Abstract
Background:
The rising global incidence of cancer has increased the demand for chemotherapy, which is a crucial treatment modality. Recent advancements in cancer treatment, including targeted agents and immunotherapy, have introduced complications owing to their specific mechanisms. However, comprehensive studies of the combined complications of these approaches are lacking.
Objectives:
This study aimed to comprehensively assess and analyze the overall incidence of anticancer drug-related complications in a nationwide patient cohort, utilizing a customized National Health Insurance Sharing Service database in Korea.
Design:
Retrospective cohort study.
Methods:
We included patients who were prescribed anticancer drugs (excluding endocrine agents) and diagnosed with cancer. For the type of cancer classification, the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10) was used and anticancer drugs were classified based on the Anatomical Therapeutic Chemical code. We classified cancer into 18 types based on the ICD-10 code and delineated cancer-related complications into 12 categories. Complications included hematological, gastrointestinal, infectious, cardiovascular, major bleeding, endocrine, neurotoxic, nephrotoxic, dermatological, pulmonary, musculoskeletal, and hepatotoxic effects.
Result:
We included 294,544 patients diagnosed with cancer and administered anticancer drugs between 2016 and 2018, with follow-up continuing until 2021. We identified 486,929 anticancer drug-related complications, with an incidence of 1843.6 per 1000 person-years (PY). Anemia was the most common complication, with a rate of 763.7 per 1000 PY, followed by febrile neutropenia (295.7) and nausea/vomiting (246.9). Several complications peaked during the first months following the initiation of anticancer drug therapy; however, herpes, skin infection, heart failure, and peripheral neuropathy peaked at 6–12 months. Among major cancers, breast cancer had the lowest overall incidence of complications. Targeted therapies revealed lower complication rates than cytotoxic chemotherapy; however, they also required careful monitoring of rash.
Conclusion:
This study highlights the importance of the proactive management of anticancer drug-related complications for patient care improvement.
Introduction
The global incidence of cancer is steadily increasing, which has driven the demand for chemotherapy as a crucial treatment modality. Chemotherapy, either alone or in combination with other therapeutic approaches, plays a pivotal role in combating various forms of cancer. Moreover, recent research from 2018 revealed that 57.7% of newly diagnosed patients with cancer required chemotherapy, including targeted therapies. Additionally, projections indicate a substantial 53% increase in the number of patients who will need chemotherapy from 2018 to 2040. 1
Although chemotherapy is effective in treating cancer, it often results in adverse events and complications. These complications can lead to treatment suspension, delays, hospital admissions, prolonged hospital stays, and emergency department visits and impose significant burdens on the healthcare system. Owing to its mechanism of action in multiple organs, traditional cancer chemotherapy is associated with a spectrum of toxicities, ranging from mild nausea and vomiting to life-threatening myelosuppression. 2 Consequently, the management of adverse events in patients receiving traditional chemotherapy remains a critical concern. 3 Advancements in anticancer drug development, targeted agents, and immunotherapy are now widely used in cancer chemotherapy and have attained promising clinical outcomes and reduced occurrence of adverse events compared to traditional chemotherapy. Despite advances in cancer chemotherapy in recent years, targeted agents and immunotherapy introduce their own set of complications that are driven by their specific mechanisms of action. 4 Notably, these treatments often require prolonged use, which can increase the likelihood of specific complications. A retrospective cohort study reported that 61.0% of the patients who received targeted therapy experienced adverse reactions, with skin damage (19.3%) being the most prevalent. 5
Immune checkpoint inhibitors, which are generally associated with a low incidence of adverse events, can cause immune-related adverse events such as endocrine toxicity, dermatologic toxicity, colitis, pulmonary toxicity, and cardiotoxicity.6–12 Dermatologic toxicity, particularly rash, has been reported in 13%–22% of patients treated with anti programmed cell death protein-1 (PD-1) agents. 10 Cardiotoxicity related to immune checkpoint inhibitors is infrequent; it is of significant concern owing to its potential to considerably increase morbidity and mortality. 8 Immune checkpoint inhibitor-induced cardiotoxicity has been reported in 4.2%–10.3% and was more prevalent among those with previous cardiovascular disease. 13
In contemporary cancer treatment, a combination of traditional chemotherapy, targeted therapy, and immunotherapy is frequently employed, yet comprehensive studies on the combined burden of complications associated with these approaches are lacking. The previous retrospective study reported overall adverse events but was limited to liver cancer or metastatic breast cancer.14,15 Most other studies have focused on identifying the adverse events caused by anticancer drugs based on their mechanisms and in patients with specific cancer types.16,17 Understanding the overall incidence of anticancer drug-related complications, categorizing them by cancer type, and classifying them according to cancer type are essential initial steps in estimating the healthcare burden of anticancer drug-related complications. Understanding the overall incidence and categorization of anticancer drug-related complications is essential in estimating the healthcare burden they pose. This foundational knowledge helps identify critical areas for intervention, informing the development of management strategies tailored to improve treatment completion rates and enhance the quality of cancer care. Therefore, this study aimed to comprehensively assess and analyze the incidence of anticancer drug-related complications, categorizing them by type and specific cancer, in the context of contemporary cancer chemotherapy.
