Abstract
Small-cell lung cancer (SCLC) is a biologically aggressive subtype of lung cancer, a lethal disease characterized by rapid tumor growth, early relapse, a strong tendency for early widespread metastasis, and high genomic instability, making it a formidable foe in modern oncology practice. While the management of non-SCLC has been revolutionized in the era of immunotherapy, progress in SCLC has been more muted. Recent randomized phase III clinical trials have combined programmed death ligand-1 inhibitors to a chemotherapy backbone and demonstrated improved survival; however, the absolute benefit observed is short months. There is an undeniable urgent need for better responses, better agents, novel therapeutic approaches, and more rational, biomarker-driven clinical trials in SCLC. In this review, we discuss the rationale and current understanding of the biology of SCLC in the modern era of immunotherapy, discuss recent advances in front-line immunotherapeutic approaches that have changed clinical practice globally, provide an overview of some of the challenges and limitations that have staggered immune checkpoint blockade in SCLC, and explore some of the novel immunotherapeutic approaches currently being investigated.
Introduction
Lung cancer remains the leading cause of cancer-related death worldwide, 1 and small-cell lung cancer (SCLC), accounting typically for 15% of cases, is a notably aggressive subtype of this disease. While the incidence of SCLC has reduced in recent years, predominantly due to changes in smoking habits, most patients with SCLC (more than 60%) present with extensive stage disease (ED-SCLC) and the 5-year overall survival (OS) remains dismal, at 7%. 2 SCLC remains a biologically aggressive disease characterized by rapid tumor growth, a strong tendency for early widespread metastasis, and high genomic instability, 3 making it a formidable clinical challenge for the modern oncologist. The management of non-small-cell lung cancer (NSCLC) has been revolutionized with the advent of molecular profiling and the development of oncogene-driven therapies,4–7 in addition to immunotherapy 8 ; however, progress in the treatment of SCLC over the decades has been more staggered.
Until recently, the standard of care for extensive-stage SCLC (ES-SCLC) was platinum-doublet chemotherapy, using cisplatin or carboplatin with etoposide or irinotecan, 9 which induces high response rates (RR) (up to 70%), with concurrent radiation therapy utilized also for limited stage disease (LS-SCLC).10,11 However, while most patients initially respond well to standard chemotherapy, relapse is inevitable, often rapid, and invariably coupled with the development of chemoresistance. 12
Second-line management typically consisted of single-agent chemotherapy (such as topotecan or irinotecan) for many years,13,14 but with limited efficacy (including RRs of 25% for platinum-sensitive disease, less than 10% for platinum-resistant disease). 14 More recently, lurbinectedin, a selective oncogenic transcription inhibitor that covalently binds DNA, generating double-strand breaks, and disrupts DNA–protein interactions and RNA transcription, has induced clinically meaningful overall response rates (ORR) and duration of response (DOR) with acceptable safety profile in second-line SCLC (ORR of 45.0%, median DOR of 6.2 months, and median OS of 11.9 months). 15 In 2020, the US Food and Drug Administration (FDA) granted accelerated approval to lurbinectedin for the treatment of adult patients with metastatic SCLC with disease progression on or after platinum-based chemotherapy based on these data.
Thus, for almost 40 years, the treatment paradigm for SCLC was halted, with several clinical trials, across decades, failing to demonstrate survival benefits over standard-of-care platinum doublet chemotherapy for ES-SCLC 16 ; however, change is on the horizon. The advent of the immunotherapy era in modern cancer care in the early 2000s and notably the remarkable responses induced by immune checkpoint inhibitors in melanoma and NSCLC17,18 have prompted new hope in SCLC.
However, while the paradigm has shifted in SCLC following two large landmark phase III trials using programmed cell death 1 (PD-1) inhibitors in combination with chemotherapy (IMpower 133 and CASPIAN19,20; Table 1), progress in SCLC has been more muted than other disease subtypes. Immune checkpoint inhibitors used in combination with chemotherapy agents in SCLC, in clinical practice, have induced prolonged durable clinical benefit, but only in a subset of patients and the absolute benefit has been modest.19,20 There is an urgent need for novel therapeutic approaches, better agents, and more rational, biomarker-driven clinical trials in SCLC. In this review, we discuss the rationale and current understanding of the biology of SCLC, we discuss recent advances in immunotherapeutic approaches that have changed clinical practice, including an overview of the challenges and limitations that have staggered development, and explore some of the novel agents targeting immuno-oncology (IO) in SCLC.
Pivotal frontline chemotherapy in combination with anti-PD-(L)1 therapy in small-cell lung cancer.
