Abstract
Background:
Renal cell carcinoma (RCC) is a common malignancy with approximately 30% of cases diagnosed at the advanced or metastatic stage. While single-agent vascular endothelial growth factor-targeted therapy has been a mainstay of treatment, data from multiple phase III trials assessing first-line immune checkpoint inhibitor (ICI) combinations have demonstrated a significant survival benefit.
Methods:
A systematic search of the published and presented literature was performed to identify phase III trials assessing ICI combination regimens in RCC using search terms ‘immune checkpoint inhibitors’ AND ‘renal cell carcinoma,’ AND ‘advanced’.
Results:
Six phase III trials showed significant benefits for ICI combinations compared with sunitinib. Nivolumab plus ipilimumab significantly improved overall survival [OS; median, 47.0 versus 26.6 months, hazard ratio (HR) = 0.68, 95% confidence interval (CI) = 0.58–0.81, p < 0.0001) and progression-free survival (PFS; median 11.6 versus 8.3 months, HR = 0.73, 95% CI = 0.61–0.87, p = 0.0004) in International Metastatic renal cell carcinoma Database Consortium intermediate and poor-risk patients. OS was also significantly improved for ICI plus tyrosine kinase inhibitor combinations regardless of risk, including pembrolizumab plus either axitinib (HR = 0.73, 95% CI = 0.60–0.88, p < 0.001) or lenvatinib (HR = 0.66, 95% CI = 0.49–0.88, p = 0.005) and nivolumab plus cabozantinib (HR = 0.66, 95% CI = 0.50–0.87, p = 0.003). No new safety signals were identified.
Conclusions:
Phase III first-line trials of ICI combinations showed survival benefits compared with a control arm of sunitinib. Global access to these combinations should be made available to patients with advanced RCC.
Keywords
Introduction
Kidney cancer is a common malignancy with over 430,000 new cases reported worldwide in 2020 resulting in approximately 180,000 deaths. 1 Renal cell carcinoma (RCC) represents the majority of kidney cancers (90–95%), with clear-cell RCC being the most common histological subtype.2,3 Approximately 30% of RCC cases are diagnosed at the advanced or metastatic stage and close to 80% of these patients have intermediate or poor-risk disease as per the International Metastatic renal cell carcinoma Database Consortium (IMDC) criteria.2–4 RCC is typified by inactivation of the von Hippel–Lindau tumor suppressor gene leading to high expression of the proangiogenic vascular endothelial growth factor (VEGF).5–7
Until recently, the mainstay of first-line therapy for advanced RCC involved the inhibition of angiogenesis with tyrosine kinase inhibitors (TKIs) against multiple receptors including those of VEGF (e.g. sunitinib and pazopanib).8–10 Historical benchmarks for median overall survival (OS) in the VEGF-targeted therapy era by IMDC risk groups have been 43, 23, and 8 months for favorable (IMDC 0), intermediate (IMDC 1 or 2), and poor-risk patients (IMDC ⩾3), respectively. 11 For years, the research landscape typically involved multiple alternative TKIs that also inhibit VEGF receptors including axitinib, cabozantinib, and lenvatinib,12–14 a monoclonal antibody (MoAb) that directly inhibits the function of VEGF (bevacizumab) 15 and inhibition of the mammalian target of rapamycin (everolimus). 16
Immune checkpoint inhibitors (ICIs) include cytotoxic T lymphocyte antigen 4 inhibitors (e.g. ipilimumab) and those against the programmed cell death protein 1 (PD-1) in peripheral tissues (e.g. nivolumab and pembrolizumab) or its ligand (PD-L1) (e.g. atezolizumab and avelumab), some of which have recently become preferred for most first-line treatment of RCC.17–23 Therapies directed at VEGF or its receptor (VEGF-directed, anti-angiogenic monoclonal antibodies or TKIs) are thought to have immunomodulatory effects including the enhancement of immune cell infiltration by normalizing tumor vasculature holding the promise of synergistic activity.24–26 In the last 3 years, results from multiple phase III trials assessing the first-line benefits of ICI combinations involving dual ICIs or ICIs in combination with VEGF-directed therapies have become available. This review will consider the safety and efficacy of these regimens in newly diagnosed advanced RCC and provide practical clinical guidance on their use in this setting.
Methods
A search of published and presented literature was conducted to identify phase III trials with outcomes assessing ICI combination regimens in RCC. PubMed (all time to 19 March 2022), the proceedings from the 2019, 2020, and 2021 American Society of Clinical Oncology (ASCO) and the European Society for Medical Oncology (ESMO) annual meetings as well as the ASCO Genitourinary Cancers Symposium were searched using the key search terms ‘immune checkpoint inhibitors’ AND ‘renal cell carcinoma’, AND ‘advanced’ OR respective aliases. A supplemental bibliographic search of review articles and pooled/meta-analyses was also conducted. In addition, directed searches were performed after the database search cutoff date to ensure that the most up-to-date reports of eligible studies were considered.
English language records were vetted at abstract level and confirmed at full text as needed. Excluded studies included those that were non-original research, preclinical, correlative science, not specific to RCC, in early stages of disease, retrospective, prospective phase I, II, IIIb, IV trials, or undefined phase, without outcomes as well as addressing non-systemic therapy combinations, and duplicate or prior reports.
Findings
The literature search identified a total of 628 records, resulting in a total of six eligible phase III trials (Preferred Reporting Items for Systematic Reviews and Meta-Analyses, Figure 1).27–32

Preferred Reporting Items for Systematic Reviews and Meta-Analyses diagram.