Methods
Data source
This study utilized a customized database sourced from the National Health Insurance Sharing Service (NHISS) in Korea that encompasses medical claims data for all patients who underwent anticancer drug treatments, excluding endocrine anticancer drugs, between 2016 and 2018. The NHISS is responsible for administering a national health insurance system that provides coverage to nearly 98% of the national population and gathers comprehensive data related to medical claims. The NHISS database contains details pertaining to prescribed medications covered by health insurance benefits, medical services, and diagnosis, all coded according to the Korean Standard Classification of Diseases, Version 6, based on the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10). 18 Therefore, this study used data on patients prescribed anticancer drugs in Korea. The reporting of this study conforms to the strengthening the reporting of observational studies in epidemiology (STROBE) guideline and is presented in Supplemental Table S1. 19
Study population
This retrospective cohort study included patients diagnosed with cancer (ICD-10 C00–C97, D37–D48) and prescribed anticancer drugs, excluding endocrine agents, between January 1, 2016, and December 31, 2018. We categorized cancers into 18 types: oral cavity and pharynx; digestive system; respiratory and intrathoracic system; bone and joints; skin; mesothelial and soft tissue; breast; genital system; urinary system; eye and orbit; brain and other nervous systems; endocrine system; lymphoma; myeloma; leukemia; multiple sites; unspecified sites; and uncertain/unknown behavior cancer (Supplemental Table S2). The index date was marked by the initial prescription of anticancer drugs following cancer diagnosis. Patients previously prescribed anticancer drugs within 1 year before the index date were excluded. Observations concluded at the end of anticancer drug use, at patient death, or on December 31, 2021. If a diagnosis of cancer and subsequent anticancer drug prescriptions occurred a year after the end of chemotherapy, individuals were re-registered as new patients. Anticancer drugs were classified based on the Anatomical Therapeutic Chemical (ATC) third level, with protein kinase inhibitors, monoclonal antibodies, antibody–drug conjugates, and platinum compounds further distinguished at the ATC fourth level (Supplemental Table S3). Anticancer drugs included chemotherapy, targeted therapy, and immunotherapy. If a drug was re-prescribed within 56 days from the end of the last anticancer drug, it was assumed that the prescription continued. For each patient, we assessed the date of each anticancer drug prescription and categorized the treatment as combination therapy if there was any overlap. When two or more anticancer drugs are given together, it’s termed combination therapy. In patients with liver cancer, transarterial chemoembolization treatments were excluded from the analysis as they were considered localized therapy.
