Abstract
Chromosomal rearrangements involving the c-ros oncogene 1 (ROS1) gene define a subset of non-small cell lung cancers highly sensitive to small-molecule tyrosine kinase inhibitors. However, little is known about the impact of different fusion partners on tyrosine kinase inhibitor efficacy. We herein describe a case of a 26-year-old never-smoker patient from southern Africa with metastatic lung adenocarcinoma driven by SLC12A2-ROS1 fusion, who had a pronounced and durable response to crizotinib. The present case underscores the importance of pursuing actionable alterations in patients with similar clinical and epidemiological characteristics. In addition, provides the second report of crizotinib activity against lung malignancies harboring the unique SLC12A2-ROS1 fusion and highlights the importance of a deeper understanding of molecular alterations in underrepresented subgroups of patients to tailor the decision-making in daily practice.
Keywords
Introduction
ROS1 gene fusions are well-described oncogenic drivers present in ~2% of non-small cell lung cancers (NSCLCs). 1 Along with never/light smoking history, higher risk of thromboembolic events and brain metastases are also associated with ROS1 rearrangement.2,3 CD74 gene is the fusion partner in approximately 40%–50% of the cases, followed by SLC34A2, EZR, SDC4, LIMA1, MSN, and TPM3;4,5 however, there are insufficient data to support differences in their prognostic or predictive roles for the available therapeutic arsenal. 5
In addition to crizotinib and entrectinib U.S. Food and Drug Administration (FDA)-approval in the frontline setting of advanced ROS1 + NSCLCs, newer agents with high central nervous system (CNS) penetration such as lorlatinib and repotrectinib (TPX-0005) have recently expanded perspectives in the management of this condition for both naïve and pretreated patients. Although ROS1 on tyrosine kinase inhibitors (TKIs) have demonstrated high response rates in prospective studies (~70%), uncertainties regarding their performance against rare and undescribed fusions persist, highlighting how reports in the literature may be of clinical use.3,5–8 Moreover, it is noteworthy that an existent knowledge gap related to a comprehensive genomic characterization of tumors arising in patients of African descent have long posed additional challenges to a more informed clinical decision regarding the optimal management of these patients.4,9,10
A young woman with metastatic lung adenocarcinoma (LAC) harboring ROS1 rearrangement showed a remarkable response to crizotinib. In her tumor cells, we identified the unique SLC12A2-ROS1 rearrangement, against which the efficacy to ROS1 TKIs is unclear. A written informed consent for this publication was obtained by the investigator.
Case presentation
A 26-year-old never-smoker female patient born in southern Africa presented, in February 2013, with a left inferior lobe mass measuring roughly 8.0 cm. A computed tomography (CT)–guided biopsy confirmed the diagnosis of LAC, and the patient underwent a left inferior lobectomy at that time. The pathology report disclosed a poorly delimited peripheral bronchioloalveolar component, with a 5.5-cm invasive neoplasm with predominant acinar pattern, also displaying papillary, micropapillary, clear cell, and signet-ring cell components, lymph node negative (AJCC 8th edition: pT3 pN0 M0).
From October to December 2013, the patient received four cycles of adjuvant cisplatin and pemetrexed, starting regular follow-up thereafter. Of note, she had also undergone a germline testing through an 84-gene panel, without any pathogenic variants. Six years later, she presented with bilateral pulmonary involvement at a chest CT scan (Figure 1(a)–(c)) and a transthoracic needle biopsy histologically confirmed recurrent LAC displaying acinar and micropapillary patterns, with positive immunostaining for TTF-1 and Napsin (Figure 2(a)–(d)). At that point, a PD-L1/TPS (programmed cell death ligand-1/tumor proportion score) of <1% was observed at Ventana PD-L1 SP263 immunohistochemical assay. Since an NGS (next-generation sequencing) Trusight Tumor 170 (Illumina®) test had not shown the presence of any driver alterations, the patient was started on carboplatin, pemetrexed, and pembrolizumab every 21 days. Nevertheless, following three cycles of chemoimmunotherapy, her respiratory symptoms worsened, and the patient came to the outpatient clinic complaining of intense cough and shortness of breath on exertion. A positron emission tomography–CT (PET-CT) scan was performed, disclosing bilateral pulmonary disease progression. Upon disease progression, in the face of very limited therapeutic options and insufficient material remaining, we decided to perform a transthoracic core biopsy and to order another NGS assay (FoundationOne®CDx test) in a new biopsy tumor sample. Due to considerable respiratory symptoms and clinical deterioration, she started on second-line paclitaxel and bevacizumab while waiting for the molecular profiling results.