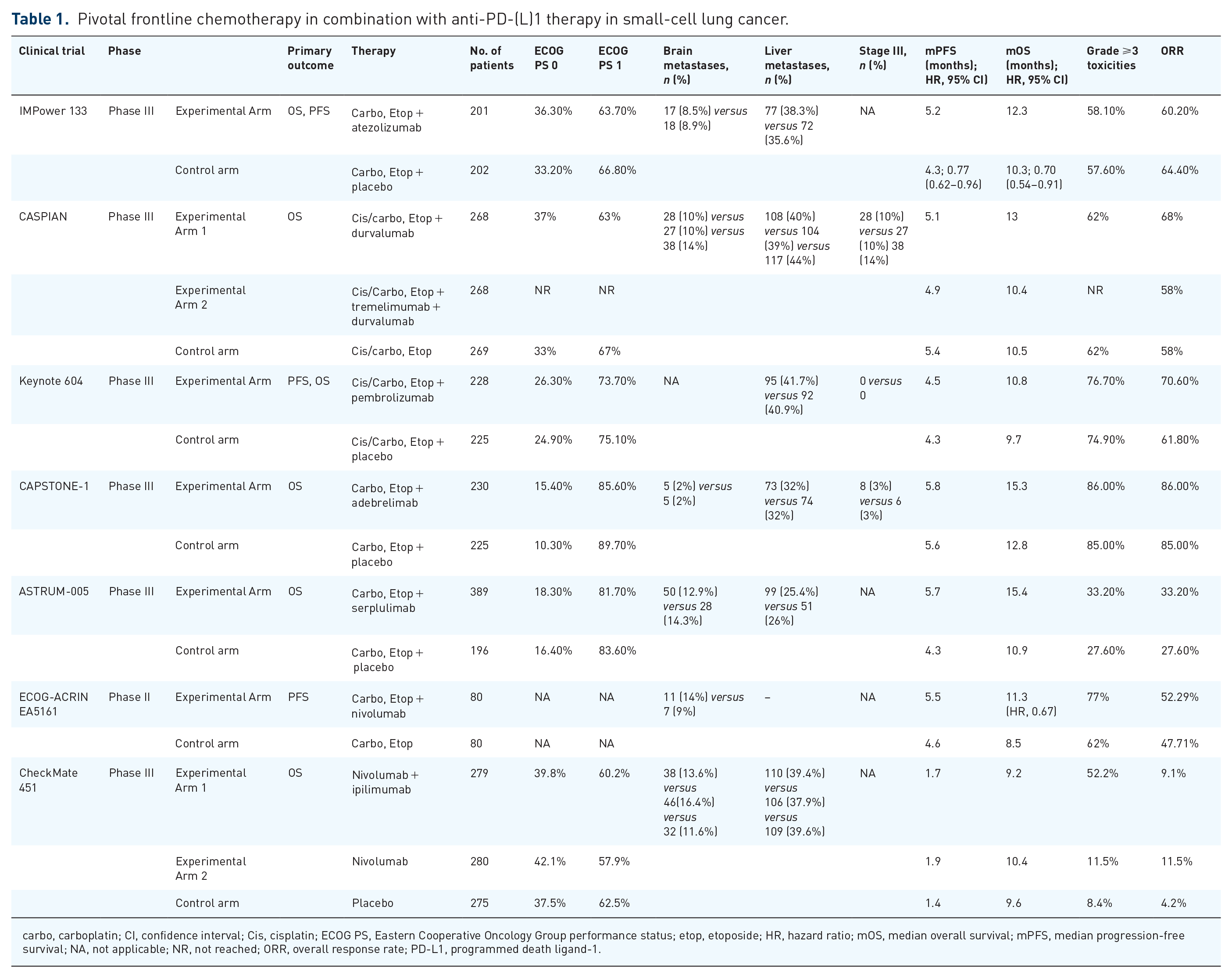
carbo, carboplatin; CI, confidence interval; Cis, cisplatin; ECOG PS, Eastern Cooperative Oncology Group performance status; etop, etoposide; HR, hazard ratio; mOS, median overall survival; mPFS, median progression-free survival; NA, not applicable; NR, not reached; ORR, overall response rate; PD-L1, programmed death ligand-1.
Immune checkpoint blockade in SCLC: rationale and recent progress
SCLC has long been hypothesized as an immunogenic disease with the potential to be exploited therapeutically. SCLC has a strong correlation with cigarette smoking, 21 high tumor mutational burden (TMB), 22 and high genomic instability, likely induced by the impairment of tumor suppressor genes RB1 and TP53 (which encode RB and p53, respectively) and exposure to tobacco carcinogens 23 ; together with well-recognized autoimmune paraneoplastic features of SCLC, such as Lambert–Eaton syndrome, 24 the successful deployment of immunotherapeutic agents in SCLC seemed a logical hypothesis. However, in clinical practice, in SCLC, responses to immune checkpoint blockade (ICB) are rare, likely due in part, to some of the following: low tumor infiltration by immune cells, most notably by cytotoxic T cells, reduced expression of human leucocyte antigens (HLAs), interferon signatures, and immune checkpoints. 25
Recently, our understanding of the biology of SCLC has been radically altered as whole-genome sequencing and transcriptional profiling have identified specific patterns in SCLC, which may account for the lack of ICB response and may pave the way for further target exploration and rationally designed clinical trials for SCLC.26–28 Important recent work from Dr Lauren Byers’ group in MD Andersen Cancer Center has identified specific patterns of transcription factor (TF) programs and immune pathway activation which define four major subtypes of SCLC that contain distinct therapeutic vulnerabilities to be exploited (Table 2). 27 In current clinical practice,10,11 SCLC is treated as a single disease entity – in fact, SCLC is a heterogeneous disease with diverse clinical heterogeneity.
Small-cell lung cancer subtypes, described by Gay et al., 27 including key TFs, and possible therapeutic approaches.

ASCL1, ATP-citrate lyase gene 1; BCL2, B-cell lymphoma 2; BTK, Bruton tyrosine kinase; DLL3, delta-like protein 3; EMT, epithelial–mesenchymal transition; NEUROD1, neurogenic differentiation 1; PARP, poly(ADP-ribose) polymerase; SCLC, small-cell lung cancer; SLFN11, schlafen family member 11; SSTR2, somatostatin receptor 2; TF, transcription factor.