Outcome definitions
Anticancer drug-related complications were classified into 12 categories: hematologic, gastrointestinal, infectious, cardiovascular, major bleeding, endocrine, neurotoxic, nephrotoxic, dermatologic, pulmonary, musculoskeletal, and hepatotoxic effects. Outcomes were defined using the criteria detailed in Supplemental Table S4 and drawn from the existing literature.20–22 Certain complications such as arrhythmias, heart failure, major bleeding, acute kidney injury, mucositis, interstitial lung disease, pulmonary hypertension, myalgia, and hepatotoxicity were identified through primary diagnosis codes associated with hospital admissions or visits to the emergency department. Anemia and thrombocytopenia were defined using transfusion codes in the absence of concurrent bleeding. Febrile neutropenia was detected through diagnosis codes or the use of specific medications, in conjunction with hospitalization or emergency department visits. Episodes of nausea/vomiting, diarrhea, and serious infections were defined as emergency department visits that accompanied the primary diagnosis code or related medication use. Opportunistic infections were determined using diagnosis codes, medication use, hospitalization, or more than two outpatient visits. A hypertensive emergency was defined as an emergency department visit that involved the use of intravenous hypotensive agents. Other conditions such as hypertension, peripheral neuropathy, hypothyroidism, and rash were identified using primary diagnosis codes and corroborated with medication records.
Complications were monitored during the course of anticancer chemotherapy and for 28 days after completion. This period was chosen because chemotherapy cycles typically span 28 days, allowing us to capture delayed adverse events influenced by the anticancer drugs. For patients who experienced disease progression and were subsequently switched to a different anticancer drug, their data was included in the overall analysis of anticancer drug-related complications. However, for toxicity analysis specific to a particular anticancer drug, we censored the data once the specific drug was discontinued and another anticancer drug was started.
Long-term complications that necessitated ongoing treatment, including new-onset hypertension, heart failure, hypothyroidism, peripheral neuropathy, rash, interstitial lung disease, and pulmonary hypertension, were recorded. Recurrent complications were tracked repeatedly if they reappeared more than 30 days after the previous occurrence. The observation period for patients who developed any of the aforementioned long-term complications was terminated at the point of diagnosis, and those with preexisting conditions of the same nature within a year prior to the index date were excluded from the analysis. An overview of this study is shown in Figure 1.

Study overview.
Statistical analysis
Continuous variables were presented as means and standard deviations, and t-tests were used to compare the groups. Categorical variables were compared using the Chi-squared test. We calculated patient days as the total sum of days each patient was observed before censoring. Additionally, patient days for long-term complications were calculated until the occurrence of each long-term complication or censoring. The incidence rate was calculated by dividing the total number of complications by the total patient days and then converting this to patient-years for standardization. Complication incidence rates were represented per 1000 person-years (PY) and adjusted for varying observation periods across complications. Statistical significance was set at p-value of less than 0.05. Analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA) and R studio version 1.4.1717 (R Foundation for Statistical Computing, Vienna, Austria), with results reported with 95% confidence intervals (CIs) and p-values.
Subgroup analyses were conducted to assess the incidence of complications among cancers, with a patient population exceeding 10,000 individuals, and among different treatment regimens, with a focus on regimens with the longest observation periods for each cancer type. Complications of immunotherapy were also analyzed for the top four cancer types for which immune checkpoint inhibitor regimens were most frequently used. Additionally, we performed subgroup analyses according to age group.
Results
Patients characteristics
This study included a cohort of 294,544 patients who were diagnosed with cancer and treated with anticancer drugs during a specified observation period. The median follow-up duration was 210 days, with an interquartile range of 121–378 days, for a total of 264,112 PY. Women comprised 48.3% of the study cohort, 36.2% of the patients were between 65 and 79 years of age, and 6.2% were 80 years of age or above. A significant portion of the patients, 79.0%, had a Charlson comorbidity index score of 4 or more. Breast cancer was the most prevalent diagnosis, affecting 16.6% of the study population, followed by colorectal (15.4%), lung (14.9%), and stomach (8.2%) cancers (Table 1).
Baseline demographics of cancer patients who received anticancer drugs.

CCI, Charlson comorbidity index; IQR, interquartile range; PNS, peripheral nerve system; ANS, autonomic nervous system; STD, standard deviation.
