Abstract
Background:
Tumor necrosis (TN) is a common feature in lung squamous cell carcinoma (LSCC), which could provide useful predictive and prognostic information.
Objectives:
This study aimed to investigate the effect of pretreatment pulmonary TN (PTN) on the prognosis of first-line anti-programmed cell death 1 (PD-1)/PD ligand 1 (PD-L1) inhibitor in advanced LSCC.
Design:
We conducted a retrospective study to analyze the association between the presence of PTN and clinical outcomes in advanced LSCC patients treated with anti-PD-1/PD-L1 inhibitors.
Methods:
Data from 240 eligible patients were collected from 27 hospitals across China between 2016 and 2020. The presence of PTN was assessed using contrast-enhanced chest computed tomography (CT) imaging at baseline. We utilized the Cox proportional-hazards regression model to analyze the association between PTN and clinical outcomes. In addition, to account for potential confounding factors and ensure comparability between groups, we employed propensity score-matching (PSM) analysis.
Results:
In the overall patient cohort, the presence of PTN was 39.6%. The median follow-up duration was 20.3 months. The positive PTN group exhibited a notably inferior median progression-free survival (PFS; 6.5 months vs 8.6 months, p = 0.012) compared to the negative PTN group. Within the Cox proportional-hazards regression model, PTN emerged as an independent predictor of unfavorable PFS (hazard ratio (HR) = 1.354, 95% confidence interval (CI): 1.002–1.830, p = 0.049). After PSM, the median PFS for the positive PTN group (6.5 months vs 8.0 months, p = 0.027) remained worse than that of the negative PTN group. Multivariate analyses also further underscored that the presence of PTN independently posed a risk for shorter PFS (HR = 1.494, 95% CI: 1.056–2.112, p = 0.023). However, no statistically significant difference in overall survival was observed between the two groups.
Conclusion:
Our study suggests that the presence of PTN on baseline contrast-enhanced chest CT is a potential negative prognostic imaging biomarker for the outcome of anti-PD-1/PD-L1 inhibitor therapy in advanced LSCC. Further studies are warranted to validate these findings and explore the underlying mechanisms.
Plain language summary
Our study focused on lung squamous cell carcinoma (LSCC) patients receiving first-line anti-PD-1/PD-L1 therapy. We explored the impact of a feature called pretreatment pulmonary tumor necrosis (PTN) on their prognosis. PTN was identified in 39.6% of patients using baseline chest CT scans. Results revealed that patients with PTN had a shorter time without disease progression (median PFS of 6.5 months compared to 8.6 months) and a higher risk of unfavorable outcomes. This suggests that PTN may serve as a negative prognostic imaging marker for anti-PD-1/PD-L1 therapy in advanced LSCC. Further research is needed to confirm and understand these findings better.
Keywords
Background
Lung cancer is a leading cause of cancer-related deaths worldwide. 1 Lung squamous cell carcinoma (LSCC) accounts for approximately 30% of all lung cancer cases. 2 Due to a lack of therapeutic targets, platinum-based chemotherapy remains the first-line standard treatment for advanced LSCC for a long period. 3 Immune checkpoint inhibitors (ICIs) targeted programmed cell death 1 (PD-1) and its ligand (PD-L1) have significantly revolutionized the treatment landscape of advanced LSCC.4,5 However, not all LSCC patients benefit from ICI treatment. Studies indicated that PD-L1 expression, tumor mutation burden (TMB), and gene profiling could serve as promising biomarkers for anti-PD-1/PD-L1 therapies in NSCLC.6–9 However, none of these biomarkers can be considered “ultimate biomarker” responses to anti-PD-1/PD-L1 antibodies and have their limitations.10,11 Therefore, the ideal non-invasive, cost-effective, and easily accessible prognostic biomarkers for ICIs are still an unmet need in clinical practice.