Gay et al. 27 used tumor expression data and non-negative matrix factorization to identify four SCLC subtypes, defined largely by differential expression of TFs ASCL1, NEUROD1, and POU2F3, or by low expression of all three TF signatures accompanied by an inflamed gene signature (SCLC-A, N, P, and I, respectively) (Table 1). SCLC-I, or ‘inflamed’ neuro-endocrine-low SCLC subtype, which lacks the expression of TFs ASCL1, NEUROD1, and POU2F3, and demonstrates high expression of epithelial–mesenchymal transition, increased immune cell infiltration, with significantly increased expression of both CD8A and CD8B, suggesting greater cytotoxic T-cell infiltration, high total immune infiltrate [absolute number of several immune cell populations were markedly increased for example, including T-cells, natural killer (NK) cells, and macrophages], enhanced antigen presentation machinery (such as increased expression of genes encoding HLAs), and consistently higher expression of an 18-gene interferon-γ-related T-cell gene expression profile, 27 which has been shown to predict response to ICB in solid tumors independent of TMB. 29 All of these features suggest a therapeutic vulnerability to checkpoint inhibition in this subtype.
Gay et al. also demonstrated that SCLC-I cells express high levels of Bruton tyrosine kinase (BTK), suggesting BTKi, such as ibrutinib, may also have some therapeutic potential. 27 Intriguingly, the group also noted that the treatment of SCLC-A patient-derived xenografts with cisplatin-based chemotherapy-induced intra-tumoral shifts toward the SCLC-I subtype, suggesting that perhaps switching of subtype could be a mechanism of acquired platinum resistance. 27 Moreover, Byers and her group demonstrated that unique therapeutic vulnerabilities are present across subtypes (Table 2); for example, in subgroup SCLC-P, which accounts for 7% of SCLC cases, 30 and had high POU2F3 expression, preclinical models were significantly more sensitive to poly(ADP-ribose) polymerase (PARP) inhibition, 27 and displayed sensitivity to anti-metabolites, such as anti-folate compounds and nucleoside analogs (Table 2). 27 SLFN11 has recently emerged as a promising predictor of sensitivity to DNA-damaging chemotherapies, and more recently, has been associated with sensitivity to PARP inhibition31–33; however, in SCLC-P models, PARP inhibitor sensitivity occurred in the context of modest and inconsistent SLFN11 expression. 27 In subgroup SCLC-A, characterized by high ASCL1 expression, high delta-like ligand-3 (DLL3) expression was noted, 27 and more responsive to platinum therapy and PARP inhibition, as well as increased susceptibility to BCL2 inhibition; SCLC-N models were highly sensitive to multiple Aurora kinase (AURK) inhibitors (Table 2). 27
Altogether, these data are thought-provoking and hypothesis generating, there is an urgent need for a greater understanding of SCLC phenotypes; SCLC-I may well derive the greatest benefit from the addition of immunotherapy to chemotherapy, and these recent data suggest that perhaps each of these subtypes demonstrate specific therapeutic vulnerabilities, ranging from PARP inhibition to inhibition of BCL-2 or AURKs, that could be exploited. It is likely that the identification and incorporation of patients demonstrating SCLC-I subtype in the setting of clinical trials and beyond may be key in unlocking the potential of ICB and beyond in SCLC.
Pivotal clinical trial data
While early activity was demonstrated using programmed death ligand-1 (PD-L1) inhibitor monotherapy in SCLC, 34 it was not until combination strategies utilizing anti-PD-L1 inhibitors with first-line chemotherapy that meaningful improved survival results were finally achieved. ICBs as monotherapy were briefly FDA approved in pretreated ED-SCLC, nivolumab in 2018, and pembrolizumab in 2019, in the third-line setting following encouraging early activity in respective trials.35,36 However, while a fraction of patients derived long-term survival benefits, the subsequent larger phase clinical trials failed to demonstrate OS benefit, and the FDA indications were consequently withdrawn. Similarly, in the maintenance setting following induction chemotherapy, treatment using single-agent ICB produced only modest activity.37,38 It was not until ICB was combined with chemotherapy that a significant survival benefit was achieved.
Front-line treatment-naïve data: PD-L1/PD-1 inhibitors
IMpower 133
IMpower 133 was the first randomized controlled trial (RCT) to demonstrate meaningful OS advantage, investigating PD-L1 inhibitor atezolizumab combined with platinum-based chemotherapy (carboplatin and etoposide) for four cycles followed by maintenance atezolizumab versus platinum-based chemotherapy alone (Table 1). 19 In total, 403 patients were randomized and a median follow-up of 13.9 months; the median OS of the atezolizumab group and placebo group was 12.3 and 10.3 months, respectively [hazard ratio (HR) 0.70; 95% confidence interval (CI) 0.54–0.91; p = 0.007] (Table 1) 19 ; atezolizumab was also associated with an improvement in median progression-free survival (PFS) (5.2 and 4.3 months; HR, 0.77; 95% CI, 0.62–0.9).
Interestingly, Dr Lauren Byer’s group retrospectively applied SCLC subtypes to IMpower 133 to explore the survival impact, and a robust expression of the SCLC-I group was noted in the study population. 27 Patients included in the IMpower 133 trial were stratified according to SCLC variants, and intriguingly a trend to higher median OS was noted using atezolizumab in combination with platinum-based chemotherapy in the SCLC-I subgroup (18 versus 10 months; HR, 0.566; 95% CI, 0.321–0.998), 27 in contrast to all other subgroups. Only modest gains were noted in the SCLC-P subgroup with the addition of atezolizumab (median OS in 9.6 versus 6 months), SCLC-A (10.9 versus 10.6 months), and SCLC-N (10.6 versus 9.4 months) (Table 1). 27
CASPIAN trial
In the CASPIAN trial, a global, randomized, open-label phase III trial, three treatment groups were included: (1) a CTLA-4 inhibitor, tremelimumab plus PD-L1 inhibitor, durvalumab plus chemotherapy, and versus (2) durvalumab plus chemotherapy versus (3) chemotherapy alone. 20 In total, 805 patients were randomized, with OS as the primary endpoint. The trial met its primary endpoint, as durvalumab plus platinum–etoposide chemotherapy significantly improved OS [median overall survival (mOS) was 13 versus 10.3 months; HR, 0.73, 95% CI, 0.59–0.91; p = 0.0047] (Table 1), though there was no significant PFS survival advantage across treatment arms. 20
There are several differences between these two practice-changing trials (Table 1): both studies deployed a standard platinum chemotherapy backbone with etoposide, though CASPIAN included either cisplatin or carboplatin, whereas IMpower 133 used only carboplatin. CASPIAN allowed up to six cycles of induction therapy, while four cycles of induction therapy were used in IMpower 133. Both studies permitted patients with brain metastases, and prophylactic cranial irradiation (PCI); however, CASPIAN allowed untreated asymptomatic brain metastases, whereas in Impower 133, all brain metastases required treatment, and PCI was permitted only in the control arm of CASPIAN, though there was no difference in the incidence on central nervous system metastases. In addition, both IMpower 133 and CASPIAN excluded consolidative thoracic radiotherapy, and both trials excluded patients with poor Eastern Cooperative Oncology Group (ECOG) performance status.
