Abstract
Myxofibrosarcoma is one of the most common types of soft tissue sarcomas occurring in the lower limbs of male older adults. Mainstay treatment is surgical intervention with chemoradiotherapy with a combination of doxorubicin and ifosfamide with brachytherapy if advanced or metastatic. There are no guidelines on the combination of immunotherapy with radiation therapy for myxofibrosarcoma. We report the first documented case of unresectable multifocal myxofibrosarcoma that was treated with pembrolizumab and radiation therapy for first-line therapy with a significant response. We also highlight genomic markers and immune radiation priming therapy, forecasting favorable response in treatment with pembrolizumab.
Introduction
Sarcomas represent a heterogeneous group of rare mesenchymal malignancies accounting for 1% of all adult cancers and exhibit extraordinary diversity of histological subtypes, clinical behavior, and genomic landscape. 1 Sarcomas occur in 2–4 people per 100,000 individuals. There are several subtypes that can arise from cartilage, muscle, blood vessels, nerves, and fat that are typically divided into soft tissue sarcomas (STS) and bone sarcomas. 2 STS predominate over bone sarcomas in a 4:1 incidence ratio with a male predominance. 3 STS anatomically occur on the lower limbs of adults with the thigh being the most common site. 3 The most common types of STS are leiomyosarcoma, undifferentiated pleomorphic sarcoma, and liposarcomas. 4 Myxofibrosarcoma (MFS) is a type of STS with an annual incidence of 5% of all STS. 5
MFS is one of the most common types of STS, typically presenting as a slow-growing, painless mass that occurs in the lower limbs of older adults in their sixth to eighth decades of life. 1 MFS generally shows an infiltrative growth pattern, extending along vascular and extra- or intramuscular fascial planes.6,7 Nearly 20–25% of MFS progress to metastatic disease. 6 Several small studies have estimated this rare STS to have a 5-year overall survival between 61% and 77%. 8 Compared to other STS, MFS is known to have a high local recurrence rate. 8 MFS tumors are histologically classified based on the Fédération Nationale des Centres de Lutte Contre le Cancer (FNCLCC) grading system for STS, which assigns grades 1 through 3, depending on tumor differentiation, mitotic count, and tumor necrosis. 9
Treatment of MFS should ideally occur at a sarcoma referral center as management outside has been associated with significantly worse clinical outcomes. 8 Treatment of STS is optimal surgery, which depends on tumor location, size, involvement of adjacent anatomical structures, and patient preference. 5
For those with metastatic or inoperable STS, high doses of external beam radiation therapy known as stereotactic body radiation therapy (SBRT) often present as an option especially in those with low metastatic disease burden. Multiple studies appreciate local control rates of greater than 90%. 10 In addition, radiation promotes antitumor immunity through tumor antigen release and increased tumor-infiltrating lymphocytes. Although rare, published case reports exist demonstrating an abscopal effect after irradiation alone of sarcomas. 11
Chemotherapy treatments for MFS remain largely extrapolated from STS studies. First-line treatment is typically intensified doxorubicin with ifosfamide for six cycles. 12 This study demonstrated an increased progression-free survival and overall response rate when compared to doxorubicin monotherapy (which was the prior first-line treatment). There is no consensus on second-line treatment; however, it generally includes gemcitabine-based treatment with response rates in this setting drop to about 10%. 13 Second- and third-line therapies also include pazopanib, a tyrosine kinase inhibitor of Vascular Endothelial Growth Factor Recepter (VEGFR), 800 mg, which demonstrated an improvement in progression-free survival as well as trabectedin and eribulin.14–17
Immunotherapy drugs called checkpoint inhibitors (CPIs) work by blocking checkpoint proteins from binding to partner proteins allowing T cells to recognize them and kill cancer cells. MFS tumors are known to display a heavy tumor microenvironment (TME), making these tumors likely responsive to immunotherapy. 18 CPIs include pembrolizumab and atezolizumab which block the Programmed death-ligand 1(PD-L1) partner protein and are active agents in pretreated, refractory MFS.19–22 Only one published report to date has demonstrated the efficacy of pembrolizumab in recurrent MFS without prior systemic therapy. 23 We present a case of a newly diagnosed, multifocal, unresectable MFS of the scalp that responded exceptionally well to pembrolizumab and radiation therapy as first-line therapy. The reporting of this study conforms to the CARE statement guidelines. 24
Case presentation
A 76-year-old man with a heavily pretreated metastatic castrate-resistant prostate cancer who recently completed a long-standing response on oral metronic cyclophosphamide treatment was discovered to have a hard lump on his left ear and the top of his scalp after completing cyclophosphamide treatment that was described as pruritic and firm. He had previously received docetaxel, abiraterone, cabazitaxel, and oral metronomic cyclophosphamide, and was undergoing treatment with lutetium Lu-177 for his prostate cancer.
