Abstract
This paper presents a patient with a novel Ig-like-III domain fibroblast growth factor receptor (FGFR2) alteration (W290_P307>C) along with CDKN2A/B alterations and a cadherin 1 (CDH1) alteration. Initial responsiveness to pazopanib monotherapy was encouraging, yet progression occurred after 7.5 months. Following progression, the molecular tumor board recommended a combination therapy approach comprising pazopanib, crizotinib, and palbociclib to target all of the changed pathways at the same time. Pazopanib was chosen to specifically target the FGFR2 alteration, while crizotinib was selected due to its potential synthetic lethality with the CDH1 alteration. In addition, the CDK4/6 inhibitor palbociclib was administered to address the CDKN2A/B alterations. The patient exhibited a remarkable and sustained response to this innovative combination. This case not only underscores the potential of tyrosine kinase inhibitors, exemplified by pazopanib, as a viable alternative for patients without access to pan-FGFR inhibitors, but it also emphasizes their efficacy beyond commonly detected point mutations and rearrangements. Notably, the outstanding response to combination therapy, including crizotinib, in a patient with a CDH1 alteration, further substantiates the preclinical evidence of synthetic lethality between crizotinib and CDH1 alterations. To our knowledge, this represents the first clinical evidence demonstrating the efficacy of crizotinib in a patient with a CDH1 alteration. Through careful dosage adjustments and consideration of individualized genomic information, this case exemplifies the power of personalized medicine in achieving favorable treatment outcomes.

Keywords
Introduction
Cholangiocarcinoma is a rare and aggressive cancer that arises from the bile ducts. Despite recent advances in diagnosing and treating cholangiocarcinoma (CCA), the prognosis for patients with this disease remains poor. A significant proportion of CCAs have somatic alterations that can be targeted. 1 Targeting the fibroblast growth factor receptor (FGFR) pathway in intrahepatic cholangiocarcinoma has grown in popularity in recent years, as FGFR2 alterations are common with studies reporting occurrence in approximately 10–16% of cases. 2
