Abstract
In recent years, with the continuous development of molecular immunology, immune checkpoint inhibitors (ICIs) have also been widely used in the treatment of gastric cancer, but they still face some challenges: The first is that only some people can benefit, the second is the treatment-related adverse events (TRAEs) that occur during treatment, and the third is the emergence of varying degrees of drug resistance with long-term use. How to overcome these challenges, combined therapy based on ICIs has become one of the important strategies. This article summarizes the clinical application of ICIs combined with chemotherapy, targeted therapy, radiotherapy, photodynamic therapy, thermotherapy, immune adjuvant, and dual immunotherapy and discusses the mechanism, and also summarizes the advantages and disadvantages of the current combination modalities and the potential research value. The aim of this study is to provide more and more optimized combination regimen for ICI combined therapy in patients with advanced gastric cancer and to provide reference for clinical and scientific research.
Keywords
Introduction
Gastric cancer is one of the common malignant tumors, with the incidence ranking fifth among global malignant tumors, accounting for approximately 5.6%, seriously affecting human health. 1 Due to the manifestation of atypical early clinical symptoms, the majority of cases are diagnosed at an advanced stage, thereby forfeiting the opportunity for radical surgery. Even among patients undergoing surgical resection, more than two-thirds are susceptible to recurrence or metastasis, culminating in a 5-year survival rate of less than 50%.2,3 The primary approach to the treatment of localized gastric cancer revolves around enhancing perioperative chemotherapy, 4 for advanced patients, commonly used treatments include stent implantation, chemotherapy, radiotherapy, and targeted therapy. In recent years, with the rapid development of immunotherapy, immune checkpoint inhibitors (ICIs) have become a research hotspot in the treatment of advanced gastric cancer.
Research on ICIs in gastric cancer
Tumor cells use multiple ways to evade the body’s immune killing response during their growth and invasion. Mediating the inhibitory immune checkpoint signaling pathway is one of its important mechanisms. ICIs function by blocking this signaling pathway to inhibit immune evasion mechanisms, resulting in the reactivation of T cells following antigen presentation, thereby achieving antitumor effects. 5 Currently, in clinical practice, the most studied immune checkpoints are cytotoxic T lymphocyte antigen-4 (CTLA-4) and programmed cell death 1 (PD-1) and programmed cell death ligand 1 (PD-L1), etc. For its mechanism, CTLA-4, known as cytotoxic T-lymphocyte-associated protein 4, serves as a shared inhibitory molecule expressed on the surface of activated T cells and regulatory T cells (Treg cells). Binding of the CTLA-4 receptor to its ligands, B7-1/B7-2, on antigen-presenting cells, inhibits the CD28-mediated T cell activation signaling pathway. Monoclonal antibodies targeting CTLA-4 effectively block its competition with CD28 for binding to B7 molecules, consequently activating the CD28 signaling pathway and diminishing the immunosuppressive Treg cell population within the tumor microenvironment. 6 PD-1, a co-inhibitory receptor, is expressed on the surface of B lymphocytes, activated CD4+ and CD8+ T lymphocytes, Treg cells, and monocytes, among other cell types. PD-1 has two ligands, PD-L1 and PD-L2, with PD-L1 being expressed on immune cells and tumor cells, while PD-L2 is primarily expressed on antigen-presenting cells. The binding of PD-L1 expressed on tumor cells to PD-1 on the surface of activated T cells generates inhibitory signals, thereby preventing T cell activation and suppressing the secretion of interferon-gamma (IFN-γ), tumor necrosis factor (TNF), interleukin-2 (IL-2), and other cytokines, thus inhibiting both cellular and humoral immunity. Blocking the PD-1/PD-L1 pathway can enhance the host’s antitumor immune response. 7
A number of large-scale experimental studies have also been conducted on the application of ICIs in advanced gastric cancer, such as KEYNOTE-012, ATTRACTION-2, KEYNOTE-059, KEYNOTE-061, which have shown that ICIs can effectively prolong patient survival, and the safety has also been recognized to a certain extent.8–11 The percentage of gastric cancer patients who derive benefit from ICI therapy is influenced by multiple factors, primarily including the biological characteristics of the tumor, the expression levels of molecular markers, and the pathological type. Predictors of the efficacy of ICI primarily include the combined positive score (CPS) and DNA mismatch repair (dMMR)/microsatellite instability-high (MSI-H). CPS is one of the key indicators for assessing whether a patient is suitable for ICIs therapy; generally, a higher CPS value is associated with better treatment response and prognosis. 