First-line ICI combinations
Six phase III trials assessed ICI combinations as first-line systemic treatment of advanced or metastatic RCC compared with the control arm of sunitinib (Table 1).27–32 One evaluated a dual ICI combination, 30 another an ICI and anti-VEGF MoAb combination, 32 and four ICI plus TKI combinations.27–29,31
Phase III trials assessing efficacy of first-line ICI combinations in advanced RCC. IRC assessed the efficacy of outcomes in intent-to-treat populations of phase III ICI combination trials in previously untreated advanced RCC summarized and ordered by date of first report of results from initial analysis. Outcomes in specific IMDC risk populations were described when used in the primary endpoint analyses. Outcomes in PD-L1-positive subgroups summarized in text only.
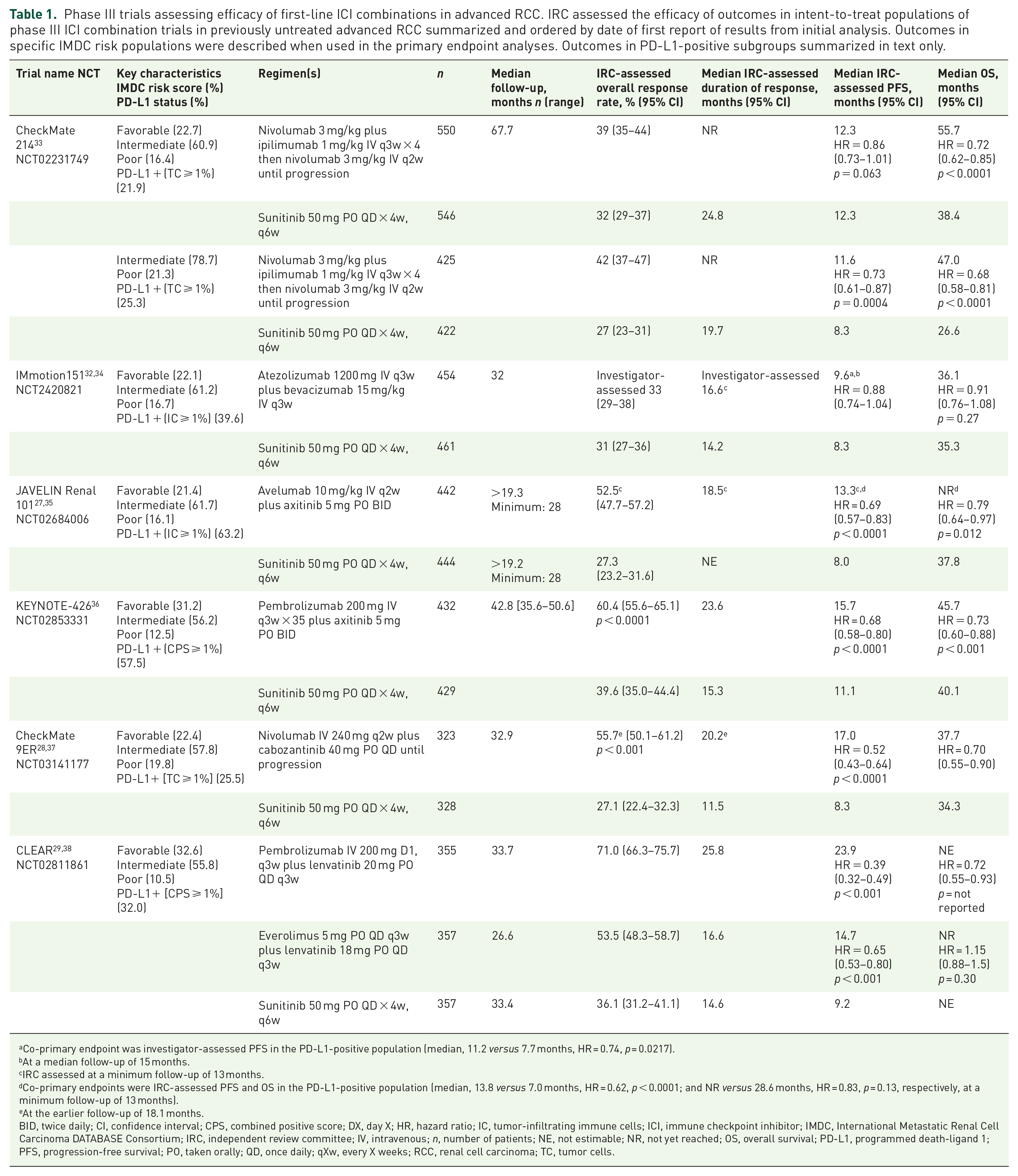
Co-primary endpoint was investigator-assessed PFS in the PD-L1-positive population (median, 11.2 versus 7.7 months, HR = 0.74, p = 0.0217).
At a median follow-up of 15 months.
IRC assessed at a minimum follow-up of 13 months.
Co-primary endpoints were IRC-assessed PFS and OS in the PD-L1-positive population (median, 13.8 versus 7.0 months, HR = 0.62, p < 0.0001; and NR versus 28.6 months, HR = 0.83, p = 0.13, respectively, at a minimum follow-up of 13 months).
At the earlier follow-up of 18.1 months.
BID, twice daily; CI, confidence interval; CPS, combined positive score; DX, day X; HR, hazard ratio; IC, tumor-infiltrating immune cells; ICI, immune checkpoint inhibitor; IMDC, International Metastatic Renal Cell Carcinoma DATABASE Consortium; IRC, independent review committee; IV, intravenous; n, number of patients; NE, not estimable; NR, not yet reached; OS, overall survival; PD-L1, programmed death-ligand 1; PFS, progression-free survival; PO, taken orally; QD, once daily; qXw, every X weeks; RCC, renal cell carcinoma; TC, tumor cells.