FGFR2 is a transmembrane receptor tyrosine kinase that regulates cell growth, differentiation, and survival. Activation occurs upon fibroblast growth factor (FGF) ligand binding to the extracellular domain, inducing structural changes that lead to receptor dimerization and intracellular tyrosine autophosphorylation. This event initiates downstream signaling through molecules like PLCγ, Grb2, and Src family kinases. Certain mutations result in constitutive activation, disrupting signaling cascades and promoting cell proliferation (Figure 1).

FGFR signaling activation and regulation.
The FGFRs have common structural characteristics such as three extracellular immunoglobulin-like domains, a transmembrane domain, and a divided intracellular tyrosine kinase domain (Figure 2).
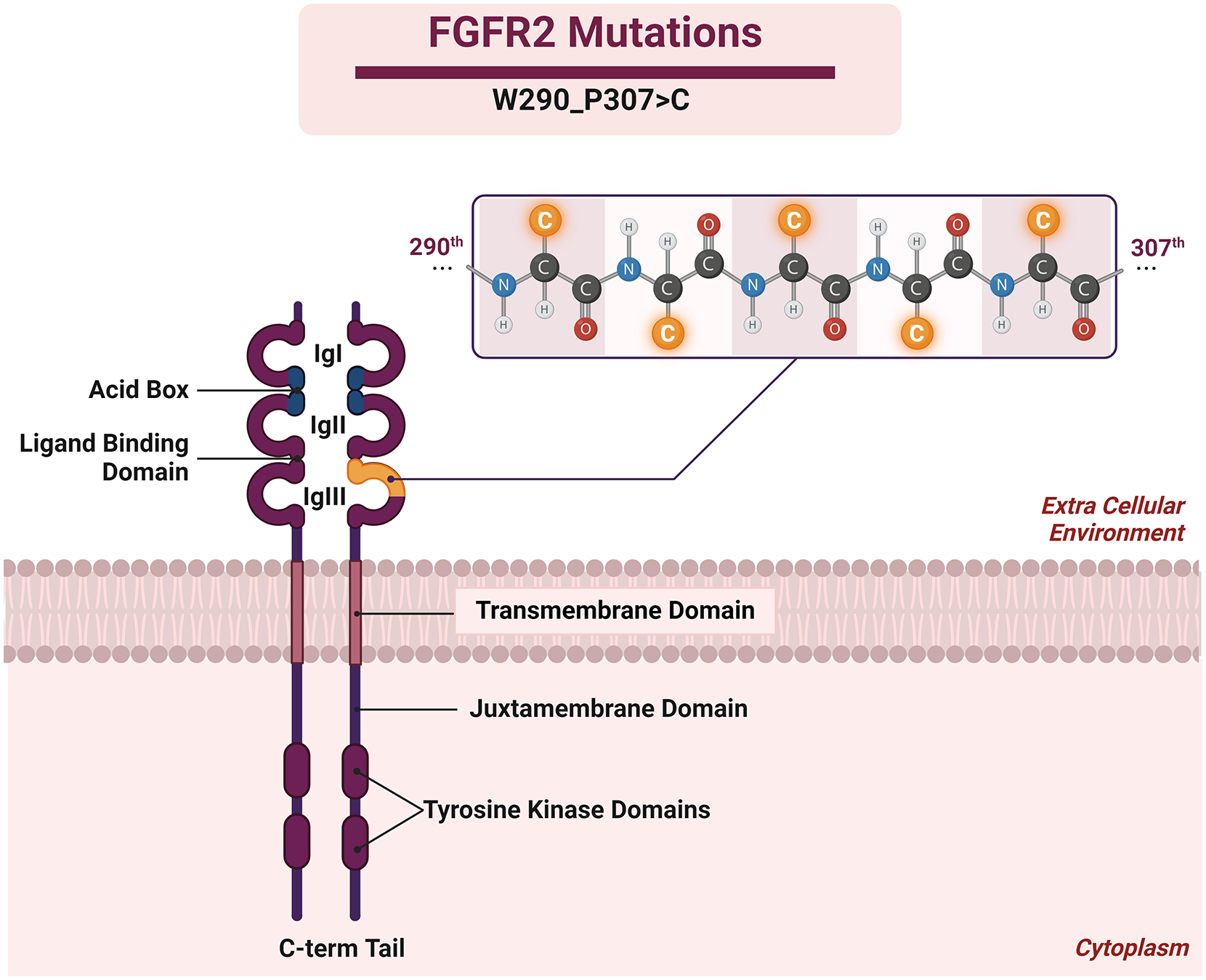
Structure of FGFR2 and novel W290_P307>C mutation.
FGFR2’s extracellular domain binds to FGFs, leading to receptor conformational changes and activation of the intracellular tyrosine kinase. The Ig-1 domain binds to FGF-1 and FGF-2, featuring a unique acidic box inhibiting receptor activation without ligand binding. Ig-2 domain facilitates receptor dimerization, crucial for activation, while Ig-3 domain triggers signal transduction by exposing the tyrosine kinase domain, initiating phosphorylation cascades. The transmembrane domain anchors FGFR2 in the cell membrane. The tyrosine kinase domain of FGFR2 is activated upon receptor dimerization, leading to the autophosphorylation of specific tyrosine residues. This triggers a downstream signaling cascade that ultimately leads to the activation of transcription factors and the expression of target genes.
The C-terminal domain of FGFR2 interacts with various intracellular signaling molecules, such as the docking protein FRS2, and plays a role in the regulation of downstream signaling pathways.
In recent years, several pan-FGFR inhibitors like pemigatinib, infigratinib, and futibatinib have emerged as promising treatment options for patients with FGFR2-altered tumors. However, they can be very expensive and may not be accessible to many patients in certain countries like Türkiye.
When studying FGFR2-targeted therapies, researchers typically investigate more common alterations such as gene rearrangements, amplifications, or point mutations associated with the gene. Although less common, some patients may have rare types of alterations in FGFR2 that could still benefit from targeted therapies, as long as the function of the alteration can be understood and interpreted.