Tumor necrosis (TN) is a common feature of many solid tumors, 12 particularly observed in LSCC. It is conventionally accepted that the tumor cavity develops from TN due to rapid tumor proliferation and consequent outgrowth of the blood supply. 13 The tumor cavity is considered indicative of a more profound level of TN. Currently, there is no uniform definition for TN assessment, which is evaluated through various methods including histological examination of tissue samples and imaging techniques such as magnetic resonance imaging or computed tomography (CT) scans. Macroscopic or radiologic forms of TN can be identified as hypo-densities on CT scans. 14 TN has been proposed as a poor prognostic factor in a variety of solid tumors, as it is thought to reflect chronic hypoxia and poor vascularity. 12 In addition, some studies showed that the presence of TN was associated with unfavorable responses to chemotherapy and radio-therapy.12,15–17 However, there are still few studies on the relationship between TN and the prognosis of anti-PD-1/PD-L1 inhibitors. Our previous study found that TN or cavity on baseline CT scan could serve as an efficacy predictor of anti-PD-1/PD-L1 inhibitor in advanced LSCC based on single-center data. 18 To date, the clinical and prognostic implication of pretreatment pulmonary TN (PTN) as an imaging biomarker for anti-PD-1/PD-L1 inhibitors in LSCC remains elusive.
Therefore, we conducted a multi-institutional, propensity score-matching (PSM) cohort analysis to investigate the effect of pretreatment PTN on the prognosis of first-line anti-PD-1/PD-L1 inhibitors in advanced LSCC.
Methods
Study population
We retrospectively collected the clinical data of advanced LSCC patients treated with anti-PD-1/PD-L1 inhibitor in monotherapy or in combination with chemotherapy as first-line treatment from 27 hospitals in China from January 2016 to December 2020. A total of 240 patients were enrolled according to the following inclusion and exclusion criteria. This study was performed in compliance with the Declaration of Helsinki, and approved by the Institutional Review Board of the Fujian Cancer Hospital (No. SQ2021-137-01). The reporting of this study conforms to the REMARK statement.
The eligibility criteria were as follows: patients diagnosed with advanced LSCC; patients treated with anti-PD-1/PD-L1 inhibitor in monotherapy or combination with chemotherapy as first-line treatment; patients underwent contrast-enhanced chest CT imaging at baseline; patients received at least one cycle of treatment and completed at least one follow-up visit; no history of previous anticancer therapy; no history of other malignancies; and diagnosed between January 2016 and December 2020.
The exclusion criteria were as follows: age at diagnosis was less than 18 years; patients with missing data on baseline clinical characteristics; and follow-up time or survival time unavailable.
Data collected and definition
Baseline clinical characteristics on patient demographics, diagnosis, treatment, and survival were collected for analysis: age, gender, Eastern Cooperative Oncology Group (ECOG) performance status (PS), smoking status, year of diagnosis; histologic subtype, the primary site, brain, liver, and bone metastases; laboratory examinations including lactate dehydrogenase (LDH) level, absolute neutrophil count, absolute lymphocyte count; PD-L1 tumor proportion score; treatment regimen, treatment response, date of relapse, cause of death, survival time, and vital status.
We defined smokers smoking ⩾20 pack-years as heavy smokers and smokers smoking ⩽20 pack-years as light smokers. The neutrophil-to-lymphocyte ratio (NLR) was calculated by dividing the absolute neutrophil count by the absolute lymphocyte count measured in peripheral blood at baseline. The assessment of TN primarily focuses on tumor lesions in the lungs. Contrast-enhanced CT was performed to detect the necrotic region of the tumor. PTN is referred to as an intratumoral area larger than 5 mm in diameter with a 10–30 HU change and no enhancement greater than 10 HU after contrast administration.18,19 The pulmonary tumor cavity that developed from PTN was seen as a lucency or low-attenuation area at CT. Contrast-enhanced chest CT images were evaluated independently by experienced radiologists from each participating institution blinded to the patient’s response to anti-PD-1/PD-L1 inhibitors. In addition, a double-check process was implemented for PTN assessment to ensure accuracy and reliability. If any tumor lesion in the lungs is assessed to have TN on enhanced CT evaluation, regardless of the number of tumor lesions, the patient is classified as TN positive. Conversely, if none of the tumor lesions in the lungs exhibit TN, the patient is classified as TN negative.
Treatment and study endpoints
All patients received anti-PD-1/PD-L1 inhibitors in monotherapy or combination with chemotherapy as first-line treatment. The therapeutic response was evaluated on contrast-enhanced CT scans according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. The primary endpoints of this study were progression-free survival (PFS) and overall survival (OS). PFS was defined as the time from treatment initiation to the first confirmation of disease progression, death from any cause, or last follow-up. OS was defined as the time from treatment initiation to the death by any cause or the last follow-up.