12 dMMR is a crucial repair mechanism within cells, responsible for correcting base-pair mismatches or insertions/deletions that may occur during DNA replication, thereby ensuring genomic stability and integrity. Tumors with deficient dMMR often produce a higher number of neoantigens, which can activate the immune system to attack the tumor. Consequently, patients with dMMR tumors may exhibit a better response to ICI therapy. MSI-H is generally associated with defects in the dMMR system and also correlates with sensitivity to ICIs.13,14 Studies indicate that dMMR/MSI tumors constitute approximately 14% of localized gastric cancers, 9% of advanced gastric cancers, and 17% among patients over the age of 70. 15 One study involving 32 patients with dMMR/MSI-H gastric/Gastroesophageal Junction (GEJ) adenocarcinoma, participants received monotherapy with ICIs (nivolumab and ipilimumab). Of these, 29 patients underwent surgery; 3 did not undergo surgical intervention and demonstrated a complete endoscopic response with tumor-free biopsies and normal computed tomography scans (2 declined surgery, and 1 had metastases at the time of inclusion). Significant remission rates were observed in gastric cancer patients with dMMR/MSI-H treated with ICIs. 16 Although ICIs bring great hope to patients, they also face some clinical challenges. The first is that only some people can benefit, the second is the treatment-related adverse events (TRAE) that occur during treatment, such as rash, colitis, immune-related hepatitis, and thyroid dysfunction,17,18 and the third is the emergence of varying degrees of drug resistance with long-term use. 19 How to improve the benefits of ICIs and reduce TRAEs in gastric cancer patients’ combination therapy has become one of the important strategies. Therefore, this article summarizes the relevant clinical research and action mechanisms of ICIs combined with chemotherapy, targeted diagnosis and treatment, radiotherapy, photodynamic therapy (PDT), hyperthermia, immune adjuvants, and dual immunotherapy (see Figure 1), aiming to provide reference for clinical treatment work and basic research.

Immune checkpoint inhibitors combined with multiple treatments.
ICIs combined with chemotherapy
Currently, chemotherapy is still the basic treatment for patients with advanced gastric cancer. 20 As a result, trials exploring the combination of ICIs with chemotherapy are continuously underway and have demonstrated certain efficacy in clinical settings. A randomized, multicenter study (NCT02746796) investigates the first-line treatment of advanced or recurrent Human Epidermal Growth Factor Receptor 2 (HER-2)-negative gastric or gastroesophageal junction cancer stages II/III, utilizing nivolumab in combination with SOX (S-1: Tegafur/Gimeracil/Oteracil, 40 mg/m2 orally twice daily for 14 days followed by 7 days off; oxaliplatin, 130 mg/m2 intravenously on day 1 every 3 weeks) or CapeOX (capecitabine, 1000 mg/m2 orally twice daily for 14 days followed by 7 days off; oxaliplatin, 130 mg/m2 intravenously on day 1 every 3 weeks). The results indicate that the median progression-free survival (PFS) for nivolumab combined with SOX was 9.8 months, and for nivolumab combined with CapeOX, the median PFS was 7.2 months. The objective response rate (ORR) demonstrated comparability in both groups, with nivolumab plus SOX at 66.7% (95% confidence interval (CI), 43.0–85.4) and nivolumab plus CapeOX at 70.6% (95% CI, 44.0–89.7). Due to the occurrence of related adverse reactions, treatment discontinuation was observed in less than 10% of cases, and no treatment-related deaths were reported. Therefore, the combination regimen of nivolumab with CapeOX/SOX can be considered as a first-line treatment option for advanced gastric cancer patients. 21 The study (NTC02494583) evaluated the efficacy of pembrolizumab in combination with chemotherapy compared to chemotherapy alone (cisplatin + capecitabine) for unresectable gastric/gastroesophageal junction cancer. The final results showed median PFS of 6.9 and 6.4 months, respectively (hazard ratio (HR) = 0.84, 95% CI, 0.70–1.02, p = 0.04); median overall survival (OS) of 12.5 and 11.1 months, respectively (HR = 0.85, 95% CI, 0.70–1.03, p = 0.05). 22 Additionally, another phase III multicenter clinical trial (NCT02625610) included a total of 499 patients with metastatic gastric/gastroesophageal junction adenocarcinoma. The patients were divided into two groups: the nivolumab combined with chemotherapy group (n = 249) and the chemotherapy group (Oxaliplatin + Capecitabine/Oxaliplatin + Leucovorin + Fluorouracil, n = 250). The results indicated a median OS of 10.4 months (95% CI, 9.1–12.0 months) and 10.9 months (95% CI, 9.6–12.4 months) for the two groups, respectively. However, the 24-month survival rates were 22.1% (95% CI, 16.8–28.0) for the combination group and 15.5% (95% CI, 11.8–20.9) for the chemotherapy alone group. In PD-L1 positive patients, the median survival for the combination group was 14.9 months (95% CI, 13.7–16.3), significantly higher than the chemotherapy alone group at 11.6 months (95% CI, 10.4–12.6). 23 For a summary of the efficacy of combination therapy, refer to Table 1.
