Abstract
Myxofibrosarcoma (MFS) is a common entity of adult soft tissue sarcomas (STS) characterized by a predilection of the extremities and a high local recurrence rate. Originally classified as a myxoid variant of malignant fibrous histiocytoma, this musculoskeletal tumor has been recognized since 2002 as a distinct histotype showing a spectrum of malignant fibroblastic lesions with myxoid stroma, pleomorphism and curvilinear vessels. Currently, the molecular pathogenesis of MFS is still poorly understood and its genomic profile exhibits a complex karyotype with a number of aberrations including amplifications, deletions and loss of function. The diagnosis is challenging due to the unavailability of specific immunohistochemical markers and is based on the analysis of cytomorphologic features. The mainstay of treatment for localized disease is represented by surgical resection, with (neo)-adjuvant radio- and chemotherapy. In the metastatic setting, chemotherapy represents the backbone of treatments, however its role is still controversial and the outcome is very poor. Recent advent of genomic profiling, targeted therapies and larger enrollment of patients in translational and clinical studies, have improved the understanding of biological behavior and clinical outcome of such a disease. This review will provide an overview of current diagnostic pitfalls and clinical management of MFS. Finally, a look at future directions will be discussed.
Keywords
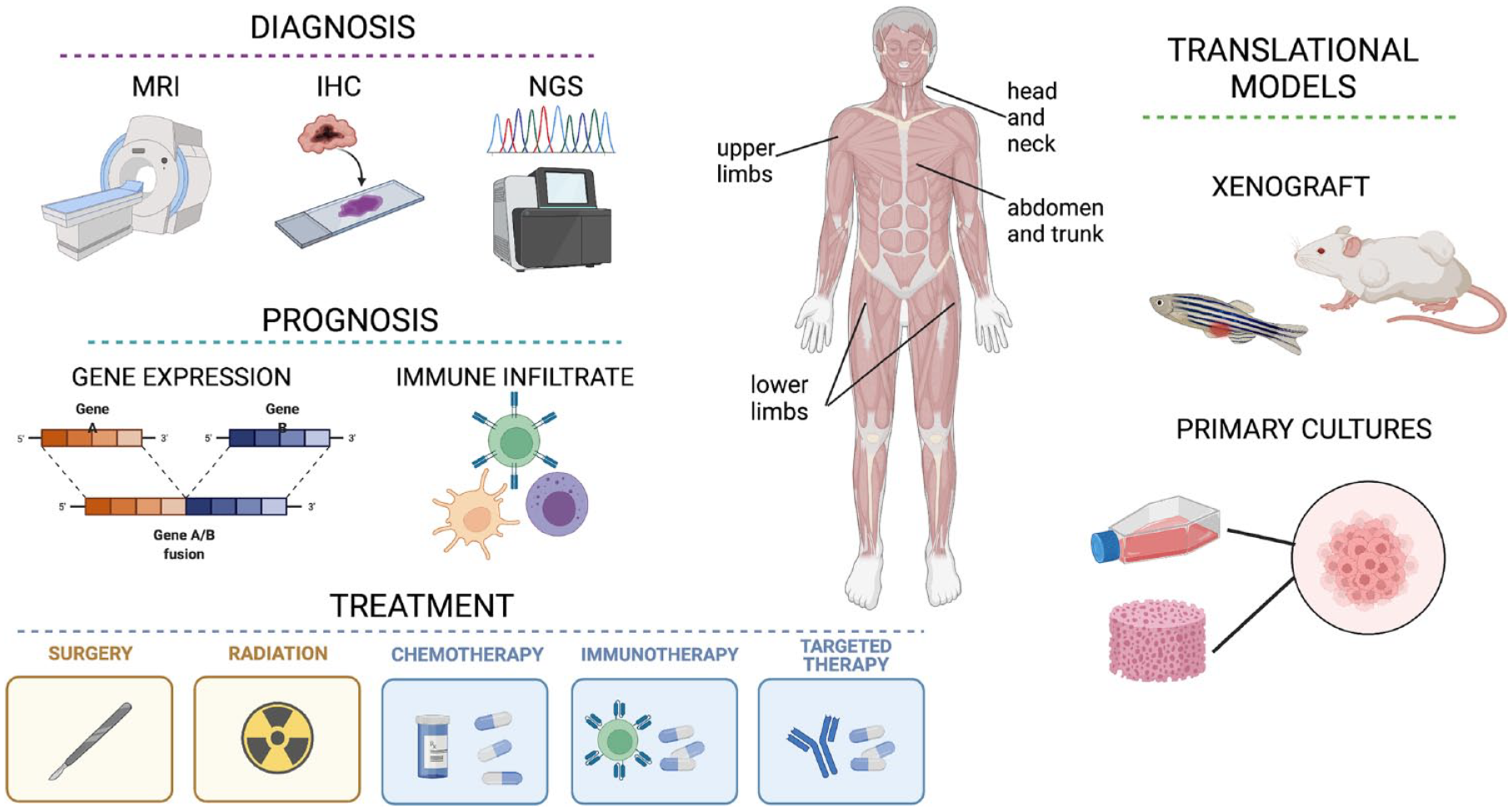
Schematic representation of the main available tools for myxofibrosarcoma (MFS) diagnosis, prognosis, treatment and translational research. MFS most commonly occurs in adults and presents as an intramuscular mass, typically located in limbs. Diagnosis mainly relies on histopathological analysis (IHC) and imaging (MRI), however in recent years also NGS proved helpful in detecting distinct molecular alterations. Prognosis has been shown to correlate with specific gene expression signatures, as well as with immune microenvironment markers and infiltrating cells. The cornerstone of treatment for localized disease is surgical resection, possibly in combination with radiotherapy, while in metastatic MFS the standard systemic treatment is based on anthracycline chemotherapy, targeted therapy such as TKIs and CDKIs or immunotherapy. In order to gain better insight on the still poorly understood pathogenesis and behaviour of MFS, several in vitro cell cultures and in vivo xenograft models have been established, representing promising tools for the improvement of MFS management.