Incidence of complications in overall cancer patients
The study documented 486,929 anticancer drug-related complications, equating to an incidence rate of 1843.6 per 1000 PY, with a 95% CI of 1838.5–1848.8. Anemia was the most common complication, with a rate of 736.7 per 1000 PY (95% CI, 733.4–739.9). Febrile neutropenia and nausea/vomiting followed, with an incidence rate of 295.7 and 246.9 per 1000 PY, respectively. The incidence rate of complications peaked during the first month following the initiation of anticancer drug therapy, registering 4282.4 per 1000 PY (95% CI, 4256.3–4308.6). Subsequently, there was a noticeable decrease in the overall incidence of complications over time, marked by rates of 2045.1 per 1000 PY (95% CI, 2031.8–2058.3) for the period of 1–3 months, 1925.0 per 1000 PY (95% CI, 1913.1–1936.9) for 3–6 months, 1906.9 per 1000 PY (95% CI, 1895.5–1918.3) for 6–12 months. Herpes infection, skin infection, heart failure, and peripheral neuropathy had the highest incidence rates at 71.0, 1.4, and 6.8 per 1000 PY, respectively, 6–12 months after the start of anticancer treatment (Table 2).
Incidence rate of anticancer drug-related complications.
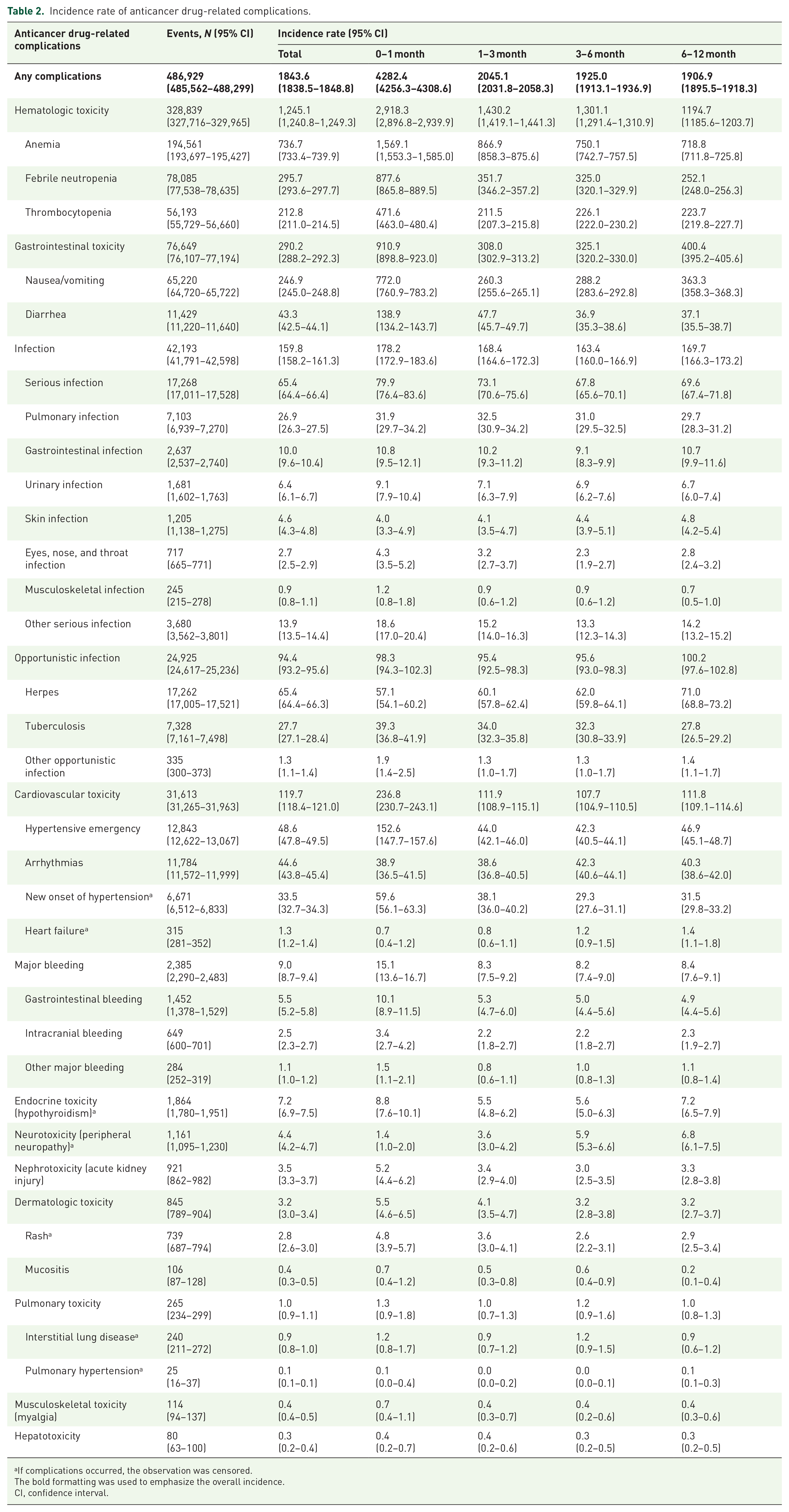
If complications occurred, the observation was censored.