Summary of the efficacy of ICIs combined with chemotherapy in gastric cancer.

Chemo, chemotherapy; CPS, combined positive score; ICI, immune checkpoint inhibitor; mOS, median overall survival; mPFS, median progression-free survival; Pt Characteristics, patient characteristics.
Traditional beliefs suggest that chemotherapy has certain side effects on the body’s immune system, such as the inhibition of tumor-killing T lymphocytes, promotion of immune tolerance, and suppression, among other effects. However, in recent years, researchers have discovered that chemotherapy may have a bidirectional regulatory effect on the immune microenvironment of the body.24–30 This includes promoting the release of immunogenic substances and cross-presentation of antigens by tumor cells, enhancing the sensitivity of the body’s immune cells. These effects are mediated by different types of chemotherapy drugs.24,25 Furthermore, studies indicate that chemotherapy drugs can induce the expression of PD-L1 in tumor cells. For example, 5-fluorouracil can enhance the expression of PD-L1 in gastrointestinal tumor cells, and combined with cisplatin, it can increase PD-L1 expression in esophageal squamous cell carcinoma tissues.26,27 Platinum-based drugs, anthracyclines, and taxanes can also enhance PD-L1 expression in ovarian cancer cells, bladder cancer cells, and liver cancer cells.28–30 This provides a basis for clinical combination therapy, suggesting that chemotherapy may enhance the responsiveness of ICIs. However, specific combination treatment regimens, beneficiary patient subgroups, and the timing of combination therapy still warrant further in-depth research.
Immunotherapy checkpoint inhibitors combined with targeted therapy
Currently, there is a growing body of research on targets that promote the progression of gastric cancer. Targets such as epidermal growth factor receptor (EGFR) and vascular endothelial growth factor receptor (VEGFR) have successfully transitioned into clinical treatments. 31 These targets are widely utilized in clinical practice, thereby altering the traditional treatment paradigm for gastric cancer.