Statistical analysis
Comparisons between categorical variables were conducted using the Chi-square test or Fisher’s exact test. The distributions of PFS and OS were estimated by the Kaplan–Meier method and comparisons were performed using the log-rank test. Univariate and multivariate analyses were performed using the Cox proportional-hazards regression model. To reduce the impact of potential confounders and selection bias, PSM analysis was used to balance the differences in baseline characteristics of patients between the positive PTN group and the negative PTN group. Matching was performed with the use of a 1:1 matching protocol without replacement, with a caliper width equal to 0.02 of the standard deviation of the logit of the propensity score. All statistical tests were two-sided, and p < 0.05 was considered statistically significant. All statistical analyses were performed using IBM SPSS Statistics, Version 25.0 (IBM, Armonk, NY, USA) and R version 3.6.1 (http://www.R-project.org).
Results
Patient characteristics and outcome
The study included 240 patients with advanced LSCC, 95 (39.6%) of whom had positive findings for PTN, and 145 (60.4%) had negative findings. The median age of the patients was 64 years (range 33–84 years), with 89.6% being female. Most patients (94.6%) had an ECOG PS of 0–1. The majority of patients (88.8%) were heavy smokers, and 72.1% of patients had an LDH level of ⩽250 IU/L. Most patients (75.4%) received anti-PD-1/PD-L1 inhibitors in monotherapy. The proportion of patients with brain, liver, and bone metastases did not differ significantly between the two groups (p > 0.05).
PSM was performed to reduce bias due to confounding variables. After PSM, 85 patients were selected from each group. The groups were well-balanced in terms of age, gender, smoking status, the presence of brain and liver metastases, LDH level, CRP level, and treatment regimen. However, there were significant differences between the groups in terms of ECOG PS (p = 0.043). Baseline characteristics of patients before and after PSM are presented in Table 1. Four typical examples of TN in chest-enhanced CT scans of LSCC are displayed in Figure 1.
Baseline characteristics before and after propensity score-matching.

CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; LDH, lactate dehydrogenase; NLR, neutrophil-to-lymphocyte ratio; PS, performance status; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease.
Bold indicates P value < 0.05.

Four typical examples of tumor necrosis in chest-enhanced CT scans of LSCC. (a–c) Representative images illustrating necrosis with associated cavitation (arrow) in lung cancer lesions. (d) Image depicting extensive low-density lesion in the lung (arrow), suggestive of necrosis in LSCC.
The median follow-up duration was 20.3 months (95% confidence interval (CI): 18.3–22.3). During the follow-up period, 79 deaths were observed, and 186 cases experienced disease progression. The median PFS for the overall patient cohort was 7.8 months (95% CI: 7.0–8.6) and the median OS was 30.5 months (95% CI: 23.7–37.3). The Kaplan–Meier curves for PFS and OS of advanced LSCC patients before and after PSM analysis are shown in Figure 2.

Kaplan–Meier curves for disease outcome of advanced lung squamous cell carcinoma patients before and after PSM analysis. (a) PFS in the overall patient cohort before PSM analysis (n = 240). (b) PFS in the PSM cohort (n = 85). (c) OS in the overall patient cohort before PSM analysis (n = 240). (d) OS in the PSM cohort (n = 85).
The prognostic impact of PTN in the overall patient cohort
In the overall patient cohort, the median PFS for patients with positive PTN group was 6.5 (95% CI: 5.4–7.6) months, compared to 8.6 (95%CI: 7.1–10.1) months for those with negative PTN (p = 0.012) (Figure 3(a)). The multivariate Cox analysis further confirmed PTN as an independent predictor of shorter PFS (hazard ratio (HR) = 1.354, 95% CI: 1.002–1.830, p = 0.049) in LSCC (Table 2). However, there was no significant difference in OS between the two groups (Figure 3(c)).