The bold formatting was used to emphasize the overall incidence.
CI, confidence interval.
Complications in major cancers
Major cancers were the most prevalent cancer type, with the number of patients exceeding 10,000 individuals, and included 8 types: breast, colorectal, lung, stomach, liver, non-Hodgkin’s lymphoma, pancreatic cancer, and leukemia. Examples of the regimens for each major cancer type are presented in Supplemental Table S5.
Among digestive cancers, colorectal, stomach, liver, and pancreatic cancers were scrutinized. The incidence rate of complications per 1000 PY was as follows: colorectal cancer, 974.6 (95% CI, 964.6–984.8); stomach cancer, 1570.4 (95% CI, 1553.5–1587.4); liver cancer, 2155.4 (95% CI, 2124.8–2186.4); and pancreatic cancer, 2235.1 (95% CI, 2204.5–2266.0) (Figure 2(a), Supplemental Table S6). Notably, colorectal cancer treatment involving a combination of antimetabolites, plant alkaloids, and vascular endothelial growth factor (VEGF) inhibitors resulted in the highest complication incidence rate of 1101.1 per 1000 PY (Supplemental Table S8). Stomach cancer had the highest complication rate, at 2986.3 per 1000 PY with plant alkaloid monotherapy, whereas the breakpoint cluster region–Abelson (BCR-ABL) tyrosine kinase inhibitor (TKI) regimens showed the lowest, at 389.1 per 1000 PY (Supplemental Table S9). Among liver cancer therapies, other protein kinase inhibitors, such as sorafenib, exhibited the lowest complication rate, with an incidence of 1419.9 per 1000 PY (Supplemental Table S10). Pancreatic cancer treatments that involved antimetabolites with epidermal growth factor receptor (EGFR) TKI showed the highest complication incidence at 2778.7 per 1000 PY (Supplemental Table S11). Antimetabolite monotherapy was administered to all four digestive cancers, and the incidences of complications associated with antimetabolite monotherapy varied significantly among digestive cancers, as illustrated in Figure 3(a). Specifically, the rates per PY were as follows: colorectal cancer at a rate of 740.1; stomach cancer, 624.9; liver cancer, 1300.1; and pancreatic cancer, 1764.1.

The incidence rate of complications among major cancers. (a) digestive cancer (b) breast cancer (c) lung cancer (d) hematologic cancer.

The incidence rate of complications by regimen type in major cancers. (a) digestive cancer (b) breast cancer (c) lung cancer (d) hematologic cancer
Breast cancer exhibited the lowest overall incidence of complications among the major cancers studied, at 895.9 per 1000 PY (95% CI, 887.3–904.6) (Figure 2(b), Supplemental Table S7). Within this category, human epidermal growth factor receptor 2 (HER2) inhibitors were associated with the lowest incidence rate of complications at 292.3 per 1000 PY (Figure 3(b), Supplemental Table S12). Interestingly, the incidence rate of arrhythmias was notably high with HER2 inhibitors, at 39.7 per 1000 PY.
Lung cancer reported a complication incidence rate of 1554.6 per 1000 PY (95% CI, 1543.0–1566.3), with pulmonary infections and toxicity being particularly prevalent (Figure 2(c), Supplemental Table S7). EGFR TKI therapy was associated with the lowest incidence rate for lung cancer, but the incidence of rash was the highest, at 21.0 per 1000 PY (Figure 3(c), Supplemental Table S13).