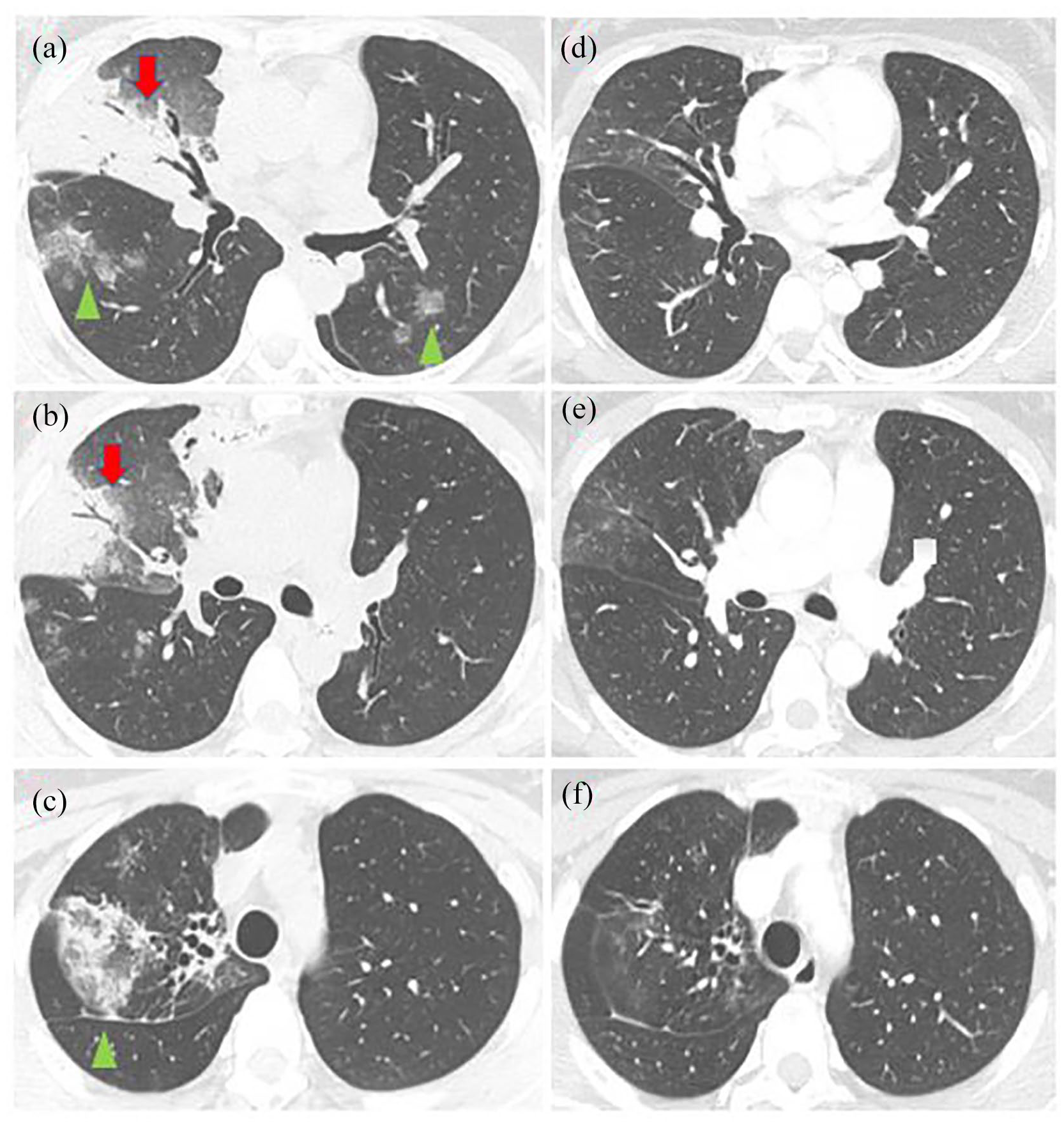
Thorax CT scan (lung window) showing

(a) Left lung transthoracic biopsy (HE 60×): adenocarcinoma infiltrating pulmonary tissue and exhibiting acinar and micropapillary architectural patterns. (b) Left lung transthoracic biopsy (HE 200×): high power view of lung adenocarcinoma showing acinar (upper left) and micropapillae (lower right). (c) Immunohistochemistry showing positivity for TTF1. (d) Immunohistochemistry disclosing positivity for Napsin.