Pazopanib (Votrient) is a second-generation oral multi-kinase inhibitor targeting Vascular Endothelial Growth Factor Receptors (VEGFRs) with high affinity and PDGFRα/β (Platelet-Derived Growth Factor Receptor), FGFR-1/2/3, and c-Kit with lower but still important affinity. 3 It is currently approved for the treatment of metastatic renal cell carcinomas and soft tissue sarcomas but preclinical studies and clinical trials have shown that pazopanib has efficacy in FGFR2-altered cancers. 4 However, the current literature predominantly emphasizes FGFR2 hotspot mutations and rearrangements, leaving a significant knowledge gap regarding its impact on a broader spectrum of FGFR2 alterations. For instance, pazopanib is effective on FGFR2-amplified cancers as well as less common fusion partners and single nucleotide variants.5,6
Cadherin 1 (CDH1) plays a critical role in cell mitosis, and its mutations can contribute to a deficiency in the E-cadherin protein. Disruptions in E-cadherin expression may trigger the overexpression of the ROS proto-oncogene 1 (ROS1), promoting the continued mitosis and proliferation of cancer cells. In cases of CDH1-deficient tumors, preclinical evidence suggests that targeting ROS1 can induce synthetic lethality. 7 Several ROS1 inhibitors, such as crizotinib (Xalkori), have been approved by the FDA for treating non-small-cell lung cancer with ROS1 rearrangements and fusions due to their significant clinical efficacy. However, the management of solid tumors with altered CDH1 expression requires further clinical evidence to support preclinical findings.
Case presentation
A 68-year-old, female, treatment-naive patient was admitted to our hospital with the diagnosis of metastatic cholangiocarcinoma exhibiting multiple bone metastases. Despite the initial recommendation for chemotherapy, the patient opted against this course of treatment. Seeking a second opinion, the patient visited our precision oncology center, where she was advised to undergo comprehensive genomic profiling to decipher potentially actionable alterations and evaluate alternative treatment options.
A tissue sample extracted from the primary liver tumor underwent comprehensive molecular profiling using hybrid capture-based next-generation sequencing by FoundationOne CDx. This extensive analysis encompassed the examination of 324 genes, including the introns of 34 genes associated with rearrangements. In addition, the sample underwent assessment for tumor mutation burden (TMB) and microsatellite instability, providing a thorough genomic characterization. Furthermore, the sample was submitted for Programmed Death-Ligand 1 (PD-L1) testing using immunohistochemistry (IHC) with the Ventana SP263 assay, conducted by Foundation Medicine.
According to the test results, W290_P307>C alteration in the FGFR2, Q677fs*7 alteration in the CDH1, and p16INK4a S56fs*51 and p14ARF Q70fs*78 alterations were detected in CDKN2A/B genes (Table 1).
Next Generation Sequencing (NGS) results of the patient.

CDH1, cadherin 1; FGFR2, fibroblast growth factor receptor; VAF, variant allele frequency.
A low PDL-1 expression (Combined Positive Score (CPS) < 1 and Tumor Proportion Score (TPS) < 1), a TMB of 2 Muts/Mb, and a stable microsatellite status were also reported by the same test. Variant allele frequencies (% VAF) of the alterations were given as 16.25% for FGFR2, 23.1% for CDH1, and 18.9% for CDKN2A/B.
Adenocarcinoma, typified by glandular formations comprising epithelial cells arranged in a single layer of cubic shape, was observed in the diagnostic evaluation. IHC results for both cytokeratin 19 and cytokeratin 7 exhibited positivity in the tumor cells. This collective finding strongly supported the diagnosis of cholangiocarcinoma, further affirming the histological characterization of the observed adenocarcinoma. The results of the histological examination are shown in Figure 3.

(a) The aspect of adenocarcinoma consists of glandular structures lined with cubic single-layer epithelial cells. (b and c) Tumor cells showed both CK 19 (b) and CK 7 (c) immunohistochemistry positivity.