Analysis of hematologic cancers, including leukemia and non-Hodgkin’s lymphoma, revealed incidence rates of 3981.2 per 1000 PY (95% CI, 3950.6–4012.1) and 4243.6 per 1000 PY (95% CI, 4203.3–4284.2) (Figure 2(d), Supplemental Table S7). Antimetabolite monotherapy was associated with the highest incidence rates in both cancer types, with non-Hodgkin’s lymphoma and leukemia reporting rates of 5173.5 and 5818.5 per 1000 PY, respectively (Figure 3(d), Supplemental Tables S14 and S15). BCR-ABL TKI therapy for leukemia was associated with the lowest complication rate, at 536.9 per 1000 PY.
Immune checkpoint inhibitor-related endocrine toxicity was highest in lung cancer, with an incidence rate of 32.8 per 1000 PY (95% CI, 26.5–40.2), followed by urinary bladder and kidney cancer (Supplemental Table S16).
Complications by age group
The incidence rate of anticancer drug-related complications by age was presented in Supplemental Table S17. The overall incidence rate of any complications was highest in the 0–17 years age group at 4479 per 1000 PY and lowest in the adult age groups at 1636.6 per 1000 PY. Hematologic toxicity was particularly high in the pediatric population, with an incidence rate of 4159.8 per 1000 PY. In contrast, the incidence rates of serious infections, cardiovascular toxicity, bleeding, and nephrotoxicity increased with age.
Discussion
In this comprehensive analysis, we evaluated anticancer drug-related complications in 294,544 patients diagnosed with cancer between 2016 and 2018, using an observation of 264,112 PYs. We observed a total of 486,929 anticancer drug-related complications, which translates to an incidence rate of 1843.6 per 1000 PY (95% CI, 1838.5–1848.8). The comparison with prior research is complicated by the fact that these studies focused on specific cancer types or chemotherapy regimens and did not include all cancer patients.
Hematologic toxicity emerged as the most common complication, with anemia being the most prevalent at 736.7 per 1000 PY, followed by febrile neutropenia, nausea/vomiting, and thrombocytopenia. This is in contrast to a retrospective cohort study of patients with five prevalent solid tumors (breast, colorectal, gastric, lung, and ovarian cancers), which found anemia in 89.5% of patients receiving chemotherapy. 23 Furthermore, another study focusing on metastatic breast cancer reported an even higher anemia incidence rate of 1366.0 per 1000 PY. 15 Our findings revealed a lower incidence rate of anemia than reported in previous studies, which may be owing to the specific focus on patients with severe anemia who required blood transfusions.
Temporal analysis of the incidence patterns revealed three distinct trends: decreasing, increasing, and stable. The high rate of complications observed within the first month of chemotherapy indicated the need for vigilant monitoring during the initial treatment phase. Complications, such as anemia, febrile neutropenia, and thrombocytopenia, presented a declining trend over time. Conversely, certain complications showed an increasing trend. Notably, the incidence rate of herpes infection escalated over time, peaking in the 6–12 month interval at 71.0 per 1000 PY (95% CI, 68.8–73.2). Similarly, the incidences of heart failure and peripheral neuropathy increased over time, reaching their highest rates within 6–12 months. This increase, observed with the prolonged use of anticancer drugs, underscores the need for caution during extended treatment durations. Finally, a subset of complications maintained stable rates over time, with no significant variations. This steady occurrence has been observed in serious infections, arrhythmias, and pulmonary toxicity, necessitating continuous monitoring throughout chemotherapy. These results indicated that the timing of toxicity monitoring should vary considering each complication. Anemia, febrile neutropenia, and thrombocytopenia needed close monitoring at the beginning of the anticancer drug. Complications, including herpes infection, heart failure, and peripheral neuropathy, were crucial to monitor 6 or more months after the initiation of the anticancer drug. Serious infections, arrhythmias, and pulmonary toxicity should be continuously monitored, regardless of the timing.
Among digestive cancers, pancreatic cancer showed the highest incidence rate of overall complications, significantly outpacing its counterparts. Notably, the incidence rates among the four types of digestive cancers displayed substantial variability, even within similar chemotherapy regimens. Antimetabolites with platinum compounds showed varying complication rates in colorectal cancer, stomach cancer, and liver cancer. This variation can be ascribed to differences in cancer stage and specific patient demographics, which notably influence complication rates. However, this also implies that more attention may be needed based on the cancer type, even with the same anticancer drug regimen.