The new comprehensive genomic panel disclosed the presence of a SLC12A2-ROS1 fusion which, at that time, had not been previously reported as an oncogenic rearrangement neither at OncoKB, Quiver, Mitelman, tumorfusions.org, cBioPortal nor at Catalog of Somatic Mutations in Cancer (COSMIC) databases. The fusion breakpoints based on GRCh37/hg19 are chr5:127492189-127492398 (intron 15) of SLC12A2 gene and chr6:117641629-117642055 (intron 35) of ROS1 gene, resulting in a fusion 5′-SLC12A2(ex1-15 NM_001046)-ROS1(ex36-43 NM_002944), with 96 supporting read pairs (Figure 3). This same sample was then subjected to RNA-based fusion panel (NGS; Trusight Tumor 170, Illumina®) that confirmed the fusion transcript of the previously reported fusion. Of note, this new biopsy disclosed acinar, micropapillary, papillary, and lepidic non-mucinous histologic patterns. We hypothesize that our first NGS assay did not identify this fusion due to low representative tumor sampling; it concurrently analyzes DNA and RNA, identifying single nucleotide variants (SNVs) and indels in 149 genes, copy number variations in 59 genes, as well as fusions in 55 genes.

Illustration of the unique SLC12A2-ROS1 fusion with specific breakpoints, showing ROS1 kinase domain preservation (illustration created with aid of Microsoft PowerPoint).
Owing to the presence of a novel ROS1 fusion (and regardless of unavailable reports in the literature pointing to crizotinib sensitivity), the patient was started on crizotinib 250 mg twice daily in March 2020. Liver function tests (LFTs) were followed weekly due to a higher risk of hepatotoxicity during the use of ROS1 TKI following immunotherapy. She experienced a fast symptomatic relief and the chest CT scan performed 4 weeks later showed marked tumor regression (Figure 1(d)–(f)). To date (December 2021), she remains in complete response, asymptomatic, with no treatment-related toxicities and attending regular visits to the outpatient clinic with follow-up imaging and blood tests. LFTs remain normal and are still followed closely. Moreover, due to the fast clinical and radiological improvement experienced in the setting of an aggressive disease (alongside the excellent tolerance to crizotinib), the patient reports that she feels safe, satisfied, and optimistic with regard to the ongoing treatment. A timeline summarizing the patient’s entire treatment is shown in Figure 4.