Despite these differences in trial design, these data confirmed PD-L1 inhibitor plus chemotherapy as a combination strategy in patients with ES-SCLC and reassuringly replicated prolonged OS results which led to subsequent FDA and EMA (European medical agency) approvals, changing global clinical practice in SCLC, ushering in a new ICB era in SCLC.
Keynote-604
In Keynote-604, a PD-1 inhibitor targeting the same PD-1/PD-L1 signaling pathway in combination with chemotherapy failed to demonstrate improvement in OS. A total of 453 patients were randomized (223 participants per group) and while a statistically significant improvement in PFS was reported (4.5 versus 4.3 months, respectively; HR, 0.75; 95% CI, 0.61–0.91; p = 0.0023), median OS was 10.8 and 9.7 months (HR, 0.80; 95% CI, 0.64–0.98; p = 0.0164) (Table 1), 39 and the combination failed to reach statistical significance for OS primary endpoint. The conflicting data of Keynote-604 highlight the urgent need for better patient selection to identify accurately those patients with SCLC who will derive benefit from immunotherapy.
ECOG-ACRIN EA5161
Another randomized trial that demonstrated efficacy with the addition of immunotherapy to chemotherapy was the ECOG-ACRIN EA5161 trial, a randomized phase II trial (n = 145) that investigated PD-1 inhibitor, nivolumab, in combination with platinum–etoposide-based treatment upfront for patients with ES-SCLC (Table 1). 40 The trial met its primary endpoint, demonstrating PFS benefit, with a median PFS of 5.5 versus 4.6 months (HR, 0.65, 95% CI, 0.46–0.91; p = 0.012); in addition, a meaningful improvement in OS was noted (11.3 versus 8.5 months; HR, 0.67, 95% CI, 0.46–0.98; p = 0.038), and an improvement in PFS and ORR (Table 1), adding further support to the strategy of chemoimmunotherapy as first-line treatment for SCLC.
Capstone-1
More recently, a novel high-affinity humanized monoclonal antibody against PD-L1, adebrelimab was investigated in combination with platinum-based chemotherapy in the CAPSTONE 1 trial (Table 1). 41 A statistical improvement in OS was reported, as the median OS was 15.3 months for the IO combination versus 12.8 months for the chemotherapy arm (HR, 0.72, 95% CI, 0.58–0.90; p = 0.0017); interestingly, a numerically longer survival was noted in this study, than in both IMPower 133 and CASPIAN,19,20 perhaps related to the patient population (Capstone-1 included more patients receiving subsequent systemic treatments, as well as less patients with brain metastases) (Table 1). 41
ASTRUM-005
The ASTRUM-005 trial, 42 an international randomized phase III trial investigated serplulimab, a novel anti-PD1 monoclonal antibody in combination with platinum-based chemotherapy and demonstrated prolonged OS in patients with ES-SCLC by 4.5 months, with significantly reduced risk of death (15.4 versus 10.9 months, respectively; HR, 0.63, 95% CI, 0.49–0.82; p < 0.001). The IO combination also favored PFS (HR, 0.48, 95% CI, 0.38–0.59), objective response rate (ORR), and DOR, and following the failure of Keynote-604 (Table 1). 39 ASTRUM-005 is the first trial to demonstrate that PD-1 inhibitor plus chemotherapy can also improve OS in ES-SCLC in the first line, suggesting serplulimab in combination with chemotherapy as another valid treatment option for first-line treatment of ES-SCLC. 42
CTLA-4 inhibition
CTLA-4 has been explored in SCLC across multiple studies, with disappointing results. While promising survival rates were initially observed investigating CTLA-4 inhibitor ipilimumab in combination with nivolumab in previously treated SCLC in CheckMate 032, 34 the randomized section of the trial failed to demonstrate improved survival between nivolumab alone and nivolumab plus ipilimumab 35 ; there were also more adverse events with the combination, and subsequent trials have also struggled to demonstrate meaningful clinical benefit.
NCT01450761
In this phase III randomized double-blind trial, NCT01450761, a phased course of ipilimumab versus placebo was combined with etoposide and platinum (carboplatin or cisplatin) in newly diagnosed ES-SCLC patients. 43 The addition of ipilimumab to chemotherapy did not prolong PFS or OS [median PFS was 4.6 months compared to 4.4 months (HR, 0.85, 95% CI, 0.75–0.97); median OS was 11 versus 10 months (HR, 0.94, 95% CI, 0.81–1.09; p = 0.3775)] (Table 1).