Molecular tumor board
Impressive responses to pazopanib have been reported in the literature for patients with activating FGFR2 alterations, including amplifications and point mutations, across various cancer types.5,8,9 In considering the patient’s treatment plan, the potential benefit from pan-FGFR inhibitors or non-selective multi-kinase inhibitors was weighed. However, the unavailability of pan-FGFR inhibitors like pemigatinib, infigratinib, or futibatinib in our country prompted the molecular tumor board to recommend pazopanib as a viable alternative. Prior to commencing pazopanib treatment patient was offered standard-of-care chemotherapy but she refused once again. In navigating the off-label use in this case, we implemented a thorough and detailed process of informed consent. The patient received comprehensive information regarding the proposed treatment, which included its off-label status, potential risks, and anticipated benefits. Explicit consent was then obtained from the patient, covering not only the treatment itself but also extending to the potential publication of case details. This comprehensive approach aimed to ensure the patient’s complete understanding and voluntary agreement, adhering to ethical standards for off-label use and subsequent publication in medical literature. After the consent was taken based on the decision of the molecular tumor board, the patient was prescribed pazopanib at a dose of 800 mg once daily with radiotherapy for bone metastases. On a PET CT scan conducted after 2 months, an impressive reduction in tumor size was detected at all tumor sites with pazopanib monotherapy [Figure 4(b) and (f) Positron Emission Tomography Computed Tomography (PET CT) Scans – Response to First Treatment].
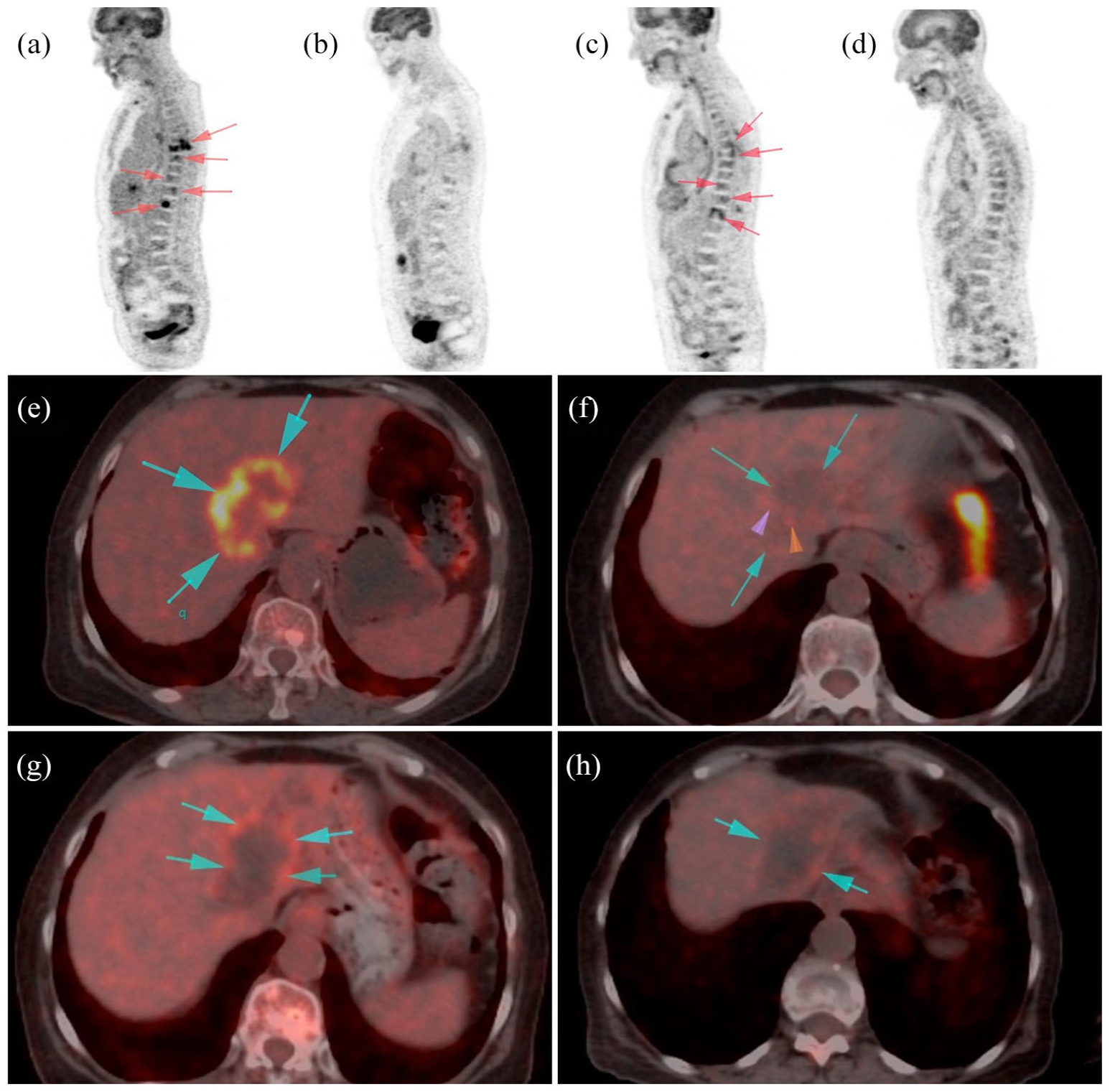
PET CT scans.