His lesions continued to grow over a week and were described as a rapidly growing painful exophytic tumor at the top of his scalp (i.e. Tumor 1) [Figure 1(a) and (b)] and another rapidly growing painful flat erythematous infiltrative tumor behind his left ear (i.e. Tumor 2) [Figure 1(c)]. He was referred to dermatology who biopsied the tumors 4 weeks after discovery of the masses.
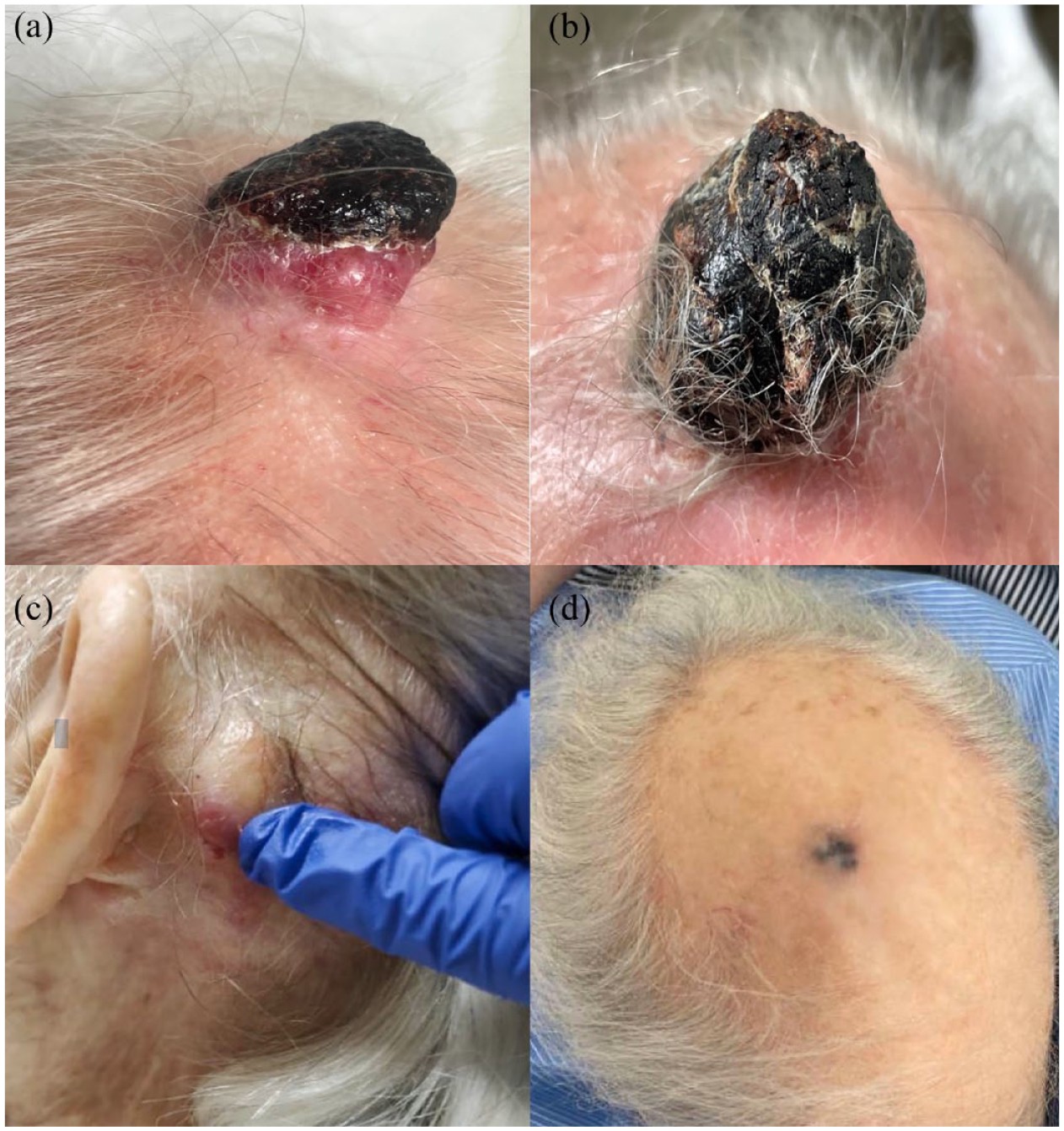
Gross appearance of patient’s tumors. (a) Tumor 1 is an exophytic tumor placed at the top of the scalp appearing anterior to the head. (b) Tumor 1 is an exophytic tumor placed at the top of the head appearing posterior to the head. (c) Tumor 2 is described as a flat erythematous infiltrative tumor behind the left ear. (d) Tumor 1 after treatment with radiation therapy and immunotherapy.