Ramucirumab is a targeted therapy that acts on the VEGFR-2. Its primary mechanism involves blocking the activation of VEGFR-2, thereby inhibiting the growth and spread of tumor blood vessels, consequently suppressing tumor growth. A clinical trial evaluating the combination of ramucirumab and pembrolizumab in the treatment of advanced or metastatic gastric cancer enrolled a total of 28 patients. The results revealed a PFS of 5.6 months (PD-L1 positive, 8.6 months; PD-L1 negative, 4.3 months) and an OS of 14.6 months (PD-L1 positive, 17.3 months; PD-L1 negative, 11.3 months). 32 Trastuzumab is a targeted therapy that acts on the HER-2. Another study assessed the efficacy of pembrolizumab combined with trastuzumab in advanced gastric cancer patients with dual HER-2 and PD-L1 positivity, demonstrating a median OS of 20.5 months and an overall response rate (ORR) of 48%. 33 Phase III clinical trials have also confirmed the clinical benefits of pembrolizumab plus trastuzumab and chemotherapy. This combination therapy can reduce the risk of disease progression or death in gastric or gastroesophageal junction adenocarcinoma patients with HER-2 positive status and a PD-L1 CPS of ⩾1. 34 Furthermore, in the treatment of advanced gastric cancer with the combination of ramucirumab and nivolumab, an 8-month follow-up revealed an ORR of 22%, a complete response rate of 24.4%, and a disease control rate (DCR) of 62.2%. 35 A single-center phase II clinical study evaluated the clinical efficacy and safety of pembrolizumab combined with trastuzumab and chemotherapy in HER-2 positive metastatic gastric cancer, achieving a remarkable ORR of 87% and a prolonged PFS of 11.3 months. 36 Lenvatinib is a multitargeted small molecule targeted therapy primarily inhibiting receptors such as VEGFR, platelet-derived growth factor receptor, and fibroblast growth factor receptor. Its main function is to inhibit tumor angiogenesis and tumor cell proliferation. Another study assessed the efficacy of lenvatinib in combination with pembrolizumab in gastric cancer patients who had failed chemotherapy, revealing an ORR of 69% and a remarkable DCR of 100%. The median PFS for the entire study cohort was 6.9 months. 37
The abovementioned studies indicate that ICIs combined with targeted therapy exhibit promising efficacy. Particularly, this combination appears to be more effective in patients with dual positivity for corresponding targets and immune checkpoints. 38 Research suggests that targeted drugs can activate immune effector cells to release IFN-γ, leading to the upregulation of PD-L1 on the surface of cancer cells. 39 Therefore, the combined application of both can synergistically enhance the body’s antitumor effects, with good safety profiles.
Immunotherapy checkpoint inhibitors combined with radiotherapy
Radiotherapy refers to the use of various types of radiation to damage the genetic material of tumor cells, thereby inhibiting and killing these cells. 