CheckMate 214 randomized 1096 patients of all risk groups 1:1 to receive nivolumab plus ipilimumab compared with sunitinib, with co-primary endpoints of OS, independent review committee (IRC)-assessed progression-free survival (PFS) and objective response rate (ORR) in IMDC intermediate and poor-risk patients (intermediate/poor risk, n = 847), as well as additional exploratory endpoints among 249 favorable-risk patients. At a median follow-up of 25.2 months, initial findings showed a statistically significantly higher OS [median not yet reached (NR) versus 26.0 months, HR = 0.63, 95% confidence interval (CI) = 0.44–0.89 p < 0.001] and increased PFS (median 11.6 versus 8.4 months, HR = 0.82, 95% CI = 0.64–1.05, p = 0.03) for the ICI combination versus sunitinib, although PFS did not reach the pre-specified threshold for significance (p = 0.009). 39 With a longer median follow-up of 67.7 months, the ICI combination continued to show significant OS (median 47.0 versus 26.6 months, HR = 0.68, 95% CI = 0.58–0.81, p < 0.0001), and PFS (median 11.6 versus 8.3 months, HR = 0.73, 95% CI = 0.61–0.87, p = 0.0004) improvements compared with sunitinib in intermediate/poor-risk population, with OS benefits also seen in the intent-to-treat (ITT) population (median 55.7 versus 38.4 months, HR = 0.72, 95% CI = 0.62–0.85, p < 0.0001) although not in favorable-risk patients (median 74.1 versus 68.4, HR = 0.94, 95% CI = 0.65–1.37, p = 0.77). 33 An improved overall response rate (42% versus 27%) and long duration of responses (DoRs, median NR versus 19.7 months) were also seen in intermediate/poor-risk patients, with reduced ORR benefit in the ITT population (39% versus 32%) and which favored sunitinib in favorable-risk patients (30% versus 52%). Median time to confirmed deterioration was significantly longer for nivolumab plus ipilimumab versus sunitinib for all scores from the 19-item Functional Assessment of Cancer Therapy-Kidney Symptom Index (FKSI-19, p < 0.05). 40 Discontinuations due to treatment-related adverse events (TRAEs) occurred in 21.8% versus 12.3% of patients, grade ⩾3 TRAEs were reported in 46.6% and 63.9%, and treatment-related deaths occurred in 1.5% versus 0.7% of patients receiving nivolumab plus ipilimumab versus sunitinib (Table 2).
Safety outcomes from clinical trials assessing first-line ICI combinations in metastatic RCC.

AEs, adverse events; ALT, alanine aminotransferase; AST, aspartate aminotransferase; IO, immunotherapy; n, number of patients; PPE, Palmar-plantar erythrodysesthesia syndrome; RCC, renal cell carcinoma; TRAE, treatment-related adverse event.
IMmotion151 randomized 915 patients with RCC and predominantly a clear-cell component (92%) 1:1 to receive atezolizumab plus bevacizumab compared with sunitinib. At median follow-ups of 15 and 24 months for PFS and OS, respectively, similar investigator-assessed ORRs and DoRs were observed between arms, with no statistically significant improvements seen in PFS or OS for the ICI plus VEGF-directed therapy combination compared with sunitinib. 32 At a longer median follow-up of 32 months, no significant improvement in the co-primary endpoint of OS was seen in ITT patients (median OS, 36.1 versus 35.3 months, HR = 0.91, 95% CI = 0.76–1.08, p = 0.27, Table 1) or in patients with PD-L1-positive tumors (median OS, 38.7 versus 31.6 months, HR = 0.85, 95% CI = 0.64–1.13, p = not reported). 34 FKSI-19 scores favored atezolizumab plus bevacizumab over sunitinib for health-related quality of life (HR = 0.68, 95% CI = 0.58–0.81). 41 Discontinuations due to TRAEs occurred in 5.3% versus 8.3% of patients, grade ⩾3 TRAEs were reported in 40.4% and 53.8%, and treatment-related deaths occurred in 1.1% versus 0.2% of patients receiving atezolizumab plus bevacizumab versus sunitinib (Table 2). 32
JAVELIN Renal 101 randomized 886 patients 1:1 to receive avelumab plus axitinib compared with sunitinib in patients with primarily PD-L1-positive tumors (63.2%). Co-primary endpoints were IRC-assessed PFS and OS among patients with PD-L1-positive tumors. With a minimum follow-up of 13 months, significant IRC-assessed PFS improvements were observed for the ICI combination over sunitinib in patients with tumors overexpressing PD-L1 and ITT patients, with a near doubling of ORRs and median DoRs not reported (Table 1). 27 At a minimum follow-up of 28 months, there was no statistically significant OS improvement for the ICI combination over sunitinib (median OS, NE versus 37.8 months, HR = 0.79, 95% CI = 0.64–0.97, p = 0.012), although further follow-up is ongoing. 35 Discontinuations due to TRAEs occurred in 7.6% versus 13.4% of patients, grade ⩾3 TRAEs were reported in 56.7% and 55.4%, and treatment-related deaths occurred in 0.7% versus 0.2% of patients in the combination and sunitinib arms, respectively (Table 2). 27