Maintenance trials: CheckMate 451
The efficacy of a maintenance strategy with checkpoint inhibitors has also been explored with anti-PD-1 inhibitors and anti-CTLA-4 agents. Based on promising RRs from CheckMate 032, CheckMate 451 a randomized phase III trial, was designed to investigate nivolumab as maintenance treatment either alone or in combination with ipilimumab after platinum-based chemotherapy in ES-SCLC. 37 The combination failed to show a survival benefit, and instead of improved survival the combination induced increased toxicities. 44 Median survival with dual blockade was 9.2 months compared to 9.6 months with placebo (HR, 0.92; 95% CI, 0.75–1.12), and monotherapy using nivolumab also failed to demonstrate improvement over placebo (10.4 months; HR, 0.84; 95% CI, 0.69–1.02). Based on these data, and other trials, there is currently no role for CTLA-4 inhibition in the treatment of ES-SCLC.
Challenges to IO success in SCLC
While undeniable progress has been made recently with immunotherapy in combination with chemotherapy for ES-SCLC, the absolute survival benefit of short months in these landmark trials suggests that more progress is urgently needed. Currently, it is thought that poor responses to ICB in SCLC are due to issues such as low tumor infiltration by immune cells, most notably by cytotoxic T cells, reduced expression of HLAs, and reduced gamma interferon signatures. 25 While these are all likely important factors, recent data have also deepened our understanding of the biology of the SCLC and suggest that selecting patients with SCLC-I, who logically express higher total immune tumor infiltrate, higher antigen presentation, and increased gamma interferon signature (Table 2), could be impactful in the setting of clinical trial design. 27 To date, clinical trials in SCLC have essentially been undertaken in an unselected SCLC patient population, not taking into account the vast heterogeneity of the biology of the disease, nor the heterogeneity in disease response. The novel classification system27,45 offers the potential for rationale patient selection, and thus rationale clinical trial design. While this new classification system has challenges, such as intra-tumoral heterogeneity and plasticity between subtypes, it also presents a significant opportunity, with huge potential in terms of thoughtful trial design, with the potential to help inform the selection of future systemic treatments. 27
Regardless, better predictive biomarkers are urgently needed: SCLC has a relatively low expression of PD-L1, typically less than 15%, compared to the roughly 60% on average positivity present in NSCLC,46,47 while effector CD8+ lymphocytes are also found to be 5.4-fold and 6-fold lower in SCLC than in adenocarcinoma and squamous cell carcinoma, respectively. 48 In IMpower 133, only 34% were evaluable for PD-L1 expression, and a survival benefit was achieved in both PD-L1-positive and PD-L1-negative patients 49 ; in CASPIAN, PD-L1 expression was evaluable in 51.6% of patients, though in those evaluable, PD-L1 expression was only noted on 5.1% of tumor cells and 22.4% of immune cells and again there was a correlation between PD-L1 expression and survival. 20 Similarly, TMB has been investigated prospectively as a predictive biomarker in SCLC in clinical trials and has repeatedly failed to demonstrate utility.20,49
SLFN11 has emerged as a promising predictor of sensitivity to DNA-damaging chemotherapies and, more recently, is associated with sensitivity to PARP inhibition,31–33 and current clinical trials are trying to prospectively validate SLFN11 as a bona fide predictive biomarker in SCLC.
The recent phase II randomized SWOG 1929 study conducted in patients with SLFN11-positive ES-SCLC, where patients with ES-SCLC expressing SLFN11 (H-score ⩾ 1, evaluated centrally at MDACC) were randomized to maintenance atezolizumab versus atezolizumab plus PARP inhibitor talazoparib following frontline chemotherapy, confirmed that maintenance atezolizumab in combination with talazoparib, improved PFS in SLFN-11 selected patients 50 ; more importantly, this study provided proof of principle that patient selection is key, and biomarker-driven trials are feasible in SCLC. Otherwise, the differential expression of the recently described four key transcription regulators, which define the four subtypes, ASCL1 (SCLC-A), NeuroD1 (SCLC-N), POU2F3 (SCLC-P), and YAP1 (SCLC-Y), is being proposed as potential biomarkers,27,45 and being explored in the context of clinical trials.
The challenge remains to translate the recent successes in clinical trial development to the SCLC population being reviewed regularly in the clinic. A practical consideration is that many patients with SCLC often require steroids for disease-related symptoms, such as symptomatic brain metastases and dyspnea, which can limit ICB efficacy. 23 The pivotal clinical trials described in this review also excluded many ‘real-life’ SCLC patients, excluding patients with poor performance status, where the potential to induce more toxicities may be greater. A challenge, many thoracic oncologists face, is whether the small OS benefit noted in the large phase III RCTs, such as IMpower 133 and CASPIAN, translates to so-called ‘real-life’ patients and whether the potential addition of toxicities induced by extra agents is worth it in SCLC patients with poor performance status. Clinical trials are currently underway to address this question; however, more therapeutic options are clearly needed to treat the different subtypes of this challenging and aggressive disease.
Another challenge in the clinic is the transformation to SCLC that occurs in a small but important subset of patients with NSCLC, typically epidermal growth factor (EGFR)-driven NSCLC. Indeed, SCLC transformation is a documented mechanism of resistance to chemotherapy, immunotherapy, and targeted therapy in NSCLC, and de novo SCLC and transformed SCLC have very different pathogenesis and tumor microenvironment. 51 While some similarities exist, such as pathological morphology, molecular characteristics, clinical manifestations, and drug sensitivity, transformed SCLC represents a new phenotype of SCLC, characterized by an aggressive disease, with limited effective treatments and dismal outcomes. While platinum-etoposide and taxane-based chemotherapy have induced clinical responses in cases of transformed SCLC, 52 documented responses to immune blockade in this population have been poor. 52 A recent study has suggested a role for PD-L1 inhibition in combination with chemotherapy and anti-angiogenic agent bevacizumab. 53 Though small in numbers, here, the median post-transformation SCLC OS of the ICB group was significantly longer than that non-ICB group (20.2 versus 7.9 months, p < 0.01). 53 Additional investigation and clinical trials in a larger cohort of patients with transformed SCLC are clearly needed to better understand the clonal evolution in transformed SCLC and to better elucidate optimal treatment strategies in this group.