Introduction
Soft tissue sarcomas (STS) represent a heterogeneous group of rare mesenchymal malignancies accounting for 1% of all adult cancers. They exhibit an extraordinary diversity in terms of histological subtypes—with more than 100 histotypes—genomic landscape with simple and complex karyotypes and clinical behavior ranging from indolent to very aggressive disease. Among them, myxofibrosarcoma (MFS) is one of the most common STS usually affecting the extremities of elderly patients. 1 Previously considered as a myxoid variant of malignant fibrous histiocytoma (MFH), it was reclassified in the World Health Organization (WHO) classification system of 2002 as a distinct entity characterized by myxoid stroma, pleomorphism, curvilinear vessels and a high propensity for local recurrence. 2 These mesenchymal lesions exhibit a predilection for the lower extremities of elderly patients but they could arise also in the upper extremities, whereas truck, head and neck, hands and feet are rarely reported. Moreover, the occurrence in the retroperitoneum and in the abdominal cavity is extremely uncommon. 1 These neoplasms affect patients in the sixth to eighth decades of life with a slight predominance in male, although exceptionally rare patients aged below 30 years have been described. 1 Macroscopic appearance is characterized by multiple variably gelatinous or firmer nodules in the superficially located lesions, while infiltrative margins are often present in deep-seated diseases. In high-grade MFS areas of tumor necrosis are described. From a molecular point of view, they exhibit highly complex karyotypes with a number of different genomic aberrations. At present no specific molecular pathology markers are available for the standard differential diagnosis, which is based on the analysis of cytomorphologic findings. Clinical manifestation of MFS is characterized by slowly enlarging and painless mass with high propensity for local recurrence. Patients affected by MFS should be managed in specialized multidisciplinary teams. Treatment options include surgery, radiotherapy (RT), and chemotherapy (CT). In the localized disease the gold standard is represented by radical surgery with the goal of achieving a complete resection of the lesion which means obtaining free margins from tumor infiltration. RT and CT could be considered as (neo)-adjuvant treatments and their impact is still debated. In metastatic disease the cornerstone of therapy is represented by CT, although with poor outcome. Recently, genomic and transcriptomic profiling have shed light on the molecular alterations of this disease through the identification of new targets with promising results. This review aims to give an update on the current diagnosis and clinical management of MFS. Finally, an overview of the research advances in translational research will be summarized.
Histopathological and clinical features
Currently, MFS is described by WHO as a spectrum of malignant fibroblastic lesions with myxoid stroma, pleomorphism and curvilinear vessels (Figure 1). It typically presents as a slow-growing and painless mass occurring in adults–mostly aged 50–70 years–with a slight preference for male sex. Myxofibrosarcoma is characterized by the highest rate of recurrence among all STS, ranging from 20–60% after 5 years, depending on the case series.3–11 Lower limbs are reported as one of the most common sites of occurrence, however MFS can arise also in trunk and head and neck region.12,13Rarely it has been reported in the retroperitoneum and abdominal cavity. 4 While superficial MFS often presents as multiple palpable nodules, the deep-seated lesions more frequently form a single mass. Myxofibrosarcoma generally shows a peripheral infiltrative growth pattern extending along vascular and fascial planes extra- or intramuscularly.8,10 From a clinical point of view, low-grade diseases exhibit a high rate of multiple local recurrences and low rate of metastatic potential. Instead, high-grade lesions show higher metastatic potential. Noteworthy, from 15–38% of MFS local recurrences evolve to higher histological grade disease with increased metastatic potential.14,15 The overall incidence of distant metastases is estimated to be between 20% and 25%.16–18 Currently, histological MFS grading relies on the general FNCLCC grading system for STS, which assigns grades 1 to 3 depending on tumor differentiation, mitotic count and tumor necrosis. However, while it is accurate to predict the probability of distant metastases (DM) and overall survival (OS), it does not predict local recurrence (LR). Moreover, the FNCLCC grading system has been generally evaluated in heterogeneous cohorts of patients including extremities and trunk/neck as well as primary/recurrent MFS. However, a recent study involving a homogeneous cohort of 229 localized MFS of the extremities showed similar LR frequency in grades 2 and 3, while grade 1 MFS showed the lowest risk of developing an LR. 11