KEYNOTE-426 randomized 861 patients 1:1 to receive the PD-1 inhibitor pembrolizumab plus axitinib compared with sunitinib. With a median follow-up of 30.6 months in the ITT population, statistically significant improvements for the ICI combination versus sunitinib were observed for the co-primary endpoint of IRC-assessed PFS (median 15.4 versus 11.1 months, HR = 0.71, 95% CI = 0.60–0.84, p < 0.0001). 31 At a longer median follow-up of 42.8 months, PFS benefit remained significant (p < 0.0001), with significant benefits also demonstrated for the co-primary endpoint of OS (median 45.7 versus 40.1 months, HR = 0.73, 95% CI = 0.60–0.88, p < 0.001) and ORR (60.4% versus 39.6%, p < 0.0001). The median DoR was numerically improved (23.6 versus 15.3 months, p = not reported) for the ICI combination compared with sunitinib. 36 Changes in mean FKSI-disease-related symptom scores from baseline to 30 weeks were similar for pembrolizumab plus axitinib versus sunitinib (−0.8 versus −0.3), with the −0.5 difference below the threshold for minimally important difference (±3 points). 42 Discontinuation of any treatment due to TRAEs occurred in 25.9% versus 10.1%, grade ⩾3 TRAEs were reported in 62.9% versus 58.1%, and treatment-related deaths occurred in 0.9% versus 1.6% of patients receiving pembrolizumab plus axitinib compared with sunitinib (Table 2). 43
CheckMate 9ER randomized 651 patients 1:1 to receive the PD-1 inhibitor nivolumab plus cabozantinib compared with sunitinib. With a median follow-up of 18.1 months, statistically significant benefits were seen in the primary endpoint of IRC-assessed PFS for nivolumab plus cabozantinib versus sunitinib in ITT patients (median PFS, 16.6 versus 8.3 months, HR = 0.51, 95% CI = 0.41–0.64, p < 0.001), as well as in the key secondary endpoint of OS (median OS, NR versus NR, HR = 0.60, 98.9% CI = 0.40–0.89, p = 0.001; Table 1), 28 which persisted with a longer median follow-up of 32.9 months (median OS, 37.7 versus 34.3 months, HR = 0.70, 95% CI = 0.55–0.90, p = NR). 37 Initial IRC-assessed ORR was also significantly improved for the combination (55.7% versus 27.1%, 95% CI = 21.7–35.6, p < 0.001), with median DoRs of 20.2 versus 11.5 months. 28 Compared with sunitinib, nivolumab plus cabozantinib was associated with improvements in FKSI-19 total score (median time to first deterioration 6.24 versus 3.48 months, HR = 0.70, 95% CI = 0.56–0.86, nominal p = 0.0007). 44 Discontinuation of any trial drug due to AEs occurred in 19.7% of patients receiving nivolumab plus cabozantinib and in 16.9% of patients receiving sunitinib, and grade ⩾3 AEs were reported in 60.6% and 50.9% of patients, respectively. One death attributed to treatment was reported in the combination arm (0.3% of patients) and two occurred in the sunitinib group (0.6%).
CLEAR randomized 1069 patients 1:1:1 to receive pembrolizumab or everolimus plus lenvatinib, with both combinations compared with sunitinib. At a median follow-up of 26.6 months, the primary endpoint of IRC-assessed PFS statistically significantly favored both the pembrolizumab (median PFS, 23.9 versus 9.2 months, HR = 0.39, 95% CI = 0.32–0.49, p < 0.001; Table 1) and everolimus combinations (median PFS, 14.7 versus 9.2 months, HR = 0.65, 95% CI = 0.53–to 0.80, p < 0.001) compared with sunitinib. 29 At a later median follow-up of approximately 33.5 months, median OS was not estimable although statistically significantly favored the pembrolizumab combination (HR = 0.72, 95% CI = 0.55–0.93, p = not reported). 38 At the earlier follow-up, OS was not improved for the everolimus combination (HR = 1.15, 95% CI = 0.88–1.50, p = .30), IRC-assessed ORRs were 71.0%, 53.5%, and 36.1%, and median DoRs were 25.8, 16.6, and 14.6 months in the pembrolizumab, everolimus, and sunitinib arms, respectively. 29 Pembrolizumab plus lenvatinib showed significant improvements in median time until definitive deterioration compared with sunitinib (30.8 versus. 27.0 months, p < 0.01) using FKSI-disease-related symptom scores. 45 Discontinuation of any treatment due to TRAEs occurred in 37.2% and 27.0% receiving pembrolizumb and everolimus versus 14.4% of patients receiving sunitinib, and grade ⩾3 TRAEs were reported in 71.6 % and 73.0 % of patients versus 58.8% in the control arm. Treatment-related deaths occurred in 1.1% and 0.8% versus 0.3% of patients receiving the pembrolizumab and everolimus combinations versus sunitinib (Table 2).
Discussion
What is the clinical benefit of ICI combination therapy in the first-line treatment of advanced RCC?
Preferred approaches for the first-line treatment of advanced RCC have shifted from TKI monotherapy, such as sunitinib or pazopanib, to combination strategies for most patients.9,10 Six phase III trials evaluating ICI combinations compared to sunitinib have been reported, including one assessing a dual ICI combination 30 and five assessing combinations of an ICI plus an anti-angiogenic agent using either a MoAb 32 or a TKI.27–29,31 At median follow-ups of approximately 20–30 months, neither of the PD-L1 combinations, atezolizumab plus bevacizumab from IMmotion151 nor avelumab plus axitinib from JAVELIN Renal 101 demonstrated OS benefit compared to sunitinib,34,35 although the final survival analyses of JAVELIN Renal 101 are awaited. 35 The rest of this discussion will therefore focus on results from PD-1 combinations, noting that these data should be interpreted in the context that trials have been reported at different timepoints of mature follow-up and that differences exist between trials with regard to IMDC risk group populations studied.