Upon attentively monitoring the patient’s condition for 7.5 months, it has become evident that she was showing signs of disease progression [Figure 4(c) and (g) PET CT Scans-Progression). Despite the initial treatment regimen, the patient’s symptoms worsened, suggesting the possible emergence of resistance mechanisms or activation of alternative pathways. Although the medical team recommended a new biopsy for another comprehensive genomic profiling (CGP), the patient chose not to pursue this course. Therefore, based on the current CGP results, the molecular tumor board conducted a thorough reassessment. Palliative radiotherapy was administered to target bone metastases that were not addressed in the previous radiotherapy and the molecular tumor board devised a new plan to manage her condition by incorporating CDK4/6 inhibitor palbociclib (75 mg, 1 day on, 1 day off – lowered from the approved dose of 125 mg PO daily for 3 weeks on and 1 week off) and crizotinib (250 mg/day – lowered from approved dose 250 mg PO bid) into her treatment protocol and reducing the dose of pazopanib to 200 mg PO daily (lowered from approved dose of 800 mg PO daily). This comprehensive and adjusted treatment protocol aimed to effectively target all of the activated pathways contributing to the progression of the disease.10,11 This type of N-of-1 approach to customized therapy has previously also been shown to be effective in several studies.12–15
Written consent was obtained from the patient prior to initiating the combination treatment. Once again, the patient exhibited a positive response to the combinatory therapy and the progression sites were absent in the follow-up PET CT. SUL (standardized uptake value corrected for lean body mass) peak value decreased by 53% compared to progression under pazopanib monotherapy and is consistent with a partial treatment response according to PERCIST (April SUL peak 2.86) post-treatment July 2023 SUL peak 1.33) [Figure 4(d) and (h) PET CT Scans-Response to Combination Therapy].
Combinational treatment was notably well tolerated by the patient. The observed side effects were primarily mild, with grade 1 asthenia based on National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE v5.0), grade 1 leukopenia based on World Health Organization criteria (version 3.2), and loss of appetite being the most notable. The absence of any grade 3 or grade 4 side effects observed in the patient’s response to this combinatory treatment instills optimism regarding its potential for further application and utilization in similar cases. The patient’s journey from diagnosis until the last follow-up is summarized in the timeline presented in Figure 5.