Novel immunotherapeutic approaches
There are several clinical trials in progress assessing next-generation immunotherapeutic agents across various settings in SCLC, both in combination with chemotherapy and beyond (Table 3). PD-1 and PD-L1 are consistently relevant immune checkpoints under investigation; in addition to newer agents engaging with these targets, other checkpoints are also being interrogated, to promote even greater antitumor immune activation and induce deeper and more meaningful clinical responses in patients with SCLC.54,55 Immunotherapeutic strategies being explored include chimeric antigen receptor (CAR) T-cells, tumor vaccines, antibody–drug conjugates (ADCs), and immunomodulators. 56
Select ongoing trials of immunotherapeutic agents in SCLC.
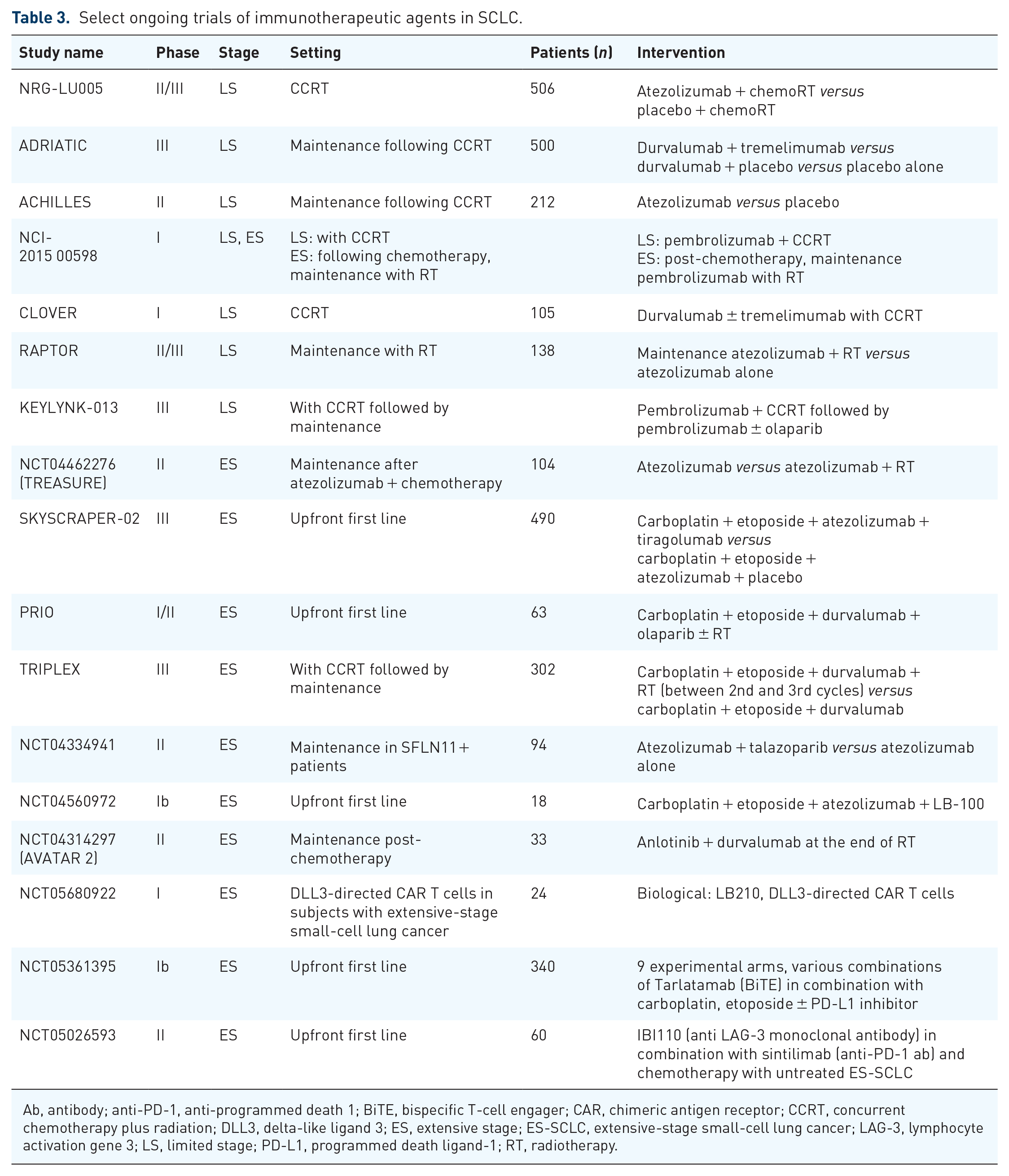
Ab, antibody; anti-PD-1, anti-programmed death 1; BiTE, bispecific T-cell engager; CAR, chimeric antigen receptor; CCRT, concurrent chemotherapy plus radiation; DLL3, delta-like ligand 3; ES, extensive stage; ES-SCLC, extensive-stage small-cell lung cancer; LAG-3, lymphocyte activation gene 3; LS, limited stage; PD-L1, programmed death ligand-1; RT, radiotherapy.