At a median follow-up of 67.7 months, the dual ICI combination of nivolumab plus ipilimumab resulted in a significant 32% reduction in risk of death in intermediate/poor-risk patients and a 28% reduction in the risk of death in ITT patients compared with sunitinib, 33 although the rates of discontinuation of any treatment due to toxicity were higher for the combination (21.8% versus 12.3%). 30 A similar benefit was seen for the PD-1 inhibitor, nivolumab, plus cabozantinib. At a median follow-up of approximately 24 months, the nivolumab combination reduced the risk of death by 34% (p = 0.003) and the risk of progression by 48% (p < 0.0001) in CheckMate 9ER. 28 These benefits were coupled with comparable rates of discontinuation due to toxicity compared with sunitinib (19.7% versus 16.9%).
Benefits were also seen for the PD-1 inhibitor pembrolizumab plus TKI combinations. At a median follow-up of 30.6 months, pembrolizumab plus axitinib reduced the risk of death by 32% (p = 0.003) and risk of progression by 29% (p < 0.001) compared with sunitinib in KEYNOTE-426. 31 Similar outcomes were also seen when pembrolizumab was combined with lenvatinib in the CLEAR study. At a median follow-up of approximately 34 months, the pembrolizumab combination reduced the risk of death by 28% compared with sunitinib (p = not reported) and the risk of progression by 61% (p < 0.001) at an earlier median follow-up of approximately 27 months.29,38 Improvements in OS seen in the KEYNOTE-426 and CLEAR studies were apparent despite the greater use of subsequent therapy in the sunitinib compared with experimental arms.46,47 Rates of discontinuation due to toxicity were higher for both pembrolizumab plus axitinib (25.9% versus 10.1%) 31 and pembrolizumab plus lenvatinib (37.2% versus 14.4%) compared with sunitinib. 29
In the absence of head-to-head trial comparisons, response outcomes may help refine selection between dual ICI or PD-1 inhibitor plus TKI combinations. The ORR reported for the dual ICI combination was 39% with an 18.2% rate of progressive disease, 48 while ORRs for PD-1 inhibitor plus TKI combinations were higher, ranging from 55.7% to 71.0%.28,29,31 The highest ORR was reported for pembrolizumab plus lenvatinib with rates of progressive disease ranging from 5.4% to 11.3%.28,29,31 A similar pattern was seen for complete responses, which ranged from 8% to 10%.28–31,49 DoRs can also be an important therapeutic consideration, potentially affording patients long-term benefit. DoRs for the PD-1 inhibitor plus TKI combinations ranged from 20 to 26 months,28,29,31 while the most durable responses were observed using the dual ICI combination, with the median DoR NR at a median follow-up of 67.7 months. 33 Furthermore, conditional survival data in intermediate/poor-risk patients treated with nivolumab/Ipilimumab showed a substantial increase in the percent probability of remaining progression-free for an additional 2 years beyond randomization (36%) compared with 3 years following randomization (90%), suggesting that responding patients have durable progression-free benefits. In patients with high tumor burden or aggressive course of disease where arresting tumor growth is clinically urgent and progression can be immediately catastrophic, an upfront approach using a PD-1 inhibitor plus a TKI may be preferred based on low rates of progressive disease seen with these regimens. All treatment decisions should consider both the evidence and patient preference and should be made in close collaborations with their physician.
What is the safety of ICI combination therapy in the first-line treatment of advanced RCC?
TKIs are commonly associated with hypertension, diarrhea, palmar-plantar erythrodysesthesia (PPE) and fatigue, and sunitinib generally requires administration daily for 4 weeks out of 6 weeks, which can increase toxicity.28–31 Any grade TRAEs rates for sunitinib ranged from 82.8% to 97.6% with grade ⩾3 TRAEs ranging from 50.9% to 63.9%. In studies demonstrating an OS gain, the addition of a PD-1 inhibitor to a TKI increased grade ⩾3 TRAEs by 4.5–12.8%,28,31 with the smallest increase seen when pembrolizumab was added to axitinib. 31 In contrast, the combination of nivolumab plus ipilimumab decreased grade ⩾3 AEs by 17.3% compared with sunitinib, with significantly longer time to confirmed deterioration for the ICI combination (FKSI-19, p < 0.05). 40 Furthermore, treatment-free survival after the dual ICI combination was over twice that of sunitinib for intermediate/poor-risk (6.9 versus 3.1 months) and three times as long for favorable-risk patients (11.0 versus 3.7 months), suggesting that the ICI combination could allow for more treatment breaks. 50 However, acute immune-related AEs were observed that required careful monitoring and carry both the risk of treatment discontinuation and ongoing management for persistent complications. 30 Overall, however, the safety profile of the dual ICI combination was consistent with previous studies in RCC and other tumor types,51–55 with dose delays, rapid diagnostic workups, appropriate timing, and the use of glucocorticoids (28.7% of patients received ⩾40 mg prednisone daily or equivalent) to manage any grade treatment-related select AEs. 48 Patient-reported outcomes were similar or significantly improved for PD-1 inhibitor plus TKI combinations compared with sunitinib.44,45
Figure 2(a) depicts grade 1/2 and grade ⩾3 TRAEs of PD-1 inhibitor plus TKI combinations that were shown to prolong survival.28,29,43 Toxicity profiles were relatively consistent across combinations, with hypertension, diarrhea, and PPE being the most common grade ⩾3 TRAEs, despite steroid use in 29% and 21% of patients in the CheckMate 214 and CheckMate 9ER trials, respectively.48,56 When the mean toxicity rates for the PD-1 inhibitor plus TKI combinations were plotted against those of the dual ICI combinations (Figure 2(b)), higher rates of hypertension, PPE, diarrhea, dysphonia, hypothyroidism, stomatitis, and decreased appetite were observed while the ICI combinations were associated with higher rates of pruritis and rash.27–31

Select TRAE rates in phase III trials of ICI combinations. (a) TKI combination trials (ICI + TKI). (b) Average ICI + TKI and ICI + ICI TRAE rates.
Do some first-line patients benefit more from ICI combinations than others?