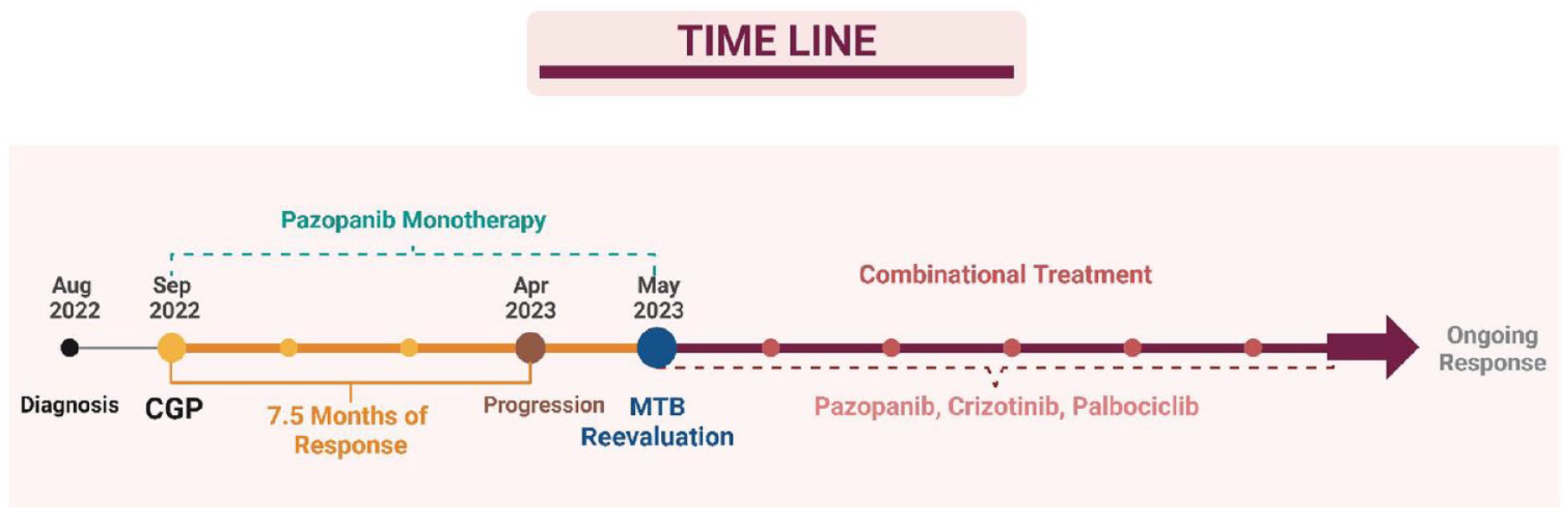
Patient’s treatment timeline summarizing the course of action.
Discussion
Upon reviewing the comprehensive genomic profiling results, we formulated a hypothesis that the patient’s carcinogenesis might be driven by the detected alteration in FGFR2. The W290_P307>C alteration in this gene leads to the substitution of all residues from tryptophan (W) at the 290th position to proline (P) at the 307th position with cysteine (C) (refer to Figure 2). Currently, this alteration lacks characterization in the existing literature, and consequently, its impact on FGFR2 remains unknown. However, by utilizing available data on nearby alterations and focusing on the affected domain and associated properties, the molecular tumor board systematically predicted the potential functionality of the uncharacterized variant.
Alterations starting at the same position, such as W290C and W290_I291delinC, have been previously defined. W290 is located in the Ig-like C2-type domain 3 of the FGFR2 protein. The W209C alteration causes cysteine to replace the tryptophan has been studied and found not to result in increased FGFR2 phosphorylation. However, it increases both ligand-dependent and ligand-independent dimerization of FGFR2. 16 Notably, cell culture studies have demonstrated that this alteration induces transformation, stimulates tumor formation in xenograft models, and enhances growth in a competition assay.16,17 The W290_I291delinC alteration, on the other hand, results in the deletion of tryptophan and isoleucine at the 290th and 291st positions, followed by the insertion of a cysteine. This specific FGFR2 alteration has been associated with increased phosphorylation, dimerization, and oncogenic transformation. 18 Since the alteration detected in our patient affected not only residues at the 290th and 291st positions but extended up to the 307th position, the molecular tumor board reasoned that it could potentially lead to FGFR2 activation.
Furthermore, point mutations and alternative splicing within the third Ig-like domain, like the alteration observed in the patient, can lead to the production of different FGFR2 isoforms with opposing antitumor or protumor functions.19,20 FGFR2b, expressed in epithelial cells, plays a pivotal role in cell division, proliferation, embryonic growth, and wound healing. Dysregulation of FGFR2b signaling is implicated in various cancers, including breast, prostate, and stomach cancers. Conversely, FGFR2c, expressed in mesenchymal cells, regulates mesenchymal cell differentiation and proliferation, with dysregulation associated with cancers like ovarian, pancreatic, and lung cancer. The context-dependent overexpression of FGFR2c or FGFR2b underscores the complexity of FGFR2 isoforms in cancer progression.