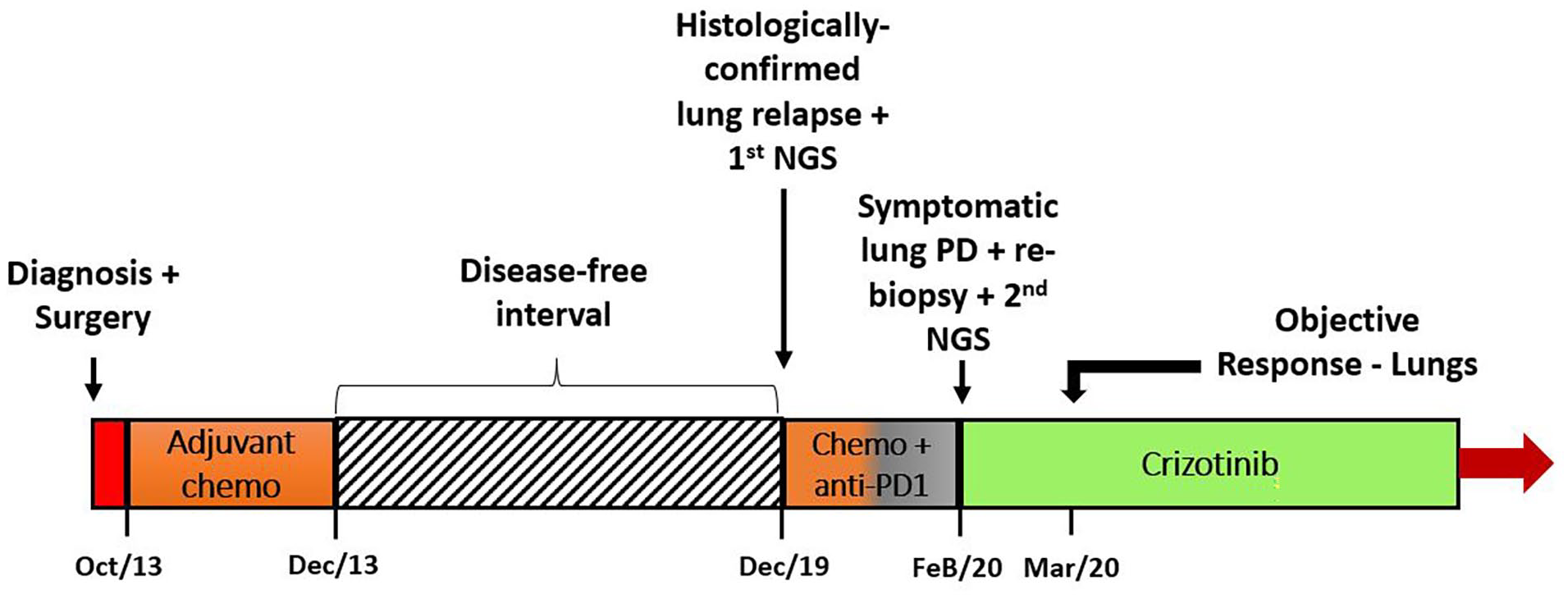
Timeline summarizing the patient’s treatment.
Discussion
ROS1 is located on chromosome 6q22 and encodes an orphan transmembrane tyrosine kinase. When fused to its upstream partners, a constitutively activated chimeric protein kinase capable of driving tumorigenesis in solid tumors is encoded.11–14 As depicted by the present case, ROS1 + NSCLCs are, predominantly, adenocarcinomas arising in young female and never-smoker patients, justifying a laborious and thorough molecular analysis searching for actionable alterations in individuals with similar characteristics. Findings reported by Kohsaka et al., 15 in which oncogenic drivers were detected in 70.4% of 125 Japanese never/light-smoker LAC patients previously considered driver-negative by conventional laboratory testing, underscore the importance of pursuing targets prior to systemic therapy initiation. Our patient experienced an utterly fast and significant clinical and radiological improvement in the setting of a chemoimmunotherapy-refractory disease right after crizotinib start, without any safety concerns for liver toxicity.