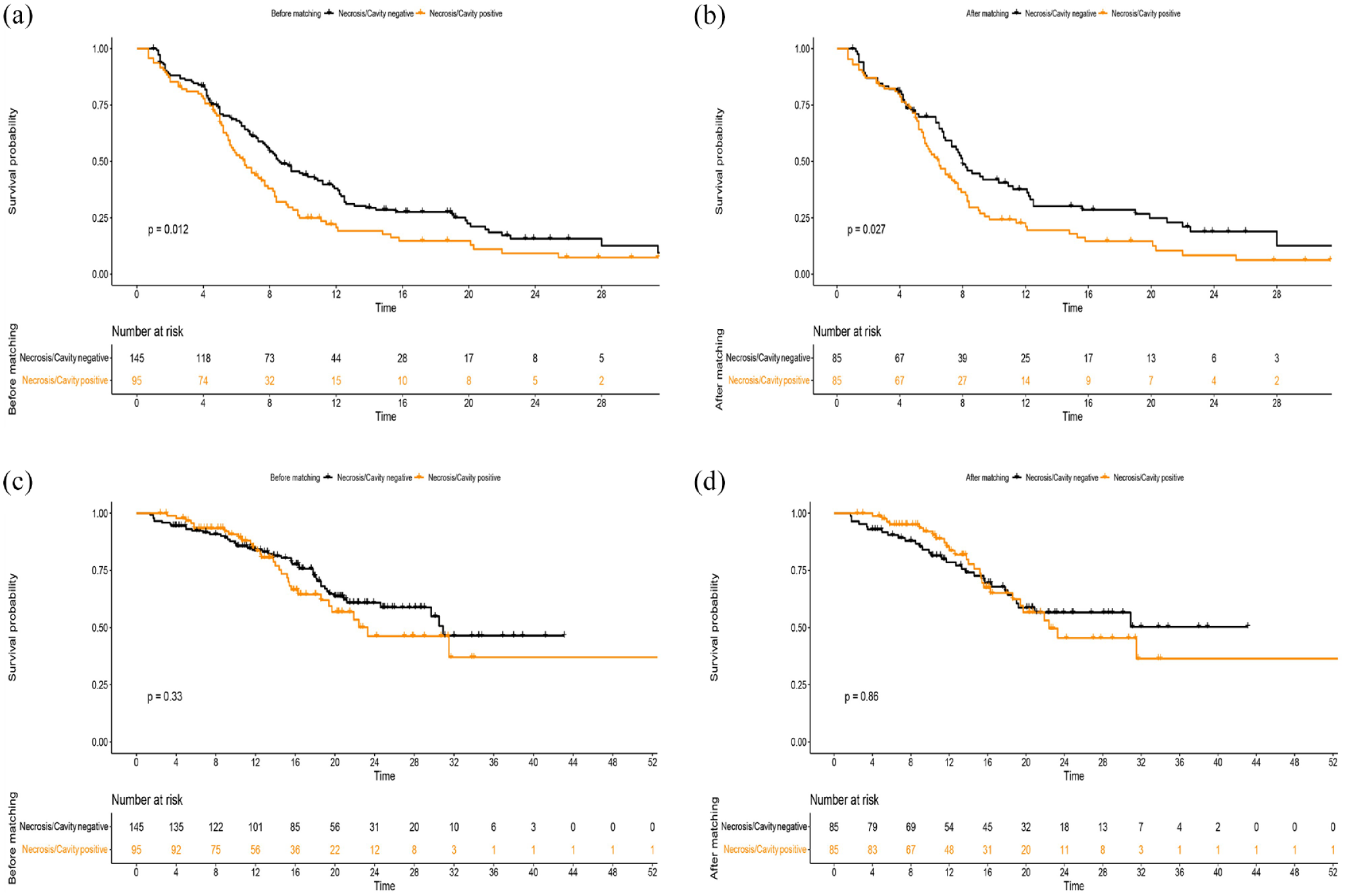
Kaplan–Meier curves for progression-free survival (a, b) and overall survival (c, d) of advanced lung squamous cell carcinoma patients stratified by present versus absent pulmonary tumor necrosis/cavity before and after matching. (a) Before matching for progression-free survival (n = 240). (b) After matching for progression-free survival (n = 85). (c) Before matching for overall survival (n = 240). (d) After matching for overall survival (n = 85).
Univariate and multivariate analyses for progression-free survival and overall survival in the overall cohort before propensity score-matching.