40 Currently, there are numerous clinical studies exploring the combination of radiotherapy with ICIs in various cancers such as lung cancer, renal cancer, bladder cancer, and more. Positive clinical outcomes have been observed in terms of DCR, ORR, and patient survival.41–43 In a phase III clinical trial (NCT02743494) evaluating the clinical efficacy of ICIs in patients with completely resected (R0) stage II or stage III esophageal or gastroesophageal junction cancer, patients received nivolumab or placebo after receiving adjuvant chemoradiotherapy. The median PFS for patients in the nivolumab group was 22.4 months (95% CI, 16.6–34.0), compared to 11.0 months (95% CI, 8.3–14.3) in the placebo group. The incidence of grades 3–4 TRAEs was 13% in the nivolumab group and 6% in the placebo group. Patients receiving nivolumab had a significantly higher PFS compared to those receiving a placebo, and the treatment was well tolerated. 44 Furthermore, in a case report of gastric cancer brain metastasis patients treated with radiotherapy combined with nivolumab, a significant reduction in distant metastatic lesions was observed. 45 Clinical trials on combination therapy are shown in Table 2.
Related studies of radiotherapy combined with ICIs in gastric cancer or solid tumors.
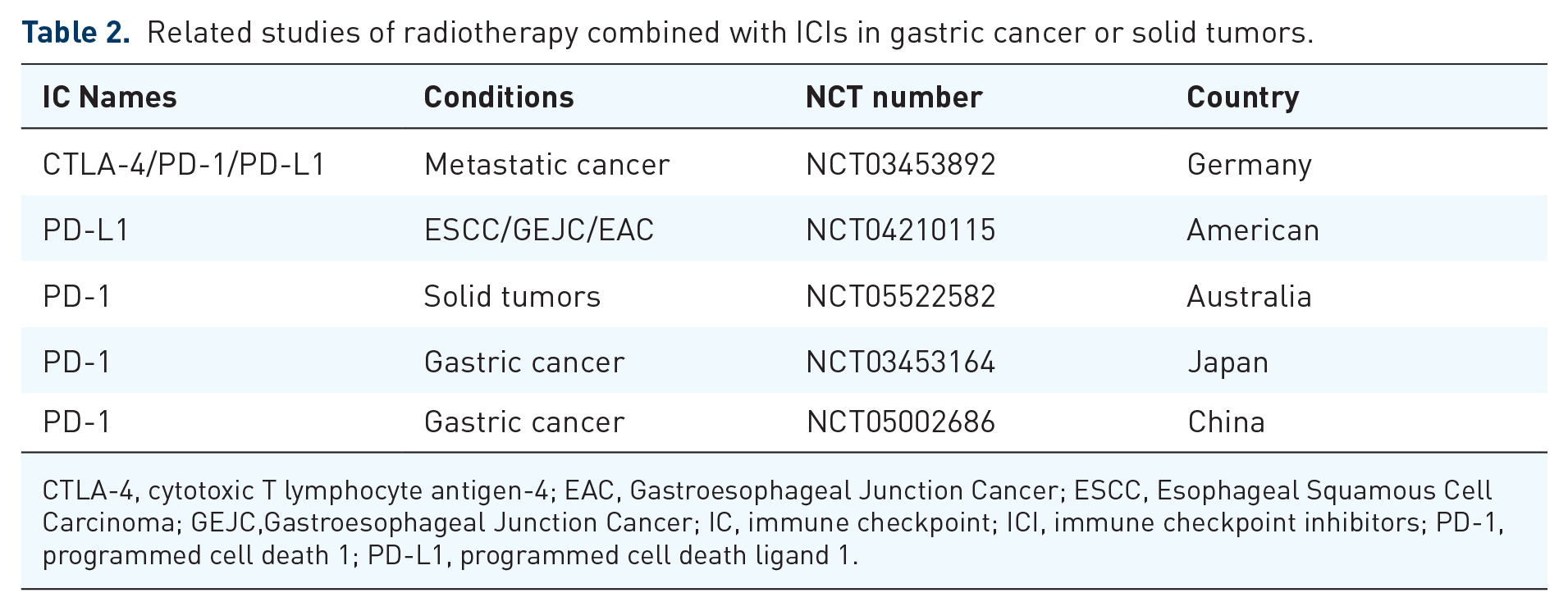
CTLA-4, cytotoxic T lymphocyte antigen-4; EAC, Gastroesophageal Junction Cancer; ESCC, Esophageal Squamous Cell Carcinoma; GEJC,Gastroesophageal Junction Cancer; IC, immune checkpoint; ICI, immune checkpoint inhibitors; PD-1, programmed cell death 1; PD-L1, programmed cell death ligand 1.
Radiotherapy directly damages cancer cells, inducing various forms of cell deaths such as apoptosis, necrosis, and autophagy-dependent cell death. 46 The death of tumor cells releases specific antigens, and the overexpression of major histocompatibility complex class I facilitates the presentation of tumor-specific antigens, thereby enhancing the recognition and cytotoxicity of cytotoxic T cells. 47 Radiotherapy can also modulate the tumor microenvironment, eliciting the release of inflammatory mediators, thereby increasing infiltration of immune cells such as T cells and natural killer (NK) cells. 48 Research indicates that postradiation, the release of fragmented double-stranded DNA from tumors can induce activation of the cyclic GMP-AMP synthase-stimulator of interferon genes pathway, subsequently activating downstream serine/threonine-protein kinase TANK-binding kinase 1, enhancing the phosphorylation of interferon regulatory factor 3, ultimately promoting the expression of PD-L1 in tumor cells. 49 Concurrently, radiation-induced double-strand DNA breaks activate the Ataxia Telangiectasia Mutated (ATM)/ATM and Rad3 Related (ATR)/Checkpoint Kinase 1 (Chk1) pathway, leading to increased expression of PD-L1 in tumor cells, thereby enhancing the efficacy of ICIs. 50
Immunotherapy checkpoint inhibitors combined with PDT
PDT is an emerging treatment modality for gastrointestinal tumors. It utilizes photosensitizers that specifically bind to tumor tissues. 51 When exposed to a specific wavelength of light, these photosensitizers undergo energy conversion, selectively killing tumor tissues in an oxygen-rich environment. 52 The primary mechanism involves the generation of reactive oxygen species activated by PDT through various signaling pathways. The three main action pathways include (1) direct damage to tumor cells; (2) injury to tumor vascular endothelial cells; (3) promotion of aseptic inflammation, inducing the body’s immune response.53,54 Due to its high selectivity, repeatability, and minimal side effects, PDT is increasingly being applied in clinical practice.