The biopsy of tumor 1 demonstrated a malignant spindled neoplasm with myxoid stroma and curvilinear blood vessels, consistent with MFS. Based on the morphology of the spindled cells, a myxoid variant of atypical fibroxanthoma or pleomorphic dermal sarcoma was considered less likely and no evidence of prostate cancer was noted [Figure 2(a)]. Meanwhile, a biopsy of tumor 2 demonstrated a malignant spindle cell neoplasm with higher cellularity, less myxoid stroma, and a similar immune profile and was favored to be the same neoplastic process as tumor 1 [Figure 2(b)]. The tumor was diffusely positive for CD10 and patchy positive for smooth muscle actin (SMA) and was negative for AE1/AE3, NKX3.1 CK5/6, S100, SOX10, and CD34 by immunohistochemical stains [Figure 2(c)–(g)]. Immunohistochemistry of PD-L1 demonstrated a tumor proportion and combined positive score of 98% [Figure 2(h)]. Genetic testing demonstrated mutations in TERT, MLH1, CDKN2A, RNF43, NRAS, NOTCH2, NF1, TP53, KMT2C, KMT2D, and NCOR1. Mutations were not present in KRAS, P10, and ALK in either of the tumors.

CT head showing the patient’s scalp tumors. (a) and (b) axial and sagittal plane respective CT images of exophytic enhancing 3.5 cm cutaneous and subcutaneous soft tissue mass overlying biparietal scalp at a vertex without evidence of periosteal reaction or osseous invasion. (c) and (d) Coronal and axial images of a 5.1 cm mass overlying the left posterior reticular and retrosigmoid scalp extending anteriorly with involvement in the superficial parotid gland. (e) and (f) Coronal and axial images of mass on the left posterior reticular and retrosigmoid scalp significantly decreased.
Staging was performed with a computed tomography (CT) head that is notable for a 3.5 cm exophytic enhancing cutaneous and subcutaneous soft tissue mass overlying the biparietal scalp at the vertex as well as a rim enchanting 5.1 cm mass overlying the left posterior reticular and rectosigmoid scalp [Figure 3(a)–(d)]. Restaging body CT scans did not demonstrate any distant metastasis of MFS but metastasis of the previously known prostate cancer-related lesions.

Pathologic images of patient’s tumors. (a) Hematoxylin and eosin (H&E) stain of tumor 1 showing hypocellular and pleomorphic spindle cells with abundant myxoid matrix, infiltrating dermis. (b) H&E stain of tumor 2 demonstrating sheets of pleomorphic spindle cells with frequent mitoses and necrosis, involving dermis. A focal myxoid matrix is present. (c)–(f) IHC studies on tumor 2 were diffusely positive for CD10, while being negative for Sox-10, S100, SMA, and Pankeratin. (h) The immunohistochemistry stain of PD-L1 is demonstrated.
A Tempus xT assay for tumor genomic profiling was obtained for both tumors. For tumor 1, the following characteristics were noted: PD-L1 tumor proportion score (TPS) was 98%, tumor mutational burden (TMB) was 59.5 m/MB, microsatellite instability (MSI) status was high, and DNA mismatch repair (dMMR) protein expression was abnormal due to the absence of PMS2. For tumor 2, PD-L1 TPS was 98%, TMB was 45.8 m/MB, and dMMR was abnormal due to the partial absence of PMS2. In addition, multiple mutations were observed in both tumors.
The patient was evaluated by a surgical team, who felt that resection of the scalp tumors would be too extensive and require significant reconstruction. His case was reviewed by a multidisciplinary tumor board, which recommended a combination of palliative radiation therapy for local control and systemic therapy. He received 25 Gy in five fractions of radiation to both scalp tumors utilizing SBRT with 6× energy. Bolus was placed around the apex lesion to improve dosimetric coverage. The dose was prescribed such that at least 85% of both lesions received the target dose which he completed before treatment with pembrolizumab (Figure 4). Given his genomic profiling results, a decision was made to proceed with first-line therapy with pembrolizumab 200 mg IV given every 21 days. During this time, he received no treatment with lutetium for his prostate cancer. After four cycles of pembrolizumab, tumor 1 fell off and the patient brought it to a clinic in a plastic bag, while tumor 2 dramatically decreased in size [Figure 1(d)]. Pathology of tumor 1 at this point demonstrated fragments of impetiginized purulent scale crust, negative for tumor. CT images show the resolution of tumor 1 with a dramatic decrease in the size of tumor 2 [Figure 2(e) and (f)]. The pain associated with the scalp tumors resolved. At the time of this report, the patient continued on pembrolizumab, which he is tolerating without any appreciable toxicity.

Dosimetry plan for radiation of tumor 1 and tumor 2.