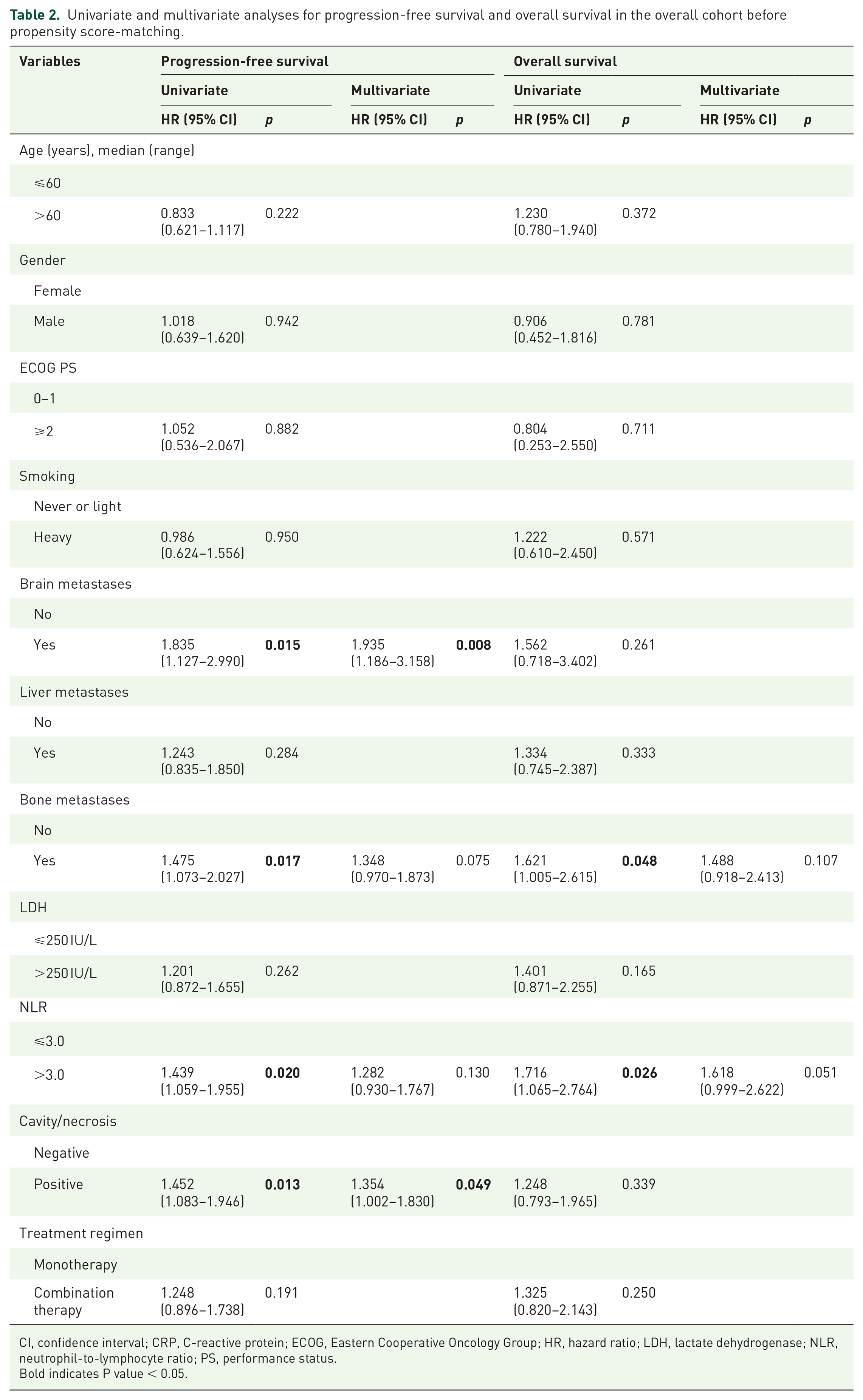
CI, confidence interval; CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; LDH, lactate dehydrogenase; NLR, neutrophil-to-lymphocyte ratio; PS, performance status.
Bold indicates P value < 0.05.
Besides the PTN, the univariate analyses indicated that brain metastases (p = 0.015) and bone metastases (p = 0.017) were associated with worse PFS. Multivariate Cox analysis indicated that the brain metastases (HR = 1.935, 95% CI: 1.186–3.158, p = 0.008) were independent risk factors for worse PFS. Meanwhile, bone metastases (p = 0.048) and NLR > 3.0 (p = 0.026) were significantly associated with worse OS. The results of univariate and multivariate analyses for PFS and OS in the overall patient cohort are shown in Table 2.
The prognostic impact of PTN in the PSM cohort
After PSM, the PTN remained significantly associated with PFS, with the median PFS of 6.5 (95%CI: 5.4−7.6) months for the positive PTN group and 8.0 (95%CI: 7.0–9.0) months for the negative PTN group (p = 0.027) (Figure 3(b)). Consistent with the results of the overall patient cohort, multivariate analyses based on the PSM cohort further demonstrated that the presence of PTN was independent risk for shorter PFS (HR = 1.494, 95% CI: 1.056–2.112, p = 0.023). Similarly, there was no significant difference in OS between the two groups after PSM (p = 0.862) (Table 3).