Aberrations in tumor suppressor genes, exemplified by CDH1, play a significant role in the functional switch of FGFR2 and the progression of the disease. Notably, in certain conditions like ulcerative colitis, activating FGFR2 mutations are observed to co-occur with CDH1 alterations. This co-occurrence has been implicated in driving FGFR2 hyperactivation, further emphasizing the interconnected and complex relationship between FGFR2 and tumor suppressor genes in disease progression. The use of small-molecule pan-FGFR inhibitors can lead to nonselective blockade of these FGFR2 isoforms, challenging the rationale behind specifically targeting FGFR2. Therefore, a multikinase inhibitor targeting FGFR2 and potential resistance pathways could represent a rational approach to maximize treatment response.
In vitro kinase assays and a case report in gastric cancer revealed that pazopanib exhibited activity against FGFR2 amplification, suggesting antitumor activity in an FGFR2-dependent manner.3,5 The activation of FGFR2 was associated with crucial downstream effectors like ERK (Extracellular Signal-Regulated Kinase) and AKT (Protein Kinase B). Upon pazopanib treatment, cells with FGFR2 amplification underwent cell cycle arrest at the G1/S phase and ultimately entered apoptosis, evidenced by elevated levels of cleaved PARP (Poly(ADP-ribose) Polymerase). Interestingly, FGFR2 amplification was the preferred mechanism of FGFR2 activation in gastric cancer, whereas mutations were more common in lung squamous cell carcinoma, lung adenocarcinoma, melanoma, endometrial cancer, and colorectal cancer. Consequently, pazopanib demonstrated the potential to impede FGFR2 mutation-driven tumor progression, showcasing substantial clinical efficacy in these diverse tumor types.
Previously, FGFR2 expression via IHC was identified as a potential biomarker for predicting treatment outcomes in metastatic or recurrent gastric cancer patients treated with pazopanib, capecitabine, and oxaliplatin. In addition, given that pazopanib inhibits not only FGFR but also other receptors with high structural homology, such as VEGFR and PDGFR, there exists the potential to concurrently target each of these receptors with a multi-tyrosine kinase inhibitor. This simultaneous targeting may contribute to a synergistic effect, enhancing the overall therapeutic impact. 21
The patient showed a positive response to pazopanib monotherapy with a good duration of 7.5 months before showing signs of progression. The observation of favorable treatment responses in this patient highlights the value of molecular tumor boards in guiding personalized treatment decisions. The ability of molecular tumor boards to foresee the functional consequences of the FGFR2 mutation emphasizes the critical importance of precision medicine in maximizing patient outcomes. However, the disease progression and the effectiveness of the combinational treatment plan raise the question of whether the initiation of palbociclib and crizotinib from the beginning would have potentially prolonged the initial response duration.
Drawing parallels with the analogy presented by Adashek et al., likening cancer to the nine-headed hydra, our strategy of simultaneously blocking all altered pathways may prove to be a more effective approach – reminiscent of cutting off all of the hydra’s heads at once. This metaphor underscores the potential efficacy of a comprehensive and simultaneous intervention targeting multiple pathways to address the complex and multifaceted nature of cancer progression. 10
Although speculative, co-administration of CDK4/6 inhibitors with pazopanib could have potentially enhanced the patient’s initial response. The hypothetical synergistic effects between these therapeutic agents might have cooperatively inhibited cancer cell proliferation and tumor progression. This combined approach could have effectively targeted both the FGFR2 alteration and the dysregulated CDKN2A/B pathway, presenting a more comprehensive and integrated treatment strategy. An example further substantiating this hypothesis is found in a study examining FGF/FGFR and co-alterations. In this study, six patients with cyclin pathway co-alterations with FGF/FGFR alterations were treated with the combination of CDK4/6 inhibitor palbociclib. The reported response rate in this cohort was an impressive 50%. Notably, one of these patients, a case of osteosarcoma, exhibited a reported response duration surpassing 52 months. 22 The combination was also found safe in terms of toxicity and tolerability. This compelling evidence supports the notion that concurrently targeting multiple pathways, as with CDK4/6 and FGFR inhibitors, may result in significant and prolonged treatment responses in certain cases.