After PDT, localized tumor necrosis ensues, inducing aseptic inflammation in the affected area and triggering the release of various inflammatory cytokines such as IL-2, IL-6, IL-8, interferon-gamma (IFN-γ), TNF-α, Damage-Associated Molecular Patterns (DAMP), Calreticulin (CRT), High Mobility Group Box 1 (HMGB1), Heat Shock Proteins (HSP), among others. This cascade of events induces the infiltration of immune cells, transforming a previously ‘cold’ tumor into a ‘hot’ tumor. Furthermore, studies indicate that PDT can stimulate the maturation of antigen-presenting cells, activate adaptive immune responses, and enhance the body’s antitumor response.55–57 In a dual tumor model of colorectal cancer (MC38 or CT26), pretreatment with CTLA-4 inhibitors before PDT significantly reduces tumor burden and extends survival compared to individual treatments. 58 Additionally, IL-6 generated from tissue damage induced by PDT can enhance the expression and stability of PD-L1 through STAT3 and JAK1.59,60 P-glycoprotein (P-gp), also known as multidrug resistance protein 1 (MDR1), is an efflux transporter that influences the absorption, distribution, and elimination of a variety of compounds. PDT also diminishes the ATPase activity of P-gp, lowering the activity of P-gp may potentially mitigate issues of drug resistance. 61 Currently, the clinical efficacy of PDT has been established in both early and advanced stages of gastric cancer.62,63 In a study by Yu et al., 40 patients treated with ICIs were retrospectively analyzed to assess the impact of PDT. Among these, five patients diagnosed with gastric adenocarcinoma underwent sample collection before and after PDT. The results demonstrated that patients in the PDT group exhibited significantly improved OS following ICI treatment compared to those in the non-PDT group. T-cell receptor analysis revealed a specific clonal expansion of cytotoxic T lymphocytes (CTLs) after PDT, while there was a reduction in Treg cells. Additionally, upregulation of the B2M gene in tumor cells after PDT was associated with increased infiltration of immune cells. 64 Furthermore, Ma et al. reported a case involving a 72-year-old male patient with gastric cancer who received combined treatment with PDT and ICIs. Following this treatment, complete disappearance of gastric cancer tissue, normalization of tumor markers, and no evidence of recurrence or metastasis were observed during a 7-month follow-up period. 65 Although comprehensive clinical data are currently lacking, the aforementioned clinical findings and case reports suggest that combining PDT with ICIs represents a viable therapeutic approach for gastric cancer, combined therapy has the potential to enhance the sensitivity of ICIs, broaden the scope of beneficiaries, and reinforce the antitumor efficacy.
Immunotherapy checkpoint inhibitors combined with thermotherapy
Thermotherapy involves elevating the local temperature of tumor tissues through various physical methods to induce a thermal effect, ultimately leading to the destruction of tumor cells. Treatment modalities include radiofrequency ablation, ultrasound thermotherapy, magnetic wave thermotherapy, among others.66,67 Studies have shown that in mouse animal models, the combination of inflammatory Radiofrequency Ablation (RFA) with immune PD-1 inhibitors significantly enhances T-cell immune responses, promoting a more sustained antitumor effect and extending survival. 68 Duffy et al. (NCT01853618) evaluated the study of RFA combined with tremelimumab in liver cancer, with results indicating 6- and 12-month PFS rates of 57.1% and 33.1%, respectively. The median PFS and median OS were 7.4 months (95% CI, 4.7–19.4 months) and 12.3 months (95% CI, 9.3–15.4 months), respectively. 69 For gastric cancer, research shows hyperthermia downregulates PLEK2/PD-L1, which further inhibits cell proliferation, invasion, and migration, promotes cell apoptosis; meanwhile, PLEK2 knockdown reverses the inhibition of T cell IFN-γ release by tumor cells and may be involved in immune regulation. 70
Thermotherapy can induce localized tumor cell damage and promote the release of various cell-derived factors, such as chemokines, CCL2/CCL5 cytokines, heat shock proteins, CD80, CD83, CD86, and inflammatory cytokine TNF-α, inducing a local inflammatory response and enhancing immune cell infiltration. 71 It can also increase the expression of activation and cytotoxic surface markers on circulating T cells and NK cells. 72 Furthermore, thermotherapy can increase the expression of PD-L1 on tumor cells. 68 Therefore, combination therapy can lead to a favorable antitumor effect. Although there are very limited reports on this approach in gastric cancer, the abovementioned studies provide a reliable basis and new strategy for our understanding of combination therapy.