Univariate and multivariate analyses for progression-free survival and overall survival in the cohort after propensity score-matching.

CI, confidence interval; CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; LDH, lactate dehydrogenase; NLR, neutrophil-to-lymphocyte ratio; PS, performance status.
Bold indicates P value < 0.05.
Univariate and multivariate Cox regression analyses of PFS and OS were also performed for other variables in the PSM cohort, as shown in Table 3. Compared with the overall patient cohort, brain metastases remained served as an independent unfavorable predictor for PFS (HR = 2.000, 95% CI: 1.125–3.557, p = 0.018). While ECOG PS ⩾2 (p = 0.074), bone metastases (p = 0.086), and NLR >3.0 (p = 0.087) were only marginally associated with worse PFS. In terms of OS, only NLR >3.0 (p = 0.028) and LDH >250 IU/L (p = 0.051) were associated with adverse prognosis. However, based on multivariate analysis, there was no independent prognostic factor for OS.
Discussion
This study aimed to investigate the effect of pretreatment PTN on the prognosis of first-line anti-PD-1/PD-L1 inhibitors in advanced LSCC. Our results showed that the presence of pretreatment PTN was significantly associated with shorter PFS in both the overall patient cohort and the PSM cohort. However, there was no significant difference in OS between the two groups.
The results of this study showed that pretreatment PTN on CT was a significant independent factor for the prognosis of anti-PD-1/PD-L1 therapy in patients with advanced or recurrent LSCC. Patients with pretreatment PTN on CT had a significantly shorter PFS than those without pretreatment PTN. The potential association between pretreatment PTN and worse response to immunotherapy may have important implications for patient selection and treatment strategies in this patient population. Assessment of pretreatment PTN might help identify cases at high risk for recurrence, predict prognosis, and develop individualized therapies. Given that CT is routinely performed in most patients with newly diagnosed LSCC, this new imaging biomarker is readily available and does not result in additional costs. Therefore, pretreatment PTN on CT may be used as a promising prognostic imaging prognostic biomarker for first-line anti-PD-1/PD-L1 therapy in advanced LSCC, which should routinely record its presence or absence. Pretreatment PTN may also need to be considered when designing clinical trials of ICI therapy and interpreting the findings. To our knowledge, this is the first time to reported that pretreatment PTN on CT is a promising prognostic imaging biomarker for first-line anti-PD-1/PD-L1 therapy in advanced LSCC based on a multi-institutional, PSM cohort analysis. Further investigation is warranted to clarify this relationship and to optimize the use of immunotherapy in LSCC patients with pretreatment PTN.
PTN is a common pathological change in treatment-naive LSCC, especially in high-grade, fast-growing large tumors, and is always located at the center of a tumor bed. 20 Some studies have investigated macroscopic or radiologic forms of necrosis,14,21–23 whereas others have examined microscopic or pathological forms of tumor cell death.12,24–27 In most cases, the massive central low-attenuation area or cavity seen on imaging tools corresponded to areas of TN in pathological specimens. 23 It is reported that contrast-enhanced CT can be used to detect TN. 28 The intratumoral necrosis was defined on CT images as a focal area of low attenuation with or without a surrounding rim of enhancement as described by King et al. 14 In another study by Song et al., they defined a hypometabolic area within the peripheral hypermetabolic area in the tumor and the absence of contrast enhancement in the center of the peripheral enhancing tumor in intravenous contrast-enhanced full-dose CT and the attenuation between 10 and 30 HU in non-intravenous contrast-enhanced low-dose CT as necrosis. 29 In the present study, we assessed TN by contrast-enhanced CT, and necrosis is referred to as an intratumoral area larger than 5 mm in diameter with a 10–30 HU change and no enhancement greater than 10 HU after contrast administration.18,19
TN detected at the histopathological or imaging evaluation has been proposed as a marker of poor prognosis in a variety of solid-organ malignancies, 12 such as renal, 13 breast, 14 lung, 15 and colorectal cancer. 16 TN has also been linked to a higher risk of tumor recurrence and lower response rates to conventional chemotherapy and radiation therapy.12,17,30,31 However, in the era of immunotherapy, relevant studies of the impact of PTN on the prognosis of ICIs in LSCC are scarce. Our previous study found that PTN on baseline CT scan serves as an efficacy predictor of anti-PD-1/PD-L1 inhibitor in advanced LSCC based on single-center data. 19 Similarly, in the present study, PTN is a valuable prognostic biomarker for worse PFS in patients with advanced LSCC who were treated with first-line anti-PD-1/PD-L1 inhibitors based on a multi-institutional, PSM cohort analysis.