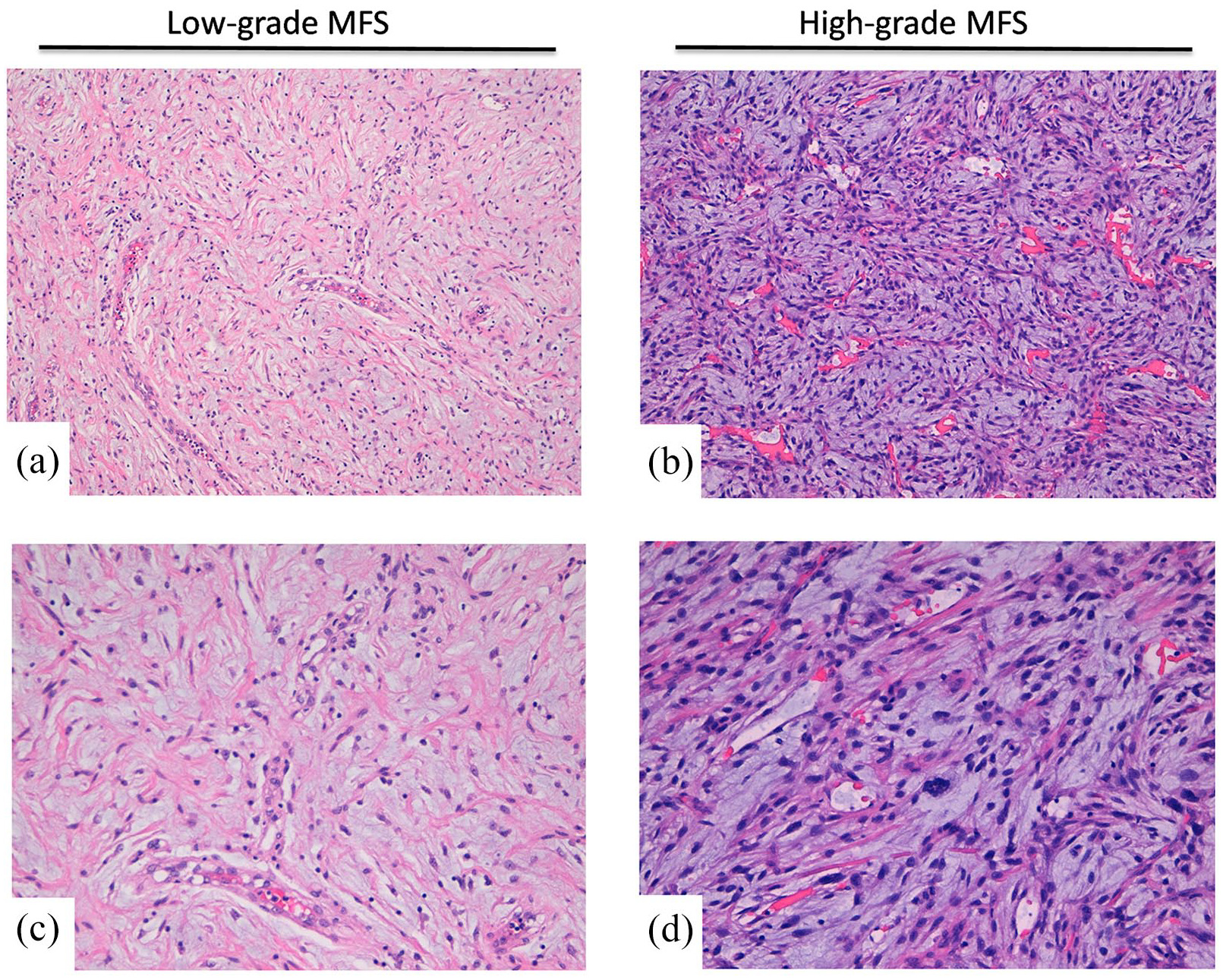
Representative histopathological images of low-grade (a,c) and high-grade (b,d) myxofibrosarcoma. H&E of typical MFS cases showing myxoid stroma, pleomorphic cells and curvilinear vessels. Upper panels show 10× magnification, lower panels represent 20× magnification.
Diagnosis and prognosis
Molecular alterations
Some STSs show recurrent genomic aberrations such as chromosomal translocations, leading to gene fusions (EWSR1-FLI1 in Ewing sarcoma, FUS-DDT3 in myxoid liposarcoma or SS18-SSX in synovial sarcoma) or point mutations (KIT and PDGFRA in gastrointestinal stromal tumors). 19 In particular, adult STSs show low somatic mutation burdens while harboring many more copy-number alterations than most other cancer types. 20 However, at present there are no specific immunohistochemical markers for the standard differential diagnosis of MFS since they are generally characterized by highly complex karyotypes and show many of the aberrations observed in other STS.9,20 Some amplifications have been reported in chromosomes 5p (rapamycin-insensitive binding partner of mTOR, CDH9, LIFR) and 1p/1q (PI4KB, ETV3, MCL1), as well as deletions of tumor-suppressor genes (CDKN2A/B, TP53) and loss-of-function mutations in NF1and PTEN. 21
A few years ago, Ogura et al. performed whole-exome sequencing (WES), RNA sequencing (RNA-seq) and methylation analysis on a MFS discovery set and then sequenced 140 targeted genes in a cohort of 99 MFSs and 17 MFSs’ data from TCGA. They reported frequent alterations of p53 signaling and cell cycle checkpoint genes in about half of cases, together with a signature of 14 driver genes (TP53, CDKN2B, CCND1, CDKN2A, KRAS, WNT11, NTRK1, MDM2, CDK6, GNAS, FOXA1, NKX2-1, SYK, JAK1) including potentially actionable therapeutic targets. Moreover, alterations to any of the cell cycle regulators were associated with poorer overall survival; however, they were not associated with local recurrence-free survival. The presence of TP53 alteration and KRAS amplification was also significantly associated with poorer overall survival. 22 In addition, expression of integrin-α10 has been correlated with worse outcome in a group of 64 primary high-grade MFSs. 23
In 2015, Emori et al. performed immunohistochemical staining on specimens from 37 MFS patients showing that CD109 expression—a TGF-β co-receptor whose frequency was approximately 10%—was associated with poor prognosis. Indeed, CD109 overexpression was significantly related to decreased overall survival, being 5-year OS rates 77% and 0% for CD109-negative and CD109-positive patients, respectively. 24 In 2017, De Vita et al. 25 performed CD109 analysis on three high-grade MFS showing its role as a promising marker for the identification of more aggressive high-grade MFS and a potential therapeutic target. In this regard, subsequent analysis evaluating CD109 expression in other STS such as undifferentiated pleomorphic sarcoma (UPS) and rhabdomyosarcoma confirmed its specificity for MFS.26,27
More recently, Sugiura et al. evaluated the prognostic value of CD34 expression status in 192 patients with MFSs and UPS. They reported that CD34 positivity is more frequent in MFS, and that CD34 status is a useful predictor of OS, being loss of expression of CD34 an adverse prognostic factor, likely because patients developed significantly more distant metastases. 28 The authors also suggest two possible mechanisms, with one being related to CD34-positive stromal fibroblastic/fibrocytic cells (CD34 + SFCs), which synthesize and remodel the extracellular matrix thereby contributing to fibrosis and tumor stroma formation. Moreover, CD34 + SFCs have been proposed to be progenitors of fibroblastic/myofibroblastic and lipomatous tumors, given that benign or low-grade malignant tumors of these types constantly express CD34. The other hypothesis is that CD34 suppresses tumor development, in which case it could be used for the development of novel therapeutic agents.