Crizotinib activity in advanced ROS1 + NSCLCs has been explored in the PROFILE 1001 trial, which recruited mostly pretreated patients to receive the dosage of 250 mg twice daily. Crizotinib showed objective response in 72% of the patients, with median progression-free survival (PFS) and overall survival (OS) of 19.3 and 51.4 months, respectively. However, since no correlation between specific fusion partners and efficacy outcomes has ever been consistently demonstrated, this still remains an open question.3,5,8,16 The present case provides the second report in the literature that NSCLCs harboring SLC12A2-ROS1 fusions can respond to second-line crizotinib as well, with our patient remaining progression-free for 21 months now. 17
Although the incidence of CNS metastasis in advanced ROS1 + NSCLCs is described as a frequent event, the precise proportion of patients with secondary involvement is conflicting, ranging from nearly 20%–40% in treatment-naïve patients to 50%–56% in crizotinib-pretreated patients according to data from prospective trials. 8 This CNS tropism, along with an inferior blood-brain barrier penetration of crizotinib (when compared to other newer-generation multi-kinase inhibitors), and the paucity of therapeutic options for crizotinib-resistant tumors, prompted the study of these modern TKIs in the context of ROS1 + malignancies. Entrectinib, lorlatinib, and repotrectinib all have demonstrated promising antitumor activity, comprising both crizotinib-resistant and naïve tumors, but no conclusive data regarding the role of different fusion upstream partners have been described for these agents as well.5,7,8,18
Even more intriguing is the scant literature regarding molecular profiling of NSCLCs arising in underrepresented subgroups, such as patients of African descent, for whom a deeper understanding would contribute enormously to a better prognostication and management. Although it is unknown whether SLC12A2-ROS1 fusion occurs more frequently in this subgroup of patients, our report reinforces the concept that not only this fusion is oncogenic but also confers sensitivity to ROS1 TKIs, which could theoretically contribute to tailor clinical decision for individuals diagnosed with lung tumors harboring this alteration.17,19 Interestingly, the case reported by Rodriguez-Antolin and colleagues displays some intriguing similarities with our case, for instance, (1) very young age of diagnosis (21 vs 26 years old, which could contribute to delay the diagnosis), (2) symptomatic and high-volume lung involvement at diagnosis of advanced disease, (3) refractoriness to frontline chemoimmunotherapy with subsequent clinical deterioration, as well as (4) fast and deep response to crizotinib. In this specific case, the ROS1 fusion breakpoint occurred in exon 36, as opposed to our case and Li et al’s 19 description (introns 35 and 34, respectively). 17 Importantly, based on the recent identification of SLC12A2-ROS1 fusion in a head and neck inflammatory myofibroblastic tumor, it is reasonable to consider that this fusion can also act as important oncogenic event in other solid tumors. 20
Finally, it is worth highlighting that stimulating specific studies and recruitment among underrepresented subgroups of patients may not only contribute to unravel and characterize novel and rare genomic alterations but also help to pave the way toward an equality with regard to data generated by clinical trials and, consequently, to address ethnic differences in upcoming guidelines.10,21
Conclusion
In summary, SLC12A2-ROS1 fusion is an uncommon oncogenic driver in NSCLCs and responds to ROS1 inhibition; our report is the second description of crizotinib activity in this setting. Whenever possible, comprehensive molecular assessment should precede treatment initiation in subgroups of NSCLC patients with higher likelihood of having oncogenic drivers.
Footnotes
Author contributions
Conception and design: all authors; Administrative support: M.P.M., L.A.T., and A.K.; Provision of study materials or patients: M.P.M., L.A.T., E.C.T.N., and A.K.; Collection and assembly of data: M.P.M. L.A.T., E.C.T.N., and M.F.S.A.R.; Data analysis and interpretation: M.P.M., E.C.T.N., L.A.T., A.K., M.F.S.A.R., K.P.S., and C.D.H.L.; Manuscript writing: All authors; Final approval of manuscript: All authors.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.F.S.A.R. has received support for attending meetings and travel expenses from Foundation Medicine. K.P.S. has received support for attending meetings and travel expenses from Astra-Zeneca. She has also received honoraria from Eurofarma. All other authors have no conflicts of interest to declare.
Ethical approval
Ethical approval to report this case was obtained from Hospital Sirio Libanes/Sociedade Beneficente De Senhoras ethical review board (HSL-RC-2021-14).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Sociedade Beneficente de Senhoras—Hospital Sírio-Libanês, São Paulo, SP, Brazil.
Informed consent
Written informed consent was obtained from the patient for her anonymized information to be published in this article.