The mechanism underlying the association between PTN and unfavorable response to immunotherapy remains unclear. However, several potential explanations have been proposed. PTN is often associated with a high tumor burden, which may correlated with a poor response to ICIs. 32 Moreover, necrotic tissues within the tumor may act as a barrier to drug delivery, leading to resistance to immunotherapy. 31 In addition, TN is considered to reflect a hypoxic microenvironment, which has been shown to play an important role in tumor development, progression, and therapy resistance. 33 Further studies are warranted to explore the underlying mechanisms of this association.
Interestingly, we did not observe a significant difference in OS between patients with positive and negative PTN. OS represents the overall survival time from the initiation of treatment until death from any cause, and it can be influenced by long-term treatment responses, disease progression, comorbidities, and other related factors. On the other hand, PFS measures the time from the initiation of treatment until disease progression or death. PFS is often more sensitive to short-term treatment responses and disease control, which might explain why PTN emerged as a prognostic factor for PFS but not for OS in our study. In addition, the follow-up duration in our study may not have been long enough to capture the full impact of PTN on OS, especially considering that OS represents a longer-term outcome compared to PFS. Furthermore, in addition to PTN, our study also identified brain metastases, bone metastases, and NLR > 3.0 as prognostic factors for worse PFS in the overall patient cohort with LSCC. These findings are consistent with previous studies.34,35 Therefore, close monitoring and early intervention may improve survival outcomes in these patients.
Several limitations of our study should be taken into consideration. First, the retrospective design of the study may have introduced selection bias and confounding factors. However, we attempted to reduce the impact of these factors by performing PSM. Second, the sample size of our study was relatively small, which may have limited the statistical power of our analyses. Third, because of the retrospective nature of this study, histopathological analysis of necrotic tumor sites on CT and genetic data, including tumor burden, were not available. In addition, due to a high amount of missing data, we ultimately did not include PD-L1, and the evaluation of necrosis in metastatic lesions and non-squamous histology was also not included in our analysis. Finally, the project concluded in 2020, which may limit the generalizability of our findings to more recent cohorts. Therefore, future studies with larger sample sizes and prospective designs are needed to validate our findings and explore the underlying mechanisms.
Conclusion
In conclusion, our study showed that pretreatment PTN was a significant predictor of worse PFS in patients with advanced LSCC. The presence of PTN may serve as a useful prognostic imaging biomarker for first-line anti-PD-1/PD-L1 therapy in patients with advanced LSCC. Future studies with larger sample sizes and longer follow-up duration are needed to confirm our findings and to explore the underlying mechanisms of this association.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241266188 – Supplemental material for Pretreatment pulmonary tumor necrosis is a promising prognostic imaging biomarker for first-line anti-PD-1/PD-L1 therapy in advanced lung squamous cell carcinoma: a multi-institutional, propensity score-matching cohort analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241266188 for Pretreatment pulmonary tumor necrosis is a promising prognostic imaging biomarker for first-line anti-PD-1/PD-L1 therapy in advanced lung squamous cell carcinoma: a multi-institutional, propensity score-matching cohort analysis by Qiaofeng Zhong, Longfeng Zhang, Lin Wu, Jun Zhao, Jianguo Sun, Yong Fang, Jin Zhou, Qian Chu, Yihong Shen, Zhenzhou Yang, Lijin Chen, Meijuan Huang, Xiaoyan Lin, Zhenhua Liu, Peng Shen, Zhijie Wang, Xin Wang, Huijuan Wang, Chengbo Han, Anwen Liu, Hongmei Zhang, Feng Ye, Wen Gao, Fang Wu, Zhengbo Song, Shengchi Chen, Chengzhi Zhou, Dingzhi Huang, Qiuyu Zhang, Xinlong Zheng, Xiaobin Zheng, Qian Miao, Kan Jiang, Zihua Zou, Yiquan Xu, Shiwen Wu, Haibo Wang, Yaping Hong, Tao Lu, Chao Li, Cheng Huang, Chuanben Chen and Gen Lin in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank all the study participants, their families as well as physicians from 27 hospitals in China who contributed to this work for their effort.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