Interestingly, an online specialized tool named Online consensus Survival analysis for MyxoFibroSarcoma (OSmfs) has been developed to evaluate the prognostic value of genes in MFS, utilizing gene expression profiling and clinical follow-up data of MFS cases from three independent cohorts, with a total of 128 Myxofibrosarcoma samples from The Cancer Genome Atlas (TCGA) and Gene Expression Omnibus (GEO) databases. The authors also took advantage of cox analysis and transcriptome data from OSmfs to select seven genes considered to be best prognostic biomarkers through overlapping and ROC analysis, concluding that Integrin Subunit α 10 (ITGA10), CD109, Cyclin Dependent Kinase 6 (CDK6), Cyclin Dependent Kinase Inhibitor 2A (CDKN2A), MET, Cyclin D1 (CCND1) and EZR predict a worst outcome for MFS patients. 29 However, diagnosis is still currently based on the analysis of cytomorphologic appearance comprising the presence of a myxoid background, nuclear atypia, pseudolipoblasts and curvilinear blood vessels. These histological characteristics also represent key features for distinguishing MFS from other sarcoma subtypes, such as liposarcoma, in cases of dubious diagnosis.30,31 In recent years, a gene expression signature named CINSARC has revealed a good prognostic capacity for metastatic outcome in sarcomas. 32 This transcriptomic signature was originally identified with microarrays from frozen tissues and includes 67 genes involved in mitosis control and chromosome integrity pathways, representing a valuable prognostic tool for predicting sarcomas’ aggressiveness with complex genetics such as MFS. 32 More recently, CINSARC has been validated also with RNA-seq from FFPE specimens, analyzing three different cohorts including 46 myxofibrosarcoma patients, and is currently being assessed in two prospective phase 3 clinical trials for stratification of therapy.33,34 In addition, CINSARC expression is associated with an increase of ploidy, intra-tumor heterogeneity, copy-number alteration, altered expression of 37 miRNAs and a decrease in DNA methylation. 35 Interestingly, a recent study integrating clinical information and RNA-seq data from 536 sarcoma samples reported subtype-specific metastasis-related genetic signatures for different types of sarcomas. In particular, it has been shown that metastasis-related signatures of UPS and MFS are mainly related to forebrain development and fatty acid degradation. 36 Furthermore, a recent publication of the Italian ACC working group evaluated the presence of fusion transcript through NGS approaches in a case study of 150 sarcoma samples including six Myxoid Fibrosarcoma. The results demonstrated that among the analyzed MFS cases, only one showed FUS-CREB3 L2 fusion and no others were detected in the remaining investigated MFS, underlying the variability in gene expression of this STS entity. 37 In particular, the FUS gene–encoding an RNA-binding protein–has been identified as a 5’-partner in other tumor-associated fusion genes such as the FUS-CHOP in myxoid/round cell liposarcoma, the FUS-ATF1 in angiomatoid fibrous histiocytoma and the FUS-ERG in Ewing sarcoma. 38 CREB3 L2 is another transcription factor gene which is a member of the old astrocyte specifically induced substance (OASIS) DNA binding and basic leucine zipper dimerization (bZIP) family. Interestingly, this fusion has been proposed to display both transactivating and oncogenic properties. 39 Besides gene expression alterations, methylation status could also represent an attractive target for analysis of sarcoma metastasis. In particular, Vargas et al. recently reported methylation changes in metastatic and recurrent disease in a cohort of 32 STS, with MFS (n = 4) showing the largest global methylation variance. In this study, low level of CNV (copy gain/loss) in MFS was reported as well as methylation alterations in several genes (MEST, C14MC, FAM136A, SNRPG, CCND2, TBX15, PLEC1, CDH15), data which are in accordance with Ogura’s study.22,40 Importance of methylation studies has been mentioned also in the only study available to date using WES and SNP arrays to investigate 20 sarcomas including five MFS, which showed a gradual increase of both nucleotide- and chromosome-level mutations. In particular, they reported that MFS has complex copy number changes but few significant single nucleotide variants. In facts only four (EGF, IDH2, PTPRB, and TP53) out of 87 mutations were shared by at least two lesions from the same patient included in COSMIC’s Cancer Gene Census, suggesting that epigenetic changes could be relevant for tumor progression in absence of genomic drivers. 41
Magnetic resonance imaging (MRI)
Preoperative MRI can be useful in order to identify high risk of local recurrence (LR), particularly in the presence of features such as high myxoid matrix content, high grade of contrast enhancement and presence of an infiltrative pattern (‘tail sign’) (Figure 2). 42 In particular, the tail sign presents as a curvilinear multidirectional projection spreading along the fascial plane, showing a similar signal intensity to that of the principal mass and the same enhancement after gadolinium-based contrast material injection. 43 (ADD SPINNATO PMID: 33724067). The tail sign represents a hallmark of MFS (especially in superficial lesions) and UPS, and it has a crucial role in surgical planning as well as in prognostic evaluation. Indeed, tail sign presence has been correlated with a higher risk of local recurrence after surgical excision and onset of distant metastases (ADD YOO PMID 24889995.). 44 Furthermore, the detection of this pattern is of great relevance for the complete and effective excision of the lesion, dictating the need for wider margins of resection to include the whole tail. 7 In addition, MRI can help in stratifying the risk for disease-specific survival, with large size of the lesion and deep location being associated with worse survival. 42 Moreover, as mentioned above, MRI features can support the establishment of surgical planning. 7 Indeed, due to these characteristics, a specific MRI grading system has recently been proposed to stratify the risk of LR and provide prognostic information. 45 This system, which relies on the evaluation of lesion sizes (both volume and maximum diameter), the ‘tail sign’ and high myxoid matrix content with MRI water-like appearance, could significantly stratify the risk of LR in MFS of the extremities.17,46

MRI patterns of myxofibrosarcoma. (a) Sagittal T1-weighted MRI shows a highly intense mass signal typical of myxoid matrix in subcutaneous tissues in the right thigh. (b) Axial T1-weighted MRI shows tail-like margin (white arrow) at caudal extent of lesion.