Discussion
MFS tumors exhibit highly complex karyotypes and a variety of genomic alterations, with a subset containing genomic features of ‘immunologically hot’ tumors that may be targetable by CPIs. 25 One study evaluated the expression of immune checkpoint biomarkers in sarcomas and found MFS to be one of the most common tumors staining positive for CD4, CD8, CD56, and FOXP3 as well as the most numerous diverse population of tumor-infiltrating lymphocytes and heavily immune-infiltrated TME. 18 In another study, MFS tumors showed high expression levels of immune TME markers, including B7-H3, TGF-β1, and TIM-3, along with a high immune infiltration score. 26 Furthermore, the increased presence of dendritic cells and natural killer cells in the TME of MFS tumors was correlated with improved disease-specific survival, proposing a role for antigen presentation in the immunological response to this type of sarcoma. 25 Meanwhile, the Cancer Genome Atlas Research Network reported an association between STS prognosis and TME signature in 206 sarcoma patients across six histological subtypes, including MFS. 1
Some clinical data support the use of CPIs for the treatment of MFS tumors, mostly case reports in patients with pretreated, refractory, or relapsed disease. For instance, one report described a patient with MFS and negative PD-L1 expression, who previously failed multiple lines of systemic therapy, and achieved a complete radiographic response of over 3 years to combined checkpoint blockade with ipilimumab and nivolumab. 27 Another report of a patient with MFS and high TMB, who progressed on multiple lines of therapy, demonstrated a complete resolution of pulmonary lesions and stable lymphadenopathy on 38 months of atezolizumab. 20 It has been hypothesized that recurrent STS tumors treated with alkylating agents may undergo hypermutation as an emerging resistance mechanism, making subsequent use of CPIs effective against these tumors. 1
However, advanced STS tumors have demonstrated low response rates to single-agent immunotherapy alone. SARC028 examined the effect of pembrolizumab in advanced soft tissue and bone sarcomas but only appreciated an 18% response rate with a median progression free survival (PFS) of 18 months in those with STS. 28 Trials examining single-agent immunotherapy for advanced leiomyosarcoma or synovial sarcoma did not appreciate a benefit regardless of PD-L1 expression.29,30 ENVASARC is an ongoing study evaluating envafolimab, a PD-L1 inhibitor, and envafolimab with ipilumumab in patients with undifferentiated pleomorphic sarcoma or MFS, with no results yet available. 31
Because response rates with immune therapy remain low in STS, one likely explanation for the dramatic response to pembrolizumab is the immune priming radiation therapy given before pembrolizumab. Radiation therapy has been found to increase the surface expression of PD-L1. 32 In addition, the effects of radiation on the TME, including dendritic cell activation, naïve CD8+ T-cell priming, and tumor CD8+ T-cell recruitment, have been well described in multiple solid tumors.33–35 A retrospective study examining 10 STS metastases that had progressed on pembrolizumab but then treated with SBRT to 21–54 Gy in three fractions and anti-PD-1 therapy concurrently. 34 Utilizing RECIST versus 1.1 criteria, they appreciated complete responses in 50% of lesions treated. They also noted regression of untreated lesions incidentally receiving 20 Gy. Despite prior pembrolizumab resistance, the radiation promoted significant regression while receiving the same immunotherapy. The tumors in this case report received 25 Gy with responses consistent with the retrospective data presented. Another case report examining carbon ion therapy followed by pembrolizumab for a large 11 cm alveolar STS appreciated 80% regression in size 10 months after finishing therapy. 36 Similarly, a case report outlining two cases of treatment-refractory STS noted a near-complete response after hypofractionated radiation therapy and nivolumab. 37 These studies support the explanation of radiation’s effect in our investigation as a synergistic treatment with checkpoint blockade.
Interestingly, the patient’s MFS developed once the patient discontinued therapy with oral metronomic cyclophosphamide for his prostate cancer, which is known to have an immune-stimulating mechanism of action via inhibition of T-regulatory cells. 38 This has not been translated over with any literature demonstrating an association with the development of STS. In addition, there is no previously published data indicating an association between MFS and prostate cancer or an association between MFS and individuals previously treated with docetaxel, cabazitaxel, abiraterone, or lutetium.
The patient in our report demonstrated an excellent, rapid response to a combination of radiation therapy and pembrolizumab for a previously untreated, de novo, multifocal, and unresectable MFS. His tumors contained high PD-L1 expression, high TMB, abnormal dMMR, and was MSI-high, which are all associated with increased responses to CPIs in solid tumors.39–42 This is the first documented case report of the successful use of a combination of immunotherapy and radiation therapy in a previously untreated MFS with favorable genomic biomarkers.
Conclusion
Although no standard-of-care therapy exists, unresectable or metastatic MFS are traditionally treated with single-agent or combination chemotherapy, although some data supporting the use of CPIs in relapsed, pre-treated tumors are available. Our case describes an effective option with radiation therapy and pembrolizumab in a patient with unresectable MFS and a favorable genomic profile on next-generation sequencing.