TIGIT
A novel therapeutic target being investigated across tumors is a T-cell immunoreceptor with immunoglobulin (Ig) and immunoreceptor tyrosine-based inhibitory motif (ITIM) domains (TIGIT). TIGIT is a co-inhibitory immune receptor present in T cells and NK cells, frequently co-expressed with PD-L1, and is an important regulator of T-cell mediated immunity. 57 To augment the antitumor effect of PD-1/PD-L1 inhibitors, preclinical models have confirmed synergistic activity when TIGIT inhibitors were used in combination with anti-PD-1 blockade. 58 The randomized phase II CITYSCAPE trial has demonstrated promising efficacy with TIGIT blockade in NSCLC, where the addition of anti-TIGIT antibody tiragolumab to atezolizumab improved PFS (HR, 0.62; 95% CI, 0.42–0.91). 59 Responses have not yet translated to SCLC: in the recent SKYSCRAPER-02 trial, a randomized, double-blinded, phase III trial, which compared tiragolumab, together with atezolizumab and platinum doublet chemotherapy with atezolizumab, the addition of tiragolumab to chemo-immunotherapy did not improve survival: OS was 13.6 versus 13.6 months (HR, 1.04, CI, 0.79–1.36; p = 0.7963), 60 though the combination was well tolerated, and no new safety signals were identified.
LAG-3
Lymphocyte-activated gene 3 (LAG-3) is a cell surface inhibitory receptor and key regulator of immune homeostasis,61,62 and another important next-generation immune checkpoint molecule being interrogated in the context of ongoing clinical trials. LAG-3 inhibitor, IBI110 combined with sintilimab and chemotherapy is one such combination strategy (NCT05026593) being explored in the clinical trial setting.
Adoptive cell therapy
Adoptive cell therapy involves the transfer of CAR-T cells directed against specific cell surface antigens, targets that are highly expressed in SCLC. Examples include CD56, delta-like protein 3 (DLL3), and CD47, which are all highly expressed in SCLC and are potential targets for CAR-T therapy. Bispecific T-cell engager (TCE) AMG 757, for example, tarlatamab, which binds both DLL3 and CD3 leading to T-cell-mediated tumor lysis, 63 has shown encouraging response durability in patients with relapsed/refractory SCLC in a phase I trial (NCT03319940). 64 The recently published open-label, multicenter, phase II trial, DeLLphi-301, has confirmed the antitumor activity of tarlatamab with durable objective responses and promising survival outcomes in patients with previously treated small-cell lung cancer. 65 In this study, 220 patients received tarlatamab either at a dose of 10 or 100 mg: an objective response occurred in 40% (97.5% CI, 20–52) of the 10 mg group and 32% (97.5% CI, 21–44) of the 100 mg group; and among those with objective response, the DOR was at least 6 months in 59% (40 of 68 patients). Adverse events, such as cytokine release syndrome, which occurred in 51% of the patients in the 10 mg group and 61% of those in the 100 mg group, remain a concern, though most of these were grade 1 or 2 in severity. 65 A dose of 10 mg was selected for subsequent tarlatamab trials moving forward. These findings are certainly promising, especially relative to the poor outcomes of clinical trials of current standard-of-care second-line treatment options.
DLL3 TCEs, which have also shown encouraging results, include HPN328 and BI764532.66,67 HHPN328 is a novel half-life extended DLL3-targeting TCE derived from the TriTAC platform, designed to minimize off-target toxicities, and an ongoing phase I/IIA study has reported encouraging results 66 : 6 of 15 patients (40%) achieved a decrease in the sum of target lesion diameters, 66 the compound was well tolerated, clinically active, and adverse events were transient, manageable, and consistent with class. BI764532 is a DLL3/CD3 T cell engaging bispecific antibody that has shown potent preclinical antitumor activity in DLL3+ cells and xenograft models, and an ongoing phase I trial has shown promising efficacy, achieving a 33% ORR in 24 SCLC patients, and clinically manageable tolerability. 67
Epigenetic therapies
Recently epigenetic reprogramming has emerged as another potential therapeutic target in SCLC, and several epigenetic modifications such as DNA methylation, chromatic accessibility, and histone modifications are being actively investigated. 68 The enhancer of zeste homolog 2 (EZH2), for example, an enzymatic subunit of polycomb repressive complex 2, is a transcription regulator highly expressed in SCLC, 69 and is associated with aberrant methylation and transcription repression; in addition, it can promote tumorigenesis, including cell cycle regulation and the down-regulation of major histocompatibility complex class 1 (MHC1) and C–C motif chemokine ligand 5 expressions. 69 The EZH2–SLFN11 axis has been shown to impact response to chemotherapy in SCLC, for example, SLFN11 enhances sensitivity to chemotherapy in pre-clinical models. 68 Valemetostat tosilate (valemetostat) is a selective dual inhibitor of EZH1/2 being explored in combination with irinotecan and topotecan in early-phase clinical trials (Table 4).
Select ongoing clinical trials with novel targeted agents under investigation in ES-SCLC.

AT, atezolizumab and talazoparib combination; CAV, cyclophosphamide, adriamycin and vincristine; CD3, cluster of differentiation 3; CI, confidence interval; CR, complete response; DLL3, delta-like ligand-3; DLT, dose limiting toxicities; ES-SCLC, extensive-stage small-cell lung cancer; HR, hazard ratio; mOS, median overall survival; mPFS, median progression-free survival; ORR, overall response rate; PDGFR, platelet-derived growth factor receptor; PK, pharmacokinetics; PR, partial response; RP2D, recommended phase 2 dose; VEGFR, vascular endothelial growth factor receptor.