It is important to note that water-like appearance can be encountered also in different types of myxoid tumor (e.g. myxoma, low-grade myxoid round-cell liposarcoma), therefore this could represent a confounding factor for radiologists in the diagnosis on unenhanced MRI. Indeed, these lesions can often resemble benign processes such as cysts, especially if located in the limbs near to the articulations, 47 or can be misdiagnosed as hematomas in case of plate-like morphology. 48 Ultrasound and contrast-enhanced MRI should be considered to address these differential diagnoses.
Treatment options
Surgery
Since local recurrence frequently occurs but distant metastasis is rare, the current standard of care for localized disease is radical resection confirmed by histological clear surgical margins combined with neoadjuvant or adjuvant radiotherapy. The management of extremity MFS favors organ preservation, with amputation only considered in extreme cases. However, despite treatments, local recurrence rates are between 50% and 60%. 21 In this regard, margin assessment is crucial to predict risk of local recurrence, even though there is still a lack of consensus on the criteria for defining adequate margins. 49 Indeed, despite taking into consideration both the margin width (quantity) and the type of anatomic barrier (quality) represent well-established criteria, 50 there are several classification schemes for reporting surgical resection margin status for STS. Generally speaking, microscopic examination of six-eight perpendicular sections from all margins below 2 cm in width is recommended. 51
The most sensitive and accurate imaging tool to assess margins in STS is represented by MRI, with the most important feature on baseline MRI being an infiltrative growth pattern. This is particularly relevant for MFS, since the high infiltration of the tail sign often lead to microscopically positive yet wide margins, representing a major risk factor for local recurrence. More recently, radiomics proved effective in predicting margin invasion in several malignancies and it holds great potential also for STS. 49
Currently, surgical procedures still rely on conventional imaging modalities such as CT and MRI to determine the location and extent of tumor burden. However, in recent years a new technique based on tumor-targeted near-infrared (NIR) fluorescence imaging, which takes advantage of specific tumor biomarkers to allow tumor identification during surgery, has been developed. 52 A study involving 34 MFS samples identified a strong overexpression of Tumor endothelial marker 1 (TEM1) in 89% of all cases, suggesting its potential use as a biomarker for targeted approaches. 53
Radiotherapy
The beneficial effect of RT on local tumor control for MFS is still unclear. Indeed, several retrospective studies reported that RT in combination with surgery is associated with a lower risk of LR in retroperitoneal STSs, while other trials did not observe significant benefit for adjuvant RT.54–57 In addition, some results suggest that MFS could be considered as a radioresistant sarcoma. 58 This could be explained by the fact that, typically, patients who undergo RT are more likely to have high-grade, deep-seated tumors, which are above 5 cm in size and excised with intralesional or narrow margins. Given that these are known risk factors for local recurrence, one may expect a higher rate of local recurrence in this group compared with the non-RT group, therefore the potential benefit could be masked. 10 Indeed, adjuvant RT is generally recommended to improve local control of the disease, particularly in high-grade STS either deep-seated or following marginal and intralesional margins. 59 Common RT schemes for STS with wide or marginal surgical margins include 50 Gy in 25 fractions or 36 Gy hyperfractionated with 1.8 Gy twice daily when combined with chemotherapy. Intralesional surgical margins are treated with higher doses of 64-66 Gy, 2 Gy/fraction, or 45 Gy with hyperfractionation.
One seminal work published in 1996 reported the results of a clinical trial with the goal of evaluating the impact of adjuvant interstitial radiation on local and systemic recurrence rates in STS. 60 This study included, among others, 19 MFH treated with postoperative RT and 20 MFH treated only with surgical resection and concluded that adjuvant RT was effective in reducing LR in resected high-grade STS but did not reduce distant metastases nor improved disease-specific survival. Later in 1998 a prospective study investigated the effect of postoperative RT on LR, overall survival and quality of life after limb-sparing resection of extremity STS, including 23 high-grade MFH and six low-grade MFH. 61 Similarly, the results showed that adjuvant RT is beneficial in preventing LR in high-risk patients. A randomized trial including 51 MFH cases aimed at comparing preoperative versus postoperative RT in STS of the limbs reported that, despite the fact that OS was slightly improved in patients who had preoperative RT, this group showed a greater risk of wound complications. 62 In 2008 Zagar et al. 63 investigated the role of RT in addition to total resection of retroperitoneal sarcoma, including three MFH, in prolonging survival. Collected data showed that neoadjuvant or adjuvant RT, compared with surgery alone, improved long-term locoregional control, distant disease free survival and overall survival. More recently, a clinical randomized trial started in 2012 with the aim to evaluate the impact of preoperative RT plus surgery versus surgery alone in 266 retroperitoneal STS patients (STRASS phase 3 study). The results reported that median abdominal recurrence-free survival was 4.5 years in the RT plus surgery group and 5.0 years in the surgery only group, concluding that preoperative RT should not be considered as standard of care treatment for retroperitoneal sarcomas. 64 Another clinical trial started in 2017 is currently investigating the role of hypofractionated RT with sequential chemotherapy in 46 patients with marginally resectable STS of extremities or trunk wall (UN-RESARC phase 2 study). The study is estimated to be completed in 2022, however no results are available yet. Taken together, these data suggest that the timing and modality of RT still remain controversial due to the lack of standardized approaches. Further clinical trials are needed to define patient selection, both in adjuvant and neoadjuvant settings.