Results from first-line trials show that ICI combinations significantly improved OS in patients with advanced RCC, although some IMDC risk groups benefited more than others. Results from subgroup analyses must be interpreted with caution as outcomes may be influenced by imbalances in baseline characteristics and studies were not designed to compare outcomes in these subgroups. IMDC risk subgroup outcomes were available for five trials (CheckMate 214, JAVELIN Renal 101, KEYNOTE-426, CheckMate 9ER, and CLEAR), which reported outcomes for intermediate/poor-risk patients representing between 66% and 78% of the ITT populations. When assessing intermediate and poor-risk groups, OS favored ICI combinations for all combinations, with relatively tight confidence intervals that did not cross unity (Figure 3(a)).27-29,31,57 In intermediate-risk patients, OS favored ICI combinations but the benefit was less pronounced with CIs crossing unity in CheckMate 9ER. 28 When assessing the favorable-risk patients, this population appeared to representing between 22% and 33% of the included studies.27–29,31,57 Benefit in these patients was less clear with OS favoring ICI combinations in some studies and sunitinib in others, with CIs that were wide and crossed unity. Assessment of OS benefit in favorable-risk patient subgroups requires longer follow-up, although initial PFS benefits appear promising for nivolumab plus cabozantinib (HR = 0.58), 58 avelumab plus axitinib (HR = 0.63) 59 and pembrolizumab plus axitinib (HR = 0.76). 36 ICI combination outcomes vary based on IMDC-risk, with the greatest benefit observed for poor-risk and intermediate/poor-risk patients.
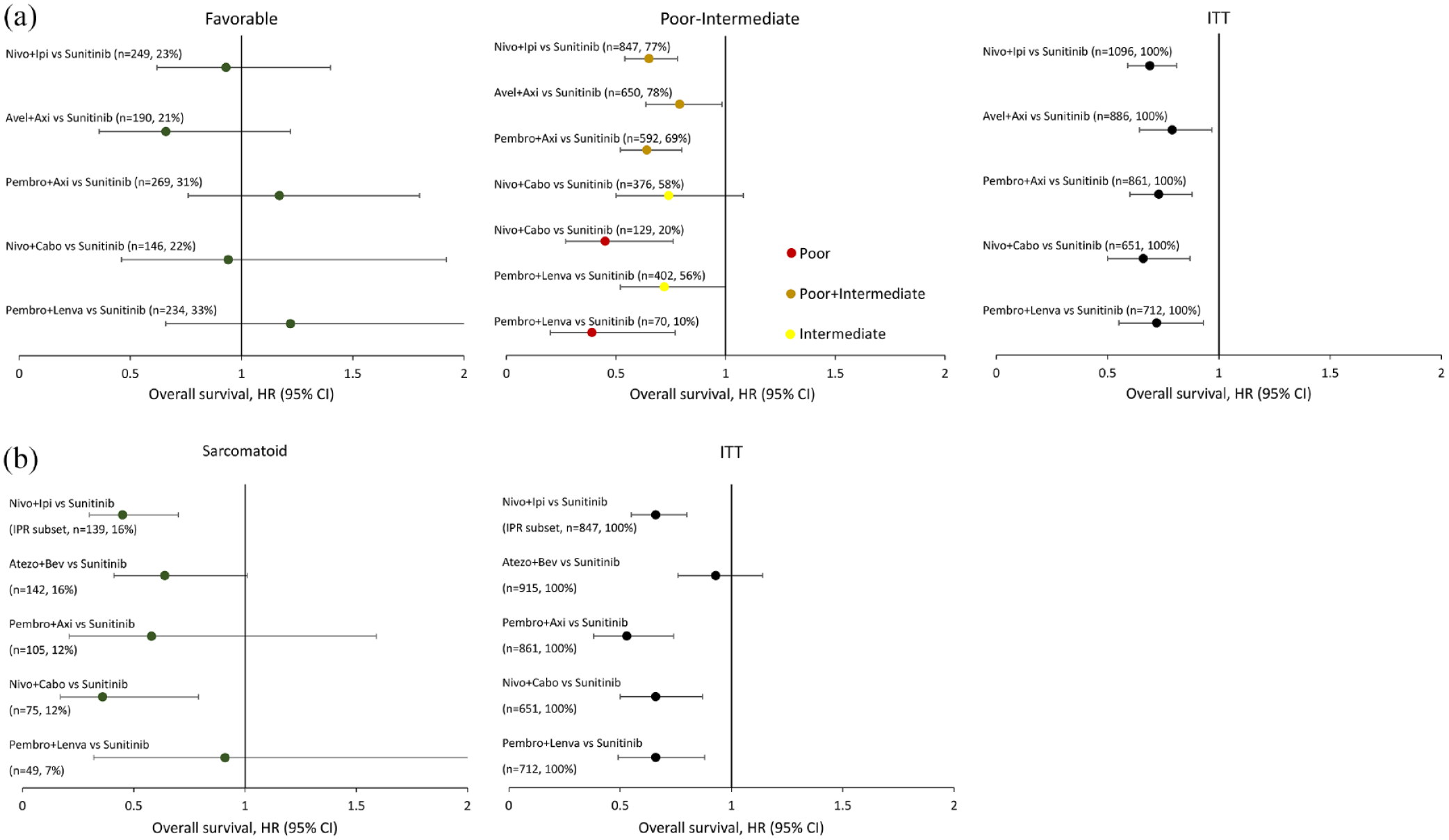
OS in select subgroups. (a) OS in IMDC risk subgroups. (b) OS in sarcomatoid subgroups.