Antibody–drug conjugates
ADCs are one of the fastest-growing classes of oncology drugs in modern drug development and through the targeting of cell surface-specific targets can harness the powers of both cytotoxic chemotherapy and targeted therapy. 79 Trophoblast cell surface antigen (TROP2) is expressed in many epithelial cancers and is highly expressed in approximately 10% of SCLC. 74 Sacituzumab Govitecan is a first-in-class ADC that targets TROP2 antigen, has shown activity in phase I/II basket trial, IMMU-132-01 (ORR of 18%, median DOR of 5.7 months, PFS of 3.7 months, and OS of 7.1 months),71,75 and is currently being explored in phase II setting (TROPiCS-03) (Table 4). Ifinatamab deruxtecan (I-DXd; DS-7300) is another ADC that has demonstrated robust and durable efficacy in patients with heavily pretreated SCLC (Table 4). 77 B7 homolog 3 (B7-H3) is a transmembrane immunoregulatory protein overexpressed in several tumor types, including SCLC; I-DXd is a novel B7-H3-directed ADC that has achieved confirmed responses in 53% of patients (including CR in 4.8%; median PFS was 4.8 months, mOS 12.2 months) (Table 4). 77 A phase II study (NCT05280470) of patients with second- or third-line ES-SCLC is currently ongoing.
Radiation therapy
The combination of immunotherapy agents with therapeutic radiation is also being explored in the clinical trial setting in SCLC (Table 3). Radiation causes neoantigen release from cancer cells, and antigen-presenting cells prime T cells to these neoantigens, allowing for T-cell-mediated cytotoxicity of cancer cells 80 ; thus, radiation therapy may potentiate synchronous immunostimulatory and immunosuppressive effects within a tumor site.81,82 However, the phase II STIMULI trial, which examined the use of consolidation nivolumab (1 mg/kg) plus ipilimumab (3 mg/kg) every 3 weeks following primary concurrent chemoradiation (CCRT) therapy did not meet its primary endpoint of PFS benefit, and consolidative ipilimumab with nivolumab was not well tolerated. 83 Given the extensive preclinical data suggesting a synergistic benefit between ICB and radiation therapy,81,84,85 further trials are ongoing (Table 3). The multi-institutional randomized phase II trial ACHILES, for example, is investigating the inclusion of atezolizumab into the consolidative phase after CCRT in LS-SCLC (NCT03540420); the ambitious three-arm phase III ADRIATIC trial RCT (NCT03703297) is investigating similar patients, using (1) consolidation durvalumab 1500 mg plus tremelimumab 75 mg (up to four doses) every 4 weeks (2), durvalumab 1500 mg plus placebo every 4 weeks (up to four doses), or (3) combination placebo every 4 weeks, followed by durvalumab single agent every 4 weeks (1 + 2) or placebo every 4 weeks (3). 86 In ES-SCLC, the Raptor trial is a phase II/III study investigating the addition of radiation to atezolizumab in patients with ES-SCLC (NCT04402788). The TRIPLEX trial is investigating the addition of thoracic radiotherapy to durvalumab (MEDI4736) plus chemotherapy in extensive-stage small-cell lung cancer (Table 3), 78 while TREASURE is a phase II trial, designed to explore the efficacy and safety of thoracic radiation to atezolizumab. 87 The early cessation of recruitment for TREASURE due to unexpected safety data, interim safety analysis demonstrating a higher rate than expected of grade 3 or more pneumonitis, 88 only highlights ongoing concerns regarding toxicities such as radiation pneumonitis.
Targeted therapies
Though beyond the scope of this review, another source of promise in the treatment of SCLC is the recent identification of potential targets for precision therapies in SCLC (Table 2), and the many novel agents are under investigation (Table 4). PARP inhibition, for example, has garnered significant interest recently in SCLC, where the high incidence of genomic aberrations observed causes an accumulation of DNA damage and genomic instability. SCLC cells can depend on functional DNA damage repair pathways, 89 and proteomic profiling has demonstrated increased levels of PARP in SCLC cells90,91; in addition, synergism has been observed with PARP inhibitors in combination with ICB in SCLC. 90 Thus, current clinical trials are investigating PARP inhibitors, in combination with DNA-damaging chemotherapy backbones plus immunotherapeutic agents, 92 as well as in combination with novel agents such as anti-TIGIT monoclonal antibodies (AdvanTIG-204, ML41257) in LS-SCLC (NCT04308785) (Table 4).
Conclusion
SCLC is a lethal disease with aggressive biology, and while progress has been slow in improving outcomes in the last three decades for patients with SCLC, recent progress has paved the way for changes in clinical practice. Several large randomized controlled phase III trials have confirmed that immunotherapeutic agents in combination with chemotherapy can improve survival in SCLC – the challenge now is to build on these modest successes and aim to induce deeper and more prolonged responses, as well as to overcome resistance mechanisms to improve patient outcomes.
Recent large-scale profiling studies have provided important biological insights into the genomic and proteomic landscapes of SCLC, with the new SCLC classification system providing a greater understanding of the heterogeneity of SCLC.23,27,93 With this increased knowledge comes the enormous potential for more thoughtful and rational clinical trial design. The recent randomized phase II SWOG 1929 study, which demonstrated improved PFS in SLFN-11 selected patients, 50 provided proof of concept, that biomarker-driven trials are feasible in SCLC. The many novel immunomodulatory agents and combination strategies currently under investigation provide further cause for optimism (Tables 3 and 4), to augment the new chemoimmunotherapy backbone.
Clinical trials to date in SCLC have essentially been undertaken in an unselected SCLC patient population. There is an urgent need to exploit the unique therapeutic vulnerabilities that are present across the different biologically distinct subtypes of SCLC.27,45 The development and subsequent incorporation of prospective predictive biomarkers in clinical trial design is imperative, and rigorous research coupled with clinical validation to optimize the efficacy of biomarkers will be key. The identification of patients of subgroup SCLC-I may also be important, as identifying potential SCLC patients who derive the most benefit from ICB and beyond is crucial. The many challenges in SCLC persist, but for the first time in decades in this deadly disease, an abundance of hope is on the horizon.