Chemotherapy
Even though administration of chemotherapy represents the standard clinical care in metastatic MFS, it is mainly used as palliative and outcome remains very poor. Indeed, no large randomized clinical trials clarifying the beneficial effect of CT in this kind of tumor have been conducted. However, data available from very few cohort studies conducted over a decade ago suggested that overall survival is not significantly increased after chemotherapy nor it is effective against distant metastases.3,4,16 Typically, the first-line chemotherapeutic treatment for recurrent and metastatic MFS includes anthracycline (doxorubicin) and ifosfamide, either alone or in combination,65,66 however its response rates in advanced STS are about 20–30%. Besides this, dacarbazine, an alkylating agent, is also frequently used in combination with doxorubicin. On second-line treatment no wide consensus has been reached yet, however it generally includes gemcitabine—due to MFS similarity with UPS that is known for being sensitive to that nucleoside analog—either alone or in combination with docetaxel. Generally, with second-line drugs response rates drop to 10%.67,68 Nevertheless, a recent study investigated the activity of gemcitabine-containing regimens for the treatment of metastatic MFS refractory to doxorubicin. A partial or complete radiological response was observed in four out of seven patients, with median PFS and OS of 8.5 months and 11.4 months respectively, showing that gemcitabine-based chemotherapy was associated with encouraging response rates. 69
Targeted therapy
Targeted therapies may represent a powerful tool to improve the current therapeutic outcome of standard CT. However, since typically sarcomas display alterations in many signaling pathways, targeting a range of pathways would result in a more effective therapy. Current targeted therapies for sarcomas aim at inhibiting cell cycle progression, sustained proliferative signaling, DNA repair, epigenetics, tumor microenvironment and angiogenesis. 70 In particular, cell cycle inhibitors (CDKIs) targeting CDK4/6 and MDM2 are widely used to target cell cycle progression, while tyrosine kinase inhibitors (TKIs), IGFR inhibitors and mTOR inhibitors aim to target growth receptors and pro-survival signaling molecules. Moreover, some drugs specifically target histone deacetylases (HDAC) to inhibit epigenetic regulators, while others inhibit Poly (ADP-ribose) polymerase (PARP) involved in DNA repair mechanism. Anti-angiogenetic therapies typically target VEGFR and PDGFR, while molecules acting on tumor microenvironment are mainly anti-PD1 or directed against macrophages.
Several clinical trials in last decades have investigated the efficacy of administering target therapy in combination with radiation or standard CT in different STS including MFS. In 2012 a phase 2 study was started to investigate the effect of alisertib—an oral adenosine triphosphate competitive inhibitor of Aurora A, a kinase which is commonly overexpressed in sarcoma—in a group of 72 patients with inoperable advanced or metastatic sarcomas. Results showed that alisertib was well tolerated, with neutropenia (42%) and leukopenia (22%) as the most common adverse events, followed by anemia (14%), platelet count decreased (14%) and oral mucositis (12%). Occasional responses, yet prolonged stable disease, were observed. Although failing to meet the primary response rate (RR) end point, twelve-week PFS progression-free survival (PFS) was promising, varying from 36–73% depending on the histotype. 71 In 2012, the results of a phase 3 study investigating the effect of pazopanib on PFS in 369 patients with metastatic nonadipocytic STS after failure of standard chemotherapy (PALETTE) were published. Median PFS was 4.6 months for pazopanib compared with 1.6 months for placebo, while OS was 12.5 months with pazopanib versus 10.7 months with placebo, with most common adverse events being fatigue, diarrhea, weight loss and hypertension. However, despite improving PFS, pazopanib treatment failed to significantly improve overall survival respect to placebo. 72 Later, in 2014 a phase 2/3 randomized study started with the aim of evaluating the effect of pazopanib plus preoperative radiation in the treatment of 81 patients with newly diagnosed nonrhabdomyosarcoma STS that could eventually be removed by surgery (PAZNTIS clinical trial). Primary completion data recently showed that adding pazopanib to neoadjuvant chemoradiotherapy improved the rate of pathological near-complete response, suggesting that this is a highly active and feasible combination in children and adults with advanced soft tissue sarcoma. The comparison of survival outcomes however requires longer follow-up. 73 A phase 1b study investigating side effects and best dose of ribociclib—a selective inh ibitor of CDK4/6—administered with doxorubicin in 16 patients with inoperable metastatic or advanced STS was initiated in 2017, with estimated completion in June 2022. No data is available yet. Another phase 1b trial started in 2020 with the aim of investigating the maximum tolerated dose and overall response rate of DCC-3014—a CSF1R inhibitor—administered concurrently with avelumab—an anti-PD-L1 antibody—in 48 patients with advanced high-grade sarcoma. Recruitment is still ongoing.