Sarcomatoid differentiation, levels of tumor PD-L1 expression, and nephrectomy status have been assessed as factors to identify patients who may benefit from ICI combination therapy. Five trials evaluated OS in patients with tumors having sarcomatoid differentiation (CheckMate 214, IMmotion151, KEYNOTE-426, CheckMate9 ER and CLEAR), with approximately 7%–16% of patients in this subgroup across trials.32,56,60–64 Survival outcomes generally favored ICI combinations compared with sunitinib,32,56,60–64 with the greatest benefit associated with the dual ICI combination and nivolumab plus cabozantinib (Figure 3(b)).56,63,64 Five of the six ICI combination trials reported outcomes based on PD-L1 expression status, with no association detected between PD-L1 status and survival (Figure S1). Four trials reported outcomes based on nephrectomy status, with none showing a clear association between this variable and survival.27,28,31,57,62
What are the factors in selecting among first-line ICI combination therapies for advanced RCC?
Four ICI combinations have demonstrated an OS benefit compared to sunitinib in patients with first-line advanced RCC and an ECOG PS ⩽2,27,28,33,35–37 although five have shown either PFS or OS benefits and are currently approved by the United States Food and Drug administration and/or the European Medicines Agency (Table S2). Treatment selection should incorporate patient and disease characteristics, IMDC risk status, treatment history prior to the onset of advanced disease, and eligibility for immunotherapy. For patients with an intermediate/poor IMDC risk, nivolumab plus ipilimumab may be a good option due to the strong and durable OS benefit in patients suitable for combination ICI therapy. All three ICI plus TKI options, pembrolizumab plus axitinib, pembrolizumab plus Lenvatinib, and nivolumab plus cabozantinib are also life-prolonging options and offer higher ORRs with the lowest progressive disease rates, although they may be associated with chronic toxicities due to extended TKI use. For patients with a favorable IMDC risk, subgroup analyses suggest that further follow-up is required (Figure 3(a)) and nivolumab plus ipilimumab has not been approved for this subgroup in many jurisdictions.
What is the state of the future for combination strategies in RCC?
The role of ICI combinations is rapidly evolving, with multiple-phase III trials underway for both advanced RCC and for adjuvant treatment (Table 3). In advanced disease, established ICI plus TKI combinations are being assessed in clear-cell component tumors with or without sarcomatoid features (PDIGREE – NCT03793166) while new ICI combinations are also being evaluated such as the TKI combinations TQB2450 plus anlotinib (NCT04523272) and toripalimab plus axitinib (NCT04394975), in addition to the PEGylated interleukin-2 bempegaldesleukin plus nivolumab (NCT03729245). The future may shift again, with triplet strategies being evaluated first-line globally using established ICIs in COSMIC-313 (NCT03937219) and belzutifan, a selective small molecule inhibitor of hypoxia-inducible factor-2α, in MK-6482-012 (NCT04736706). In the adjuvant setting, dual ICI combinations including nivolumab plus ipilimumab (NCT03138512), durvalumab plus tremelimumab (NCT03288532), and pembrolizumab plus belzutifan (NCT05239728) are being assessed as in patients with intermediate/high risk of relapse. The role of ICI combinations is rapidly evolving and ongoing trials will inform optimal use across the disease trajectory.
Ongoing Phase III Clinical Trials of Immune Checkpoint Inhibitor Combination Therapy in Early Stage or First-Line Advanced Renal Cell Carcinoma.
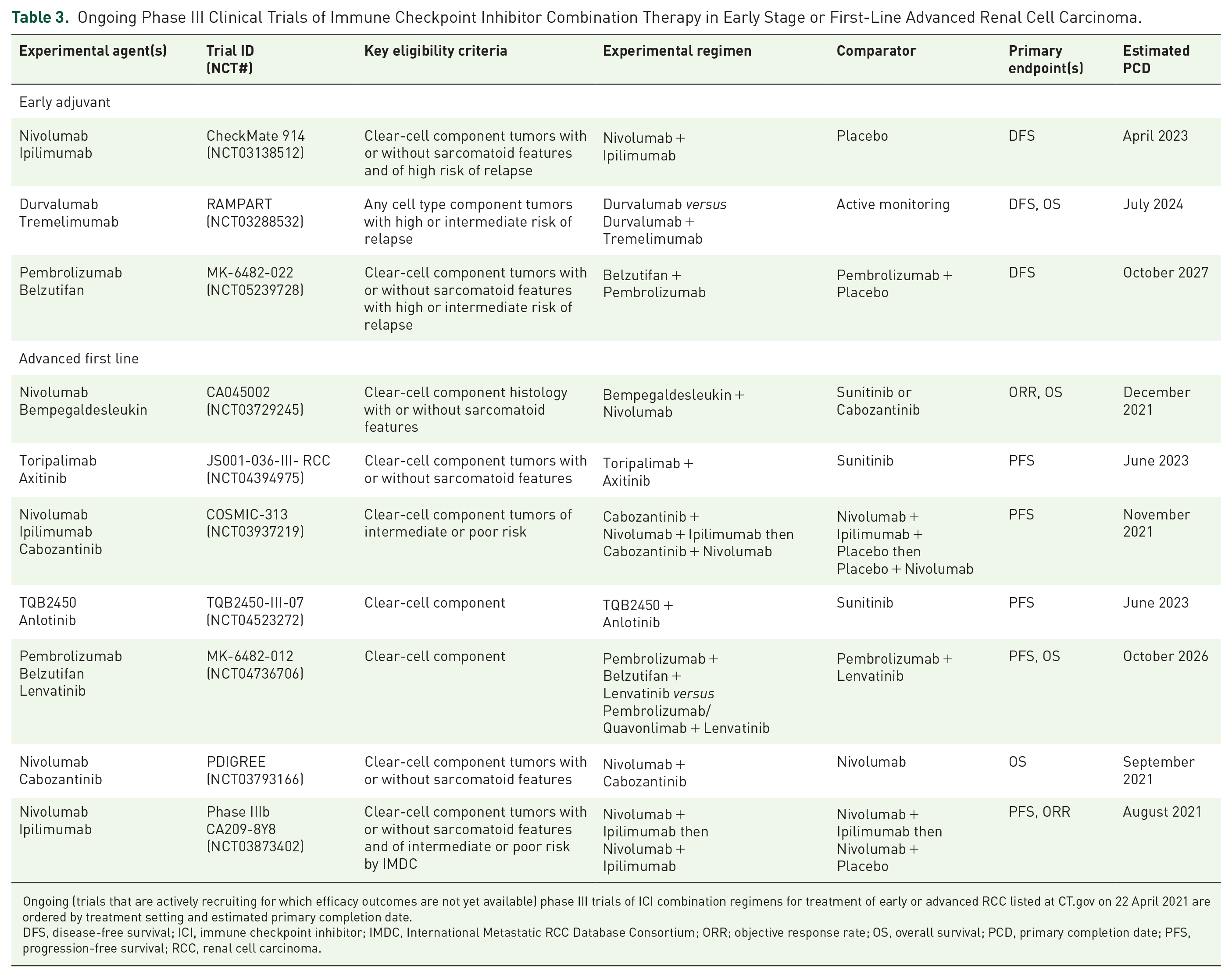
Ongoing (trials that are actively recruiting for which efficacy outcomes are not yet available) phase III trials of ICI combination regimens for treatment of early or advanced RCC listed at CT.gov on 22 April 2021 are ordered by treatment setting and estimated primary completion date.