Among the top eight cancers studied, breast cancer had the lowest incidence of complications. This contrasts with the higher rates reported in previous studies that focused on metastatic breast cancer and suggests that the scope of the study population and definitions of complications significantly affect the rates observed. 15 The HER2 inhibitor had the lowest complication rate but exhibited a cardiotoxicity incidence comparable to or higher than that of the other regimens. Additionally, HER2 inhibitors induced cardiovascular toxicity by overproduction of reactive oxygen species, which was dose-independent and reversible.24,25 Cardiovascular toxicity associated with HER2 inhibitors was similar to or higher than that associated with other chemotherapies; therefore, close monitoring is required during the administration of HER2 inhibitors.
Lung cancer presented a unique complication profile, marked by a significant incidence of pulmonary infections, dermatological toxicity, and pulmonary toxicity. Dermatologic toxicity is a common side effect associated with EGFR TKI inhibitors used in lung cancer and was observed to occur approximately 10 times more frequently in lung cancer than in other cancer types in our study. 26 Moreover, the highest incidence of peripheral neuropathy was observed with the use of plant alkaloids in combination with platinum compounds. These drugs are associated with chemotherapy-induced peripheral neuropathy, which requires enhanced vigilance.27,28
Leukemia and non-Hodgkin’s lymphoma exhibited higher incidences of hematologic toxicities compared to those observed in solid tumors. The high incidence of hematologic toxicities was considered to be due to the characteristics of hematologic cancer and the use of high doses of cytotoxic anticancer drugs. 29
In immune checkpoint inhibitor-related hypothyroidism was approximately 10 times higher compared to other regimens. The higher incidence of hypothyroidism with immune checkpoint inhibitors warrants close monitoring. Additionally, the incidence of dermatologic toxicity was higher than in traditional chemotherapy but not as high as EGFR TKI inhibitors. The complications related to immune checkpoint inhibitors vary depending on the cancer type, indicating extra caution in cancers that typically have a higher risk of toxicity. 10 However, anti-PD-1/PD-L1 were drugs that had recently begun to be used for cancer treatment. In Korea, the PD-1/PD-L1 inhibitor was covered by insurance in August 2017 for lung cancer, January 2018 for urinary bladder cancer, and February 2018 for melanoma. Therefore, only immunotherapy prescribed after those dates was included. This study included the number of patients who received immunotherapy: 5451 with 2923.7 patient-years for lung cancer, 516 with 328.0 patient-years for urinary bladder cancer, 508 with 347.0 patient-years for melanoma, and 139 with 77.7 patient-years for kidney cancer. Consequently, some complications may not have been accurately estimated due to the insufficient number of patients to evaluate the complications of immunotherapy.
Our study also highlighted generally lower complication rates associated with targeted therapies, despite the need for careful monitoring of specific side effects such as hypertension and major bleeding, particularly with drugs such as sorafenib. The potential of sorafenib to induce hypertension and bleeding, which has been attributed to its anti-angiogenic action, was reflected in a meta-analysis that identified a sorafenib-induced hypertension incidence of 23.4%.30,31 These findings suggest the critical need for attentive monitoring to effectively manage mechanism-specific side effects.
Although there is a difference in incidence depending on age, it is considered that the cancer itself or chemotherapy has a greater impact. The high incidence of hematologic toxicity in children is believed to be due to the administration of powerful cytotoxic drugs for the treatment of pediatric hematologic malignancies rather than their young age. This suggests that while age-specific factors play a role, the type of cancer and the intensity of the treatment regimen are significant determinants of complication rates.
It is important to note that the side effect profile and complication rates can differ significantly based on the specific anticancer agents used. For example, some targeted therapies may have different complication rates compared to traditional chemotherapy agents. Therefore, healthcare providers should interpret the data within the specific context of the patient’s treatment regimen and adjust monitoring and management strategies accordingly.