Alternative strategies
High-intensity focused ultrasound (HIFU) is a noninvasive heating technique that allows ablation of the central tumor zone inducing thermal coagulation necrosis, thus resulting in precise and localized ablation. Moreover, it also allows prolonged hyperthermia of the tumor periphery. When performed under magnetic resonance guidance (MR-HIFU), in vivo mapping of the induced temperature change is used as a feedback for accurate heating of the tissue over an extended time period. 74 A recent study reported a case of MFS of the right upper arm, which relapsed 7 months after surgical procedure but was completely ablated after 5 cycles of low power cumulative high-intensity focused ultrasound (HIFU) treatments, without complications. The patient has been disease-free with a high quality of life for more than 30 months, indicating that HIFU ablation might be a safe, minimally invasive therapy for recurrent myxofibrosarcoma. 75 An additional application for HIFU is in combination with MR for image-guided drug delivery. Indeed, Hijnen et al. 74 investigated MR-HIFU thermal therapy with complementary intravascular doxorubicin delivery in rhabdomyosarcoma rat tumor model, showing that the combination of hyperthermia-induced drug delivery followed by ablation resulted in a homogeneous drug distribution and the highest therapeutic effect due to direct induction of thermal necrosis in the tumor core and efficient drug delivery to the tumor rim. Another minimally invasive alternative for locoregional treatment of primary or secondary tumors is represented by percutaneous image-guided cryoablation. This option could be particularly relevant for patients having recurrences or unresectable lesions and can be used both as palliative treatment to reduce disease-related pain or as a curative treatment to achieve effective local disease control. 76 Back in 1999, a phase I trial investigating feasibility and safety of cryosurgical ablation of soft tissue sarcomas reported that complications associated with this treatment were minor and transient and that the procedure was well tolerated by patients. 77 More recently, cryoablation has been exploited for the treatment of muscolo-skeletal tumors and metastases, reporting low morbidity (thanks to the preservation of collagens structures), excellent tolerance and effective local control. 78 One major advantage of this technique is represented by the possibility of intraprocedural monitoring of the ablation area, as an ice ball, to ensure accuracy. Furthermore, it can treat irregularly shaped lesions by using multiple synergistic probes, and also it can be applied repeatedly. 79 However, indication of this treatment in sarcomas is still limited due to the scarcity of available data. Moreover, standardized selection criteria for indication of cryoablation is still lacking, despite some features such as adequate distance to skin and to neurovascular structures should be considered to avoid necrosis. 80
Immunotherapy
From an immunogenomic point of view, sarcomas carrying complex karyotypes such as MFS are known to display a heavily immune infiltrated tumor microenvironment (TME), making these tumors likely to be responsive to immunotherapy. 81 Indeed, sarcomas with a greater number of mutations are genetically heterogeneous and may provide multiple potential neoantigens for the host’s immune system. This results in an increased amount of tumor-infiltrating lymphocytes (TILs), as well as aberrant expression of immune checkpoint markers, thus representing promising candidates for ICI therapies. However, overall response rates are lower than observed in other tumors, highlighting the generally low immunogenicity of STS, resulting in an immune cell-poor TME, with a consecutive lack of targetable molecules. 82 Several immunotherapeutic approaches have been studied in sarcomas, such as immunomodulatory agents (IL-2, IFNα), cancer vaccines, adoptive cell therapy (which relies on isolation and reinfusion of tumor-specific T cells from the patient after ex vivo expansion) and immune checkpoint inhibitors (aimed at restoring the normal immune response against tumor which is often blocked by inhibitory ligands released by the tumor itself). 83 Recently, The Cancer Genome Atlas Research Network reported the association between prognosis and immune microenvironment signature in 206 STS patients across six histological subtypes, including MFS. Myxofibrosarcoma showed high expression levels of immune microenvironment markers such as B7-H3, TGF-β1 and TIM-3, together with a high immune infiltration score. Moreover, the presence of dendritic cells (DCs) and the immune infiltration of natural killer (NK) cells were correlated with improved disease-specific survival (DSS), suggesting the role of antigen presentation in the immunological response to this sarcoma. 20 In a recently published clinico-pathological study, Smolle et al. showed how T-regulatory cells can predict clinical outcome in STS of extremities and trunk, including 78 MFS patients. In detail, they found higher levels of PD-L1, PD-1 and any TIL phenotype in MFS compared with other STS studies, as well as significantly higher immune checkpoint marker levels. Moreover, they reported that the presence of regulatory T cells (Tregs) was associated with increased LR risk, irrespective of margins. 82
A few years ago, the results of the first multicenter, two-cohort, single arm phase 2 trial were published in which the safety and activity of pembrolizumab—an anti-PD-1 antibody—were assessed in advanced STS (SARC028 trial). Despite MFS not being included in the enrollment given to funding limitations, the UPS subtype was included and 40% of these patients showed an objective response, suggesting that these immunotherapy agents should be specifically explored in MFS as well. 84 Indeed, in April 2020, a phase 2 study started with the purpose of assessing if giving pembrolizumab in combination with the chemotherapy drugs melphalan and dactinomycin, delivered directly to the affected arm or leg using a technique called isolated limb infusion (ILI), is a safe treatment that can delay disease progression in patients with locally advanced or metastatic extremity sarcoma. Progression-free survival data will be available upon study completion scheduled for April 2023 (NCT04332874). Another study investigating both local and systemic immunotherapeutic effect of an anti-PD-1 antibody—nivolumab—administered before surgery in combination with BO-112—a nanoplexed form of polyinosinic: polycytidylic acid (poly I:C)—started in October 2020 by recruiting patients with resectable STS. The first data on safety of this combination as a treatment in patients undergoing preoperative radiotherapy before surgical resection is expected to be available at the beginning of 2024 (NCT04420975). In December 2020, a phase 2 pivotal randomized study also started aiming to investigate the safety and effectiveness of envafolimab—an anti-PD-L1 antibody—plus or minus ipilimumab—an anti-CTLA-4 antibody—in 160 patients with inoperable or metastatic UPS or MFS who have progressed on one or two lines of chemotherapy (ENVASARC clinical trial). Data about objective response rate (primary outcome) are still not available since study completion is scheduled for July 2022, however preliminary data from the first 20 enrolled patients showed that envafolimab has been well tolerated as a single agent and when combined with ipilimumab [TRACON Pharmaceuticals, Inc. June 1, 2021. https://bit.ly/3uLHM7U].