Immunotherapy checkpoint inhibitors combined with immune adjuvant
Gastric cancer patients often experience poor nutritional status and weakened immune defenses due to obstruction, while prolonged tumor burden leads to T cell exhaustion. 73 Immunoadjuvants are small molecular peptides or glycoproteins that regulate the body’s immune system, aiming to enhance immune responses by activating both innate and adaptive immunity. 74 CpG oligodeoxynucleotide (CpG-ODN) is an agonist of Toll-like receptor 9 (TLR9) that activates immune responses by inducing co-stimulatory molecules CD80 and CD86, as well as cytokines TNF-α, IL-6, IL-12, and type I IFN in dendritic cells through TLR9.75–77 Additionally, injecting CpG-ODN into the tumor site can induce local tumor cell death, release more tumor antigens into the tumor microenvironment, activate dendritic cells for antigen uptake and presentation, and initiate specific cytotoxic CD8+ T cell responses to promote tumor destruction.75–77 Researchers utilized CpG-ODN and the gastric cancer-specific antigen MG7 to construct nanovaccine and conducted in vivo antitumor experiments. It was shown that the tumor masses formed in the mice immunized with coencapsulated nanovaccine (0.0825 g) markedly smaller (p < 0.01) than those formed in the mice immunized with nanovaccine encapsulated with antigen peptide alone (0.4465 g). A tumor inhibiting rate as high as 82.5% of the coencapsulated nanovaccine was obtained, while nanovaccine encapsulated with peptide only could not achieve the same effect (28.5%) (p < 0.01). 78 Pseudomonas aeruginosa is a Gram-negative bacterium that has been combined with mannose-sensitive hemagglutination to chemically inactivate P. aeruginosa-mannose-sensitive hemagglutinin (PA-MSHA). It has been proven by related experiments that it has a broad-spectrum immunomodulatory effect and has an obvious killing effect on tumor cells.79,80 Researchers constructed a patient-derived xenograft model of gastric cancer in mice to compare the efficacy of PD-1 inhibitors alone, PD-1 inhibitors in combination with PA-MSHA, and PD-1 inhibitors combined with PA-MSHA and bevacizumab. Study found that PD-1 inhibitors alone could not significantly inhibit the growth of transplanted tumors in mice. PD-1 inhibitors combined with bevacizumab, anti-PD-1 mAb combined with bevacizumab and PA-MSHA could all significantly inhibit tumor growth in mice, and the combination of three drugs presented the highest tumor inhibition rate. Anti-PD-1 monoclonal antibody combined with bevacizumab and PA-MSHA could significantly upregulate the number of T helper 1 cells, CD8 + T cells, and Type I tumor-associated macrophages (TAMs), while downregulate the number of Th2-type cells, myeloid-derived suppressor cells, Treg cells, and Type II TAMs. 81
The above studies indicate that immunoadjuvants play a role in reshaping the tumor microenvironment, and their combination with ICIs can optimize their antitumor effects. Currently, numerous clinical trials, such as NCT03618641, NCT03507699, NCT03007732, NCT04050085, conducted in melanoma, colorectal cancer, prostate cancer, and pancreatic cancer, respectively, the effect potential of combination therapy cannot be underestimated. Therefore, this approach could be a viable strategy for application in gastric cancer.