DFS, disease-free survival; ICI, immune checkpoint inhibitor; IMDC, International Metastatic RCC Database Consortium; ORR; objective response rate; OS, overall survival; PCD, primary completion date; PFS, progression-free survival; RCC, renal cell carcinoma.
Summary
Recent outcomes from first-line ICI combination trials have reported OS benefit compared to sunitinib in advanced RCC, all of which present efficacious treatment options in this setting depending on IMDC risk status. The dual ICI combination nivolumab plus ipilimumab demonstrated a robust and durable OS benefit with a relatively favorable safety profile compared to sunitinib in IMDC intermediate or poor-risk patients. Pembrolizumab plus axitinib or lenvatinib and nivolumab plus cabozantinib demonstrated an OS benefit in patients regardless of IMDC risk. Research into novel therapies and to elucidate the role of ICI combinations in earlier lines of treatment are ongoing and will help inform the optimal role of these combinations in this rapidly evolving treatment landscape.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221108685 – Supplemental material for Evolving landscape of first-line combination therapy in advanced renal cancer: a systematic review
Supplemental material, sj-docx-1-tam-10.1177_17588359221108685 for Evolving landscape of first-line combination therapy in advanced renal cancer: a systematic review by Aly-Khan A. Lalani, Daniel Y. C. Heng, Naveen S. Basappa, Lori Wood, Nayyer Iqbal, Deanna McLeod, Denis Soulières and Christian Kollmannsberger in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359221108685 – Supplemental material for Evolving landscape of first-line combination therapy in advanced renal cancer: a systematic review
Supplemental material, sj-docx-2-tam-10.1177_17588359221108685 for Evolving landscape of first-line combination therapy in advanced renal cancer: a systematic review by Aly-Khan A. Lalani, Daniel Y. C. Heng, Naveen S. Basappa, Lori Wood, Nayyer Iqbal, Deanna McLeod, Denis Soulières and Christian Kollmannsberger in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank Ilidio Martins and Paul Card from Kaleidoscope Strategic Inc. for their research and editorial support as well as Eisai Limited (CA), Ipsen Canada, Merck Canada Inc. and Pfizer Canada Inc. for funding this review.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by unrestricted educational grants from Eisai Limited (CA), Ipsen Canada, Merck Canada Inc. and Pfizer Canada Inc. No discussion or viewing of review content was permitted with sponsors at any stage of review development.
Competing Interests
A-K.A.L. has received honoraria from AbbVie, Astellas, Bayer, BMS, Eisai, Ipsen, Janssen, Merck, Novartis, Pfizer, Roche, and TerSera, and received research funding from BMS, BioCanRx, Novartis, Roche, Ipsen, and EMD Serrono.
D.Y.C.H. has served in consultancy or advisory role for Pfizer, Novartis, BMS, Merck, Ipsen, Exilexis, and Eisai; and has received research funding from Pfizer, Novartis, BMS, Merck, and Ipsen.
N.S.B. has served in consultancy or advisory role for Merck, BMS, Eisai, Ipsen, EMD Serono, Roche, and Pfizer; and has received honoraria from Merck, BMS, Eisai, Ipsen, EMD Serono, Roche, and Pfizer.
L.W. has received research funding from Merck, Roche, BMS, Astrazeneca, and Pfizer.
N.I. has nothing to disclose.
D.M. has nothing to disclose.
D.S. has served in consultancy or advisory role for Merck, Novartis, Pfizer, Adlai-Nortye, and BMS; has received honoraria from Merck, BMS, Pfizer, Adlai-Nortye; and has received research funding from Pfizer, BMS, Merck, GSK, and Adlai-Nortye
C.K. has served in consultancy or advisory role for Pfizer, Merck, Eisai, Ipsen, EMD, Astellas, Janssen, Bayer, and BMS, and has received honoraria from BMS, Ipsen, Astellas, Merck, and Pfizer.
Disclaimer
This review was prepared according to ICMJE standards with editorial assistance from Kaleidoscope Strategic Inc.
Availability of data and materials
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