A recent study reported excellent long-term response—complete response for over 3 years and without disease on nivolumab maintenance—to ipilimumab and nivolumab treatment in a MFS patient with no expression of PD-L1 who had previously failed multiple lines of systemic treatment. This result suggests that PD-1 blockade can provide long-term disease control for patients with advanced STS in real-world clinical practice, irrespective of PD-L1 status. 85 Similarly, a patient with refractory high-grade MFS with pulmonary metastasis was treated with camrelizumab—a PD-1 inhibitor—due to 40–50% PD-L1 positivity in his pulmonary tissues. After 6 months of immunotherapy, the size of pulmonary lesions showed marked shrinkage, and after an 18 months follow-up the patient remained in good condition with no progression of disease, indicating that PD-1 inhibition is a promising treatment option for effective tumor control. 86 Moreover, another anti-PD-L1 antibody—atezolizumab—has recently shown the ability to provide durable response in a patient with metastatic MFS still progressing after sixth-line therapy with temozolomide, an oral imidazo-tetrazine alkylating agent. There is emerging evidence that recurrent STS treated with alkylating agents often gain hypermutation as a means of developing resistance, and that checkpoint inhibitors are subsequently effective in these tumors. Indeed, at the time of recurrence after treatment with temozolomide, the tumor showed high tumor mutational burden (TMB), with 889 mutations identified per megabase (Mb). Currently, the patient demonstrates stable disease after 22 cycles of atezolizumab, suggesting that checkpoint inhibition may represent an effective therapy in STS patients with high TMB as a consequence of alternate systemic therapy resistance. In addition, these data highlight the role of TMB as promising marker of response to PD-1/PD-L1 inhibitors, regardless of cancer type or PD-1/PD-L1 expression. 87
Translational research achievements: In vivo and in vitro models
Given that intra- and inter-tumor cytogenetic heterogeneity and clonal evolution are common events in MFS, the availability of appropriate in vitro cell systems is of particular relevance. However, only few patient-derived MFS cell lines have been reported to date. In a recent work, Lohberger et al. isolated a novel MFS cell line with two subclones named MUG-Myx2a and MUG-Myx2b. The parental tumor tissue and both MUG-Myx2 cell lines showed the same STR profile, however Myx2a clone showed higher proliferation activity, faster migration and enhanced tumorigenicity. Moreover, this clone also showed an additional PTEN mutation, as well as unique CNVs that distinguish the two cell lines. 88 In our lab, three primary cultures derived from high-grade MFS have been established and their molecular and pharmacological profiles were characterized. An overexpression of CD109 was observed in all three MFS-derived cell cultures, in line with previously published data.24,25 In addition, the establishment of MFS patient-derived cultures have been confirmed culturing the cells in 3D collagen-based scaffolds, which represents a powerful tool to assess the cytomorphological features with H&E staining, since no specific immunohistochemical markers for the standard differential diagnosis of MFS are currently available. Later, another MFS patient-derived primary cell line (IM-MFS-1) was established and further genetically characterized through Comparative Genomic Hybridization (CGH) array, showing a complex molecular karyotype with a high number of cytogenetic alterations particularly in chromosome 7 and 13 in the portions containing BRAF and RB1 genes. 89 Recently, Tsuchiya et al. isolated a MFS-derived cell culture (NCC-MFS3-C1) showing copy number alterations, spheroid formation, aggressive invasion as well as sensitivity to bortezomib and romidepsin, similarly to what has been reported for NCC-MFS2-C1 and NCC-MFS1-C1 cell lines previously isolated.90–92
Among in vivo models, the subcutaneous implant of patients-derived cell lines in immunocompromised mice (xenografts) represents a powerful tool to study the different forms of MFS that occur in patients (superficial, deep and with lung metastasis).93,94 This model has been used to obtain proof of concepts on the efficacy of drugs in MFS as well as to understand key biological issues in MFS tumorigenesis.23,88,90,95–100 As far as we know, a single report of a mouse model of deep-seated MFS has been reported. Krause et al. 101 implanted 5 × 105 OH931 MFS cells in a muscle of the thigh of athymic mice obtaining a tumor with comparable morphology to the primary lesion, with pleomorphic mononuclear and multinucleated cells with a complex karyotype immersed in a myxoid matrix. Also, there is a single model reported for metastasis in which, after RAC and mTOR inhibitors treatment, the authors detected a reduction in the luminescence in the lung consistent with a reduction of the size of the metastases. 23 However, a major limitation of cell line-derived mouse xenografts is that they do not maintain the complexity of tumor microenvironment of the original lesion, which can only be obtained with heterotransplanted tumors.
Also patient-derived xenograft (PDX) mice models showed to maintain the histologic and molecular features of the donor tissue and proved to be a valuable tool in preclinical compound testing. In recent years, Cornillie et al. 102 established and characterized a panel of PDX models representing different STS subtypes, including seven MFS. In a following work, they also tested some of these PDX models for drug sensitivity and reported that combining doxorubicin and anti-PDGFRA did not reduce tumor burden, though a mild inhibition of proliferation was observed in MFS model UZLX-STS59. 103 Another option is represented by patient-derived orthotopic xenograft (PDOX) mouse model, which is advantageous over subcutaneous-transplant models which are growing ectopically under the skin. Kiyuna et al. recently established a PDOX of MFS through subcutaneous transplantation in nude mice and subsequent orthotopic implantation. They took advantage of this PDOX to test effective drug combinations and reported the efficacy of a combination of irinotecan with temozolomide or cisplatin in inhibiting tumor growth. 104
Future perspectives
Despite the fact that many efforts have been put into sarcoma research in recent years, the advances made in unraveling the pathogenesis and development of MFS have not led to a substantial improvement of the clinical outcome. This failure highly depends on the greatly heterogeneous karyotype and clone variability between different patients and different areas within the same tumor. Therefore, molecular analyses aimed at detecting specific expression signatures may represent a powerful tool to identify druggable targets. In this context, 3D cell-based cultures and in vivo xenografts are valuable models to gain better insight in the pathology features and to identify effective drug combinations. In addition, encouraging results are coming from immunotherapy-based clinical trials. In conclusion, therapeutic strategies which target multiple pathways and combine different treatment approaches are the most promising tool for MFS management.
Footnotes
Acknowledgements
The authors gratefully acknowledge Sarah Frances for editing the paper.
Ethics approval
This work does not require ethics approval by an Ethic committee and written informed consent and signed by the patients due to its design (Review).
Consent for publication
Patients signed informed consent regarding publishing their data and photographs.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Italian Ministry of Health (GR-2016-47040236, RF-2018-80041236).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