Application of dual ICIs
With the successful clinical outcomes achieved by dual immune checkpoints in lung cancer, 82 renal cancer, 83 and melanoma, 84 exploration is underway in gastric cancer as well. The Check Mate-032 study enrolled a total of 160 patients with locally advanced or metastatic chemotherapy-resistant gastric cancer, esophageal cancer, or gastroesophageal junction cancer. Among them, 59 received nivolumab at a dose of 3 mg/kg, 49 received nivolumab at 1 mg/kg in combination with ipilimumab at 3 mg/kg, and 52 received nivolumab at 3 mg/kg in combination with ipilimumab at 1 mg/kg. The results indicated ORRs of 12%, 24%, and 8%, respectively. Treatment responses were observed regardless of the tumor cell PD-L1 expression levels. The median follow-up times for the three groups were 28, 24, and 22 months, with 12-month PFS rates of 8%, 17%, and 10%, and 12-month OS rates of 39%, 35%, and 24%, respectively. Adverse events related to treatment of grade 3/4 were reported by 17%, 47%, and 27% of patients in the three groups. 85 Nivolumab and ipilimumab-based neoadjuvant therapy is feasible and associated with no unexpected toxicity and a high PCR rate in patients with dMMR/MSI-H resectable gastric/GEJ adenocarcinoma, 2 patients received neoadjuvant immunotherapy (27 patients completed all cycles). Twenty-nine patients underwent surgery; three did not have surgery and had complete endoscopic response with tumor-free biopsies and a normal computed tomography scan (two refused surgery and one had metastasis at inclusion). 16 Nivolumab and nivolumab combined with ipilimumab demonstrated clinically significant antitumor activity, durable responses, encouraging long-term OS, and manageable safety profiles in patients with chemotherapy-resistant esophagogastric cancer. Currently, a number of clinical trials have been conducted on the study of dual ICIs in gastric cancer (see Table 3); therefore, dual ICIs are also a strategy for adjuvant treatment of gastric cancer.
Related research on dual ICIs combination therapy for gastric cancer or solid tumors.

IC, immune checkpoint; ICI, immune checkpoint inhibitor.
Summary
There are numerous approaches to combining ICIs, encompassing chemotherapy, targeted therapy, radiotherapy, PDT, thermotherapy, immune adjuvants, and dual immunotherapy. Each combination offers distinct advantages, as detailed in Table 4. Combining chemotherapy addresses the limited reactivity of standalone ICIs, enhancing treatment sensitivity. The synergy of combined targeted therapy, particularly in patients with dual positive target genes, is notably significant. Immune adjuvants regulate the body’s immune response, amplifying the efficacy of ICIs in combination therapy. Radiotherapy, PDT, and thermotherapy activate the body’s immune response, alter the tumor microenvironment, diminish drug-resistant protein activity, and elevate tissue PD-L1 expression levels. Combination therapy not only directly eradicates tumors but also generates distant effects, overcoming the challenges of low response rates and drug resistance associated with ICIs. Consequently, the integration of ICIs with the aforementioned three physical therapies emerges as a pivotal treatment modality for advanced gastric cancer. Radiotherapy, PDT, and thermotherapy each offer specific advantages. For instance, radiotherapy’s sensitivity to squamous cell carcinoma makes it an optimal choice for esophagogastric junction squamous cell carcinoma. In cases of large exophytic tumors and liver metastases, the unique characteristics of PDT may leave unirradiated areas due to treatment specifics and thickness limitations, favoring the selection of thermotherapy therapy. For elderly patients, those in poor physical condition, ulcerative-type tumors, and severe obstructions unable to tolerate chemotherapy and radiotherapy’s side effects, PDT can be used as one of the treatments. Therefore, tailoring different combination strategies based on individual patient and tumor characteristics is imperative to achieve optimal antitumor efficacy.
Potential research value of combination therapy with ICIs.

cGAS-STING, cyclic GMP-AMP synthase-stimulator of interferon genes; ICI, immune checkpoint inhibitors; NK, natural killer; PD-L1, programmed cell death ligand 1; PDT, photodynamic therapy; P-gp, P-glycoprotein.
The potential of ICIs combination therapy cannot be underestimated. In clinical practice, determining the optimal selection, timing, sequencing, and frequency of ICIs combination therapy to achieve the best PFS, OS, minimal adverse reactions, and enhanced cost-effectiveness poses a critical challenge. This warrants extensive experimental research. In the realm of physical therapy integration, each treatment modality presents its own set of advantages and disadvantages. Exploring how to judiciously choose physical therapy in clinical practice is also a subject deserving of in-depth study. Currently, both domestically and internationally, there is a robust focus on research into the development of highly responsive immune checkpoints, and the identification of markers indicative of therapeutic responsiveness to ICIs remains a prominent research area. It is anticipated that with the continued advancement of science and technology, treatments based on ICIs will unfold broader prospects, delivering positive outcomes to an increasing number of patients.