Although comprehensive, this analysis has some limitations. First, there is the potential for inaccuracies in incidence estimates that stem from the reliance on claims data and the absence of laboratory values. To mitigate this, we supplemented our analysis with medication use and procedural codes in patients for whom diagnostic codes alone were insufficient. Additionally, the methodology used to track toxicities in our study may not have captured all toxicities. To address this, we defined complications using the criteria established in previous studies.14,20–22 Despite this, we could not include non-emergency complications, such as alopecia, which do not require hospitalization or emergency visits but significantly impact the patient’s quality of life. Also, it is important to note that our study did not account for the exacerbation of preexisting conditions. While increased medication use might indicate a worsening of conditions such as hypertension, hyperglycemia, or anemia, the absence of laboratory and physical examination data makes it difficult to assess these exacerbations accurately. Consequently, we excluded patients with these preexisting conditions from our analysis.
Moreover, distinguishing complications caused by chemotherapy from those caused by the cancer itself, particularly within a retrospective study design, is challenging. By focusing on complications that occurred within 28 days after chemotherapy, we aimed to attribute these complications to the anticancer drugs. However, it is possible that some complications arising from the cancer itself during or shortly after the treatment period were not fully distinguishable. This limitation should be considered when interpreting our findings.
Second, our study did not account for the severity of cancer or the specific lines of chemotherapy, both of which could have influenced the observed complication rates. Furthermore, appropriate supportive care was administered before anticancer treatment or dose intensity was not reflected in the occurrence of complications.
Third, due to the limitations of claims data, our study could not perform analyses based on common terminology criteria for adverse events (CTCAE) grading. While many complications that led to emergency department visits or hospitalizations are likely to be CTCAE grade 3 or higher, we cannot exclude the possibility that patients visited the emergency department for lower-grade complications (CTCAE grade 1 or 2).
Fourth, we included only chemotherapy-naïve patients to avoid the confounding effects of prior chemotherapy regimens. This exclusion criterion may limit the generalizability of our findings to the broader population of cancer patients, many of whom may have received prior treatments. We did not adjust for the effects of comorbidities, which could influence the incidence and severity of chemotherapy-related complications. This approach was chosen to reflect the real-world cancer treatment environment, but it may limit the ability to isolate the impact of chemotherapy alone on complication rates.
Lastly, we categorized chemotherapy into ATC third or fourth levels for comprehensive analysis. Even within the same class of anticancer drugs, there may be differences in the incidence of complications, and specific adverse events unique to each drug may exist. This study did not analyze the incidence of complications within each anticancer drug, thus we did not elucidate these characteristics. However, it’s considered that some level of distinction can be made due to the varied types of carcinoma utilized. For example, in breast cancer treatment, taxanes are more common than vinca alkaloids, so the results can be interpreted as taxanes. Additionally, by analyzing the comprehensive complication rate across all cancer types, this study provides direction for more detailed comparative analyses in future research.
Conclusion
Our comprehensive analysis revealed a notable overall incidence of anticancer drug-related complications, of 1843.6 per 1000 PY. Among these, hematological toxicities were the most common, followed by gastrointestinal toxicities and infections. The first month of chemotherapy marked the highest overall risk period, emphasizing the need for early and vigilant monitoring. Over the course of treatment, complications such as herpes infection, heart failure, and peripheral neuropathy increased, whereas others remained constant, suggesting that a tailored monitoring approach is crucial. Targeted therapies have lower complication rates than cytotoxic chemotherapy; however, they also require the careful monitoring of specific adverse events. Our study findings underscore the importance of the proactive management of anticancer drug-related complications for improving patient care.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241272970 – Supplemental material for Comprehensive analysis of nationwide anticancer drug-related complications in Korea: incidence, types, and cancer-specific considerations in contemporary oncology
Supplemental material, sj-docx-1-tam-10.1177_17588359241272970 for Comprehensive analysis of nationwide anticancer drug-related complications in Korea: incidence, types, and cancer-specific considerations in contemporary oncology by Jonghyun Jeong, Soyoung Park, Kyu-Nam Heo, Soh Mee Park, Sangil Min, Young-Mi Ah, Ji Min Han and Ju-Yeun Lee in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