The CDH1 gene encodes the cell adhesion protein E-cadherin, a critical component for cell–cell adhesion and the prevention of tumorigenesis. Mutations in the CDH1 gene can result in the loss of E-cadherin expression, potentially contributing to tumorigenesis. In vitro studies have revealed that crizotinib induces cell death in CDH1-mutant breast cancer cells, and this effect is boosted when E-cadherin expression is compromised. 6 This suggests a synthetic lethality between CDH1 and ROS1, signifying that the simultaneous depletion of both genes has a more significant impact on cell death than the loss of either gene alone. Increased cell proliferation and invasion triggered by the loss of E-cadherin caused by a CDH1 mutation can be further boosted by the activation of ROS1. Crizotinib can intervene in this process by targeting ROS1, thereby reducing proliferation and invasion caused by the confluence of CDH1 mutation and ROS1 activation.
Crizotinib is already approved to treat ROS1-altered lung cancer, 23 and there is potential for its repurposing to treat other cancer types with CDH1 alterations. 24 Ongoing clinical trials of ROS1 inhibitors in CDH1 mutant breast and diffuse gastric cancer patients indicate a potential expansion of their therapeutic application (ClinicalTrials.gov identifier: NCT03620643). These inhibitors may be beneficial in patients with CDH1 mutations, offering a more targeted and effective treatment strategy in the future.
In this specific case, the addition of crizotinib could have further enhanced the treatment strategy. By incorporating crizotinib, all the identified alterations and pathways thought to be involved in tumorigenesis are comprehensively addressed, potentially providing a more robust and synergistic therapeutic approach.
In the present landscape, drug approvals still predominantly operate within an organ-based framework, although a transformative shift is undeniably underway. For precision oncology clinics, steering cancer treatments guided by genomic data, there exists a notable reliance on off-label recommendations around the globe. 25 Estimates even suggest that as many as 71% of adult cancer patients receive drugs off-label during their treatment. 26
During the shift toward the ‘tumor-agnostic era’, where guidelines are moving beyond the focus on specific organs, a period of evolution is being experienced. Guidelines are adapting to incorporate the intricacies of personalized treatment plans guided by precision oncology principles. This transition involves a departure from traditional organ-based approaches, inevitably leading to more off-label applications in treatment planning. This case report serves as an illuminating example in this changing landscape, showcasing how oncological care is dynamically transforming to align with the principles of precision medicine to address the unique needs of each patient.
Patient update
According to the most recent assessment, the patient continues her daily activities without any reported issues or adverse effects.
Conclusion
This case report underscores the significance of a personalized and precision-oriented therapeutic approach in the management of advanced cholangiocarcinoma. The utilization of pazopanib as an alternative in the absence of readily accessible pan-FGFR inhibitors demonstrates the adaptability required in diverse clinical settings. The exploration of less common FGFR2 alterations expands our understanding of targeted therapies beyond prevalent mutations. The integration of co-targeting agents in response to disease progression, particularly with palbociclib for CDKN2A/B alterations and crizotinib for a CDH1 alteration, emphasizes the dynamic nature of precision oncology. The observed synthetic lethality between crizotinib and CDH1 alterations further supports the translation of preclinical evidence into clinical settings. This case report not only contributes to the evolving landscape of cholangiocarcinoma treatment but also underscores the pivotal role of molecular tumor boards and dosage adjustments in optimizing therapeutic outcomes for patients with intricate genomic profiles. In conclusion, the explorative nature of the discussed treatment necessitates validation by a tumor board before implementation. Thorough consideration, guided by the limited existing data on this therapeutic association, emphasizes the importance of cautious application and the need for further research in shaping a comprehensive and adaptable approach to precision oncology for addressing the complexity of genomic alterations.
Footnotes
Acknowledgements
We would like to express our gratitude to Prof. Dr Ayşim Büge Öz for her contribution to the histologic examination and Assoc. Prof. Dr Lebriz Uslu from Istanbul University Cerrahpasa Medicine Faculty for her assistance in evaluating the response based on PERCIST criteria. Figures 1, 2, and 5 were created with ![]() by the first author Esranur Aydşn.
by the first author Esranur Aydşn.
