate that this discrepancy may be related to the sample source. In this study, a large proportion of the samples were surgical, whereas biopsy samples, which may contain fewer tumor cells, could be limited in their ability to qualitatively detect cancer. Consequently, the detection of C-MET protein overexpression might sometimes be omitted. Our findings revealed that while a significant proportion of NSCLC patients with C-MET overexpression were adenocarcinoma, a smaller subset (13.2%) presented with other histological subtypes. This highlights the necessity of conducting C-MET overexpression testing for all NSCLC patients, regardless of their histological or clinical characteristics.
Upon further narrowing down the cohort to those patients with positive C-MET overexpression (IHC 3+) and analyzing potential influencing clinical factors, our study revealed that female patients had a higher rate of C-MET overexpression positivity compared to male patients. Additionally, lung adenocarcinoma patients exhibited significantly higher C-MET positivity rates than other histological subtypes. These findings are aligned with previous research.17,19 Moreover, our study found that early-stage patients were more likely to exhibit positive C-MET overexpression. However, Zeng et al. 20 reported that C-MET overexpression correlated with advanced tumor stages and played a crucial role in the metastasis and progression of colorectal cancer. Conversely, Bubendorf et al. 17 concluded that C-MET protein overexpression did not significantly correlate with tumor pathological staging. We speculate that our outcomes might be associated with the predominance of early-stage cases in our cohort.
It is worth noting that, in our study, 4326 patients had sufficient tissue samples for genetic testing. Within this cohort of NSCLC patients, we explored the heterogeneity of various levels C-MET overexpression among nine major oncogenic driver genes. The findings revealed that compared to the wild-type gene status, C-MET overexpression was more prevalent among patients with gene alterations, particularly in those with C-MET overexpression positivity (IHC 3+). Furthermore, NSCLC patients harboring EGFR, ALK, or KRAS abnormalities were more likely to exhibit positive C-MET overexpression (IHC 3+). Previous studies have suggested that MET amplification, following by MET protein overexpression, could potentially induce resistance to epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) in NSCLC. 21 Additionally, there is evidence indicating that MET alterations may be one of the mechanisms of resistance to ALK inhibitors. 22 Moving forward, it will be crucial to incorporate the detection of C-MET protein overexpression via IHC in NSCLC patients with these gene abnormalities to optimize clinical treatment decisions effectively.
Interestingly, our results revealed that the intensity of C-MET overexpression in IHC varied with different driver gene alterations. Patients with EGFR, ALK, KRAS, ROS1, BRAF, and PIK3CA alterations exhibited an increasing trend in C-MET overexpression. This trend was particularly notable in NSCLC patients with ALK abnormalities, with the highest occurrence of C-MET overexpression positivity (IHC 3+), exceeding half of the cases. Studies have indicated a significant increase in MET expression in ALK-rearranged NSCLC.23,24 Furthermore, MET alterations could be one of the mechanisms of resistance to ALK inhibitors. 22 Therefore, further investigation of treatment strategies will be essential when managing cases where C-MET overexpression coexists with ALK abnormalities.
Finally, based on our study results, the OS associated with C-MET overexpression (IHC 2+) and C-MET overexpression (IHC 3+) was better, consistent with previous research findings. 25 However, some studies have indicated a poorer prognosis associated with C-MET overexpression in NSCLC6,18,26 or reported no correlation with prognosis. 17 Our study further refined the survival analysis across different levels of C-MET overexpression in NSCLC patients with driver mutations, especially within various EGFR subgroups. The results also indicated better OS associated with C-MET overexpression (IHC 2+, 3+). This observation may be influenced by our earlier findings, suggesting a higher prevalence of C-MET overexpression in early-stage NSCLC. Tsakonas et al. 27 similarly suggested that a C-MET overexpression H-score ⩾20 served as a positive prognostic biomarker for OS in early-stage NSCLC, and this benefit appeared to be closely related to adjuvant chemotherapy. Although COX multivariable analysis among various factors, including gene mutations, did not show a statistically significant impact of C-MET overexpression on OS, the survival trend has not changed, and our study’s large sample size enhances credibility and representativeness to a considerable extent.
However, this study has several limitations that warrant consideration. First, our study was conducted within a single institution. Additionally, while our study encompassed a large sample size over an extended period, many patients did not receive complete long-term treatment at our institution, posing challenges in tracking their comprehensive treatment histories. Currently, we have conducted preliminary analysis on OS, but analysis on progression-free survival is still ongoing. Furthermore, there is a lack of standardized international evaluations for C-MET overexpression via IHC. 18 Therefore, future multicenter large-scale studies will be essential to further validate the heterogeneity of C-MET overexpression and its impact on survival prognosis in NSCLC, as well as to determine its potential as a prognostic and predictive marker in NSCLC.
Conclusion
In conclusion, our study reflected the clinical–pathological characteristics, survival outcomes, and associations with major driver gene alterations in a large cohort of NSCLC patients with C-MET overexpression in China. Female, early-stage, adenocarcinoma, and driver mutations were identified as independent factors related to positive C-MET overexpression (IHC 3+). NSCLC patients with C-MET overexpression exhibited better OS, particularly those with high expression (IHC 2+, 3+). Our findings provide a factual data foundation for supporting the consideration of C-MET overexpression as a prognostic and predictive marker in NSCLC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241279715 – Supplemental material for Landscape of C-MET overexpression in non-small cell lung cancer: a large-scale study of clinicomolecular features and prognosis based on Chinese data
Supplemental material, sj-docx-1-tam-10.1177_17588359241279715 for Landscape of C-MET overexpression in non-small cell lung cancer: a large-scale study of clinicomolecular features and prognosis based on Chinese data by Shuting Zhan, Jianfu Li, Bo Cheng, Caichen Li, Yi Feng, Lei Fan, Shan Xiong, Wenchuang Zeng, Qi Cai, Yang Xiang, Huiting Wang, Chunyan Li, Peiling Chen, Xin Zheng, Wenhai Fu, Zhexue Hao, Jianxing He and Wenhua Liang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
